mass number The atomic number tells us the

mass number The atomic number tells us: Ø the number of protons in the nucleus of an atom. The mass number tells us: atomic number Ø the mass of the atom (protons + neutrons). Sometimes, scientists want to find out the total mass of an element or a compound. The total mass of an element is called the RELATIVE ATOMIC MASS (Ar). The total mass of a compound is called the RELATIVE FORMULA MASS (Mr).

21/12/2021 Task – 3 minutes! Find the relative atomic masses from the periodic table (inside front cover of your workbook): 12 C O 16 Ca 40 Cl 35. 5 N 14 H S Al Fe 1 32 27 56

Relative formula mass, Mr The relative formula mass of a compound is the relative atomic masses of all the elements in the compound added together. E. g. water H 2 O: Relative atomic mass of O = 16 Relative atomic mass of H = 1 Therefore Mr for water = 16 + (2 x 1) = 18 Work out Mr for the following compounds: 1) HCl H=1, Cl=35 so Mr = 36 2) Na. OH Na=23, O=16, H=1 so Mr = 40 3) Mg. Cl 2 Mg=24, Cl=35 so Mr = 24+(2 x 35) = 94 4) H 2 SO 4 H=1, S=32, O=16 so Mr = (2 x 1)+32+(4 x 16) = 98 5) K 2 CO 3 K=39, C=12, O=16 so Mr = (2 x 39)+12+(3 x 16) = 138
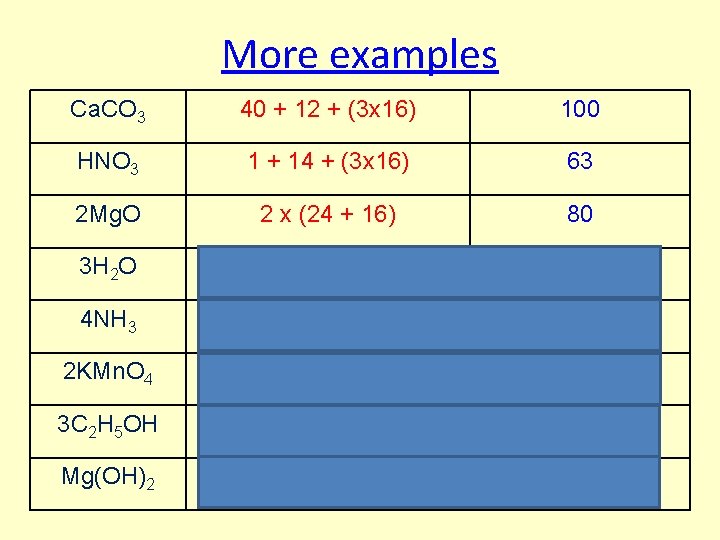
More examples Ca. CO 3 40 + 12 + (3 x 16) 100 HNO 3 1 + 14 + (3 x 16) 63 2 Mg. O 2 x (24 + 16) 80 3 H 2 O 3 x ((2 x 1) + 16) 54 4 NH 3 4 x (14+(3 x 1)) 68 2 KMn. O 4 2 x (39+55+(16 x 4)) 316 3 C 2 H 5 OH 3 x((2 x 12)+(5 x 1)+16+1)) 138 Mg(OH)2 24+ ((16+1)x 2) 58

Calculate the Percentage by mass 1. CO 2 (Oxygen) 72. 7% Relative Formula Mass of CO 2 = 12 + (2 x 16) = 44 g/mol. In CO 2 there’s 2 x Oxygen: 2 x 16 = 32. To find percentage by mass: (32/44) x 100 = 72. 7%

Calculate the Percentage by mass 1. CO 2 (Oxygen) 72. 7% 2. Ca. Cl 2 (Calcium) 36% 3. H 2 SO 4 (Sulfur) 32. 7% 4. Al 2 O 3 (Aluminium) 52. 9% 5. Fe(NO 3)3 (Oxygen) 59. 5%
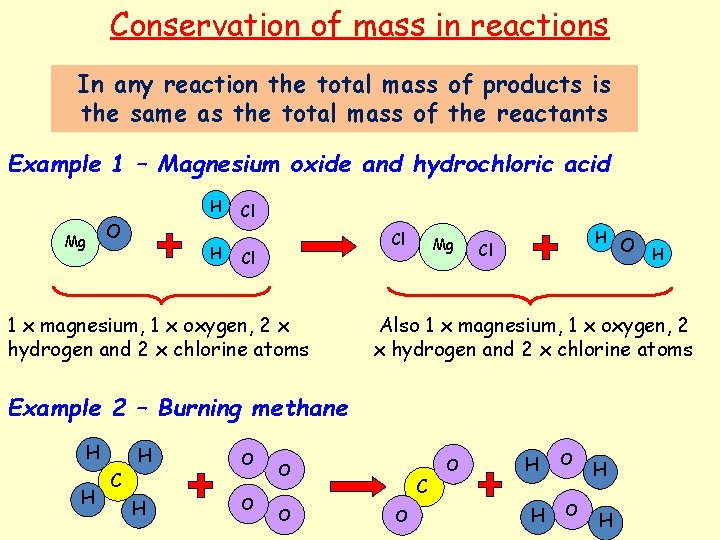
Conservation of mass in reactions In any reaction the total mass of products is the same as the total mass of the reactants Example 1 – Magnesium oxide and hydrochloric acid H Mg O H Cl Cl Cl 1 x magnesium, 1 x oxygen, 2 x hydrogen and 2 x chlorine atoms Mg H Cl Also 1 x magnesium, 1 x oxygen, 2 x hydrogen and 2 x chlorine atoms Example 2 – Burning methane H H H C H O O O H C O O H H

Calculating the mass of a product E. g. what mass of magnesium oxide is produced when 60 g of magnesium is burned in air? e Step 1: READ the equation: 2 Mg. O ig n or 2 Mg + O 2 Reacting ratio 2 Mg makes 2 Mg. O Step 2: WORK OUT the relative formula masses (Mr): 2 Mg = 2 x 24 = 48 2 Mg. O = 2 x (24+16) = 80 Step 3: LEARN and APPLY the following 3 points: 1) 48 g of Mg makes 80 g of Mg. O 2) 1 g of Mg makes 80/48 = 1. 66 g of Mg. O 3) 60 g of Mg makes 1. 66 x 60 = 100 g of Mg. O

re re 2) What mass of hydrochloric acid is needed to produce 11. 1 g calcium chloride? reacting ratio 2 HCl Mr: 2 HCl = 2 x (1 + 35. 5) ig no Ca. Cl 2 + 2 H 2 O ig no Ca(OH)2 + 2 HCl makes 1 Ca. Cl 2 = 40 + (2 x 35. 5) = 73 = 111 g Ca. Cl 2 produced by 73 g HCl 1 g of Ca. Cl 2 produced by (73/111)= 0. 66 so 11. 1 g produced by 0. 66 x 11. 1 = 7. 3 g of HCl
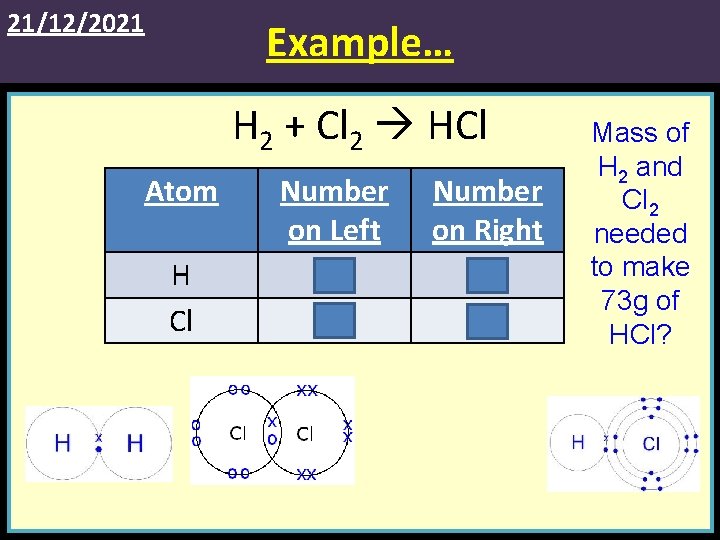
21/12/2021 Example… H 2 + Cl 2 HCl Atom H Cl Number on Left 2 2 Number on Right 1 1 Mass of H 2 and Cl 2 needed to make 73 g of HCl?
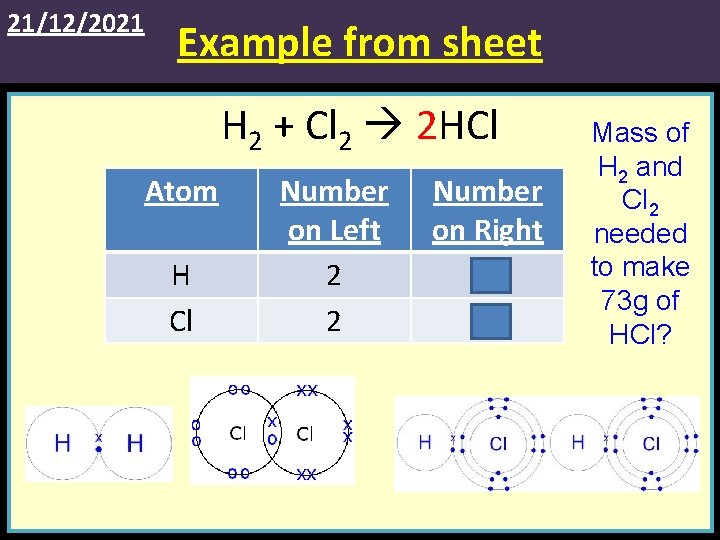
21/12/2021 Example from sheet H 2 + Cl 2 2 HCl Atom H Cl Number on Left 2 2 Number on Right 2 2 Mass of H 2 and Cl 2 needed to make 73 g of HCl?

21/12/2021 Example from sheet H 2 + Cl 2 2 HCl React Rel. Formula Molar ant/Pr Mass (Mr) Ratio oduct H 2 2 x 1 = 2 g/mol 1 Cl 2 HCl 35. 5 x 2 = 71 g/mol 1+35. 5 = 36. 5 g/mol Mr x Molar Ratio 2 g/mol 1 71 g/mol 2 2 x 36. 5 g/mol = 73 g/mol What if I wanted to make only 7. 3 g of HCl?

21/12/2021 Example from sheet H 2 + Cl 2 2 HCl Reacta nt/Pro duct Molar Ratio Mr x Molar Ratio H 2 1 2 g/mol 2 x 0. 1 = 0. 2 g Cl 2 HCl 1 2 71 g/mol 2 x 36. 5 g/mol = 73 g/mol 71 x 0. 1 = 7. 1 g 7. 3/73 = 0. 1 What if I wanted to make only 7. 3 g of HCl? Divide the mass you want by (Mr x Molar Ratio) to find a factor to multiply by!

21/12/2021 • Pg 63 - 67 Workbook Qs!
- Slides: 14