Matter and Atoms Vocabulary Matter Anything that takes
Matter and Atoms
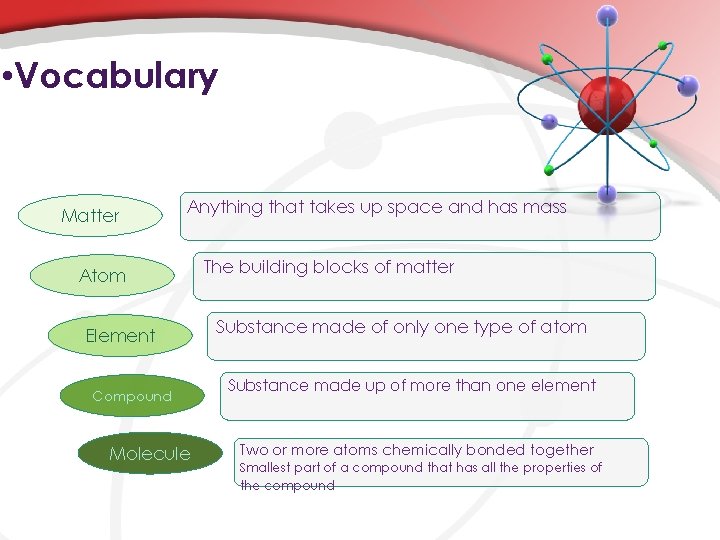
• Vocabulary Matter Anything that takes up space and has mass Atom Element Compound Molecule The building blocks of matter Substance made of only one type of atom Substance made up of more than one element Two or more atoms chemically bonded together Smallest part of a compound that has all the properties of the compound
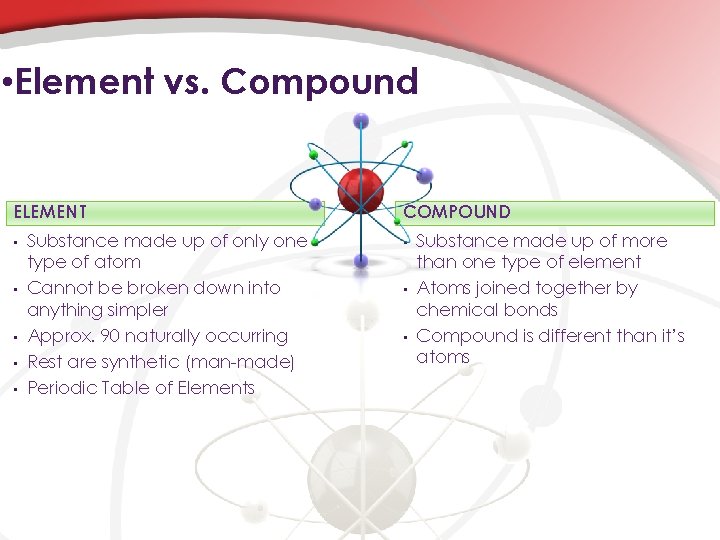
• Element vs. Compound ELEMENT • • • Substance made up of only one type of atom Cannot be broken down into anything simpler Approx. 90 naturally occurring Rest are synthetic (man-made) Periodic Table of Elements COMPOUND • • • Substance made up of more than one type of element Atoms joined together by chemical bonds Compound is different than it’s atoms

Chemical Formulas • A combination of chemical symbols and numbers to represent a substance • Subscript – shows the number of atoms of each element H 2 0 • CO 2 C 6 H 12 O 6 O 3 If only one atom of an element is present, no subscript is used Na. Cl CO
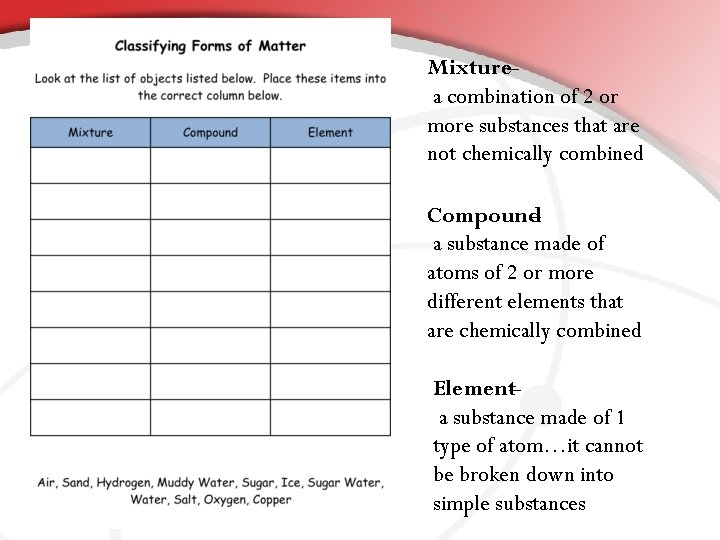
Mixture– a combination of 2 or more substances that are not chemically combined Compound – a substance made of atoms of 2 or more different elements that are chemically combined Element– a substance made of 1 type of atom…it cannot be broken down into simple substances

Properties of Matter Physical Properties • • Properties that are often very easy to observe or measure Help to determine use(s) Chemical Properties • • • Texture Color Shape Melting Point Boiling Point Hardness Strength Density State of Matter (solid/liquid/gas) Magnetism Conduct electricity Usually not as easy to observe Describe how a substance reacts Describes how a substance changes into a new substance Reactivity – the ability of a substance to combine chemically with another substance Flammability – the ability to burn

Changes of Matter Physical Changes • A change of matter from one form to another without a change in chemical properties • Do not change a substances identity Chemical Changes • A change that occurs when a substance changes composition by forming one or more new substances • Form new substances that have different properties • Cannot be reversed by physical changes
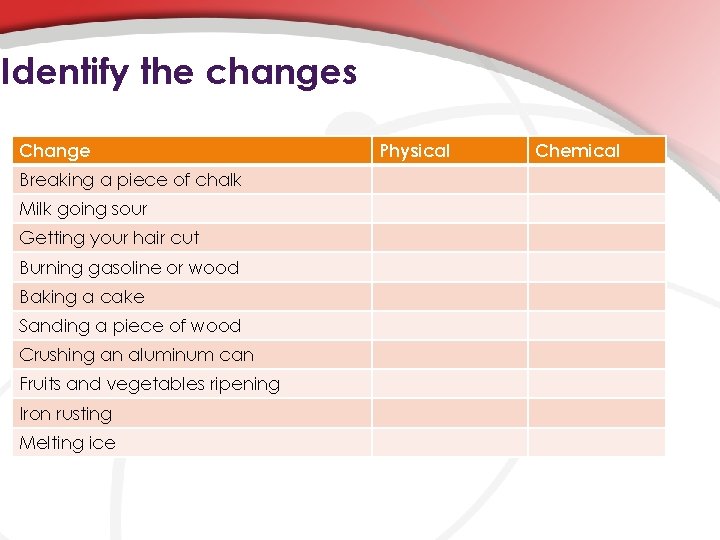
Identify the changes Change Breaking a piece of chalk Milk going sour Getting your hair cut Burning gasoline or wood Baking a cake Sanding a piece of wood Crushing an aluminum can Fruits and vegetables ripening Iron rusting Melting ice Physical Chemical
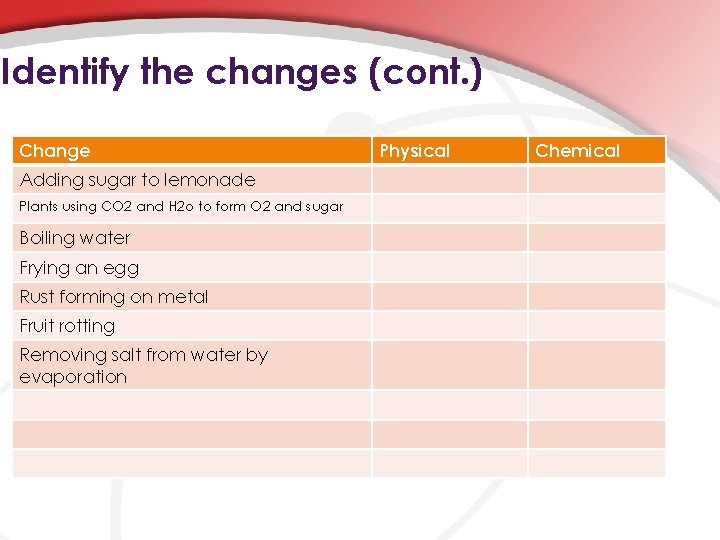
Identify the changes (cont. ) Change Adding sugar to lemonade Plants using CO 2 and H 2 o to form O 2 and sugar Boiling water Frying an egg Rust forming on metal Fruit rotting Removing salt from water by evaporation Physical Chemical

MATTER AND ENERGY

Kinetic Theory of Matter • All matter is made of atoms and molecules that act like tiny particles. • These tiny particles are always in motion. • • • The higher the temperature of the substance, the faster the particles move. The lower the temperature, the slower the particles move. At the same temperature: • • More massive (heavier) particles move slower Less massive (lighter) particles move faster

STATES OF MATTER

States of Matter Solids • • • Have a definite shape and volume Rigid Particles have almost no freedom to change position Particles held closely together Particles vibrate Liquids • • Change shape, not volume Particles can slide past one another Particles move more rapidly than in a solid Flow freely Gases • • Free to spread in all directions Expand to fill in available space Particles move rapidly Change both shape and volume

Plasma • Most common state of matter • 99% of known matter in universe • Sun and stars are made up of this • A state of matter that starts as a gas and then becomes ionized

ENERGY

Energy • The ability to change or move matter. • The ability to do work. • Kinetic Energy – the energy of motion • Thermal Energy – the total kinetic energy of the particles that make up an object. Also depends upon the # of particles in a substance. KINETIC ENERGY = • THERMAL ENERGY Temperature – the measure of average kinetic energy KINETIC ENERGY = TEMPERATURE

Energy • • The temperature of a substance is NOT determined by how much of the substance you have Example: Decide higher, lower, or equal in the following example Teapot Mug of tea Amount of Tea Temp. (Avg. kinetic energy) Total Kinetic Energy

ENERGY AND CHANGES OF STATE

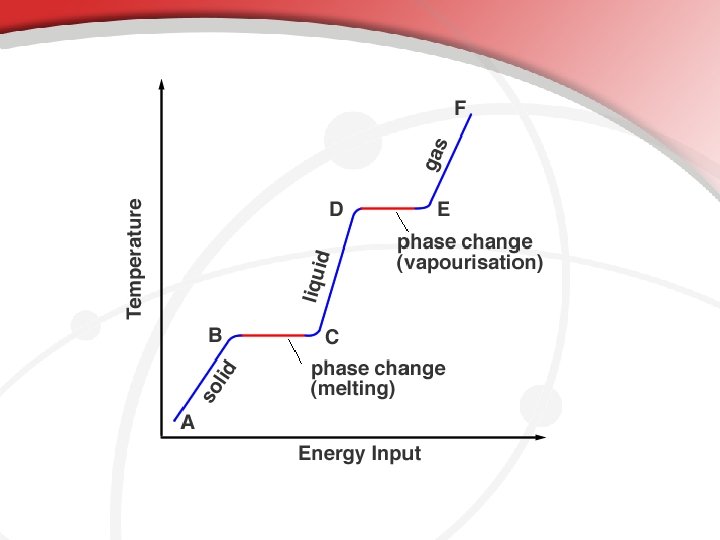
Energy & Changes of State • • Add energy = particles move faster Remove energy = particles move slower Endothermic Reactions – require energy from outside Melting – change of a substance from a solid to a liquid • Melting Point Evaporation – change of a substance from a liquid to a gas • Boiling Point Sublimation – change of a substance from a solid directly to a gas Exothermic Reactions – energy is released into the environment Condensation – change of a substance from a gas to a liquid • Condensation Point (Dew Point) Freezing – change of a substance from a liquid to a solid • Freezing Point

CONSERVATION OF MASS AND ENERGY

Mass cannot be created or destroyed In a chemical or physical change: • Matter changes • The total mass stays the same Law of Conservation of Mass • Mass cannot be created or destroyed • Mass of the reactants = mass of the products Law of Conservation of Energy • Energy may be converted to another form • Total amount of energy present before and after the change is the same • Energy cannot be created or destroyed
ATOMIC STRUCTURE

Atomic Theory Atom – • • Tiny units that determine the properties of all matter • Literally means “unable to divide” • The building blocks of matter Democritus (Greek philosopher) - 4 th century BC • • Suggested that the universe was made of invisible units that he named atoms and that the movement of the atoms caused the changes in matter that are observed. John Dalton (English schoolteacher) – 1808 • Proposed Atomic Theory • Atoms could not be divided • All atoms of a given element are exactly alike • Atoms can join together to make compounds
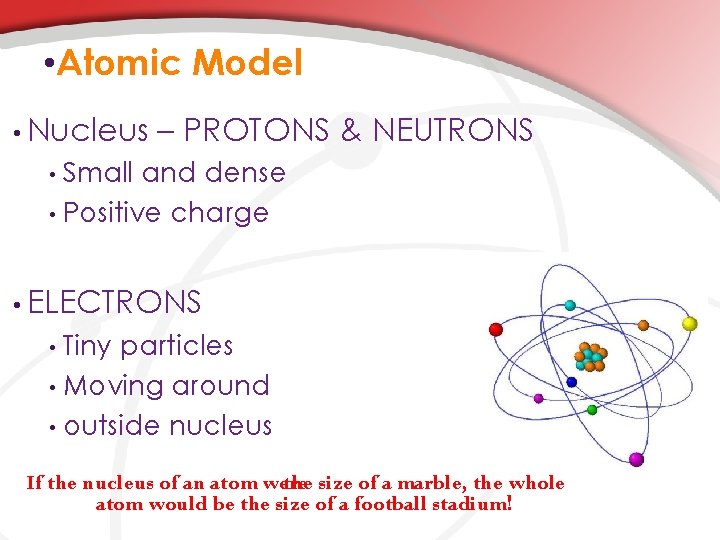
• Atomic Model • Nucleus – PROTONS & NEUTRONS Small and dense • Positive charge • • ELECTRONS Tiny particles • Moving around • outside nucleus • If the nucleus of an atom were the size of a marble, the whole atom would be the size of a football stadium!

• Subatomic Particles Particle Proton Neutron Electron Charge Location Mass (amu)

• Electron Extras • Close to Nucleus = Low Energy • Far from Nucleus = High Energy • *Behive

• How Many Protons? • Number of Protons = Atomic Number
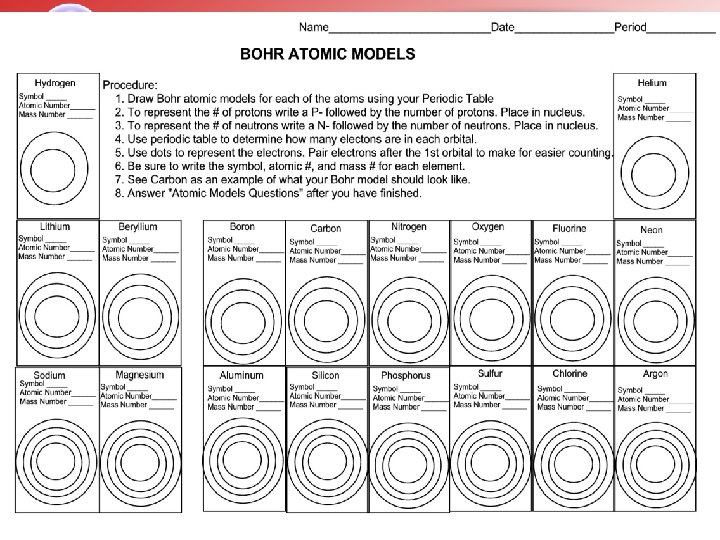

Atomic Model Information • Electron Shells (Orbits) can hold the following • 1 st Orbit – 2 electrons • 2 nd Orbit – 8 electrons • 3 rd Orbit – 18 electrons • 4 th Orbit – 32 electrons Valence Electrons – 2 n 2 an electron that is found in the outermost shell (orbit) of an atom and that determines the atoms chemical properties and its ability to form bonds.

• How Many Electrons? • Neutral Atom: Electrons = Protons • So in a Neutral Atom: Atomic Number = Protons = Electrons • BUT!!! Electrons can be lost or gained. Creates an ION. Ions have a +(positive) or –(negative) charge.

• How Many Neutrons? • Neutron numbers can differ in the same elements. ISOTOPE • Math formula • Atomic Mass = protons + neutrons • Periodic Table

- Slides: 33