Massive Transfusion Protocols The use of Thromboelastography and

Massive Transfusion Protocols: The use of Thromboelastography and Tranexamic Acid Ariana Beck, M. D. PGY 2

Objectives n Overview and background information • What is massive transfusion and why do we need a protocol ? n n n Coagulopathy of trauma- recent ideas on platelet dysfunction TEG guided resuscitation The use of tranexamic acid (TXA) in massive transfusion: • CRASH-2 trial • MATTERs paper

Overview and general information n According to the CDC, unintentional injury is the leading cause of death in persons ages 1 -44 years in the USA Most recent data from 2010 [Nunez TC et al, 2010; the CDC, CDC 2010]

Overview and general information n These unintentional injuries are primarily composed of motor vehicle collision, and firearm injuries (by homicide and suicide) or more generally, trauma • 4 of the top 5 causes of unintentional injuries are trauma related • Trauma is the leading cause of death in ages 5 -24 Most recent data from 2010 [Nunez TC et al, 2010; the CDC, CDC 2010]

Overview and general information n Hemorrhagic shock and exsanguination are responsible for up to 50% of deaths in the first 24 hours after injury and greater than 80% of deaths in the operating room • Death typically occurs quickly within 6 hours of injury n n Massive transfusion is defined as receiving 10 or more units of packed red blood cells over a period of 24 hours 3 -5% of civilian trauma patients will require massive transfusion • These patients consume up to 75% of all blood products at urban trauma centers n The logistics of preparing the products and delivering them in a expedient manner necessitates a defined protocol with coordination of the personnel from multiple departments [Nunez TC et al, 2010] 2010

Overview and general information: Why do we need MTPs and what are they? ER n n Trauma ICU Emergency medicine, trauma surgery, critical care, transfusion, nursing, hematology and anesthesia are involved in the care of trauma patients and should be involved in creating and carrying out these protocols Massive transfusion requires the immediate availability of certain products n n OR Prethawed AB fresh frozen plasma, O-/O+ red blood cells, and platelets Without a defined, set protocol, delays are inevitable and adversely affect patient outcomes Additionally, transfusing products in the correct volume and ratio has been shown to decrease overall usage and improve survival MTPs are essentially a protocol for the rapid release of emergency blood products [Nunez TC et al, 2010] 2010

Overview and general information: Why do we need MTPs and what are they? n n n • What happens? Attending trauma surgeon or anesthesiologist directly calls the blood bank to activate the MTP and gives basic demographic information including the approximate age and gender of the patient as well and the location that the products should be delivered to Typically a clinical decision is made (not based on lab values) n A type and screen is sent so the conversion to type specific products may be made n Basically, the blood bank sends appropriately packaged products ready for transfusion in appropriate amounts in a timely manner until the MTP is stopped by the clinician The implementation of MTPs has been shown to have significant positive affect on trauma outcomes and allows products to be released immediately in the appropriate volume and ratio [Nunez TC et al, 2010] Nunez TC et al, 2010

Trauma associated coagulopathy n n Acute coagulopathy of trauma (ACOT) is characterized by non-surgical bleeding that occurs with and without the appropriate concentrations of coagulation factors. One in four severely injured patients presents with ACOT Associated with higher transfusion rates, longer ICU stays, increased mortality Independent of iatrogenic/ dilutional causes [Nunez TC et al, 2010; Napolitano et al, 2013; Maegele et al, 2013]

Trauma associated coagulopathy n Protein C pathway occurs when tissue injury is associated with hypoperfusion • n ACOT may also be related to the disruption of the vascular endothelium and its glycocalyx • • • n The glycocalyx is a negatively charged antiadhesive/ anticoagulant surface layer that protects endothelium Endothelial damage leads to glycocalyx degradation which leads to the release of heparin like substances into circulation => autoheparinization Endothelial injury also leads to increased TPA The combination of increased TPA and protein C activation can lead to fibrinolysis • n Downstream anticoagulant effect Present in 7 -20% of trauma patients and associated with increased mortality The consumption of coagulation factors also drives this process [Maegele et al, 2013]

Trauma associated coagulopathy n n n Does platelet dysfunction play a role? Wolhauer et al explored this aspect of ACOT in the Journal of the American College of Surgery Conventional coagulation assays like PT and PTT are unable to determine platelet function A CBC can provide information on platelet quantity, but not on functional status The authors “hypothesized that early platelet dysfunction is prevalent after severe injury and can be evaluated in a point of care setting using thrombelastography (TEG) based platelet functional analysis to measure the ability of platelets to assemble into a stable thrombus with different platelet activators” [Wohlauer et al, 2012] 2012

Trauma associated coagulopathy n Enrolled 51 consecutive trauma patients who were determine to be at risk for post injury coagulopathy • Risk: Blunt and penetrating injuries with a pre-hospital systolic blood pressure less than 90 mm. Hg, penetrating gunshot wounds to the torso, stab wounds to the torso requiring endotracheal intubation, amputation proximal to the wrist or ankle, a Glasgow Coma Scale (GCS) less than 8 or respiratory compromised with presumed thoracic, abdominal or pelvic injury, inter-hospital transfers requiring blood transfusion to maintain vital signs or when the attending physician suspects the patient is likely to require urgent operative intervention n n 39 healthy volunteers were enrolled Whole blood was collected from the trauma patients in the field or within 30 minutes of arrival to the ED Whole blood was also collected from the healthy volunteers Platelet mapping was performed within 2 hour of sample collection [Wohlauer et al, 2012] 2012

Trauma associated coagulopathy n Platelet mapping was done by TEG • The TEG platelet functional assay was used which measured the reactivity of platelets to the activators arachidonic acid (AA) and ADP • In trauma patients overall, the mean ADP inhibition of platelet function was 86. 1% as compared to 4. 2% in healthy volunteers (p<0. 0001) • The mean AA inhibition of platelet function was 44. 9% compared to 0. 5 % in healthy volunteer (p<0. 0001) [Wohlauer et al, 2013] 2013
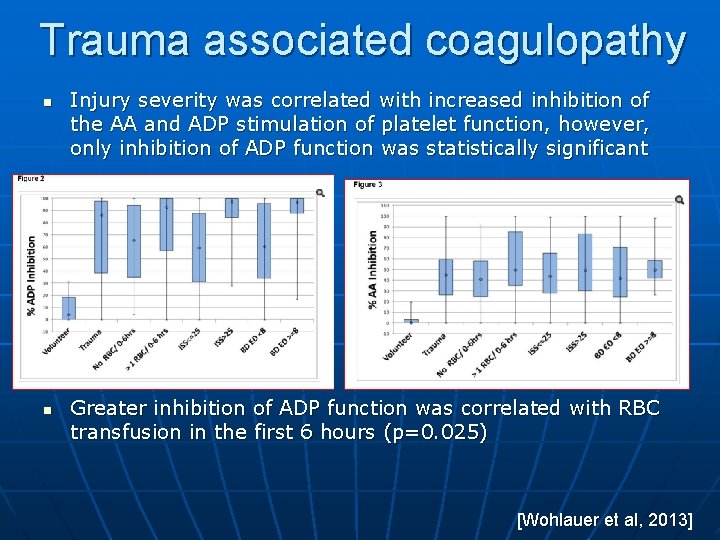
Trauma associated coagulopathy n n Injury severity was correlated with increased inhibition of the AA and ADP stimulation of platelet function, however, only inhibition of ADP function was statistically significant Greater inhibition of ADP function was correlated with RBC transfusion in the first 6 hours (p=0. 025) [Wohlauer et al, 2013] 2013

Trauma associated coagulopathy n Authors concluded that platelet dysfunction occurs after major trauma, before substantial fluid or blood administration • Platelets were dysfunctional, despite being adequate in number • Platelet hyperactivation leads to decreased platelet response to subsequent stimulation n “exhausted platelet syndrome” n n RBC transfusion, as a surrogate for shock, correlated with increasing severity of platelet dysfunction The authors also state that TEG provides an advantage when compared to traditional tests and supports a multidisciplinary approach to manage massively transfused patients • TEG platelet mapping correlates well with light transmittance aggregometry (LTA) which is the gold standard of platelet function assays and is labor intensive and time consuming [Wohlauer et al, 2013] 2013

TEG in Trauma

TEG in trauma n In a study looking at 1974 consecutive trauma patients, Holcomb et al showed that rapid-TEG correlated with conventional coagulation tests (PT, PTT, INR, platelet count, and fibrinogen level) • Activated clotting time (ACT) predicted red blood cell transfusion and the a-angle predicted massive RBC transfusion better than PT, PTT and INR • The a-angle was also superior to fibrinogen for predicting plasma transfusion • The maximal amplitude (MA) was superior to platelet count in predicting platelet transfusion • The LY 30 (rate of amplitude reduction 30 minutes after the MA is reached) documented fibrinolysis n This group concluded that r-TEG data is superior to conventional coagulation tests and better identifies patients with increased risk of need for transfusion and fibrinolysis [Holcomb et al, 2012]

TEG guided resuscitation vs MTPs n n Tapia and colleagues looked at MTP vs TEG guided resuscitation in the Journal of Trauma and Acute Care Surgery Prior to the institution of an MTP, Ben Taub General Hospital, a level 1 trauma center in Houston used TEG guided resuscitation n n This method was in place for nearly 10 years FFP and/or platelets were given in conjunction with RBCs based on TEG results obtained in the operating room or ICU • In October of 2009, an MTP was implemented with a 1: 1: 1 ratio of blood products • The authors hypothesized that TEG guided resuscitation was equivalent to MTP guided transfusion [Tapia et al, 2013] 2013

TEG in Trauma n n n Authors queried their database for patients who received 6 units of more RBCs and 10 units of more of RBC in the first 24 hours of admission from January 2008 to June 2010 (21 months before and 21 months after the switch to MTP) and performed a retrospective analysis Primary outcome was 30 day mortality Secondary outcomes included early (<24 hours) and delayed (>24 hours) deaths, total volume transfused and individual volumes of RBCs, FFP and platelets transfused in the first 24 hours, as well as ICU and ventilator days and crystalloid volume received [Holcomb et al, 2012] 2012

TEG guided resuscitation vs MTPs n n n n Patient demographics were similar between the groups There were no significant differences in ICU and ventilator days between the groups In patients with blunt trauma who received >6 units of RBCs, the ratio of RBCs to FFP decreased after MTP implementation There was decreased crystalloid use in all groups post MTP implementation In the group receiving >6 units of RBCs, there was no significant difference in 30 day mortality between the pre and post MTP cohorts There was also no significant difference in 30 day mortality for patients receiving 10 units of more with blunt trauma However, in patients with penetrating trauma who received >10 units of RBCs, 30 day mortality was increased post MTP implementation (33% vs 54%) • These patients also had increased early deaths n Volume of RBCs transfused did not differ between TEG and MTP cohorts • The only group in which FFP increased was blunt trauma patients who received >10 units of RBCs [Tapia et al, 2013] 2013

TEG guided resuscitation vs MTPs n Authors concluded that in their patient population, TEG guided resuscitation is superior to MTP in massively transfused patients with penetrating trauma receiving >10 units of RBC • TEG guided therapy seemed to provide the optimal ratio of products through laboratory analysis rather than prescribed transfusion • The use of “TEG in conjunction with MTP may help elucidate those patients who require more aggressive resuscitation because of their unique physiologic response to injury” n n Some patients are more susceptible to coagulopathy and others have a unique ability to withstand injury TEG can be used as a marker of the ability to mount a homeostatic response to injury • Authors propose that TEG should guide correction of coagulopathy at the point of care while MTP should be used as a system based infrastructure to provide the logistical support of blood availability and transport [Tapia et al, 2013] 2013

TEG use in conjunction with MTPs • This publication from Copenhagen University Hospital basically describes a combined approach to massive transfusion n Their MTP encompasses transfusion packages and TEG guided therapy • Prospective, observational cohort study • Patient’s admitted to the trauma center have blood sampled for TEG analysis immediately upon arrival along with samples for type and screen and conventional lab testing • TEG sampling is done by the blood bank and is played in real time on a computer screen in the trauma center • During resuscitation, blood samples are sent for TEG analysis every 30 minutes [Johansson et al, TRansfusion 2013] 2013

TEG use in conjunction with MTPs • For patients with hemostatic instability, blood products are transfused in a fixed ratio via transfusion packets with focused intervention via TEG data as it becomes available • Additionally, tranexamic acid , cryoprecipitate, and/ or fibrinogen concentrate is given in accordance with the result of the TEG analysis n Table 1 of the paper shows this goal directed therapy • A 24/7 on call consulting physician with expertise in hemostasis and transfusion medicine is activated by the blood bank staff [Johansson et al, 2013] 2013

TEG use in conjunction with MTPs n This study was essentially descriptive • “ The blood bank provides the necessary blood products immediately available, performs the TEG analysis, and when needed, assists in interpretation of TEG results and in applying the goal-directed hemostatic protocols” • They “infer that the relatively low number of bleeding deaths observed in this study reflect the use, at our hospital of early administration of plasma, PLTs, and hemostatic agents based on TEG-protocols. ” [Johansson et al, 2013] 2013

Tranexamic Acid Use in Trauma
The use of tranexamic acid in trauma: CRASH 2 trial lysine n n Tranexamic acid Synthetic derivative of the amino acid lysine Inhibits fibrinolysis by blocking the lysine binding site on plasminogen • No interaction of plasminogen with fibrin, no dissolution of fibrin clot n Has been shown to reduce the need for blood transfusion in elective surgeries • Has been available for 20 years • FDA approved for the treatment of menorrhagia and for the reduction or treatment of bleeding during tooth extraction in hemophilia patients [Crash-2 cont. 2010]

The use of tranexamic acid in trauma: CRASH 2 trial n Because the hemostatic responses in surgery are similar to that of trauma, tranexamic acid may reduce bleeding and mortality in trauma patients • Additionally, hyperfibrinolysis is a common feature of trauma associated coagulopathy n The purpose of the CRASH-2 trial was to assess the effect of early administration of tranexamic acid on death, vascular occlusive events, and blood transfusions in trauma patients with significant bleeding [Crash-2 cont. 2010]

n Methods: Tranexamic acid: CRASH 2 trial • Randomized control trial undertaken in 274 hospitals in 40 countries using placebo and tranexamic acid • First patient was enrolled in May of 2005 • Eligibility was defined as adult trauma patients with significant hemorrhage, or who were considered to be at significant risk for hemorrhage within 8 hours of injury n n Systolic blood pressure <90 mm. Hg, heat rate of >110 beats per minute or both Clinical, not lab based decisions • Patients were included and randomized if the treating physician was unsure about whether to use tranexamic acid n Excluded if the use of tranexamic acid was contraindicated or if there was a clear indication for use • Where reliable telephone service was available, patients were randomized by a telephone randomization service that used a minimization algorithm which balanced for sex, age, time since injury, type of injury and other factors [Crash-2 cont. 2010]

Tranexamic acid: CRASH 2 trial n n n Patients then either received a 1 g loading dose of tranexamic acid or 0. 9% saline (placebo) followed by infusion of an additional 1 gram of treatment (or matched placebo) over 8 hours The primary outcome was death within 4 weeks of injury Secondary outcomes included vascular occlusion, surgical intervention, receipt of blood transfusion and the units of blood transfused • Outcomes were recorded for up to 4 weeks if the patient was still hospitalized [Crash-2 cont. 2010]
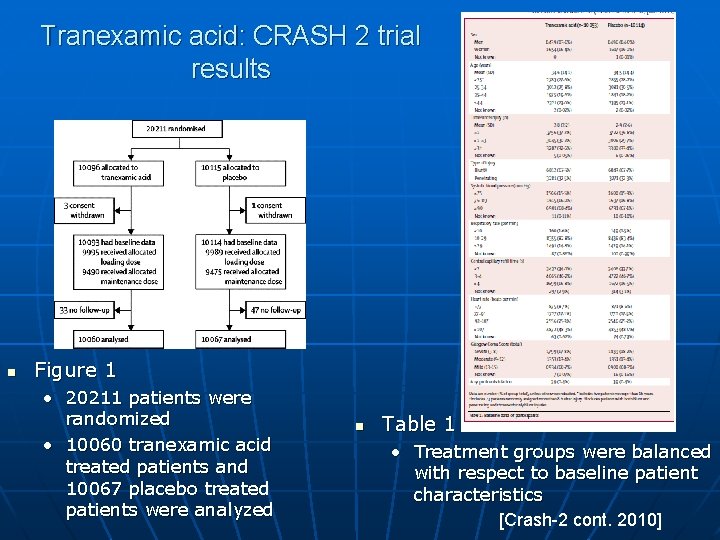
Tranexamic acid: CRASH 2 trial results n Figure 1 • 20211 patients were randomized • 10060 tranexamic acid treated patients and 10067 placebo treated patients were analyzed n Table 1 • Treatment groups were balanced with respect to baseline patient characteristics [Crash-2 cont. 2010]

Tranexamic acid: CRASH 2 trial results n Table 2 • All cause mortality was significantly reduced with tranexamic acid treatment • The risk of death due to bleeding was also significantly reduced • Death from multiorgan failure, head injury and other causes did not differ [Crash-2 cont. 2010]
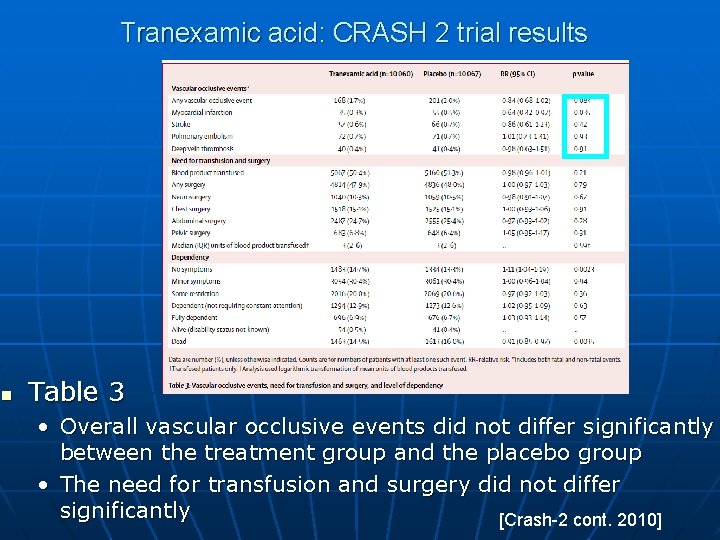
Tranexamic acid: CRASH 2 trial results n Table 3 • Overall vascular occlusive events did not differ significantly between the treatment group and the placebo group • The need for transfusion and surgery did not differ significantly [Crash-2 cont. 2010]

Tranexamic acid: CRASH 2 trial n n n The results show that the administration of tranexamic acid early in trauma is beneficial with a decrease in risk of death from bleeding and decreased overall mortality with no apparent increase in vascular occlusive events There was no significant reduction in the receipt and amount of blood transfused Further questions left unanswered • Does not explain tranexamic acid accomplishes this n n Assumed due to antifibrinolysis, however, fibrinolytic activity was not measured Also, there was no significant reduction in the receipt and amount of blood transfused • The dose of tranexamic acid used was based on studies of the drug in surgical patients n n n Studies in cardiac and surgical patients have shown no significant changes in outcomes based on dose Would a higher dose of tranexamic acid have a greater treatment effect in trauma patients? In patient’s with intracranial hemorrhage, could the use of tranexamic acid improve outcomes? [Crash-2 cont. 2010]

Tranexamic acid: CRASH 2 trial n In 2011, the results were analyzed again • Authors looked at the effect of treatment on death due to bleeding subdivided by 4 baseline characteristics n n Time from injury to treatment Severity of hemorrhage as assessed by SBP Glascow coma scale Type of injury [Crash-2 cont, 2011]

Tranexamic acid: CRASH 2 trial n Death due to bleeding was significantly reduced with treatment of TXA when patients were treated <1 hour after injury and when patients were treated from 1 to 3 hours after injury • Death due to bleeding was increased when patients were treated >3 hours after injury • There was no heterogeneity for the other 3 subgroups n These results suggest that tranexamic acid should be given to bleeding trauma patients as soon as possible • In patients admitted late after injury tranexamic acid is less effective and may be harmful [Crash-2 cont, 2011]

Putting it all together: MATTERs Study n An additional limitation of the CRASH-2 trial is the lack of a standard RBC transfusion protocol • Most of the study sites lacked MTPs and the availability of similar resuscitation strategies n Authors of the MATTERs study undertook a retrospective observational study comparing TXA administration with no TXA administration in patients receiving at least 1 unit of blood at a military surgical hospital [Morrison et al, 2012]

Putting it all together: MATTERs Study n The UK Defense Medical Service has used TXA as part of their MTP since 2010 (at the time of publication, the US Combat Casualty Care program deferred use) • Prior to 2010, TXA was administered at the discretion of treating physicians or in some instances, following the demonstration of fibrinolysis on TEG • Since 2010, TXA has been administered to patient’s requiring emergency blood products n n n A retrospective cohort study was performed using patients treated at a single surgical hospital in southern Afghanistan Patients were separated into 2 groups based on whether or not they received TXA Patients who received 10 units of more of RBCs within 24 hours were identified as the massive transfusion cohort and separated based on TXA use The primary endpoints were 24 and 48 hours and in hospital mortality (up to 30 days at any hospital in the aeromedical evacuation chain) Secondary endpoints included transfusion requirements and coagulation parameters (PT, PTT) • Thrombotic events were an additional endpoint [Morrison et al, 2012]
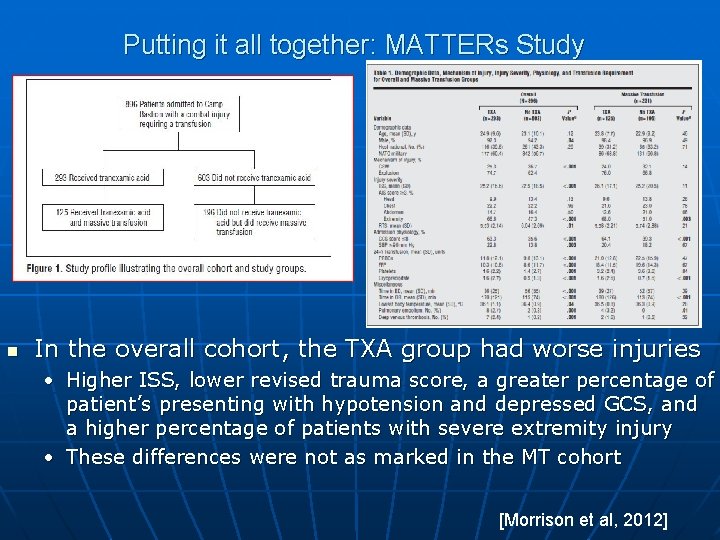
Putting it all together: MATTERs Study n In the overall cohort, the TXA group had worse injuries • Higher ISS, lower revised trauma score, a greater percentage of patient’s presenting with hypotension and depressed GCS, and a higher percentage of patients with severe extremity injury • These differences were not as marked in the MT cohort [Morrison et al, 2012]
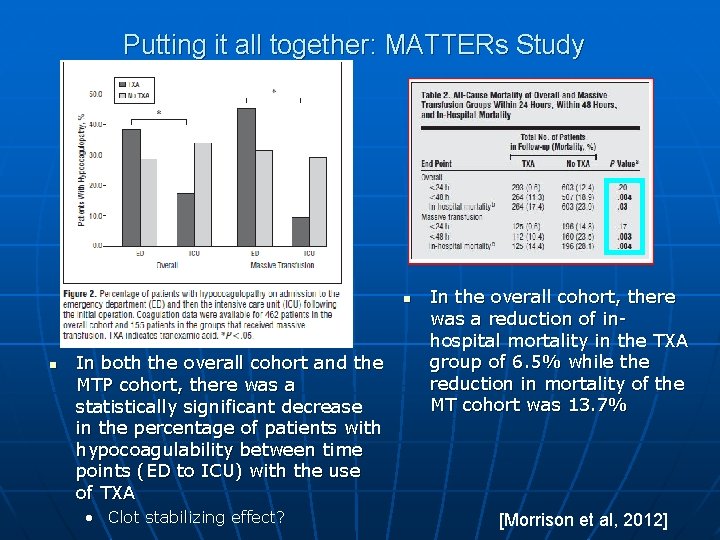
Putting it all together: MATTERs Study n n In both the overall cohort and the MTP cohort, there was a statistically significant decrease in the percentage of patients with hypocoagulability between time points (ED to ICU) with the use of TXA • Clot stabilizing effect? In the overall cohort, there was a reduction of inhospital mortality in the TXA group of 6. 5% while the reduction in mortality of the MT cohort was 13. 7% [Morrison et al, 2012]
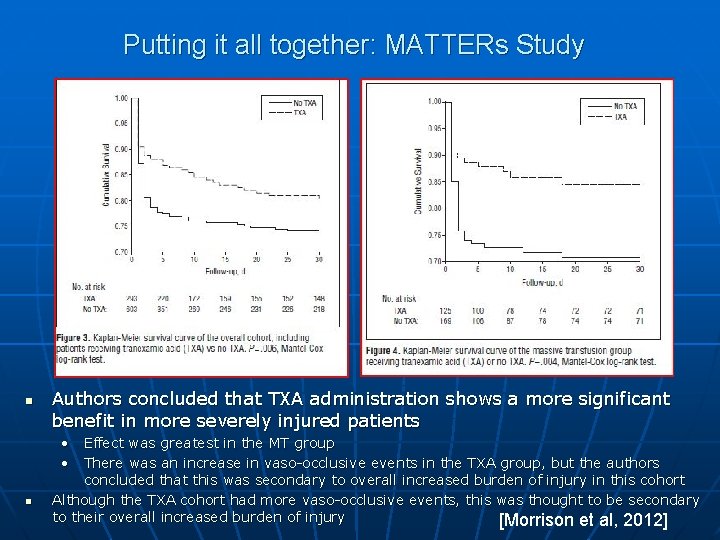
Putting it all together: MATTERs Study n n Authors concluded that TXA administration shows a more significant benefit in more severely injured patients • Effect was greatest in the MT group • There was an increase in vaso-occlusive events in the TXA group, but the authors concluded that this was secondary to overall increased burden of injury in this cohort Although the TXA cohort had more vaso-occlusive events, this was thought to be secondary to their overall increased burden of injury [Morrison et al, 2012]

Conclusions about TXA n n n TXA is associated with a reduction in all cause mortality in adult trauma patients with signs of bleeding TXA has the greatest impact if given in the fist hour after injury but can be beneficial if given between 1 and 3 hours after injury TXA should not be given after 3 hours after injury • Increased mortality TXA is not associated with increased risk of vascular occlusive events TXA administration is beneficial in massively transfused patients [Napolitano et al, 2013] 2013

Tranexamic acid: Future directions n n There are some interesting proposed new uses for TXA In a study protocol published in 2013, authors aim to see if ultra-early and short-term administration of the TXA as an add-on to standard subarachnoid hemorrhage management, leads to better functional outcomes in a prospective, randomized control trial [Germans et al 2013]

References n n n n The centers for disease control and prevention http: //www. cdc. gov/injury/wisqars/pdf/10 LCID_All_Deaths_By_Age_Group_2010 -a. pdf Nunez TC, Young PP, Holcomb JB, Cotton BA. Creation, implementation, and maturation of a massive transfusion protocol for the exsanguinating trauma patient. J Trauma. 2010 Jun; 68(6): 1498 -505. CRASH-2 trial collaborators, Shakur H, Roberts I, Bautista R, Caballero J, Coats T, Dewan Y, El-Sayed H, Gogichaishvili T, Gupta S, Herrera J, Hunt B, Iribhogbe P, Izurieta M, Khamis H, Komolafe E, Marrero MA, Mejía-Mantilla J, Miranda J, Morales C, Olaomi O, Olldashi F, Perel P, Peto R, Ramana PV, Ravi RR, Yutthakasemsunt S. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebocontrolled trial. Lancet. 2010 Jul 3; 376(9734): 23 -32. CRASH-2 collaborators, Roberts I, Shakur H, Afolabi A, Brohi K, Coats T, Dewan Y, Gando S, Guyatt G, Hunt BJ, Morales C, Perel P, Prieto-Merino D, Woolley T. The importance of early treatment with tranexamic acid in bleeding trauma patients: an exploratory analysis of the CRASH-2 randomised controlled trial. Lancet. 2011 Mar 26; 377(9771): 1096 -101, 1101. e 1 -2. Germans MR, Post R, Coert BA, Rinkel GJ, Vandertop WP, Verbaan D. Ultra-early tranexamic acid after subarachnoid hemorrhage (ULTRA): study protocol for a randomized controlled trial. Trials. 2013 May 16; 14: 143. Morrison JJ, Dubose JJ, Rasmussen TE, Midwinter MJ. Military Application of Tranexamic Acid in Trauma Emergency Resuscitation (MATTERs) Study. Arch Surg. 2012 Feb; 147(2): 113 -9. Morrison JJ, Ross JD, Poon H, Midwinter MJ, Jansen JO. Intra-operative correction of acidosis, coagulopathy and hypothermia in combat casualties with severe haemorrhagic shock. Anaesthesia. 2013 Aug; 68(8): 846 -50. Napolitano LM, Cohen MJ, Cotton BA, Schreiber MA, Moore EE. Tranexamic acid in trauma: how should we use it? J Trauma Acute Care Surg. 2013 Jun; 74(6): 1575 -86.

References cont. n n n Wohlauer MV, Moore EE, Thomas S, Sauaia A, Evans E, Harr J, Silliman CC, Ploplis V, Castellino FJ, Walsh M. Early platelet dysfunction: an unrecognized role in the acute coagulopathy of trauma. J Am Coll Surg. 2012 May; 214(5): 739 -46. Maegele M, Schöchl H, Cohen MJ. An Up-date on the Coagulopathy of Trauma. Shock. 2013 Nov 4. Tapia NM, Chang A, Norman M, Welsh F, Scott B, Wall MJ Jr, Mattox KL, Suliburk J. TEG-guided resuscitation is superior to standardized MTP resuscitation in massively transfused penetrating trauma patients. J Trauma Acute Care Surg. 2013 Feb; 74(2): 378 -85; discussion 385 -6. Johansson PI, Sørensen AM, Larsen CF, Windeløv NA, Stensballe J, Perner A, Rasmussen LS, Ostrowski SR. Low hemorrhage-related mortality in trauma patients in a Level I trauma center employing transfusion packages and early thromboelastography-directed hemostatic resuscitation with plasma and platelets. Transfusion. 2013 Apr 25. Holcomb JB, Minei KM, Scerbo ML, Radwan ZA, Wade CE, Kozar RA, Gill BS, Albarado R, Mc. Nutt MK, Khan S, Adams PR, Mc. Carthy JJ, Cotton BA. Admission rapid thrombelastography can replace conventional coagulation tests in the emergency department: experience with 1974 consecutive trauma patients. Ann Surg. 2012 Sep; 256(3): 476 -86.
- Slides: 43