Mass Spectrometer Basics and Spectra Interpretation MS Basics

Mass Spectrometer Basics and Spectra Interpretation

MS Basics n What does a mass spectrometer do? A mass spectrometer produces charged particles (ions) from the chemical substances that are to be analyzed. The mass spectrometer then uses electric and magnetic fields to measure the mass ("weight„ or more precisely the Mass/charge ratio, m/z or m/e) of the charged particles. Mass spectra are typically presented as a „bar“ type graph. n What does the mass tell us? Let us use water (H 2 O) as an example. A water molecule consists of two hydrogen's (H) and one oxygen (O). The total mass of a water molecule is the sum of the mass of two hydrogen's (approximately 1 atomic mass unit per hydrogen) and one oxygen (approximately 16 atomic mass units per oxygen): 16 O ~ 16 amu 2 H: 2 amu (atomic mass units) + O: 16 amu ______ = H 2 O: 18 amu = Electron ~ 0 AMU = Neutron ~ 1 AMU = Proton 11/1/2020 MS basics and spectra interpretation ~ 1 AMU 2

MS basics Let's suppose that we put some water vapor into the mass spectrometer. A very small amount of water is all that is needed -- the water is introduced into a vacuum chamber (the "ion source") of the mass spectrometer. If we shoot a beam of electrons through the water vapor, some of the electrons will hit water molecules and knock off an electron. If we lose a (negatively charged) electron from the (neutral) water molecule, the water will be left with a net positive charge. In other words, we have produced charged particles, or "ions" from the water: H 2 O + 1 (fast) electron --> [H 2 O]+ + 2 electrons Some of the collisions between the water molecules and the electrons will be so hard that the water molecules will be broken into smaller pieces, or "fragments ". For water, the only possible fragments are [OH]+, O+ and H+. The mass spectrum of water will show peaks that can be assigned to masses of 1, 16, 17, und 18 amu. The relative abundance and the fragmentation pattern are like a finger print of a substance. 11/1/2020 MS basics and spectra interpretation 3

MS basics Molecule + electron Example: CO 2 + e- (70 e. V) 11/1/2020 Ionization of molecule Fragmentation of molecule Multiple charged ions Isotopic effects are shown CO 2+ 44 m/e C+ O+ CO+ 12 m/e 16 m/e 18 m/e CO 2++ 22 m/e 12 C 16 O 18 O 46 m/e MS basics and spectra interpretation 4
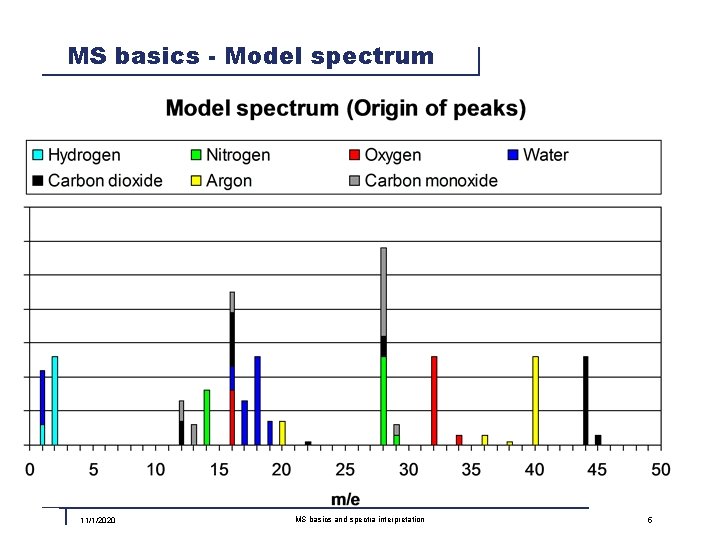
MS basics - Model spectrum 11/1/2020 MS basics and spectra interpretation 5
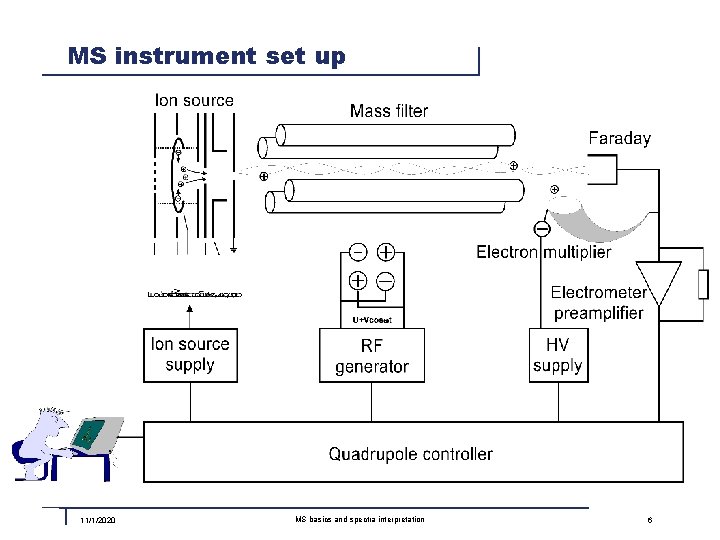
MS instrument set up 11/1/2020 MS basics and spectra interpretation 6

MS set up n General To ensure filament longevity, ion mobility and detector operation the analyzer has to be kept under vacuum conditions. n Ion source The filament emits electrons which – on their path to the positive formation chamber – collide with gas particles and knock out electrons. In this process the gas particles become positively charged, they are ionized. The extraction orifice extracts the ions from the formation chamber, and the focus lens ensures ideal conditions at the entrance to the mass filter. n Mass filter The RF and DC voltages for separating the particles are available on the two rod pairs of the quadrupole mass filter. 11/1/2020 MS basics and spectra interpretation 7

MS set up – cont. n Ion detection, Electron multiplier The ions that have passed the filter impinge on the Faraday collector where they release their charge. The very small resulting current is the input to the electrometer preamplifier. If a secondary electron multiplier (Channeltron) is configured and switched on, the negative high voltage on the input of the latter attracts the ions. On impact they release electrons and these in turn release additional electrons which leads to an amplification in the magnitude of several powers of ten. The Channeltron output is connected in parallel to the Faraday collector. n Electrometer preamplifier This instrument converts very small currents (10 -15. . . 10 -5 A) from the Faraday or Channeltron to a voltage which is subsequently processed in the QC 200 quadrupole controller. n Quadrupole contoller Controls all other modules and processes their measurement data. 11/1/2020 MS basics and spectra interpretation 8

Spectra interpretation "a mass spectrometrist is someone, who figures out what something is, by smashing it with a hammer and looking at the pieces" 11/1/2020 MS basics and spectra interpretation 9

Measurement of pure Argon Information from spectra library 11/1/2020 MS basics and spectra interpretation 10
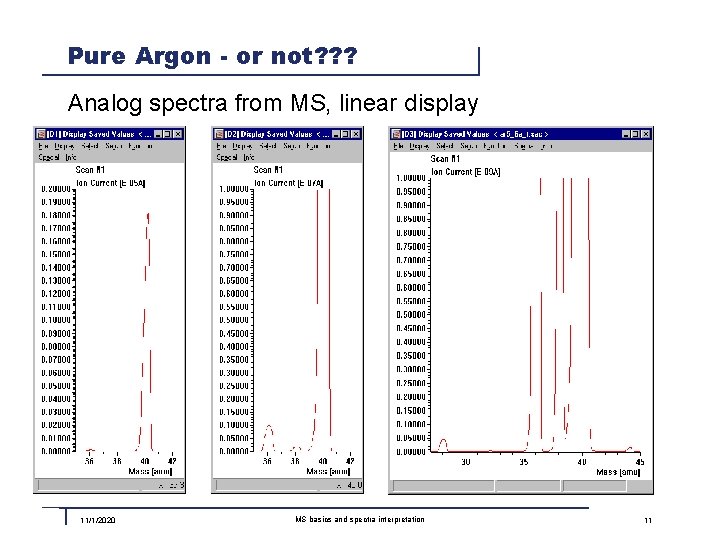
Pure Argon - or not? ? ? Analog spectra from MS, linear display 11/1/2020 MS basics and spectra interpretation 11
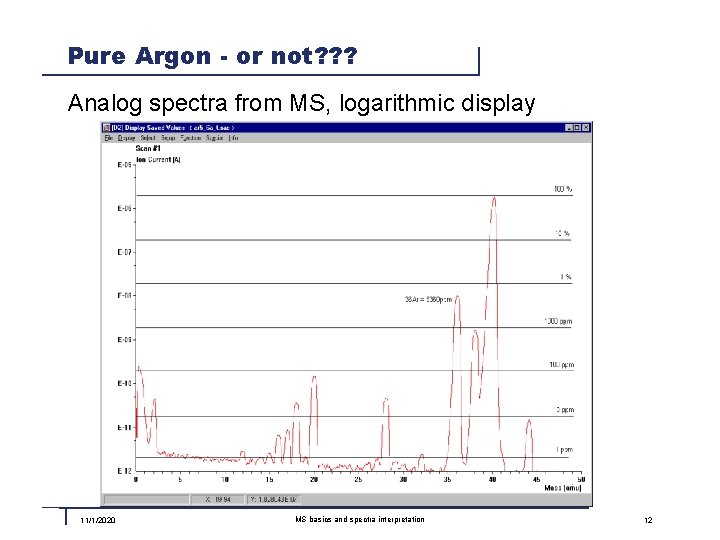
Pure Argon - or not? ? ? Analog spectra from MS, logarithmic display 11/1/2020 MS basics and spectra interpretation 12

Pure Argon - Quality 5. 6 In the MS we see more then just a peak at mass 40. Why? ? ? n 36, 38: Argon has stabile Isotopes with Mass 36 und 38 n 20: Argon is partially double ionized in the ion source and appears as Ar++ The following peaks are system related (background): n 18: Water vapor from the vacuum chamber, sealings, etc. n 14, 28: Nitrogen from the vacuum chamber, sealings, etc. n 16, 32: Oxygen from the vacuum chamber, sealings, etc. n 44: Carbon dioxide from the vacuum chamber, sealings, etc. n 1, 2: Hydrogen from the vacuum chamber and from water vapor All these are just traces in the level of <100 ppm! 11/1/2020 MS basics and spectra interpretation 13
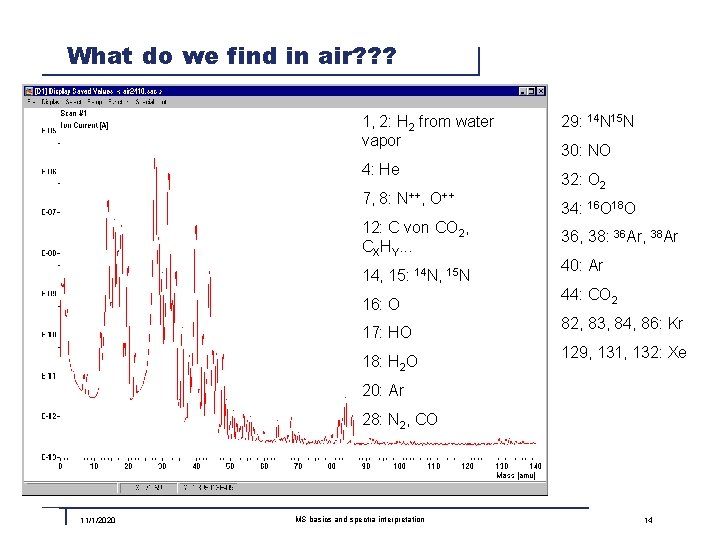
What do we find in air? ? ? 1, 2: H 2 from water vapor 4: He 7, 8: N++, O++ 12: C von CO 2, CXHY. . . 14, 15: 14 N, 15 N 16: O 17: HO 18: H 2 O 29: 14 N 15 N 30: NO 32: O 2 34: 16 O 18 O 36, 38: 36 Ar, 38 Ar 40: Ar 44: CO 2 82, 83, 84, 86: Kr 129, 131, 132: Xe 20: Ar 28: N 2, CO 11/1/2020 MS basics and spectra interpretation 14

Possible errors The last slide showed the most simple interpretation. In detail it can be much more difficult. n 16: Could be CH 4 instead of O n 17: Could be NH 3 instead of HO n 20: Could be Ne instead of Ar++ n 29: Could be C 2 H 5 (Ethanol) instead of 14 N 15 N n 36: Could be HCl instead of 36 Ar n 44: Could be N 2 O instead of CO 2 Knowledge about the sample and possible reactions are necessary for an exact interpretation of the measurements. Mass spectra are reproducible, spectra libraries (e. g. Wiley, NIST) can help with the identification of unknown substances. 11/1/2020 MS basics and spectra interpretation 15

List of fragments part 1 Add. masses Possible origin Fragment Masse (m/e) 1 H+ H+ H+ H 2 O Cx. Hy 2 18, 17, 16 12, 13, 14, 26, 27 etc. 2 H 2+ He+ C++ C++ H 2 He He CO CO 2 Cx. Hy 1 4 2 12, 28, 29 12, 28, 44 12, 13, 14, 26, 27 etc. N++ O++ C+ C+ C+ N 2 O 2 H 2 O CO CO 2 Cx. Hy 14, 28, 29 16, 32, 34 16, 17, 18 28, 29, 44 13, 14, 26, 27 etc. 13 CH+ Cx. Hy 12, 14, 26, 27 etc. 14 N+ N+ CH 2+ N 2 NH 3 Cx. Hy 28, 29 15, 16, 17 12, 13, 26, 27 etc. CO++ CO 28, 29 4 6 7 8 12 11/1/2020 15 CH 3+ NH+ Cx. Hy NH 3 12, 13, 14, 26, 27 etc. 14, 16, 17 16 O+ O+ CH 4+ NH 2+ OH+ NH 3+ H 2 O+ F+ F+ HF+ 20 Ne+ Ar++ CO 2++ 22 Ne+ C 2 H 2+ C 2 H 3+ N 2+ C 2 H 4+ CO+ C 2 H 5+ 14 N 15 N+ O 2 H 2 O CH 4 NH 3 H 2 O F 2 HF HF 20 Ne Ar CO 2 22 Ne Cx. Hy N 2 Cx. Hy CO CO 2 Cx. Hy N 2 32, 34 17, 18 12, 13, 14, 15, 17 16, 18 14, 15, 16 16, 17 38 20 19 22 40 6, 12, 28, 29, 44 20 12, 13, 14, 26, 27 12, 13, 14, 24, 26 7, 14, 29 12, 13, 14, 26 6, 12, 29, 44 12, 13, 14, 26, 27, 28 7, 14, 28 17 18 19 20 22 24 26 27 28 29 MS basics and spectra interpretation 16

List of fragments part 2 30 31 32 34 35 36 37 38 39 40 41 42 43 C 2 H 6+ C 2 H 6 NO+ NO C 2 H 2 OH+C 2 H 3 OH O 2+ O 2 S+ H 2 S S+ SO 2 H 232 S+ H 2 S 34 S + H 2 S 16 O 18 O+ O 2 35 Cl+ Cl 2 35 Cl+ HCL H 35 Cl+ HCl 36 Ar+ Ar 37 Cl+ Cl 2 37 Cl+ HCl 37 H Cl+ HCl C 3 H 2+ Cx. Hy 38 Ar+ Ar C 3 H 3+ Cx. Hy 39 K+ K Ar+ Ar C 3 H 4+ Cx. Hy C 3 H 5+ Cx. Hy 41 K+ K C 3 H 6+ Cx. Hy C 3 H 7+ Cx. Hy C 2 H 3 O+ C 2 H 5 OH 11/1/2020 12, 13, 14, 26, 27, 28, 29 14, 16 26, 28, 29 8, 16, 34 33, 34 34, 64 32, 33, 34 8, 16, 32 37, 70, 72, 74 36, 37, 38 35, 37, 38 20, 38, 40 35, 70, 72, 74 35, 36, 38 35, 36, 37 12, 13, 14, 26, 27, 28 20, 36, 40 12, 13, 14, 26, 27, 28, 38 41 36, 38 12, 13, 14, 24, 26, 27, 28, 38, 39 39 12, 13, 14, 24, 26, 27, 28, 38, 39 31, 44, 45, 46 44 C 3 H 8+ C 3 H 8 CO 2+ CO 2 C 2 H 4 OH+ C 2 H 5 OH (alcohol) N 2 O+ N 2 O 45 C 2 H 5 O+ C 2 H 5 OH (alcohol) 13 CO + CO 2 2 46 NO 2+ NO 2 C 2 H 5 OH+ C 2 H 5 OH (alcohol) 48 SO+ SO 2 55 C 4 H 7+ Cx. Hy 38, 39 57 C 4 H 9+ Cx. Hy 38, 39 58 (CH 3)2 CO+, C 3 H 6 O (acetone) 64 SO 2+ SO 2 77 C 6 H 5+ Phenyl 78 C 6 H 6+ C 6 H 6 (benzene) 185 92. 5 Re++ Rhenium (filament) 93. 5 187 Re++ Rhenium (filament) 149 Phthalic ester (softening agent) 182 W+ Tungsten (filament) 183 W+ Tungsten (filament) 184 W+ Tungsten (filament) 185 Re+ Rhenium (filament) 187 Re+ Rhenium (filament) MS basics and spectra interpretation 41, 42, 43 6, 12, 28, 29 31, 43, 45, 46 14, 16, 28 31, 43, 44, 46 6, 12, 28, 29, 44 14, 16 31, 43, 44, 45 32, 64 12, 13, 14, 24, 26, 27, 28, 43 32, 48 50, 51, 52 93. 5, 187 92. 5, 187 183, 184, 186 182, 183, 186 187 185 17

Literature in the web n Basics http: //science. widener. edu/svb/massspec. pdf http: //www. chem. arizona. edu/massspec/ http: //www. jeol. com/ms/ n Web library NIST http: //webbook. nist. gov/ n General http: //www. sisweb. com/mstools. htm 11/1/2020 MS basics and spectra interpretation 18

Books n 11/1/2020 Spectra interpretation Title: A beginner‘s guide to mass spectral interpretation Author: Terrence A. Lee Publisher: John Wiley & Sons ISBN 0 -471 -97629 -6 MS basics and spectra interpretation 19
- Slides: 19