Matter and Change Chapter 1 Branches of Chemistry

Matter and Change Chapter 1
Branches of Chemistry • Organic Chemistry • Inorganic Chemistry • Physical Chemistry • Analytical Chemistry • Biochemistry • Theoretical Chemistry …. and many more!

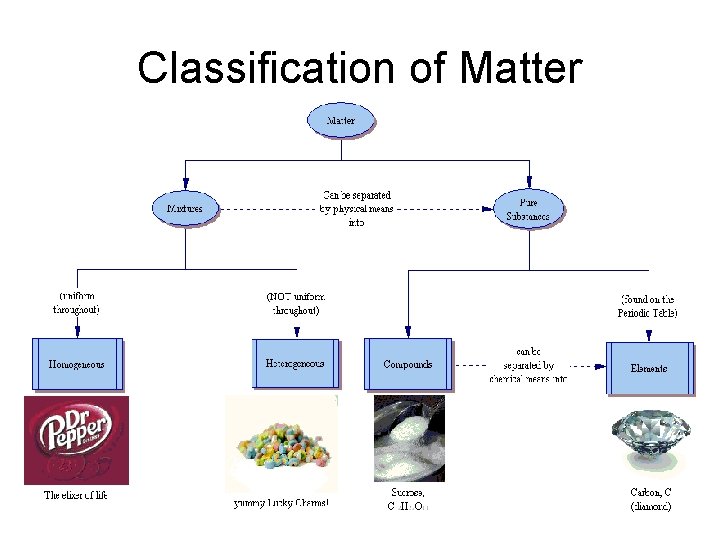
Matter anything that has mass and takes up space pure substances – compounds and elements mixtures – two or more pure substances mixed in the same container…not bonded

Pure Substances Element – A pure substance made of only one kind of atom Atom – An atom is the smallest unit of an element that maintains the properties of that element

Pure Substances • Compound – A substance that is made from the atoms of two or more elements that are chemically bonded. Molecule – the smallest unit of an element or compound that retains all of the properties of that element or compound

Huh? How can a molecule describe an element?
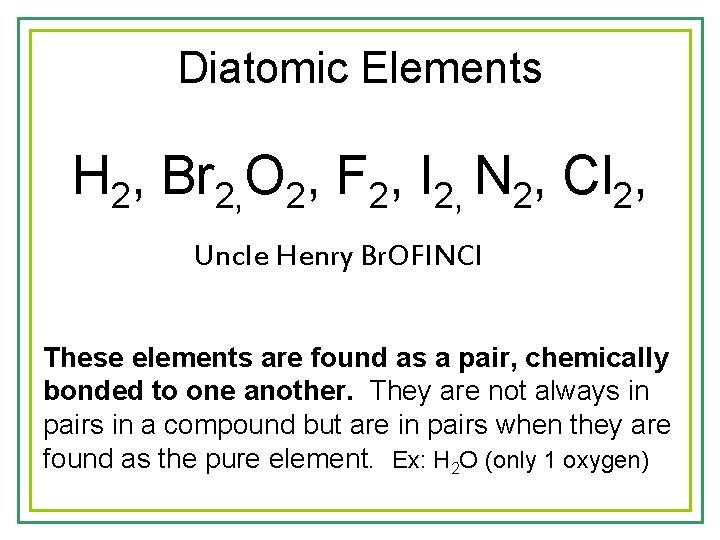
Diatomic Elements H 2, Br 2, O 2, F 2, I 2, N 2, Cl 2, Uncle Henry Br. OFINCl These elements are found as a pair, chemically bonded to one another. They are not always in pairs in a compound but are in pairs when they are found as the pure element. Ex: H 2 O (only 1 oxygen)
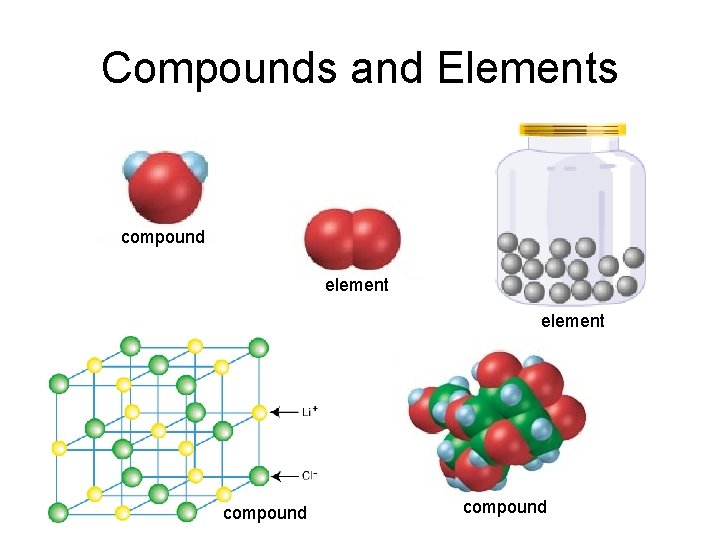
Compounds and Elements compound element compound
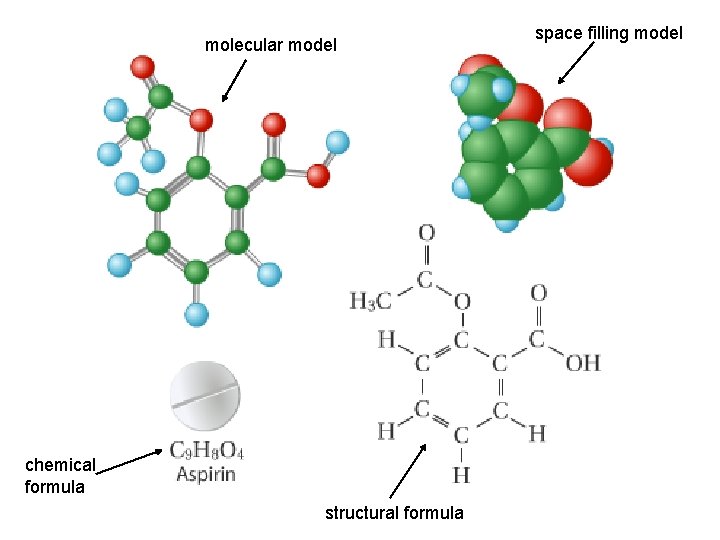
molecular model chemical formula structural formula space filling model

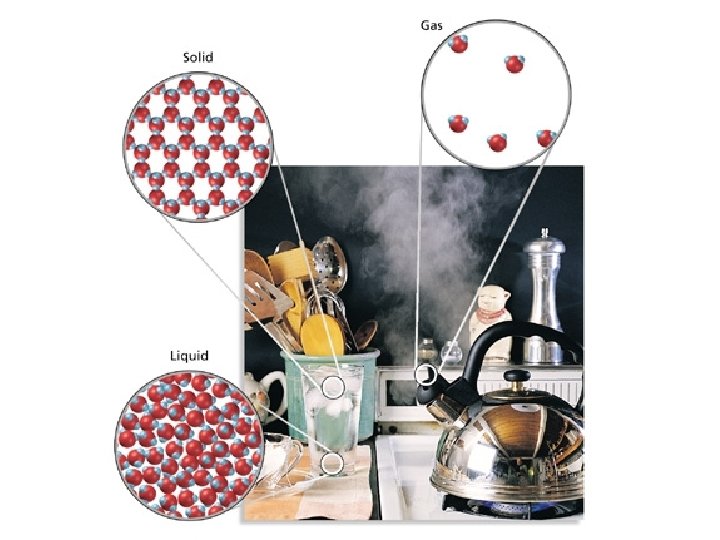
SHAPE • Solid • definite shape VOLUME • definite volume • Liquid • indefinite shape • definite volume • Gas • indefinite volume • indefinite shape

States of Matter • The particles of a solid are packed together in relatively fixed positions. • The particles have strong attractive forces and low energy. • (s)

Solids • Solids are in a relatively fixed position. • Solids have only vibrational movements around fixed points. • Solids have definite shape and volume. • Solids are almost incompressible. • Solids do not diffuse (practically).

Solids • Solids are either crystalline or amorphous. crystalline – consist of crystals, particles arranged in an orderly, geometric repeating pattern amorphous – Greek for “without shape”; consist of particles, randomly arranged
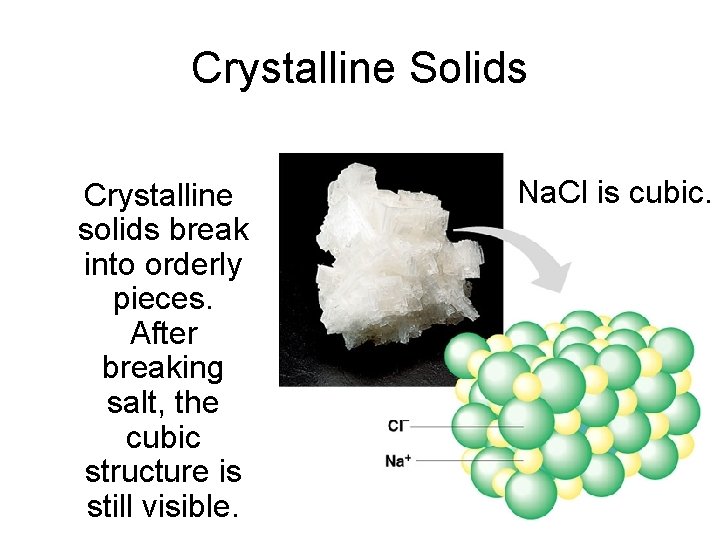
Crystalline Solids Crystalline solids break into orderly pieces. After breaking salt, the cubic structure is still visible. Na. Cl is cubic.
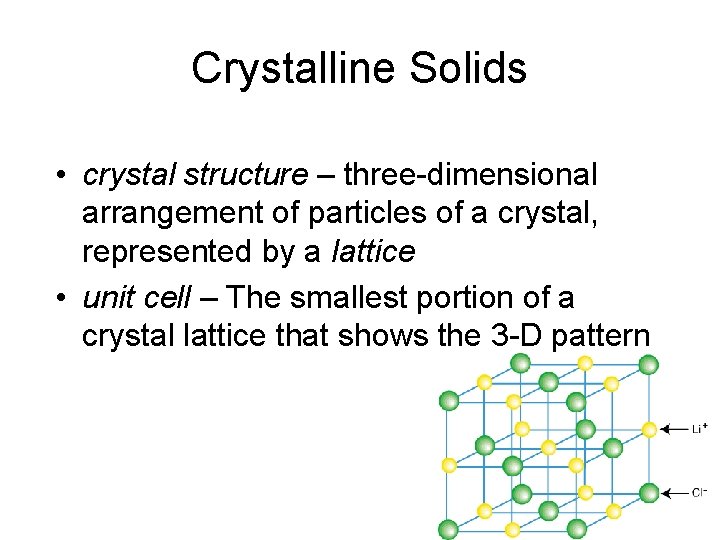
Crystalline Solids • crystal structure – three-dimensional arrangement of particles of a crystal, represented by a lattice • unit cell – The smallest portion of a crystal lattice that shows the 3 -D pattern

Crystalline Systems

Amorphous Solids Amorphous solids break into random pieces. They usually shatter into irregular shapes. Most plastics are amorphous.

Amorphous Solids • The freezing point of amorphous solids can vary according to how slowly the material cools. (Ex: butter) • http: //math. ucr. edu/home/baez/physics/ General/Glass/glass. html

States of Matter • The particles of a liquid are close together but can most past one another. • These particles have more energy than those of a solid, and this energy is what allows a liquid to flow. • Pure liquid = (l) • A liquid solution = (aq)

Liquids LIQUIDS… • have a definite volume and take on the shape of their container (unlike gases) • have a high density • are not compressed well (brake fluid) • diffuse (like food coloring in water) • have surface tension • are fluids (but fluid ¹ liquid)

Liquids surface tension – a force that tends to pull adjacent parts of a liquid’s surface together, thereby decreasing surface area to the smallest possible size. Surface tension results from attractive forces between the particles in the liquid. The stronger the attractive force, the higher the surface tension.
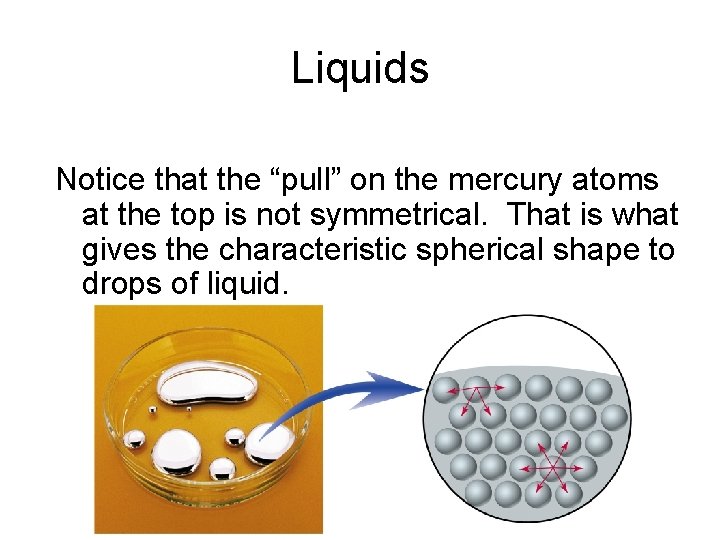
Liquids Notice that the “pull” on the mercury atoms at the top is not symmetrical. That is what gives the characteristic spherical shape to drops of liquid.

Liquids viscosity – the resistance of a liquid to flow high viscosity = “thick” liquid low viscosity = “thin” liquid Liquids with stronger intermolecular forces have higher viscosity. An increase in temperature will decrease the viscosity.

Liquids • volatile liquid - a liquid that evaporates readily at low temperatures The higher the volatility of a liquid, the weaker the intermolecular forces of attraction between their particles. An increase in T will increase evaporation.
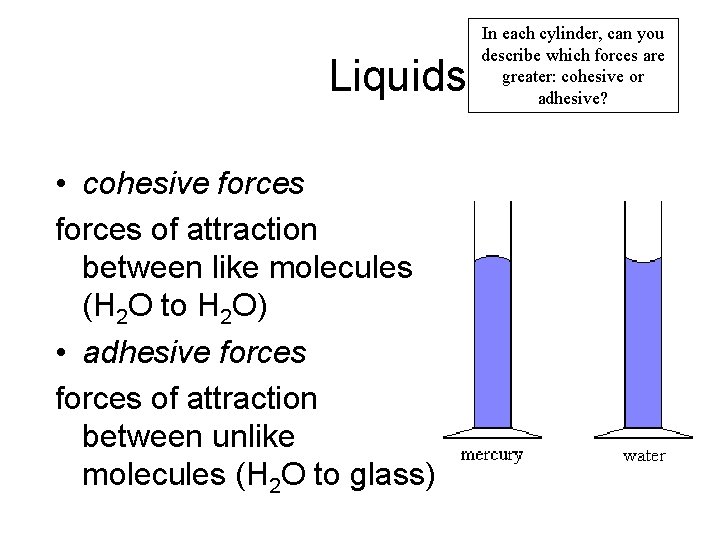
Liquids • cohesive forces of attraction between like molecules (H 2 O to H 2 O) • adhesive forces of attraction between unlike molecules (H 2 O to glass) In each cylinder, can you describe which forces are greater: cohesive or adhesive?

Liquids • capillary action - the attraction of the surface of a liquid to the surface of a solid (adhesive forces) Many liquids will “creep” along a solid, like water does to paper or cloth fibers until the pull of gravity is too much for it to overcome.

States of Matter • The particles of a gas move rapidly past one another, and are at a great distance apart from one another compared to their size. • These particles have very high energy. • (g)

States of Matter - Plasma • A plasma is a very high temperature physical state of matter in which atoms lose their electrons. • Plasma is found in fluorescent light bulbs and plasma Televisions.
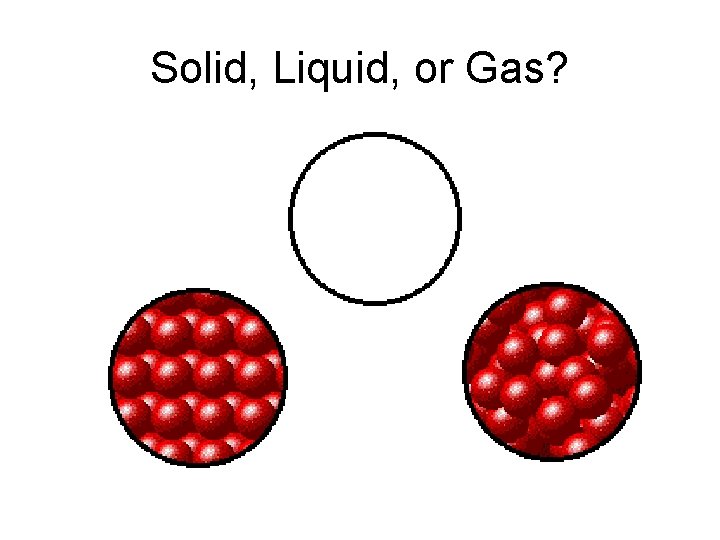
Solid, Liquid, or Gas?
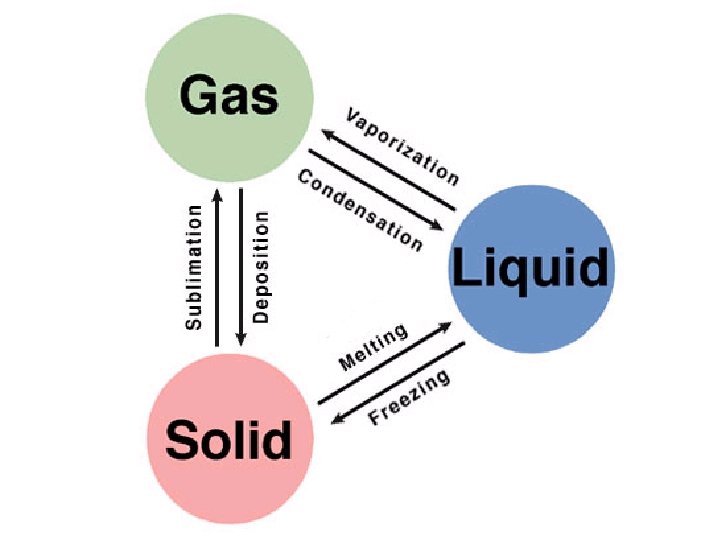
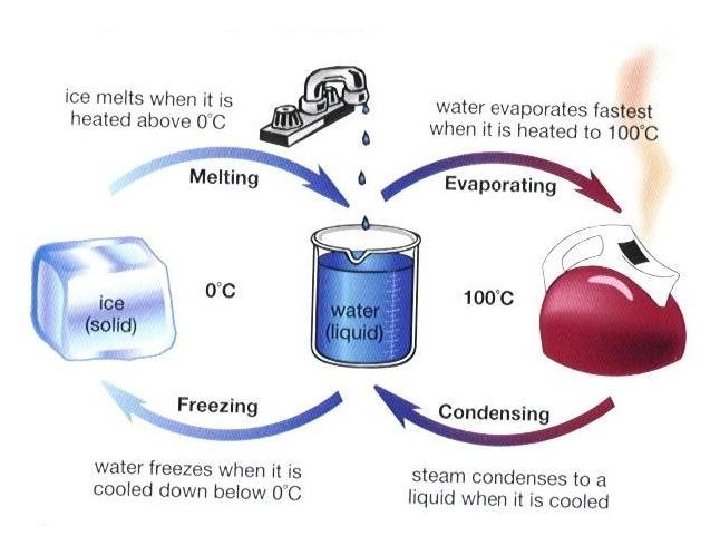


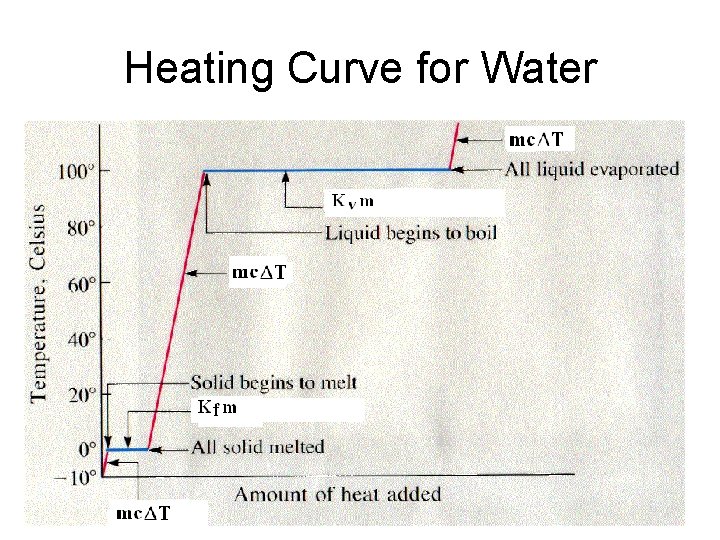
The Kinetic Theory of Heat and Temperature When a phase change is occurring, the temperature does not change, only the position of the particles. (PE) When something is being heated and it is not changing phase, the temperature will rise. (KE)

Potential Energy Differences PE – “energy of position”

PE vs. KE • Only differences in kinetic energy are reflected by temperature differences. • Difference in potential energy are NOT reflected in temperature differences. THERE IS NO TEMPERATURE CHANGE DURING A PHASE CHANGE.

Phase Changes • During solidification or melting DH = Kfm Kf = heat of fusion, the amount of heat needed to melt/freeze 1 g of a substance • During boiling or condensing DH = Kvm Kv = heat of vaporization, the amount of heat needed to boil/condense 1 g of a substance

Phase Changes • Kf for water = 333 J/g • Kv for water = 2260 J/g
Heating Curve for Water
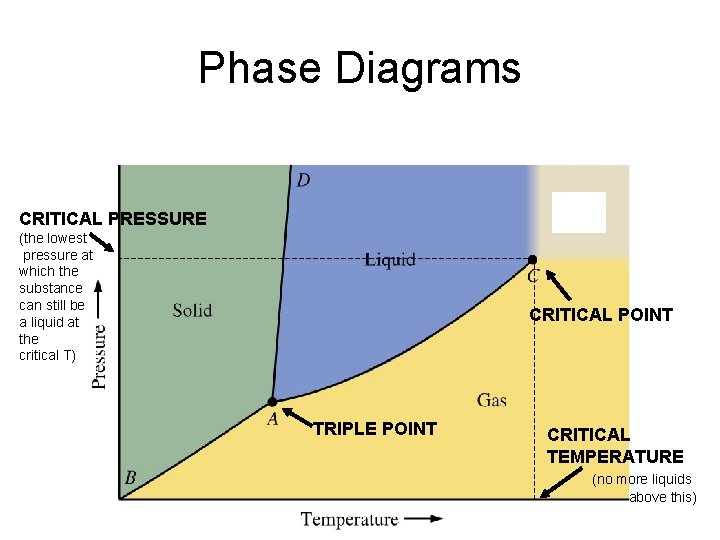
Phase Diagrams CRITICAL PRESSURE (the lowest pressure at which the substance can still be a liquid at the critical T) CRITICAL POINT TRIPLE POINT CRITICAL TEMPERATURE (no more liquids above this)
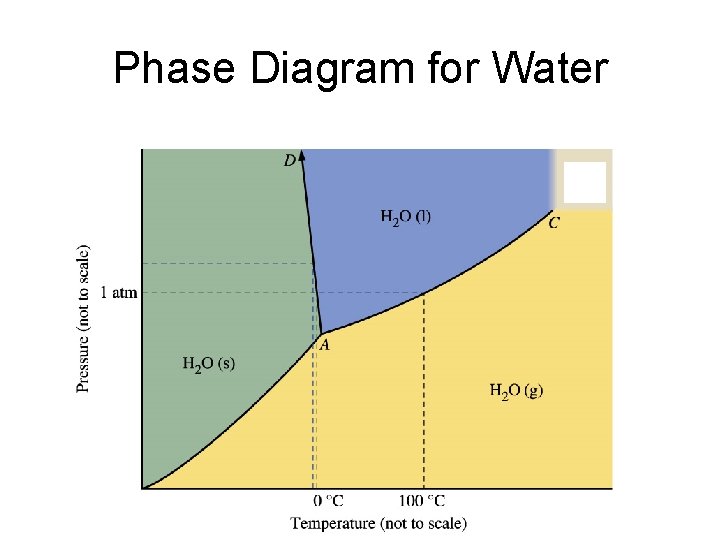
Phase Diagram for Water
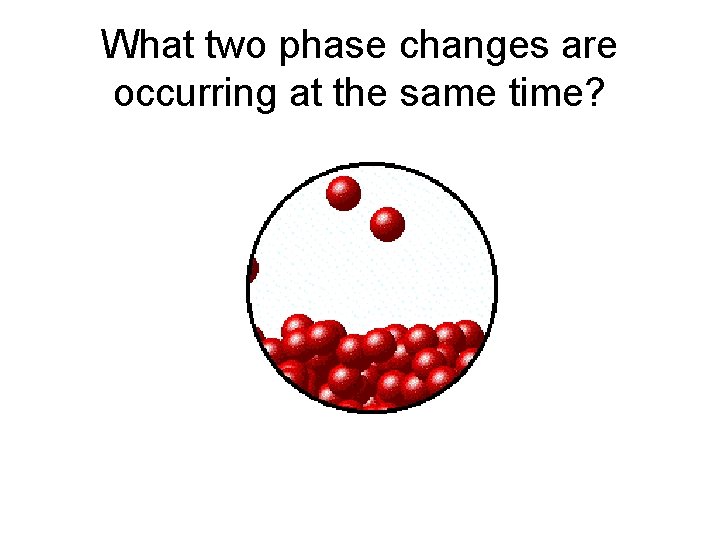
What two phase changes are occurring at the same time?

Physical Properties • physical property – a characteristic that can be observed or measured without changing the identity of the substance melting point mass state of matter color

Physical Changes • physical change – a change in a substance that does not involve a change in the identity of the substance breaking cutting boiling dissolving tearing
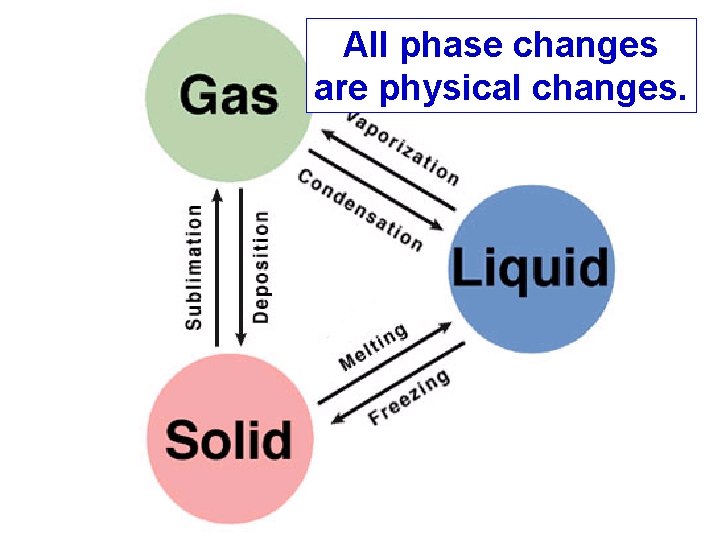
All phase changes are physical changes.

Chemical Properties • chemical property – a characteristic that can be observed or measured with a change in the identity of the substance flammability reacts with an acid reacts with oxygen

Chemical Changes • chemical change – a change in a substance that does involve a change in the identity of the substance color change gas released (often with an odor) energy change (light, heat, …) precipitate formed …four good indications of a chemical change.

Precipitate aqueous – dissolved in water • precipitate – a solid formed from two aqueous solutions during a chemical reaction

Extensive vs. Intensive Properties • extensive – DOES depend on the amount of matter present Examples: mass, volume, amount of energy within a substance • intensive – does NOT depend on the amount of matter present Examples: color, melting point, density, luster If mass and volume are extensive properties, why is density an intensive property?
Classification of Matter
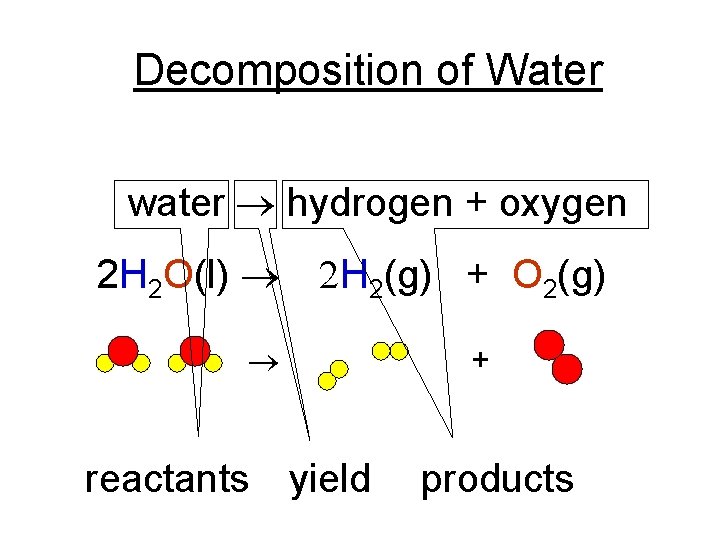
Decomposition of Water water ® hydrogen + oxygen 2 H 2 O(l) ® 2 H 2(g) + O 2(g) ® reactants yield + products

Hoffman Apparatus • The Hoffman Apparatus is used to decompose water into its pure elements. This is a chemical change. The new substances (hydrogen gas and oxygen gas) did not retain the properties of the original substance (water). • Watch this video!

Homogeneous Mixtures the same throughout…each sample contains the same ratio of ingredients (Also called solutions) Heterogeneous Mixtures different throughout…each sample contains a different ratio of ingredients

Separation of Mixtures density PHYSICAL changes only magnetism by hand evaporation filtration chromatography

Separation of elemental Fe magnetism This doesn’t work with iron that is bound into a compound, only with elemental Fe.
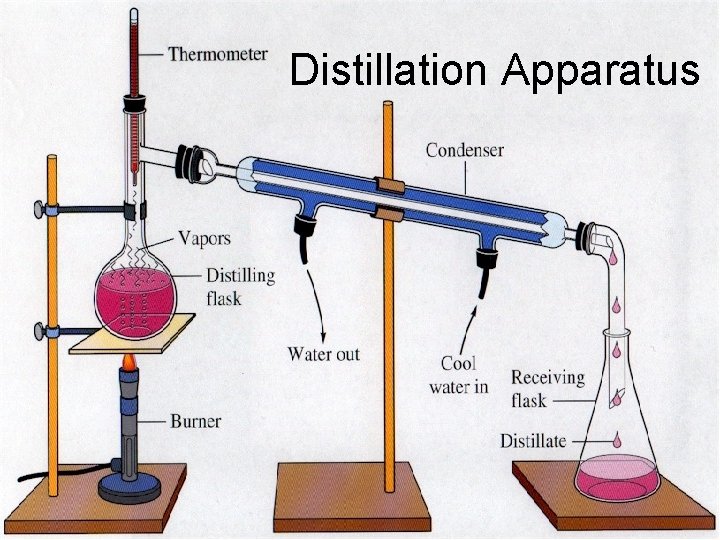
Distillation Apparatus
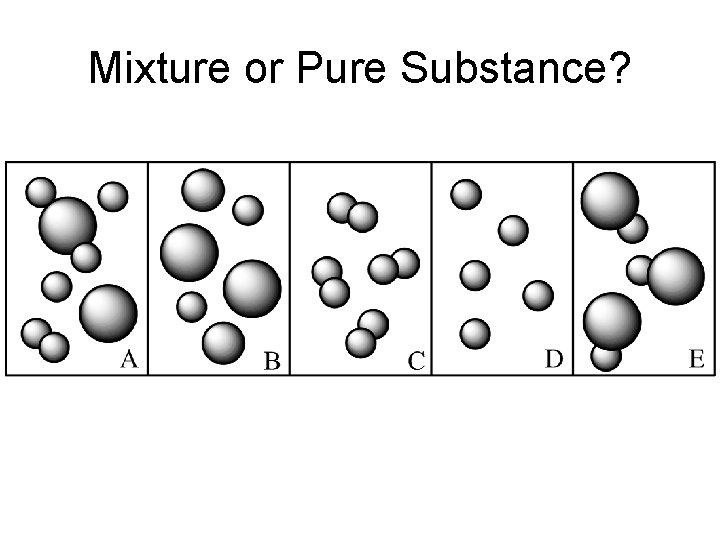
Mixture or Pure Substance?

Homogeneous Mixture, Heterogeneous Mixture or Pure Substance?
- Slides: 58