Materials and Membranes for Megawatt Scale Energy Storage

Materials and Membranes for Megawatt Scale Energy Storage Jagjit Nanda nandaj@ornl. gov Ethan Self, Frank Delnick, G. Yang, M. Lehman, Tomonori Saito, Landon Tyler, T. Zawodzinski Office of Electricity, Peer Review Sept 23 -26 th Albuquerque, NM ORNL is managed by UT-Battelle, LLC for the US Department of Energy
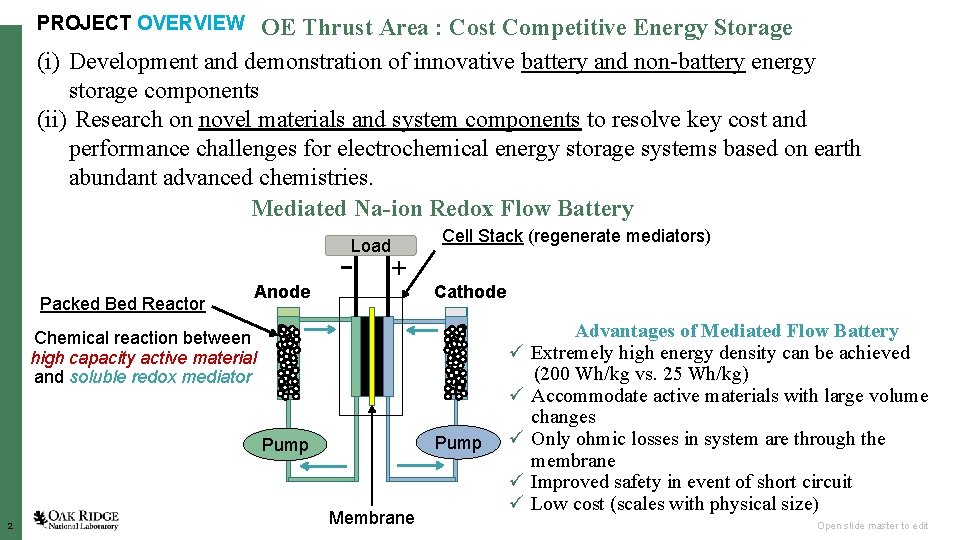
PROJECT OVERVIEW OE Thrust Area : Cost Competitive Energy Storage (i) Development and demonstration of innovative battery and non-battery energy storage components (ii) Research on novel materials and system components to resolve key cost and performance challenges for electrochemical energy storage systems based on earth abundant advanced chemistries. Mediated Na-ion Redox Flow Battery Load Packed Bed Reactor Anode - Cell Stack (regenerate mediators) + Cathode Chemical reaction between high capacity active material and soluble redox mediator Pump 2 Membrane Advantages of Mediated Flow Battery ü Extremely high energy density can be achieved (200 Wh/kg vs. 25 Wh/kg) ü Accommodate active materials with large volume changes ü Only ohmic losses in system are through the membrane ü Improved safety in event of short circuit ü Low cost (scales with physical size) Open slide master to edit

PROJECT TEAM • Redox Flow and Membrane Development - ORNL Ethan Self, Frank Delnick, Guang Yang, Tomonori Saito, Michelle Lehman, Landon Tyler Kun Lou, Thomas Zawodzinski • Na-ion Electrode Materials – Prof. David Mitlin, University of Texas at Austin • Vijay Murugesan and Wei Wang, PNNL – Membrane Characterization • R. Mukundan, LANL – Ceramic composite membranes • Vish Viswanathan and P. Balducci, PNNL - Techno-economic Modeling • Eric Spoerke and Cy Fujimoto, Sandia – Membranes for RFB’s 3 Open slide master to edit
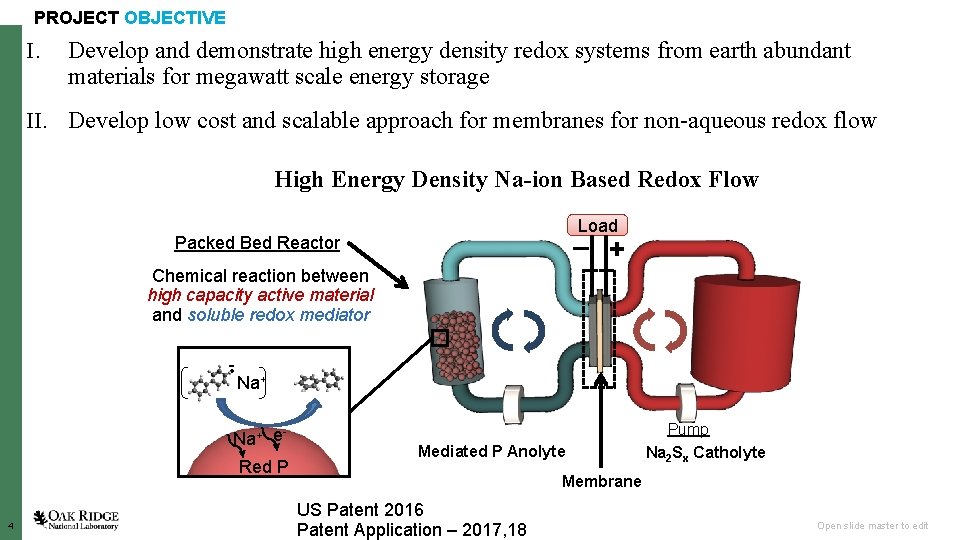
PROJECT OBJECTIVE I. Develop and demonstrate high energy density redox systems from earth abundant materials for megawatt scale energy storage II. Develop low cost and scalable approach for membranes for non-aqueous redox flow High Energy Density Na-ion Based Redox Flow Load – + Packed Bed Reactor Chemical reaction between high capacity active material and soluble redox mediator • - Na+ e Red P 4 Mediated P Anolyte Pump Na 2 Sx Catholyte Membrane US Patent 2016 Patent Application – 2017, 18 Open slide master to edit

PROJECT OBJECTIVE Low Cost Membrane Development for Na-ion NRFB Developed and tested low cost membranes for non-aqueous RFBs – PEO-CMC plasticized with tetraglyme – Crosslinked PEO – PEO Reinforced with Glass Fiber Mat PEO-Glass Fiber Composite Stretchable Ionomer Crosslinked PEO Target Membrane Resistivity < 20 ohm/cm 2 Mechanical Strength > 100 MPa Minimal Chemical Crossover 5 Open slide master to edit

PROJECT METRICS AND MILESTONES • Test and benchmark phosphorus-Na-polysulfide NRFB redox flow cell • Synthesis, fabrication and optimization of sodium-ion based membranes based on cross- link PEO and polystyrene (PS) block copolymers suitable for mediated and other organic flow batteries. Conductivity > 10 -4 S/cm; ion-transference number > 0. 7; Youngs modulus > 10 MPa • Evaluate mechanical durability of membranes in redox flow cells. Evaluate chemical/electrochemical stability of membranes in situ using NMR, Raman, and complex impedance. Target membrane resistivity < 20 Ohm/cm 2. • Test compatibility and chemical cross-over of sodium-based polymer membrane separators for radical anion mediated flow batteries Workshop organized to discuss targets and metrics on Non-Aqueous Redox Flow Batteries, Santa Fe, NM January 2019 – Jointly with OE and 4 National Laboratories – LANL-ORNLPNNL-Sandia 6 Open slide master to edit
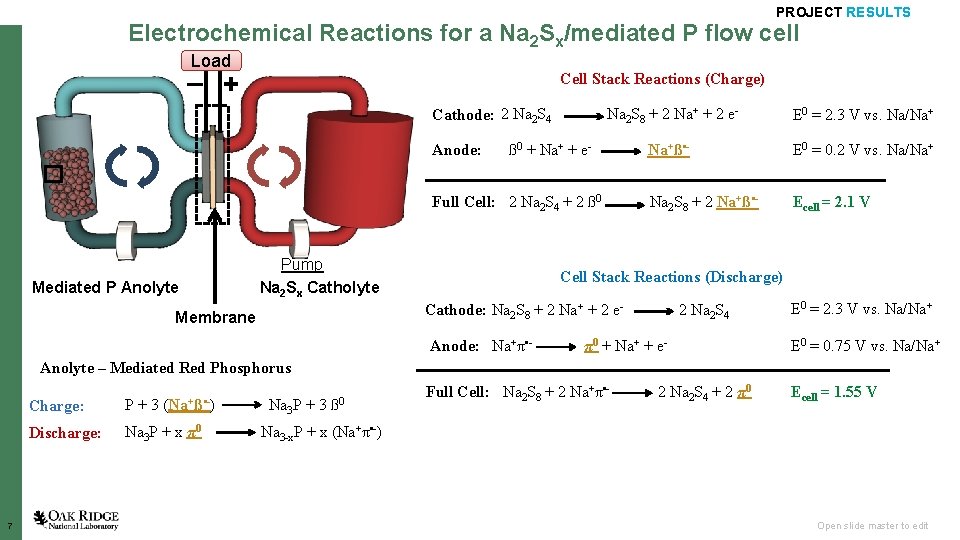
PROJECT RESULTS Electrochemical Reactions for a Na 2 Sx/mediated P flow cell Load – + Cell Stack Reactions (Charge) Cathode: 2 Na 2 S 4 Na 2 S 8 + 2 Na+ + 2 e- E 0 = 2. 3 V vs. Na/Na+ Anode: E 0 = 0. 2 V vs. Na/Na+ ß 0 + Na+ + e- Na+ß • - Full Cell: 2 Na 2 S 4 + 2 ß 0 Na 2 S 8 + 2 Na+ß • - Mediated P Anolyte Pump Na 2 Sx Catholyte Membrane Ecell = 2. 1 V Cell Stack Reactions (Discharge) Cathode: Na 2 S 8 + 2 Na+ + 2 e- 2 Na 2 S 4 E 0 = 2. 3 V vs. Na/Na+ Anode: Na+π • - π0 + Na+ + e- E 0 = 0. 75 V vs. Na/Na+ Full Cell: Na 2 S 8 + 2 Na+π • - 2 Na 2 S 4 + 2 π0 Ecell = 1. 55 V Anolyte – Mediated Red Phosphorus 7 Charge: P + 3 (Na+ß • -) Na 3 P + 3 ß 0 Discharge: Na 3 P + x π0 Na 3 -x. P + x (Na+π • -) Open slide master to edit
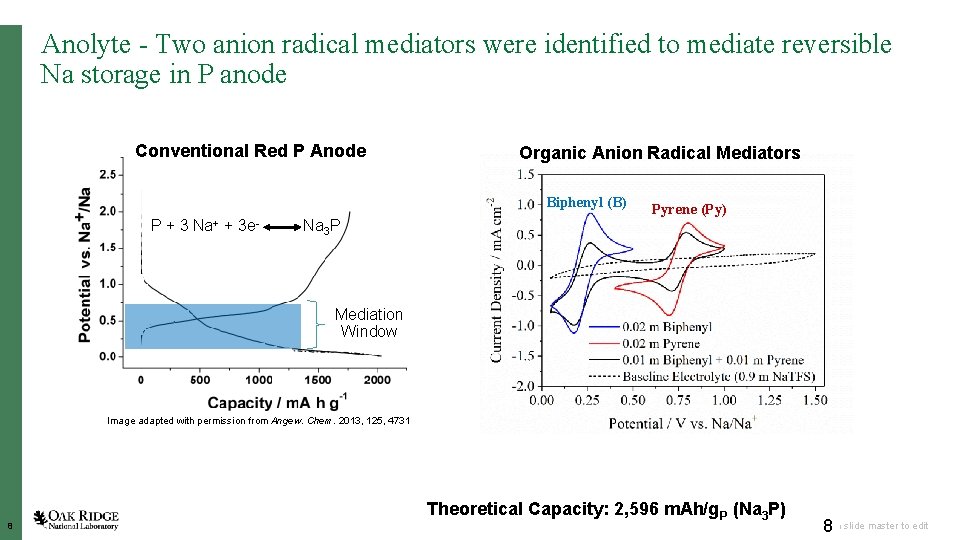
Anolyte - Two anion radical mediators were identified to mediate reversible Na storage in P anode Conventional Red P Anode Organic Anion Radical Mediators Biphenyl (B) P+3 Na+ + 3 e- Pyrene (Py) Na 3 P Mediation Window Image adapted with permission from Angew. Chem. 2013, 125, 4731 8 Theoretical Capacity: 2, 596 m. Ah/g. P (Na 3 P) 8 Open slide master to edit
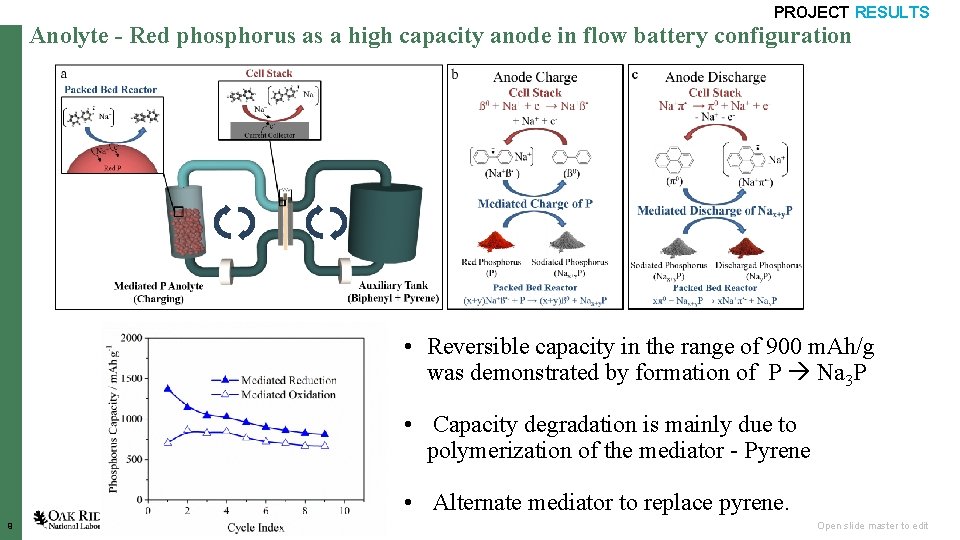
PROJECT RESULTS Anolyte - Red phosphorus as a high capacity anode in flow battery configuration • Reversible capacity in the range of 900 m. Ah/g was demonstrated by formation of P Na 3 P • Capacity degradation is mainly due to polymerization of the mediator - Pyrene • Alternate mediator to replace pyrene. 9 Open slide master to edit
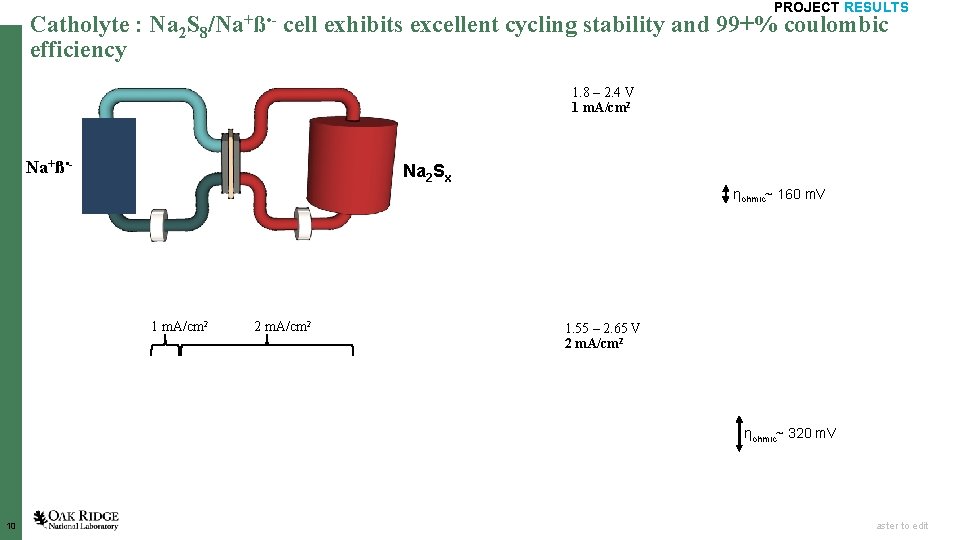
Catholyte : efficiency Na 2 S 8/Na+ß • - PROJECT RESULTS cell exhibits excellent cycling stability and 99+% coulombic 1. 8 – 2. 4 V 1 m. A/cm 2 Na+ß • - Na 2 Sx ηohmic~ 160 m. V 1 m. A/cm 2 2 m. A/cm 2 1. 55 – 2. 65 V 2 m. A/cm 2 ηohmic~ 320 m. V 10 Open slide master to edit
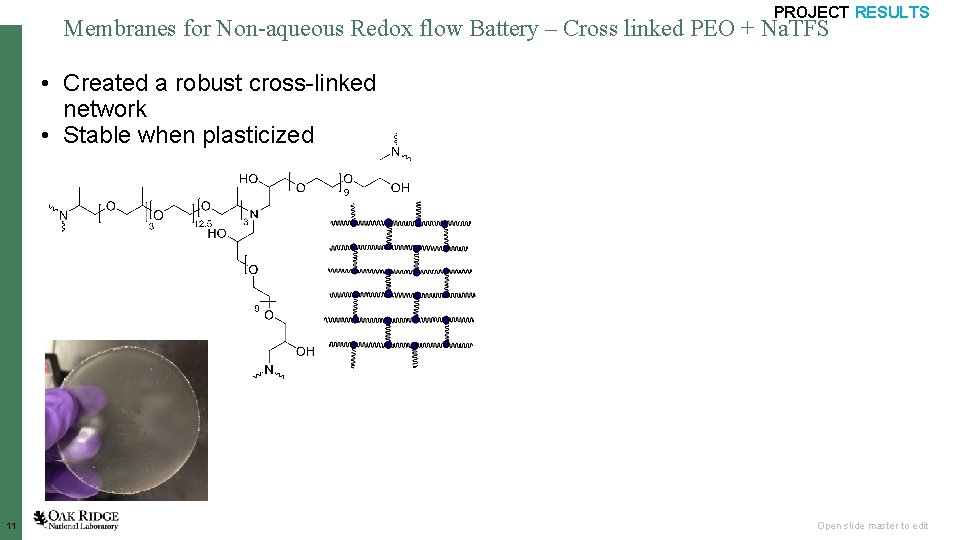
PROJECT RESULTS Membranes for Non-aqueous Redox flow Battery – Cross linked PEO + Na. TFS • Created a robust cross-linked network • Stable when plasticized Dry 11 Plasticized Open slide master to edit
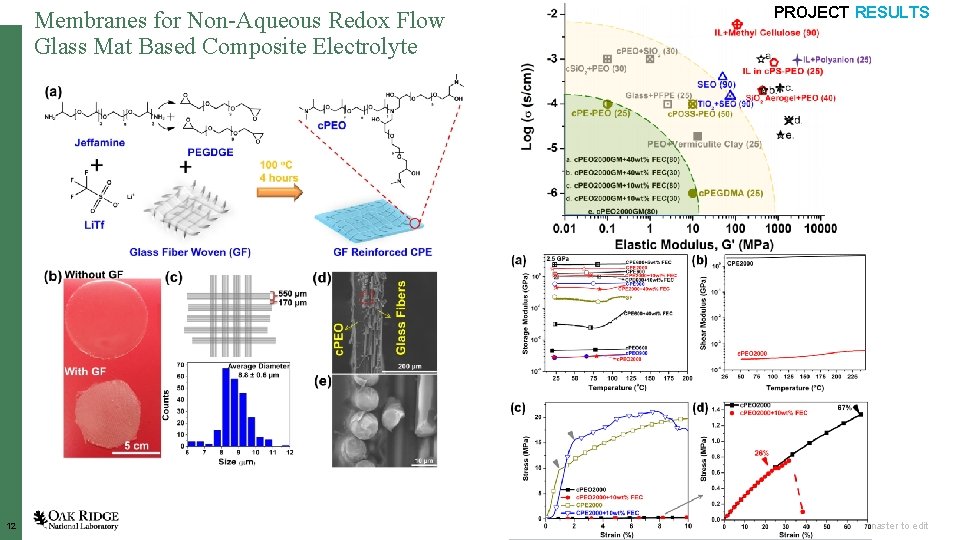
Membranes for Non-Aqueous Redox Flow Glass Mat Based Composite Electrolyte 12 PROJECT RESULTS Open slide master to edit

Insitu Crossover measurements using UV-Vis Spectroscopy PROJECT RESULTS Calibration using Standard Materials Redox Species: tetraethylammonium vanadium(III) acetylacetonate (TEA-V(acac)3) Solvent: Acetonitrile Membrane: TEA-substituted membrane (3 M Company) 13 Concentration of TEA-V(acac)3) increases as time elapses Open slide master to edit
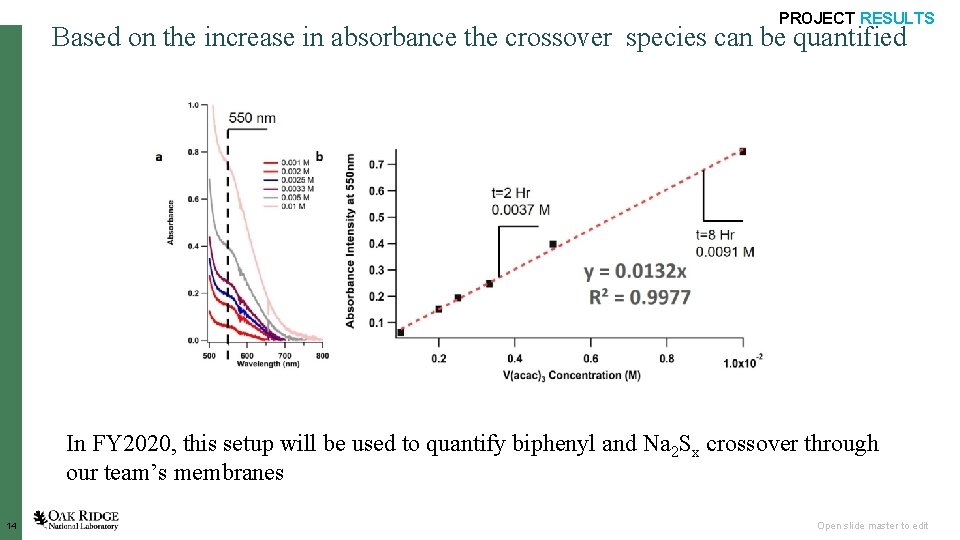
PROJECT RESULTS Based on the increase in absorbance the crossover species can be quantified In FY 2020, this setup will be used to quantify biphenyl and Na 2 Sx crossover through our team’s membranes 14 Open slide master to edit

LOOKING FORWARD • Find alternate redox mediator to replace Pyrene • Evaluate performance at higher Na 2 S 8 concentrations (>1 M) and higher current densities. • Quantify overpotential losses (activation, ohmic, concentration) using AC impedance • P-Na 2 S full cell testing • Membrane cross-over test 15 Open slide master to edit

PROJECT RESULTS Patents and Publications 1. M. L. Lehmann, G. Yang, D. Gilmer, K. S. Han, E. C. Self, R. E. Ruther, S. Ge, B. Li, V. Murugesan, A. P. Sokolov, F. M. Delnick, J. Nanda, T. Saito, Crosslinking of Poly(Ethylene Oxide) Membranes Enables Mechanical Robustness and Improved Sodium-Ion Conductivity with Plasticization Energy Storage Materials 2019, 21, 85 -96. 2. R. E. Ruther, J. Nanda, S. Greenbaum, Ion transport and association study of glyme-based electrolytes with lithium and sodium salts, Electrochimica Acta 2019, 304, 239 3. E. C. Self, F. M. Delnick, R. E. Ruther, J. Nanda, High Capacity Organic Radical Mediated Phosphorus Anode for Sodium Based Redox Flow Batteries ACS Energy Lett. 2019 (Accepted with revision ) 4. F. M. Delnick, J. Nanda, E. C. Self, High Capacity Organic Radical Mediated Phosphorous Anode for Redox Flow Batteries, United States Patent Application No. 62/701, 859, Filed on July 23, 2019. 5. F. M. Delnick, D. Ingersoll, C. Liang. Polyarene mediators for mediated redox flow battery, US Patent No. 9, 859, 583 B 2, 2018. Invited Presentation in National and International Conference – 4 16 Open slide master to edit

PROJECT CONTACTS This work is supported by Dr. Imre Gyuk, Manager, Energy Storage Program, Office of Electricity, Department of Energy Michael Starke, Thomas King – ORNL Vince Sprenkle – PNNL Babu Chalamela Name of the Presenter – Jagjit Nanda E-mail – nandaj@ornl. gov Phone-865 -241 -8361 17 17 Open slide master to edit

Supporting Information 18 Open slide master to edit

A custom redox flow cell was designed to demonstrate a mediated red phosphorus anode Cell Components: Working/Auxiliary Electrodes: Porous Ni foam Reference Electrode: Na in 1 m Na. TFS (TEGDME) Membrane: Na+ß” Al 2 O 3 Ceramic (Ionotec, 45 x 1. 5 mm 3) Purified Ar Serpentine Flow Channels Syringe Pump 19 ORNL IP August 2017 Membrane Flow Cell (connected to potentiostat) Reference Electrode Port Syringe Pump 19 Open slide master to edit
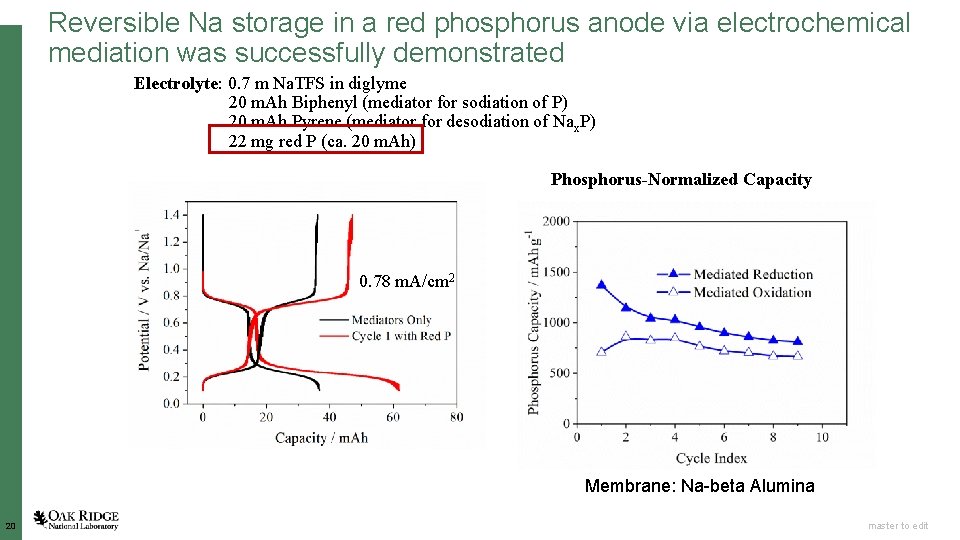
Reversible Na storage in a red phosphorus anode via electrochemical mediation was successfully demonstrated Electrolyte: 0. 7 m Na. TFS in diglyme 20 m. Ah Biphenyl (mediator for sodiation of P) 20 m. Ah Pyrene (mediator for desodiation of Nax. P) 22 mg red P (ca. 20 m. Ah) Phosphorus-Normalized Capacity 0. 78 m. A/cm 2 Membrane: Na-beta Alumina 20 Open slide master to edit
- Slides: 20