Mass Spectrometry Ionization Techniques and Mass Analyzers Ionization







































- Slides: 51

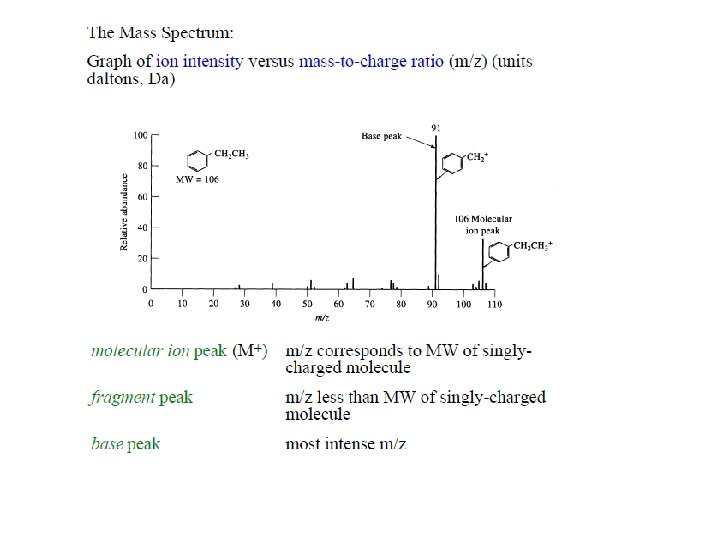
Mass Spectrometry Ionization Techniques and Mass Analyzers



Ionization Techniques Gas-Phase Methods • Electron Impact (EI) • Chemical Ionization (CI) Desorption Methods • Matrix-Assisted Laser Desorption Ionization (MALDI) • Fast Atom Bombardment (FAB) Spray Methods • Electrospray (ESI) • Atmospheric Pressure Chemical Ionization (APCI)

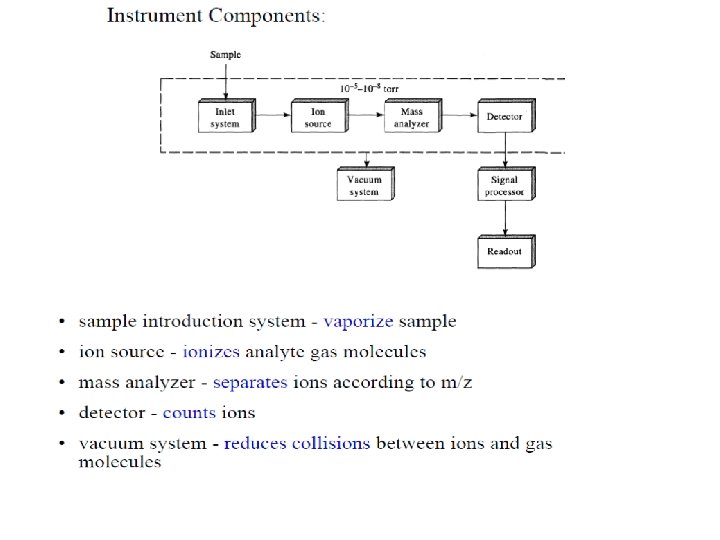
Ion Sources

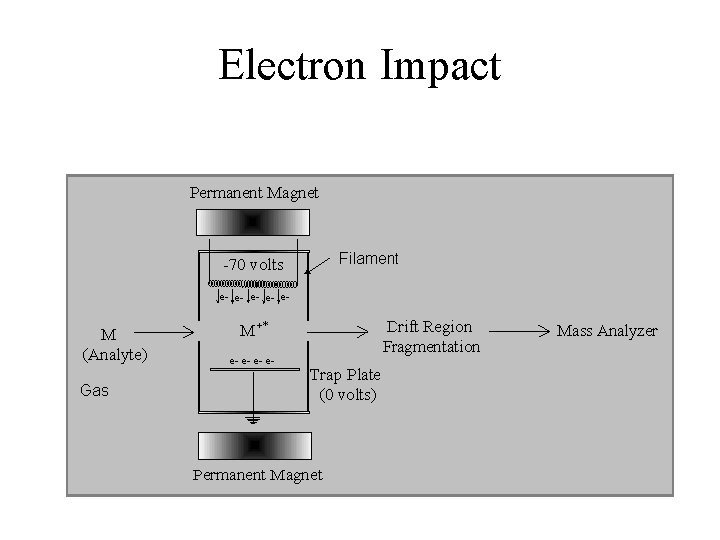
Electron Impact




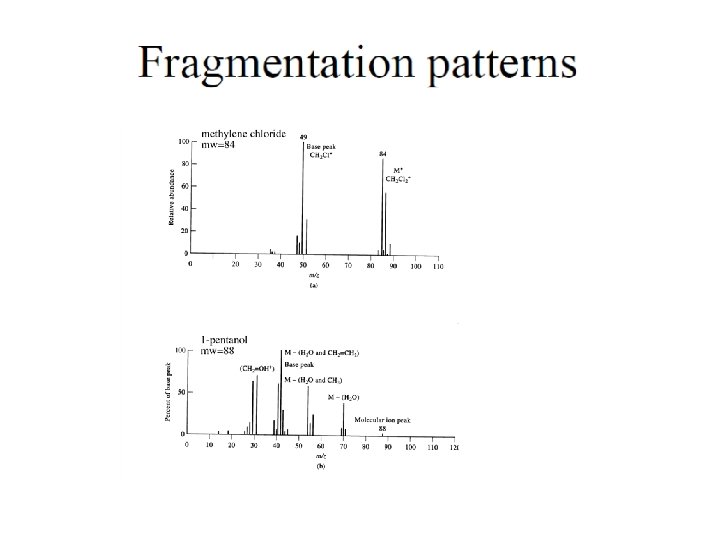
Electron Impact (low picomole) • • • Advantages Well-Established Fragmentation Libraries No Supression Insoluble Samples Interface to GC Non-Polar Samples • • • Disadvantages No Parent Identification Need Volatile Sample Need Thermal Stability No Interface to LC Low Mass Compounds (<1000 amu) Solids Probe Requires Skilled Operator

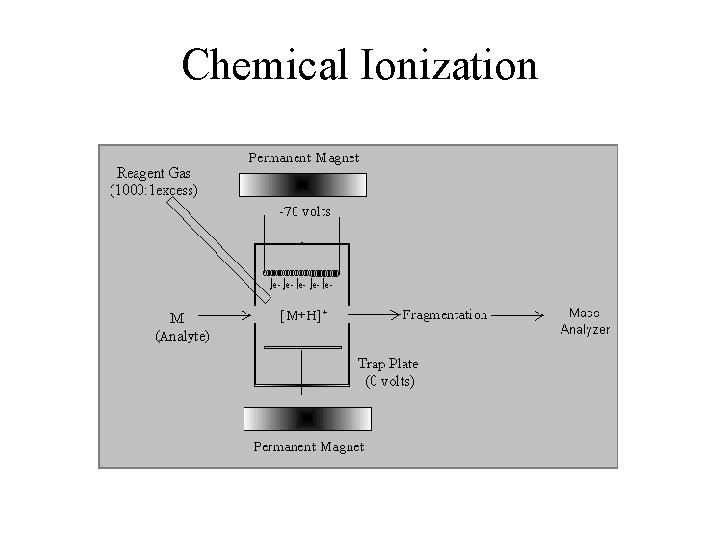
Chemical Ionization


Chemical Ionization Advantages • Parent Ion • Interface to GC • Insoluble Samples (low picomole) • • • Disadvantages No Fragment Library Need Volatile Sample Need Thermal Stability Quantitation Difficult Low Mass Compounds (<1000 amu) Solids Probe Requires Skilled Operator

Field Desorption Ionization Features • Wire probe • Electric current (107 – 108 V/cm) • Chemical ionization plasma or an electron impact ionisation • Non-homogeneity of the wire size and directional orientation provide high energy distribution • Spread over a small range.

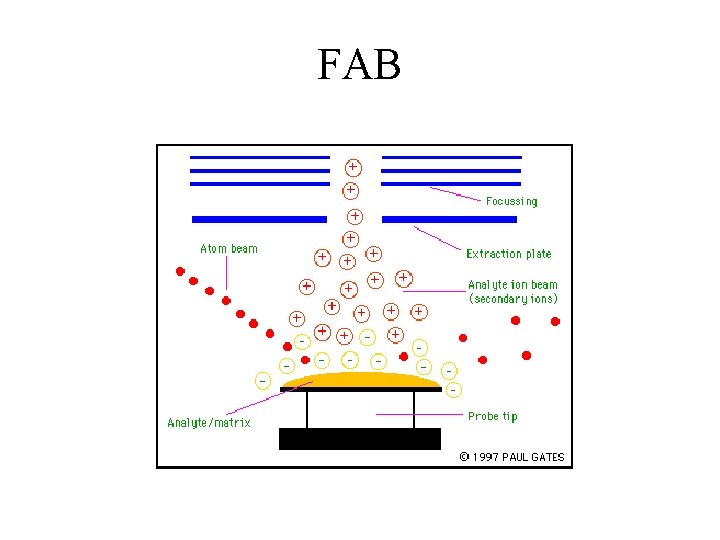
FAB

Fast Atom Bombardment Technique 1. 2. 3. 4. 5. 6. 7. 8. 9. Argon gas of high pressure is placed in between the ionizer and the sample. It is ionized by a hot filament type ion source Ar+ is accelerated in an electric field and focused into a beam that bombards the sample The Ar+ provides high kinetic energy and undergoes charge exchange with the solvent of sample solution Ar+ + S Ar + S+ The solvents (non volatile) used are glycerol, monothioglycerol, 2, 4 dipentylphenol, m-nitrobenzylalcohol A monolayer of material is completely sputtered in a matter of seconds that the sample surface need be continuously renewed Secondary ion MS technique is used for ionisation of sample. The bombarding Argon beam ionises the solvent. These ions react with the surrounding solvent molecules to produce (Sn + H)+ The sample undergoes proton transfer, hydride transfer, or ion-pair reaction with the reactant ion (Sn + H)+ to produce pseudomolecular ions such as (M+H)+, (M-H)+, and (M+S+H)+

FAB (nanomole) Advantages • Parent Ion • High Mass Compounds (10, 000 amu) • Thermally Labile Compounds (R. T. ) • • • Disadvantages No Fragment Library Solubility in Matrix (MNBA, Glycerol) Quantitation Difficult Needs Highly Skilled Operator Relatively Low Sensitivity

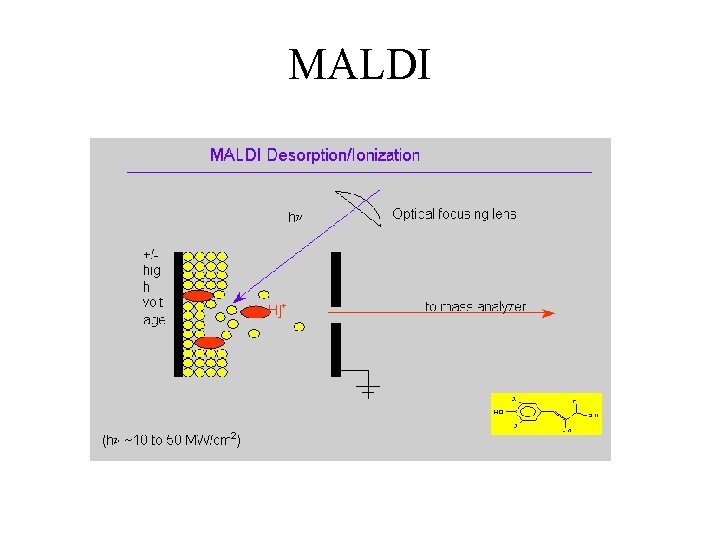
MALDI

MALDI


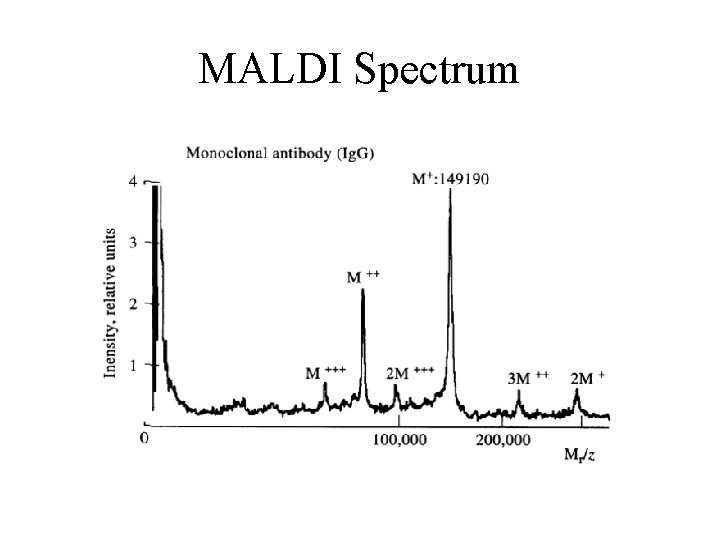
MALDI Spectrum

MALDI

Matrix Assisted Laser Desorption Ionisation (MALDI) (low femtomole) • • Advantages Parent Ion High Mass Compounds (>100, 000 amu) Thermally Labile Compounds (R. T. ) Easy to Operate Disadvantages • No Fragment Library • Wide variety of matrices • Quantitation Difficult

Electron Spray Ionisation

ESI


Mass Spectrum of ESI

ESI MS

ESI • • • (low femtomole to zeptomole) Advantages Disadvantages Parent Ion • No Fragmentation High Mass Compounds • Need Polar Sample (>100, 000 amu) • Need Solubility in Polar Thermally Labile Solvent (Me. OH, ACN, Compounds (<0º C) H 2 O, Acetone are best) Easy to Operate • Sensitive to Salts Interface to HPLC • Supression Zeptomole sensitivity with nanospray

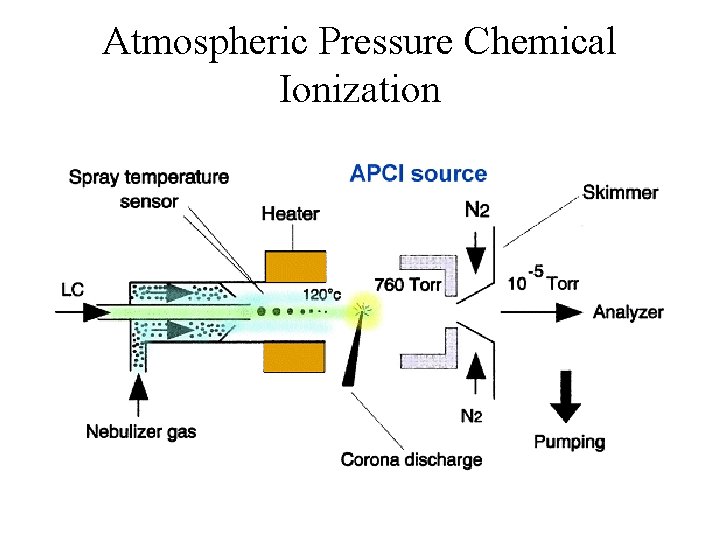
Atmospheric Pressure Chemical Ionization

APCI (high femtomole) • • • Advantages Parent Ion Insensitive to Salts Interface to HPLC Can use Normal Phase Solvents Handles High Flow Rates Disadvantages • Need Volatile Sample • Need Thermal Stability

Other Techniques Desorption • Plasma desorption • Laser desorption • Ion Bombardment Fourier Transform MS • FT-ICR • Rapid scan-Ion cyclotron resonance Tandem Mass spectrometry • Triple quadrupole MS • Quadrupole FT MS • Inductively Coupled Plasma MS • Secondary ion MS • Ion Microprobe mass analyser

TRIPLE QUADRUPOLE MASS SPECTROMETER

Triple Quadrupole Mass Spectrometry • • Ions from normal source First quadrupole – mass filter for sp. Mass sepn Second fragmentation done Second Qp ie rf only Qp – focuses scattered ions Third Qp – provides final scanned spectrum A 3 D fragmentation spectrum is obtained Each Qp provides each parent ion

Quadrupole FT-MS • + or – ions produced • Focussed into Qp mass analysers • Transmitted ions are accelerated into a superconducting solenoid magnet • Trapped in an ion cyclotron resonance cell • Detected by FT-MS

ICP-MS • ICP works on nebulisation, hydride generation, electrothermal vaporisation • Tail part of ICP is introduced into vacuum system

Mass Analyzers • • • Double Focusing Magnetic Sector Quadrupole Mass Filter Quadrupole Ion Trap Linear Time-of-Flight (TOF) Reflectron TOF Fourier Transform Ion Cyclotron Resonance (FT-ICR-MS)

Mass Analyzer Resolution

Double-Focusing Magnetic Sector


Double-Focusing Magnetic Sector Advantages • Very High Resolution (60, 000) • High Accuracy (<5 ppm) • 10, 000 Mass Range • • Disadvantages Very Expensive Requires Skilled Operator Difficult to Interface to ESI Low resolution MS/MS without multiple analyzers

Quadrupole Mass Filter

Quadrupole Mass Filter Advantages • Inexpensive • Easily Interfaced to Many Ionization Methods • • • Disadvantages Low Resolution (<4000) Low Accuracy (>100 ppm) MS/MS requires multiple analyzers Low Mass Range (<4000) Slow Scanning

Quadrupole Ion Trap

Quadrupole Ion Trap Advantages • Inexpensive • Easily Interfaced to Many Ionization Methods • MS/MS in one analyzer • • • Disadvantages Low Resolution (<4000) Low Accuracy (>100 ppm) Space Charging Causes Mass Shifts Low Mass Range (<4000) Slow Scanning

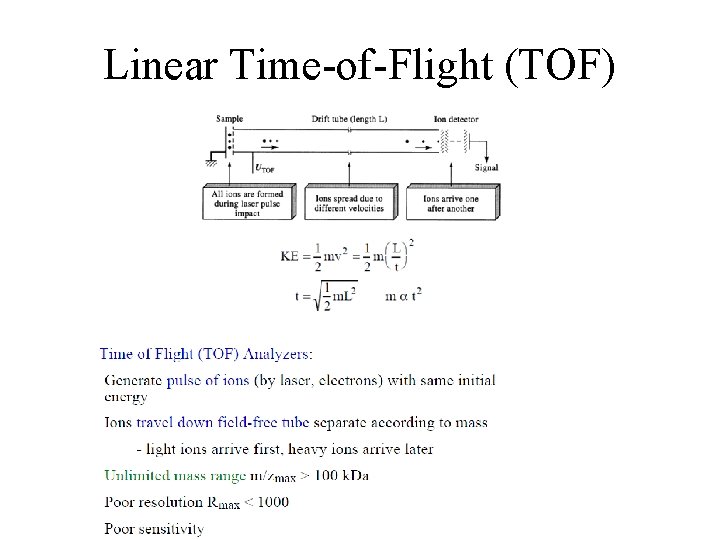
Linear Time-of-Flight (TOF)

Linear Time-of-Flight (TOF) Advantages • Extremely High Mass Range (>1 MDa) • Fast Scanning Disadvantages • Low Resolution (4000) • Low Accuracy (>200 ppm) • MS/MS not possible

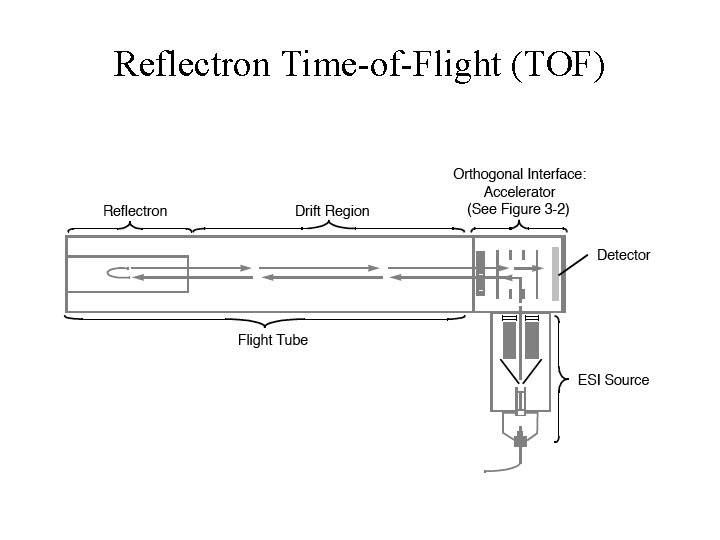
Reflectron Time-of-Flight (TOF)

Reflectron Time-of-Flight (TOF) • • Advantages High Resolution (>20, 000 in some models) High Accuracy (<5 ppm) 10, 000 Mass Range Fast Scanning Disadvantages • Low Resolution for MS/MS (PSD)

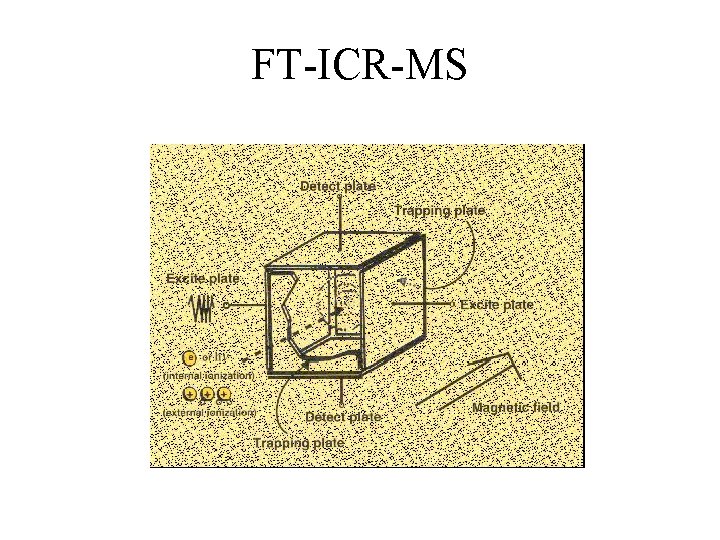
FT-ICR-MS

FT-ICR-MS Advantages • Extremely High Resolution (>500, 000) • Very Good Accuracy (<1 ppm) • MS/MS in one analyzer Disadvantages • Expensive • Requires Superconducting Magnet • Slow MS/MS