Mass Spectrometry Electron Ionization and Chemical Ionization Mass














- Slides: 24

Mass Spectrometry Electron Ionization and Chemical Ionization

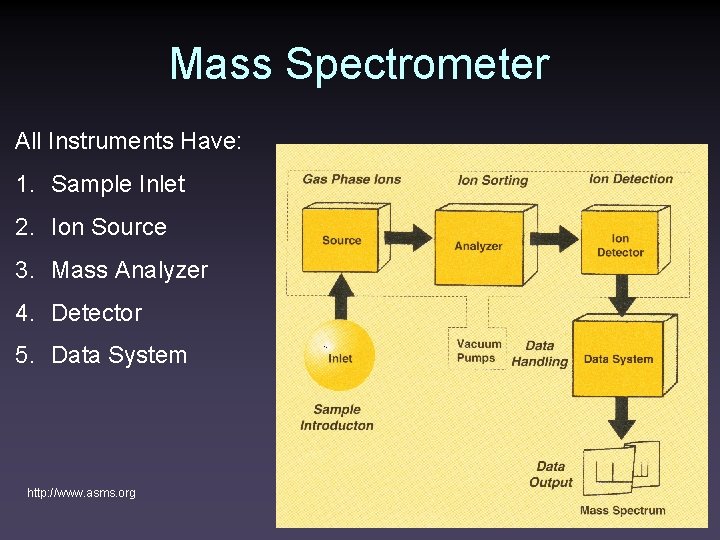
Mass Spectrometer All Instruments Have: 1. Sample Inlet 2. Ion Source 3. Mass Analyzer 4. Detector 5. Data System http: //www. asms. org

Ionization Techniques Gas-Phase Methods • Electron Ionization (EI) • Chemical Ionization (CI) Desorption Methods • Matrix-Assisted Laser Desorption Ionization (MALDI) • Fast Atom Bombardment (FAB) Spray Methods • Electrospray (ESI) • Atmospheric Pressure Chemical Ionization (APCI)

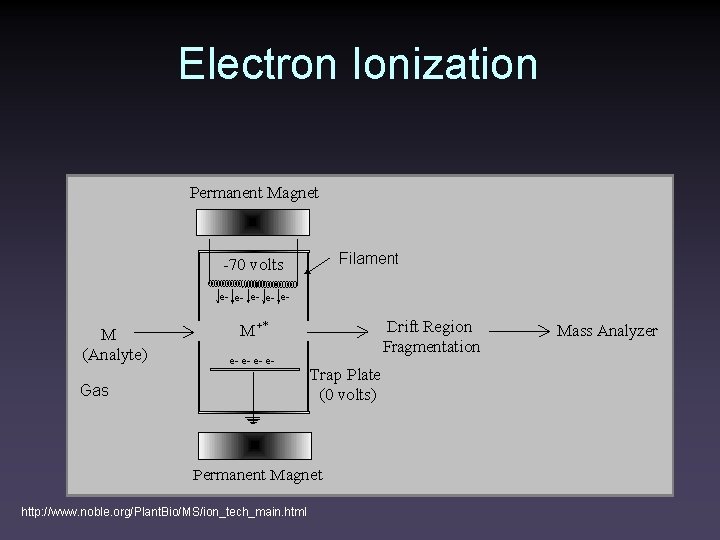
Electron Ionization http: //www. noble. org/Plant. Bio/MS/ion_tech_main. html

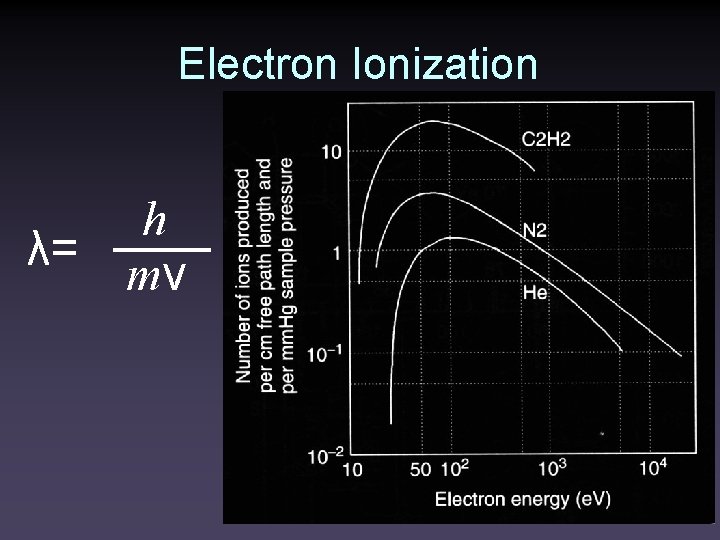
Electron Ionization λ= h mv

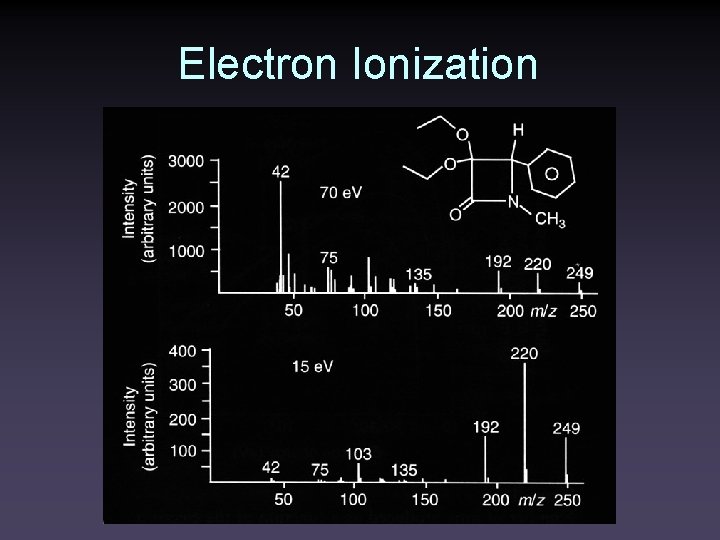
Electron Ionization • Samples must be vaporized in the ion source • Typically 1 of 1000 molecules entering the source is ionized • 10 -20 e. V of energy is imparted to the molecule • ~10 e. V is enough to ionize most molecules • Up to 230 kcal/mol is left to cause fragmentation

Electron Ionization

Electron Ionization (low picomole) • • • Advantages Well-Established Fragmentation Libraries No Supression Insoluble Samples Interface to GC Non-Polar Samples • • • Disadvantages Parent Identification Need Volatile Sample Need Thermal Stability No Interface to LC Low Mass Compounds (<1000 amu) Solids Probe Requires Skilled Operator

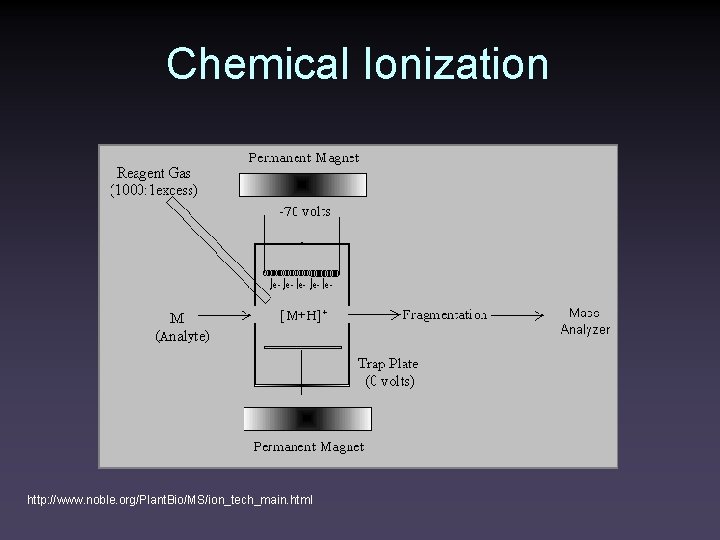
Chemical Ionization http: //www. noble. org/Plant. Bio/MS/ion_tech_main. html

Chemical Ionization • Reagent gas is introduced into the source at ~0. 5 torr • Reagent gas is preferentially ionized. Ions react mostly with neutral reagent gas • Reactions occurring depend on the nature of the reagent gas • Ions in the reagent gas plasma react with the analyte

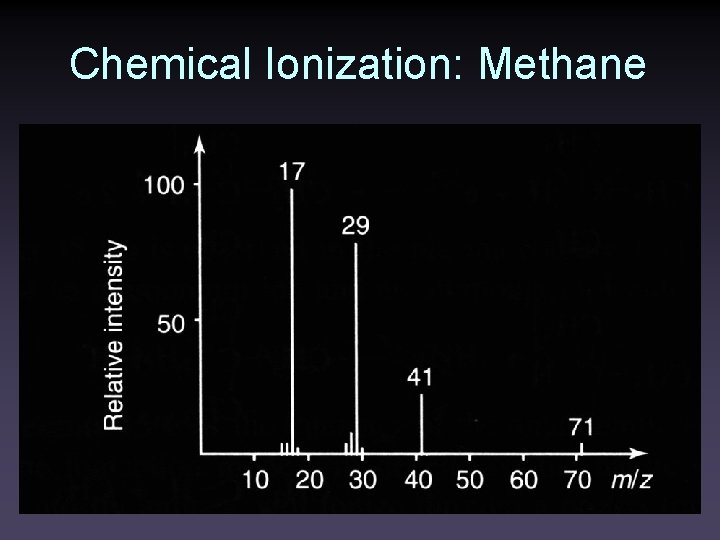
Chemical Ionization: Methane • Methane primarily forms CH 4+ • with CH 2+ • and CH 3+ • CH 4+ • + CH 4 → CH 5+ + CH 3 (m/z 17) • CH 2+ • + CH 4 → C 2 H 3+ + H 2 + H • • C 2 H 3+ + CH 4 → C 3 H 5+ + H 2 (m/z 41) • CH 3+ + CH 4 → C 2 H 5+ + H 2 (m/z 29)

Chemical Ionization: Methane

Chemical Ionization: Methane • Ions other than saturated hydrocarbons react via proton transfer • CH 5+ + M → MH+ + CH 4 (or via C 2 H 5+ or C 3 H 5+) • For saturated hydrocarbons, hydride abstractions is common • CH 5+ + RH → R+ + CH 4 + H 2 • For polar molecules, adducts can form • CH 3+ + M → (M+CH 3)+ • MH+, R+, and adducts are pseudomolecular ions.

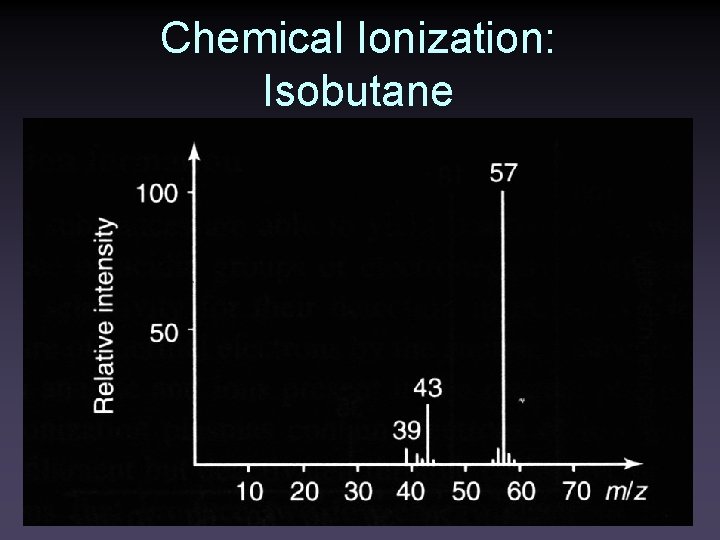
Chemical Ionization: Isobutane

Chemical Ionization: Isobutane

Chemical Ionization: Isobutane • Reacts through Proton Transfer • C 4 H 9+ + M → MH+ + C 4 H 8 • For saturated hydrocarbons, no reaction • For polar molecules, adducts can form • C 4 H 9+ + M → (M+C 4 H 9)+ • Lack of reaction with hydrocarbons can be used for selective detection of compounds in mixtures containing hydrocarbons • Less fragmentation is observed with isobutane. (molecular species is more reliably formed)

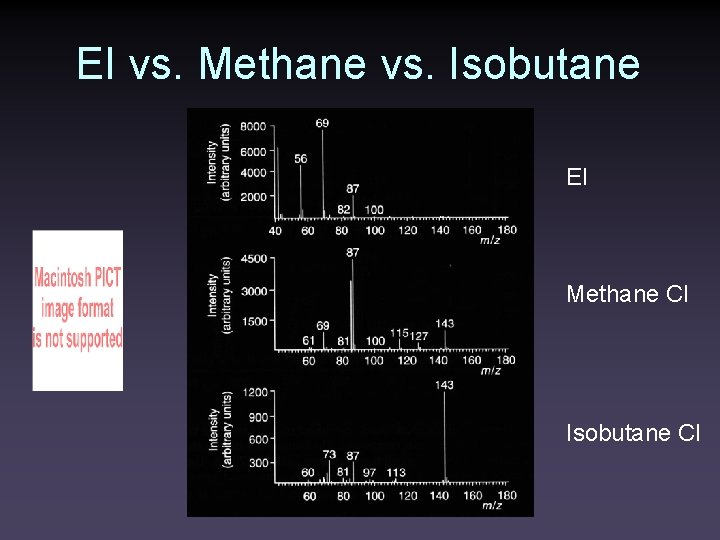
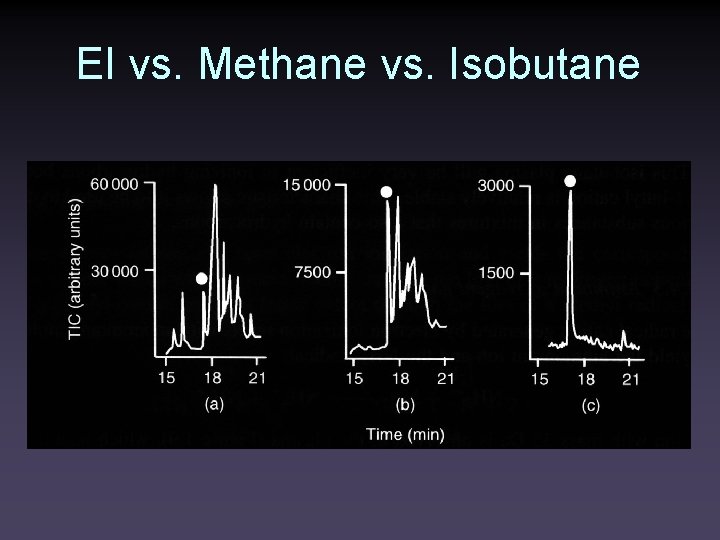
EI vs. Methane vs. Isobutane EI Methane CI Isobutane CI

EI vs. Methane vs. Isobutane

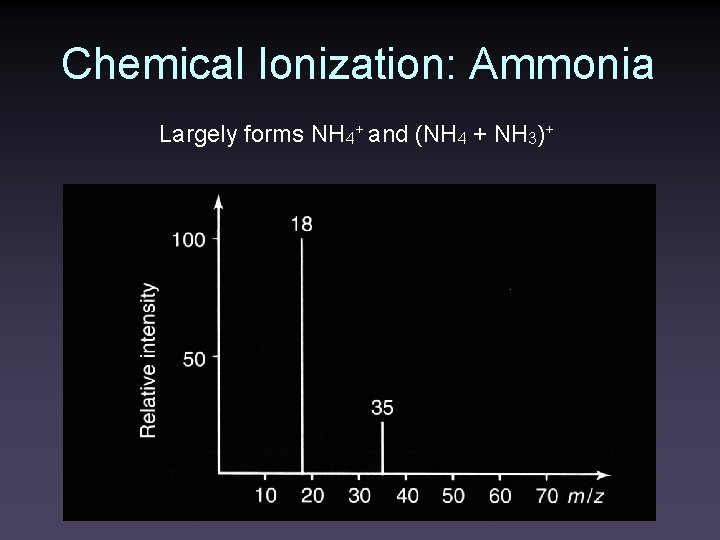
Chemical Ionization: Ammonia Largely forms NH 4+ and (NH 4 + NH 3)+

Chemical Ionization: Ammonia • Reacts through Proton Transfer with basic molecules • NH 4+ + M → MH+ + NH 3 • Less basic molecules will form adducts • NH 4+ + M → (M+NH 4)+ • Many molecules with intermediate basicity, both ions will be observed • For saturated hydrocarbons, no reaction • Less fragmentation is observed with ammonia. (molecular species is most reliably formed)

Chemical Ionization: Negative Ions • Low energy electrons are present in the CI plasma • These can attach to molecules with high electron affinities • There are two principal pathways • AB + e- → AB- • (associative resonance capture) • AB + e- → A • + B- (dissociative resonance capture) • Deprotonation can also occur if a basic ion is formed in the reagent gas plasma

Chemical Ionization: Negative Ions • A common negative CI gas is a mixture of N 2 O/CH 4 in a ratio 25/75 • N 2 O + e- → N 2 O- • • N 2 O- • → N 2 + O- • • O- • + CH 4 → CH 3 • + OH • This mixture yields thermal electrons for electron capture and hydroxide for deprotonation of acidic molecules

Chemical Ionization Proton Affinity ∆H˚acid CH 4 543 1749 H 2 O 691 1634 NH 3 854 1689 CH 3 OH 754 1592 C 2 H 5 OH 776 1583 IPA 793 1573 t-Bu. OH 803 1568 Aniline 921 1533

Chemical Ionization (low picomole) Advantages • Molecular Ion • Interface to GC • Insoluble Samples • • • Disadvantages No Fragment Library Need Volatile Sample Need Thermal Stability Quantitation Difficult Low Mass Compounds (<1000 amu) Solids Probe Requires Skilled Operator