MASS TRANSFER 1 UNIT 1 INRODUCTION Introduction Three





































- Slides: 43

MASS TRANSFER - 1

UNIT : 1 INRODUCTION

Introduction : • • Three fundamental transfer processes: Momentum transfer Heat transfer Mass transfer

Mass transfer : • Mass transfer is the net movement of mass from one location, usually meaning stream, phase, fraction or component, to another. • Mass transfer occurs in many processes, such as absorption, evaporation, drying, precipitation, membrane filtration, and distillation.


Driving force :

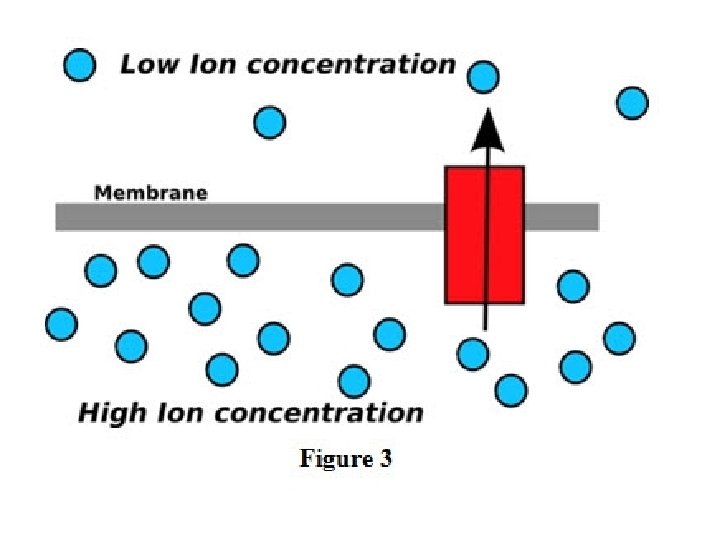
• Mass transfer may occur in a gas mixture, a liquid solution or solid. • Mass transfer occurs whenever there is a gradient in the concentration of a species.

Definition of Concentration : • Number of molecules of each species present per unit volume (molecules/m 3) • ii) Molar concentration of species i = Number of moles of i per unit volume (kmol/m 3) • iii) Mass concentration = Mass of i per unit volume (kg/m 3)

Classification of mass transfer: • • • Gas-gas contact Gas-solid contact Gas-liquid contact Liquid-solid contact Solid-solid contact

UNIT : 2 DIFFUSION

Diffusion phenomena • Fick’s law: linear relation between the rate of diffusion of chemical species and the concentration gradient of that species. • Thermal diffusion: Diffusion due to a temperature gradient. Usually negligible unless the temperature gradient is very large. • Pressure diffusion: Diffusion due to a pressure gradient. Usually negligible unless the pressure gradient is very large

Forced diffusion: • Diffusion due to external force acting on a molecule. Forced diffusion occurs when an electrical field is imposed on an electrolyte (for example, in charging an automobile battery)Knudsen diffusion: Diffusion phenomena occur in porous solids

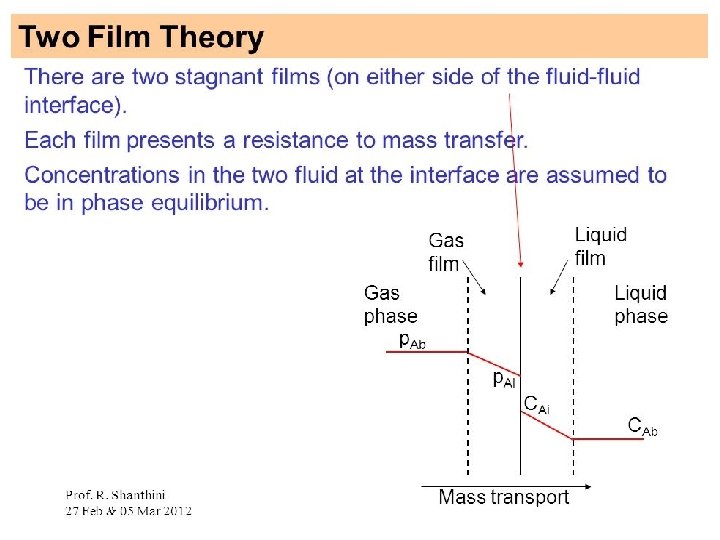
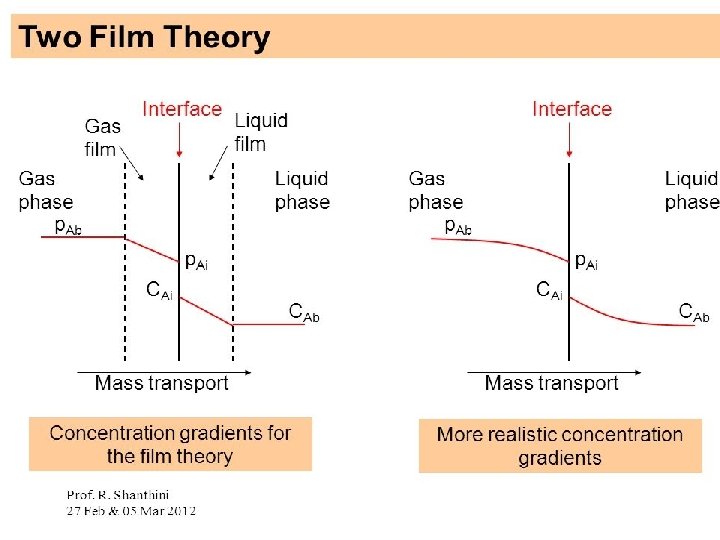
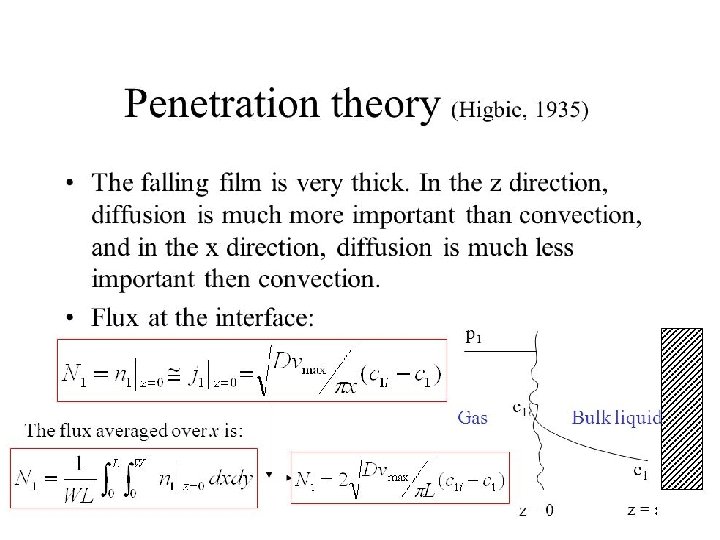
• Whenever there is concentration difference in a medium, nature tends to equalize things by forcing a flow from the high to the low concentration region. • The molecular transport process of mass is characterized by the general equation: Rate of transfer process = driving force resistance

Modes of Mass transfer The two modes of mass transfer: • molecular diffusion • Convective mass transfer

Molecular Diffusion : • The diffusion of molecules when the whole bulk fluid is not moving but stationary. Diffusion of molecules is due to a concentration gradient. • The general Fick’s Law Equation for binary mixture of A and B • c = total concentration of A and B [kgmol (A + B)/m 3] • x. A= mole fraction of A in the mixture of A and B

Molecular diffusion equation : • Fick’s Law • is the molar flux of component A in the z direction in kg mol A/s. m 2. • is the molecular diffusivity of the molecule A in B in m 2/s • is the concentration of A in kg mol/m 3. • z is the distance of diffusion in m

Fick’s Law: • Molecular diffusion or molecular transport can be defined as the transfer or movement of individual molecules through a fluid by mean of the random, individual movements of the molecules












UNIT : 3 DRYING













