Marketing Deck OCTOBER 2020 Marketing Deck OCTOBER 2020

Marketing Deck OCTOBER 2020

Marketing Deck OCTOBER 2020 www. e 25 bio. com E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

E 25 Bio Antigen Testing for Pandemic Control : E 25 Bio is a unique, research-driven biotechnology company focused on the affordable, early field detection of viral pandemics without the need for medical facilities anywhere in the world. Quality Science Rigor Trust Reach Responsiveness www. e 25 bio. com E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

E 25 Bio develops, produces, and sells rapid diagnostic tests for viral fevers such as COVID 19, dengue, Zika, and chikungunya. Our One. Lab technology platform enables data mapping of testing results in real-time through a proprietary integrated phone app. Our products enable consumers to confirm Our primary markets are direct-to-consumer, C O M P A N Y P R E S E N T A T I O N illness in 20 minutes or less through lab-based pharmacy, physician office lab (POL), hospitals, and self-administered testing kits. clinical and reference labs globally. We are: Socially minded Innovative Data-driven Profitable Scalable Efficient Founded by scientists aiming to solve complex public health problems Attractive margins sustainable through testing solutions at affordable rates worldwide www. e 25 bio. com Fast scientific response to emerging pandemics and proactive scientific development using our large library of antibodies Fast-growing revenue opportunities based on easy -to-use products that can be sold anywhere and produce data directly from consumers Merged testing and data mapping to support public health solutions globally Partnered for global production, supply, and distribution E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

Founding Team COMPANY PRESENTATION Dr. Irene Bosch Dr. Bobby Brooke Herrera Dr. Lee Gehrke Bosch’s 25 -year career has focused on epidemic viruses, in particular tropical mosquito-borne viral infections. Completed a Postdoc at Dana Farber Cancer Institute. Herrera developed T-cell based diagnostic tools for Zika, dengue, and Ebola viruses that were implemented in hospitals and labs in West Africa and LATAM. Completed a Postdoc at Harvard Medical School focused on antibody engineering for therapeutic development. Hermann von Helmholtz Professor of Health Sciences and Technology at the MIT Institute of Medical Engineering and Science Co-Founder & CTO Adjunct Professor, Mt. Sinai School of Medicine Ph. D in Molecular Biology, Harvard University www. e 25 bio. com Co-Founder & CSO Visiting Scientist, Harvard T. H. Chan School of Public Health Co-Founder & Scientific Advisor Professor of Immunobiology at Harvard Medical School Member of Harvard and MIT faculty since 1982 Ph. D in Biological Sciences in Public Health (Immunology and Infectious Diseases) , Harvard University E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

COVID-19 DART: Overview • Instrument-free COVID-19 DART cassette-based test • Sample type: respiratory (nasal or nasopharyngeal) • Time to result: 15 minutes • Ability to scale product to 10 M+ units per month • FDA review: pending www. e 25 bio. com E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
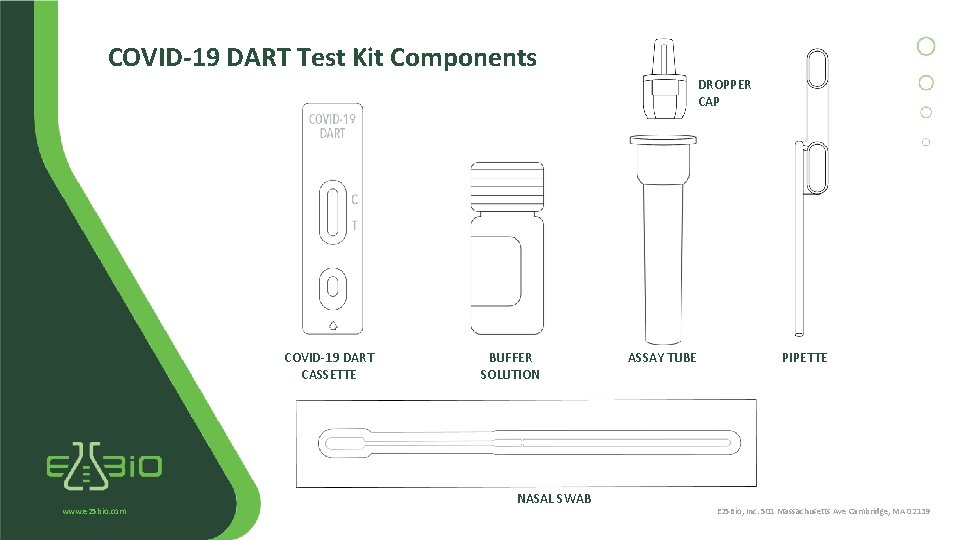
COVID-19 DART Test Kit Components DROPPER CAP COVID-19 DART CASSETTE www. e 25 bio. com BUFFER SOLUTION NASAL SWAB ASSAY TUBE PIPETTE E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
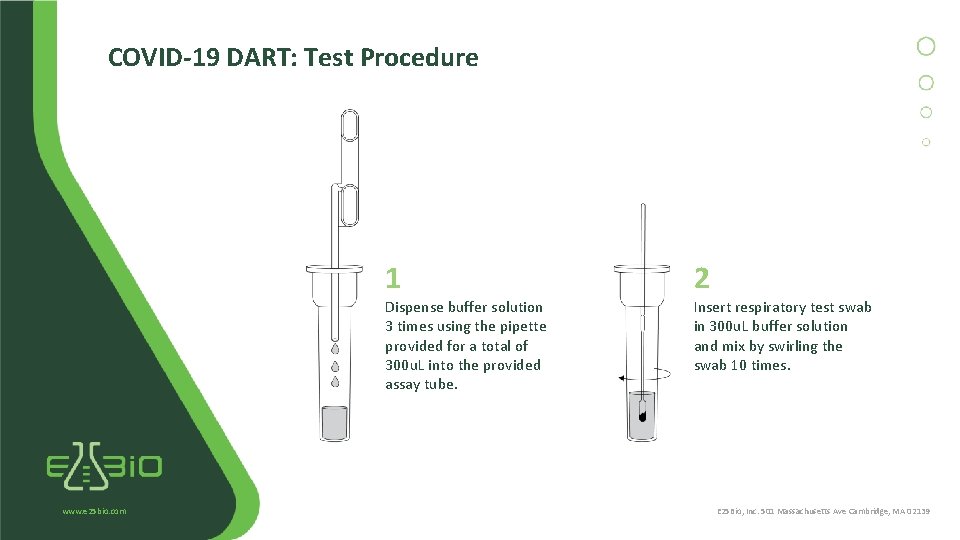
COVID-19 DART: Test Procedure www. e 25 bio. com 1 2 Dispense buffer solution 3 times using the pipette provided for a total of 300 u. L into the provided assay tube. Insert respiratory test swab in 300 u. L buffer solution and mix by swirling the swab 10 times. E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
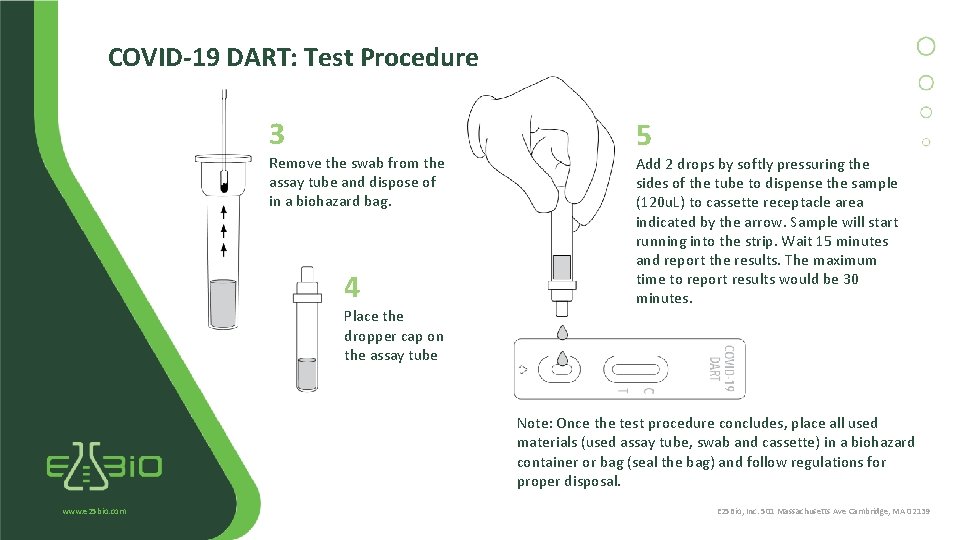
COVID-19 DART: Test Procedure 3 Remove the swab from the assay tube and dispose of in a biohazard bag. 4 Place the dropper cap on the assay tube 5 Add 2 drops by softly pressuring the sides of the tube to dispense the sample (120 u. L) to cassette receptacle area indicated by the arrow. Sample will start running into the strip. Wait 15 minutes and report the results. The maximum time to report results would be 30 minutes. Note: Once the test procedure concludes, place all used materials (used assay tube, swab and cassette) in a biohazard container or bag (seal the bag) and follow regulations for proper disposal. www. e 25 bio. com E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
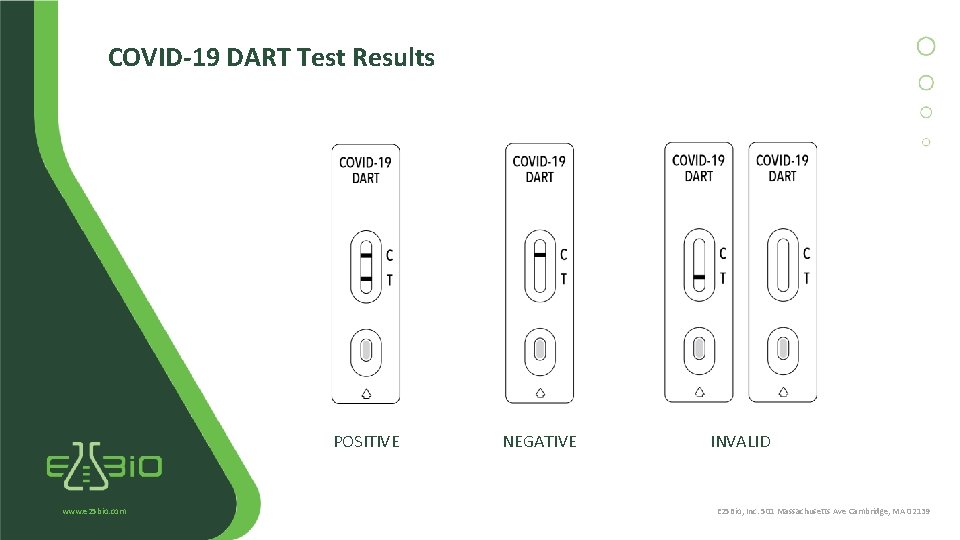
COVID-19 DART Test Results POSITIVE www. e 25 bio. com NEGATIVE INVALID E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
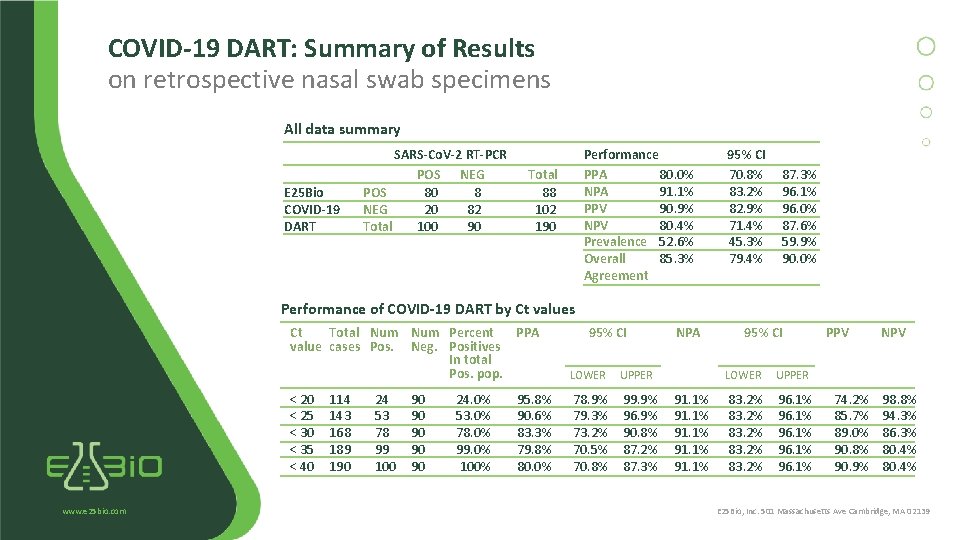
COVID-19 DART: Summary of Results on retrospective nasal swab specimens All data summary E 25 Bio COVID-19 DART SARS-Co. V-2 RT-PCR POS NEG POS 80 8 NEG 20 82 Total 100 90 Performance PPA 80. 0% NPA 91. 1% PPV 90. 9% NPV 80. 4% Prevalence 52. 6% Overall 85. 3% Agreement Total 88 102 190 95% CI 70. 8% 83. 2% 82. 9% 71. 4% 45. 3% 79. 4% 87. 3% 96. 1% 96. 0% 87. 6% 59. 9% 90. 0% Performance of COVID-19 DART by Ct values www. e 25 bio. com Ct Total Num Percent PPA value cases Pos. Neg. Positives In total Pos. pop. LOWER UPPER < 20 < 25 < 30 < 35 < 40 78. 9% 79. 3% 73. 2% 70. 5% 70. 8% 99. 9% 96. 9% 90. 8% 87. 2% 87. 3% 114 143 168 189 190 24 53 78 99 100 90 90 90 24. 0% 53. 0% 78. 0% 99. 0% 100% 95. 8% 90. 6% 83. 3% 79. 8% 80. 0% 95% CI NPA 91. 1% 95% CI LOWER UPPER 83. 2% 96. 1% PPV 74. 2% 85. 7% 89. 0% 90. 8% 90. 9% NPV 98. 8% 94. 3% 86. 3% 80. 4% E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
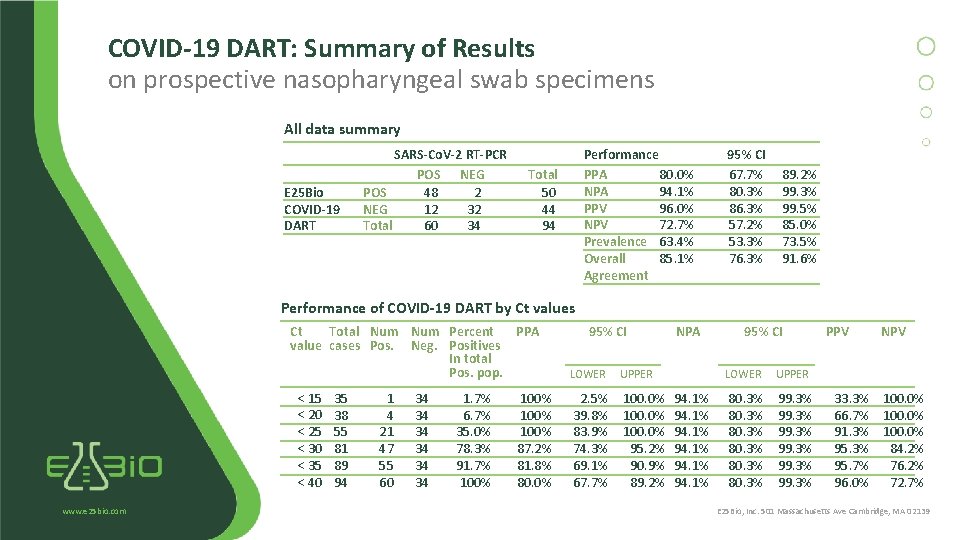
COVID-19 DART: Summary of Results on prospective nasopharyngeal swab specimens All data summary E 25 Bio COVID-19 DART SARS-Co. V-2 RT-PCR POS NEG POS 48 2 NEG 12 32 Total 60 34 Performance PPA 80. 0% NPA 94. 1% PPV 96. 0% NPV 72. 7% Prevalence 63. 4% Overall 85. 1% Agreement Total 50 44 94 95% CI 67. 7% 80. 3% 86. 3% 57. 2% 53. 3% 76. 3% 89. 2% 99. 3% 99. 5% 85. 0% 73. 5% 91. 6% Performance of COVID-19 DART by Ct values Ct Total Num Percent PPA value cases Pos. Neg. Positives In total Pos. pop. < 15 < 20 < 25 < 30 < 35 < 40 www. e 25 bio. com 35 38 55 81 89 94 1 4 21 47 55 60 34 34 34 1. 7% 6. 7% 35. 0% 78. 3% 91. 7% 100% 87. 2% 81. 8% 80. 0% 95% CI LOWER UPPER 2. 5% 39. 8% 83. 9% 74. 3% 69. 1% 67. 7% 100. 0% 95. 2% 90. 9% 89. 2% NPA 94. 1% 95% CI LOWER UPPER 80. 3% 99. 3% PPV NPV 33. 3% 100. 0% 66. 7% 100. 0% 91. 3% 100. 0% 95. 3% 84. 2% 95. 7% 76. 2% 96. 0% 72. 7% E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

COVID-19 DART: Summary of Results on prospective self-administered nasal swab specimens All data summary E 25 Bio COVID-19 DART www. e 25 bio. com SARS-Co. V-2 RT-PCR POS NEG POS 1 NEG 85 Total 86 Total 1 85 86 Performance PPA 98. 8% NPA 0% PPV 100% NPV 0% Prevalence Overall Agreement 95% CI 93. 7% N/A 0% - 99. 97% N/A 4. 20% - E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

COVID-19 DART: Summary of Analytical Performance Pathogens with no cross-reactivity and no interference Pathogens Tested Test Concentration Human coronavirus 229 E 1 X 105 PFU/m. L Human coronavirus OC 43 1 X 105 PFU/m. L Human coronavirus NL 63 9. 87 x 103 PFU/m. L MERS-coronavirus 7930 PFU/m. L Adenovirus 1 X 105 PFU/m. L Human Metapneumovirus (h. MPV) 1 X 105 PFU/m. L Parainfluenza virus 1 1 X 105 PFU/m. L Parainfluenza virus 2 1 X 105 PFU/m. L Parainfluenza virus 3 1 X 105 PFU/m. L Parainfluenza virus 4 1 X 105 PFU/m. L Influenza A H 3 N 2 8. 82 X 104 PFU/m. L Influenza A H 1 N 1 1 X 105 PFU/m. L Influenza B 3. 24 X 104 PFU/m. L Enterovirus 1 X 105 PFU/m. L www. e 25 bio. com Pathogens Tested Test Concentration Respiratory syncytial virus 1 X 105 PFU/m. L Rhinovirus 3. 95 X 105 PFU/m. L Haemophilus influenza 1 X 106 CFU/m. L Streptococcus pneumoniae 1 X 106 CFU/m. L Streptococcus pyogenes 1 X 106 CFU/m. L Candida albicans 1 X 106 CFU/m. L Bordetella pertussis 1 X 106 CFU/m. L Mycoplasma pneumoniae 1 X 106 CFU/m. L Chlamydia pneumoniae 1 X 106 CFU/m. L Legionella pneumophila 1 X 106 CFU/m. L Mycobacterium tuberculosis 1 X 106 CFU/m. L Pneumocystis jirovecii (PJP) 1 X 106 CFU/m. L Pneudomonas aeruginosa 1 X 106 CFU/m. L Staphylococcus epidemidis 1 X 106 CFU/m. L Streptococcus salivarius 1 X 106 CFU/m. L Pooled human nasal wash 15%v/v E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

Quality E 25 Bio Science : Antigen Testing for Pandemic Control www. e 25 bio. com Rigor Trust Reach Responsiveness E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139

THANKS! info@e 25 bio. com www. E 25 BIO. com | info@E 25 BIO. com E 25 Bio, Inc. 501 Massachusetts Ave Cambridge, MA 02139
- Slides: 16