Matter and Change Chapter 2 Matter Definitions Anything


























- Slides: 31

Matter and Change Chapter 2

• Matter Definitions – Anything that has ____ and takes up _____ • Volume – Space the object ______ • What would you use to determine the volume of… – Book? Air? Water? • Mass – ______ of matter contained in an object. • What do we use in lab to determine mass?

Mass vs. Weight • Are they the same? ? ? • Big difference… – Weight depends on the location in the ______. It will change, mass does not change no matter the location. • Weight is defined as – The force produced by ______ acting on mass

Solid Liquid Gas

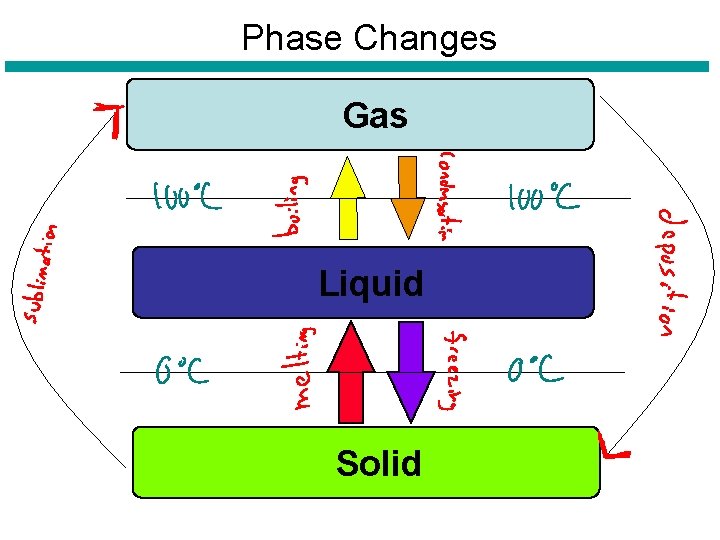
Phase Changes Gas Liquid Solid

What makes up matter • All matter is composed of about 118 different kinds of _____s. • Atom – Smallest unit of an element that maintains the properties of that element.

Pure Substance single element or compound that has definite chemical & physical properties • Element – Contains only ____ atom – Can not be broken down by chemical means or physical means • Compound – Made up of atoms of two or more elements joined by a _____bond – Can be broken down by chemical means, cannot be broken down by physical means

Elements exist as: • Single atoms – Example: He in balloon (monatomic gas) • Molecules – ____ or more atoms combine in a definite ratio. • Examples Br 2, I 2, N 2, Cl 2, H 2, O 2, F 2 -all diatomic molecules

Allotrope • One of a number of different molecular forms of an element. – Examples • O 2 is oxygen • O 3 is ozone • Carbon you know as _____ and _____

Compounds • Are represented by formulas. • Molecular formulas – Tells only what makes up a compound • Structural formulas – Shows how the atoms are connected

Mixture vs. Pure Substance A pure substance has only ____ kind of element or ______. Where as a mixture has more than one kind of ______ or molecule.

Mixtures vs. Compound • Mixture • Compound • ____ chemically joined • Reflect the properties of the substances it contains • Mixture’s components can vary in proportions • Chemically joined • Has properties different from elements that make it up • Has a ______ composition

Types of Mixtures Homogeneous Mixtures in which the composition is uniform throughout. (i. e. the phases cannot be distinguished. ) Called a _________!!! Heterogeneous Mixtures in which the composition is not uniform throughout. (i. e. the phases can be distinguished. ) Known as a ________!!!

Separation of Matter Elements: Can only be broken down by _________ reactions. Compounds: Can only be broken down by _________ reactions. Mixtures (hetero) and Solutions (homogeneous): Can be broken down by ___________ means.

Physical Means of Separation Filtration: Separates mixtures by particle size. Used to separate solids from liquids

Physical Means of Separation Distillation: Separates liquid mixtures by differences in boiling point. Txtbk p 34 http: //www. dit. ie/DIT/science/chemistry/rsccomp/competition 00/distillation/labdistillation. JPG



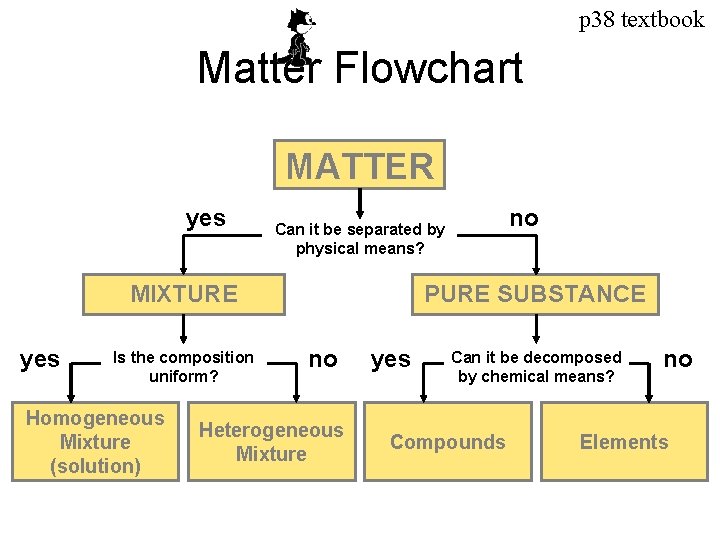
p 38 textbook Matter Flowchart MATTER yes MIXTURE yes Is the composition uniform? Homogeneous Mixture (solution) no Can it be separated by physical means? PURE SUBSTANCE no Heterogeneous Mixture yes Can it be decomposed by chemical means? Compounds no Elements

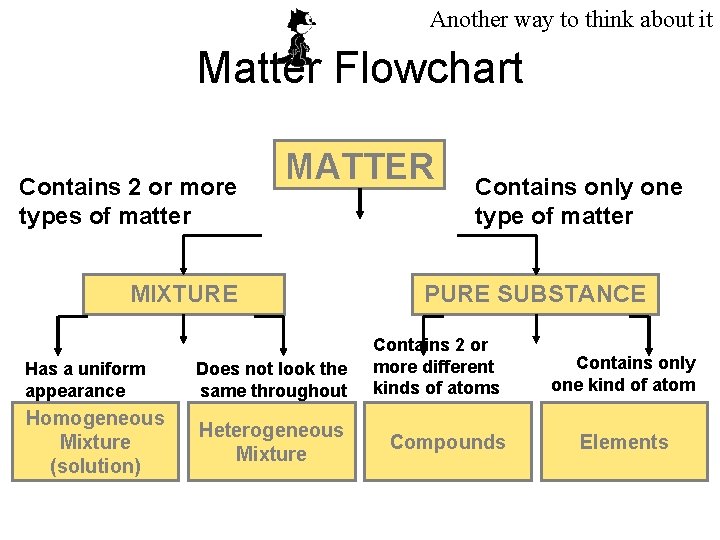
Another way to think about it Matter Flowchart Contains 2 or more types of matter MATTER MIXTURE Has a uniform appearance Does not look the same throughout Homogeneous Mixture (solution) Heterogeneous Mixture Contains only one type of matter PURE SUBSTANCE Contains 2 or more different kinds of atoms Compounds Contains only one kind of atom Elements

Words to know and use • • • Mixture Pure substance Element/Atom Compound/Molecule Homogeneous (solution) Heterogeneous (mixture) Allotrope Alloy Filtration Distillation

• What is wrong with the phrase… – “heterogeneous compound” – “pure mixture”

Properties of Matter (How we describe matter) • Physical – Can be determined without ______ the nature of the substance. – Examples: • Density, hardness, color • Chemical – Describes a substance’s ______ to participate in a chemical reaction. – Ability to change – Examples: • Reactivity, Stability, Flammability • Reactivity with oxygen, decomposes with light

Types of Physical Properties • Extensive- depends on the _____ of matter • Ex: volume, mass, amount of energy • Intensive- _____ depend on the amount • Ex: density, boiling point, ability to conduct

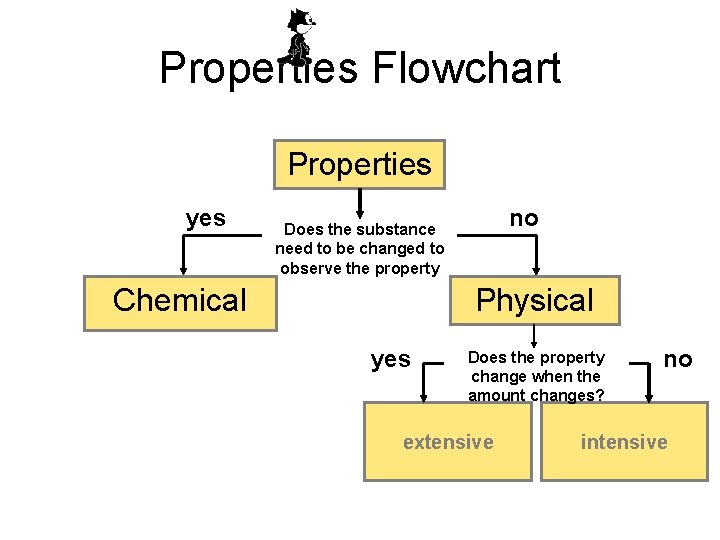
Properties Flowchart Properties yes no Does the substance need to be changed to observe the property Chemical Physical yes Does the property change when the amount changes? extensive no intensive

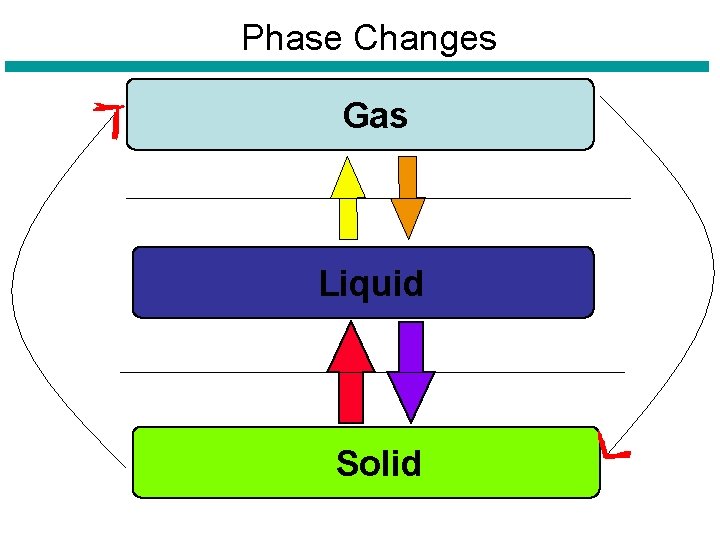
Changes of Matter • Physical Change – Identity (or chemical makeup) of a substance does not change, however the ____, ______ and ______ of particles may change. – Examples: • • Crushing Ripping Bending Phase changes

Phase Changes Gas Liquid Solid

Changes of Matter • Chemical Change – Identities of substances changes and ____ substances form. • Examples: – ______

5 Indicators of a Chemical Change: • Evolution of a gas • Odor change • Production of heat/light • Color change • Formation of a precipitate


Reactions • A reaction has – Reactant (left side) • What ____ in the reaction – Product (right side) • What is ______ Example of a chemical: Burning Dollar CH 3 OH + O 2 CO 2 + H 2 O – Law of Conservation of Mass • Mass is neither created nor destroyed in a chemical reaction – Almost true