Materials Science Chapter 5 Phase Equilibria Phase Diagram

Materials Science Chapter 5 Phase Equilibria

Phase Diagram • A collection of curves showing solubility limits. • In one side of the curve a single phase is present as unsaturated solution (liquid or solid) • Beyond the curve the solubility limit is exceeded, i. e. 2 nd phase exists and a mixture of two phases are present. • Useful to know the phases in equilibrium, alloy composition at a desired temperature, Chemical composition of each phase and calculation of the quantity of phase present.

Solutions and Mixtures • Different components combine to form solutions or mixtures. • A solution is a phase with more than one component. • A Mixture is a material with more than one component. • It is possible to have a mixture with more than one solution, e. g. lead-tin solder
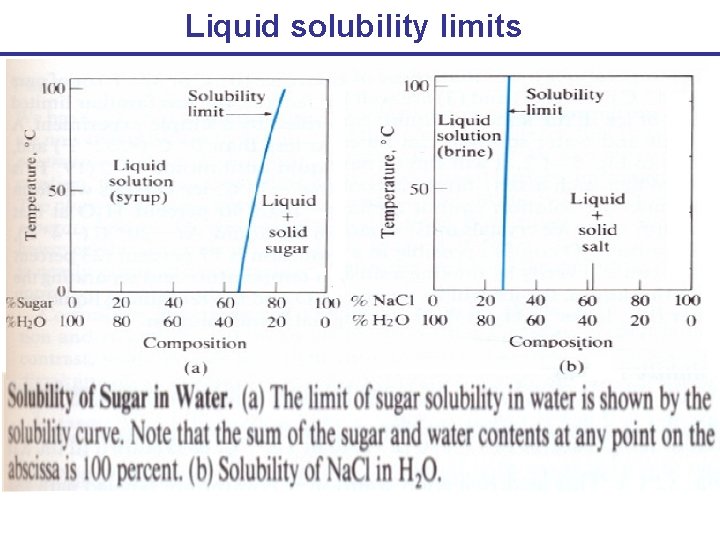
Liquid solubility limits
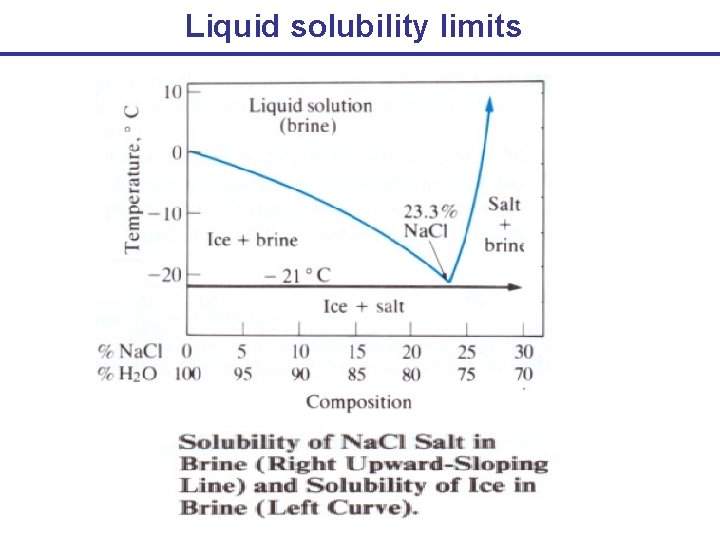
Liquid solubility limits
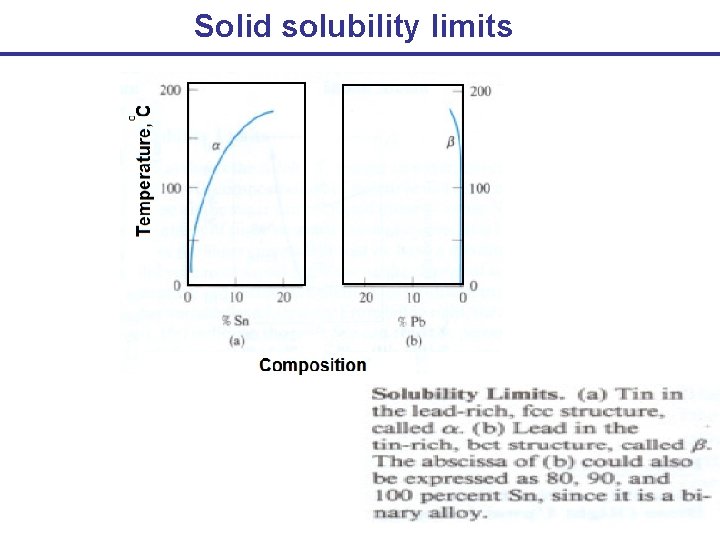
Solid solubility limits

Definitions • Phase: part of material that is distinct from others in structure or composition. • Phase boundary: is a region between two phases locates the discontinuinity in structure to from separate phases. • Solution: is a phase with more than one component. • Mixture: a material with more than one phase. • Component: a constituent of a phase with a definite chemical composition.
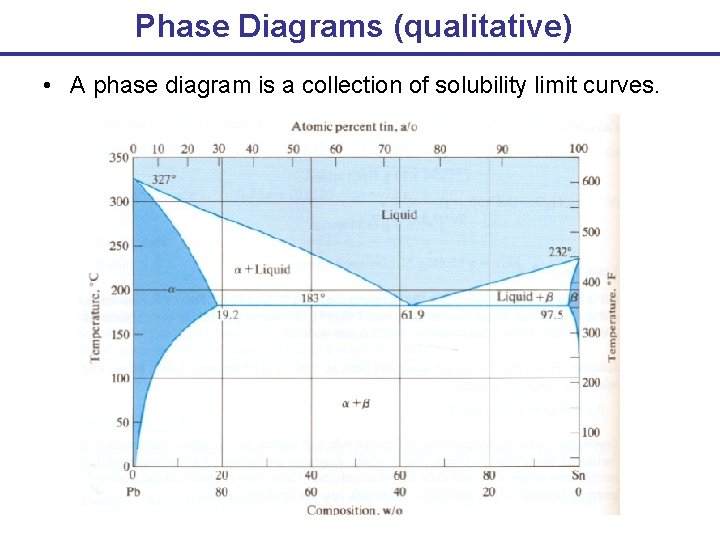
Phase Diagrams (qualitative) • A phase diagram is a collection of solubility limit curves.
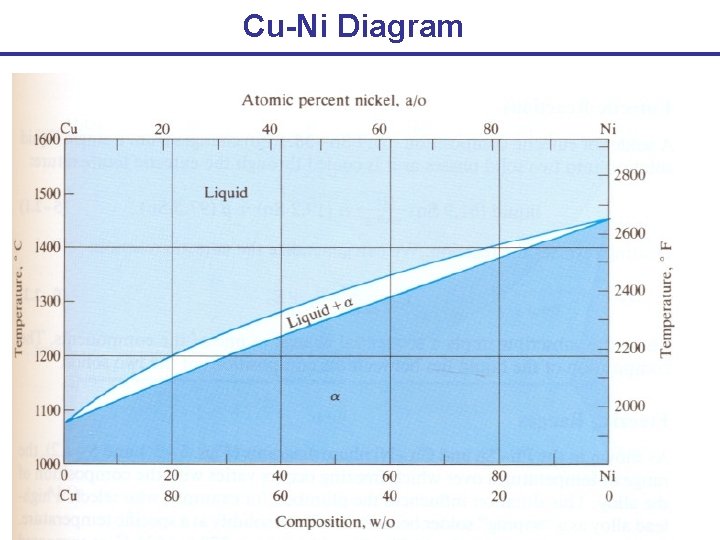
Cu-Ni Diagram

Eutectic Temperature and Composition • Pure lead melts at 327 o. C; pure tin melts at 232 o. C. • Alloys of lead and tin melt at lower temperatures. • Example: 60 -40 solder which is widely used for electrical connections. An alloy of this composition melts most readily and thus facilitates production with the a minimum possible damage to adjacent circuit components. • The above alloy is an eutectic alloy. • The eutectic temperature is 183 o. C; the eutectic composition is 38. 1 pb and 61. 9 Sn. • The eutectic composition and the eutectic temperature exist at the intersection of the two solubility curves that limit the composition of the liquid.

Eutectic Reactions

Freezing Ranges • The range of temperatures over which freezing occurs. It changes with the composition. • Liquidus: the locus of temperatures above which all compositions are liquids. • Solidus: the locus of temperatures below which all compositions are solids. • Liquidus and solidus meet at the eutectic temperature and composition.
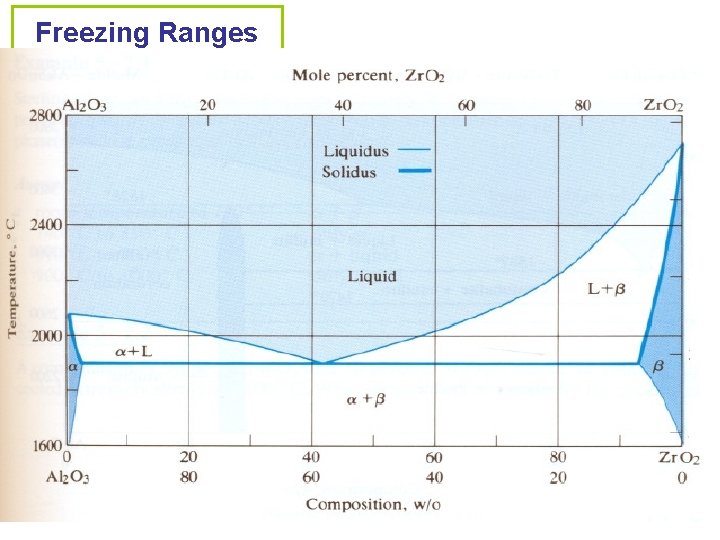
Freezing Ranges
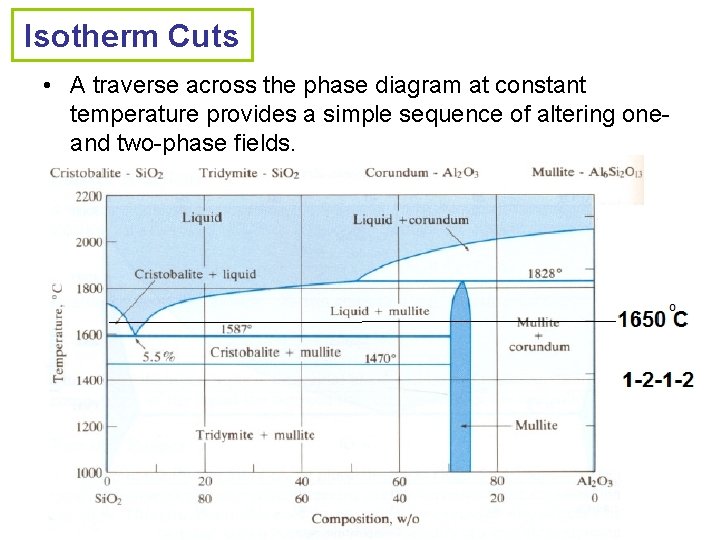
Isotherm Cuts • A traverse across the phase diagram at constant temperature provides a simple sequence of altering oneand two-phase fields.

Phase Names and Lables • Brass: is the phase name of an fcc solid solution of zinc in copper. • Bronze: is an fcc alloy of tin in copper. • Sterling Silver: 92. 5 Ag and 7. 5 Cu. • More often phases are labled with greek letters such as , , , etc. .
- Slides: 15