Mark Pap TECHNOLOGY Focal Points Bio Sci Con

Mark. Pap® TECHNOLOGY Focal Points Bio. Sci. Con, Inc. 12/3/2020 1

Focal Points Ø Technology Overview Ø Company Overview Ø Mark. Pap® Products Ø Pap Test Market 12/3/2020 2
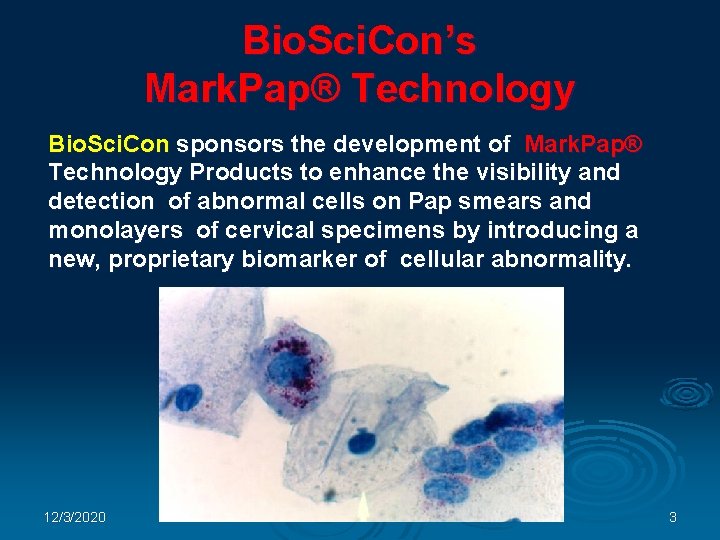
Bio. Sci. Con’s Mark. Pap® Technology Bio. Sci. Con sponsors the development of Mark. Pap® Technology Products to enhance the visibility and detection of abnormal cells on Pap smears and monolayers of cervical specimens by introducing a new, proprietary biomarker of cellular abnormality. : 12/3/2020 3
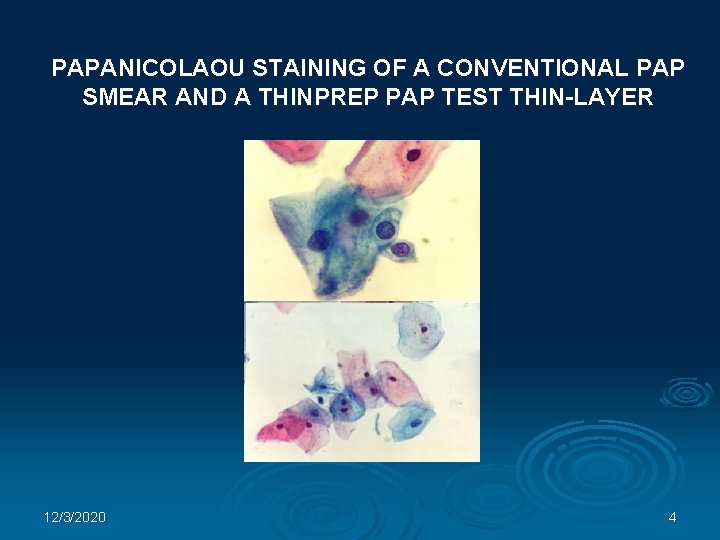
PAPANICOLAOU STAINING OF A CONVENTIONAL PAP SMEAR AND A THINPREP PAP TEST THIN-LAYER 12/3/2020 4

Why Enhancing Pap Test? Ø 50 M Pap tests per year in the U. S Ø 15% false negatives Ø The use of biomarker may reduce false negative readings over 50% Ø 13, 000 new cervical cancers per year in the US Ø If Mark. Pap® Test were available, these numbers would have been different: 12, 000 new cases, 7% false negatives 12/3/2020 5
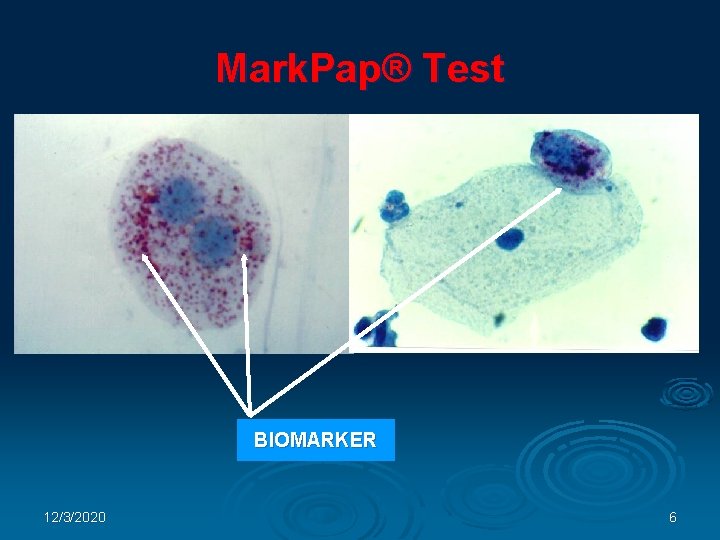
Mark. Pap® Test BIOMARKER 12/3/2020 6
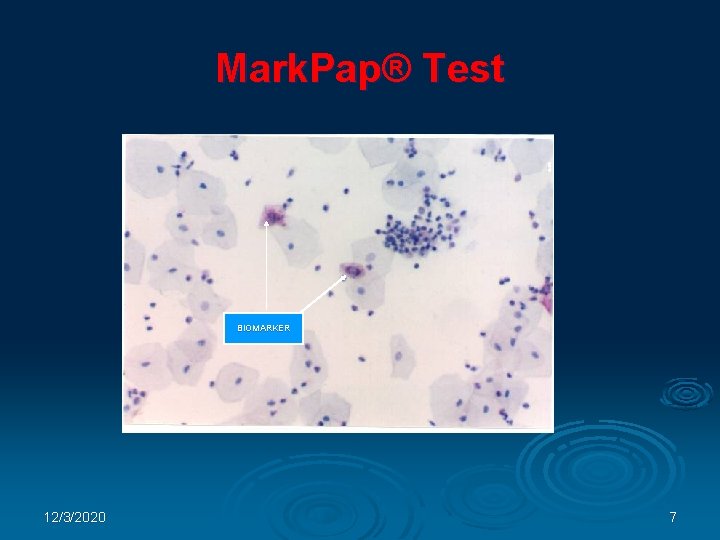
Mark. Pap® Test BIOMARKER 12/3/2020 7

Mark. Pap® Product Family Mark. Pap® Test Phase 2/3 Clinical Trial Service available for research purposes only Mark. Pap® Research Kit Beta-testing Commercially available for research purposes only Mark. Pap® Solution Commercially available for research purposes only Mark. Pap® Processor Under development Mark. Pap® Workstation Under development 12/3/2020 8

Bio. Sci. Con’s Mission Statement Our primary mission is to bring the Mark. Pap® test to the public as soon as possible and save women lives. The Mark. Pap® family of products will be manufactured and sold through partnerships with established medical device providers. 12/3/2020 9

Company Information Biomedical Science Consulting Company , Bio. Sci. Con, Inc. is a woman-owned small business with Bio. Sci. Con Center located in Rockville, MD with two laboratory locations (Clarksburg and Bethesda, MD), and seven clinical sites (universities, women ‘s health centers and OB/GYN doctors offices) throughout the state of Maryland The District of Columbia. Bio. Sci. Con was incorporated in 1996. The Mark. Pap® product development is supported in part by SBIR, NIH Phase-1 and Phase-2 grants and the Montgomery County TGP Grant/Loan. 12/3/2020 10

Mark. Pap® Products Commercially Available Now Ø Mark. Pap® TEST: Method and service. Ø Mark. Pap® RESEARCH KIT : A set of reagents, instructions and control designed to facilitate the use of the test. Ø Kit Accessories: Cytopreservative solution for optimal enzyme and cellular preservation in extended period of time. 12/3/2020 11

Mark. Pap® Research Kit 12/3/2020 12

Mark. Pap® Solution 12/3/2020 13

Product Features, Advantages and Benefits Visual Biomarker 1. Reduces false negative reading to less than 4%. Ø 2. Increases accuracy (sensitivity/specificity) above 90%. Ø 3. Doubles “true” positives (cytology standard). Ø 4. Reduces screening time to 3 min, re-screen time to 1 min per slide. Ø Ø Red, granular deposit inside abnormal cervical squamous cells. 12/3/2020 14
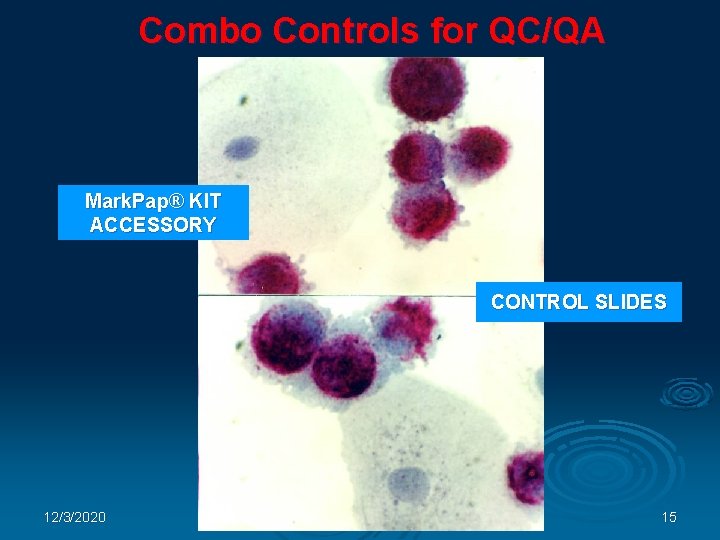
Combo Controls for QC/QA Mark. Pap® KIT ACCESSORY CONTROL SLIDES 12/3/2020 15

Product Features, Advantages and Benefits Control Slides Ø Ø Mark. Pap® Test Kit contains COMBO control slides. Ø Ø Ø 12/3/2020 COMBO control slides serve as QC/QA Specimen processing easily controlled Inter-laboratory consistency Reduced liability 16

Product Features, Advantages and Benefits Low Cost Ø Inexpensive, simple test. Ø Total cost (material and screening) lower then Pap. Much less expensive then Thin. Prep Pap test. 12/3/2020 17

Product Features, Advantages and Benefits Same infrastructure as Pap Test Ø Immediately applicable. Based on the existing Pap test infrastructure. Ø Same lab space, equipment and personnel (cytoprep, cytotech, pathologist). Simple and customer friendly. Few hours training needed for Mark. Pap® test. Ø This is an “enchanced Pap-test” and all currently used classifications (TBS, Bethesda 2001) apply. 12/3/2020 18

Success Ingredients Intellectual property Ø Ø Ø Parent patent “CAP-PAP TEST” (November 2000) Patent pending “Cap-Pap Test Kit” (January 2003) PCT: “MPSystem” (January 2004) Trademark: “Mark. Pap®” (April 2004) Copyright: “Mark. Pap® System” (December 2003) Know-how: Clinical Laboratory Trials’ Results 12/3/2020 19
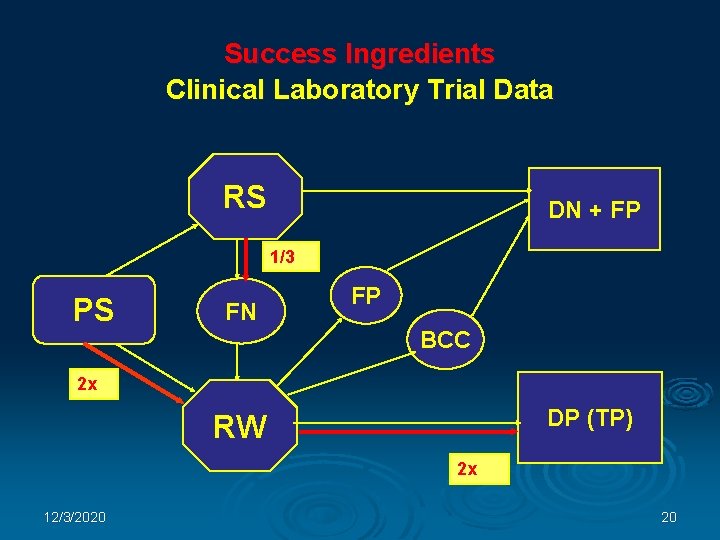
Success Ingredients Clinical Laboratory Trial Data RS DN + FP 1/3 PS FN FP BCC 2 x DP (TP) RW 2 x 12/3/2020 20

Cervical cancer is a preventable disease IF detected on time Mark. Pap® test improvements Ø < 4% false negatives Ø Doubles “true” positives Ø Increases accuracy >90% Ø Low cost—simple Ø Increases productivity Ø Lowers liability (QC/QA) 12/3/2020 21

Pap Test Market Trends Ø Increase outreach among under-served minorities in the United States. Ø Global expansion into developing countries. Ø Development of simple, low cost methods to service large populations. Ø Industrial standards and Quality Assurance. 12/3/2020 22

Customers Ø Cytopathology laboratories (~3, 400) Ø Research laboratories Ø HMO Ø Women’s Health Centers Ø Doctor’s offices Ø Do. D Ø WHO 12/3/2020 23

Market Opportunity U. S. A Only WORLD Ø 50 M tests annually in the U. S. Ø Women at risk: Ø 3. 6 M (7%) cytologically abnormal tests Ø Ø 15% - 20% false negatives Ø 13, 000 newly diagnosed cervical cancer cases Half a million women develop cervical cancer annually of whom more then 50% will die. About 80% of all these women are from the developing world. Kills more women then childbirth. Ø 4, 500 cervical cancer deaths Ø 40 -50% HPV infected 12/3/2020 Potential markets: Ø EU: 42 M Ø World: 110 M Ø At risk: 2 billion 24

Product Pipeline Ø Mark. Pap® Test: Clinical trials phase II/III. Investigational Plan for IDE study in development. Ø Mark. Pap® Kit, Mark. Pap® Solution, COMBO Controls Ø All products are available in the US for research only. Not for diagnostic use. They are available for global marketing. 12/3/2020 25

Contact Information Bio. Sci. Con, Inc. 14905 Forest Landing Circle Rockville, MD 20850 -2934 P/F: (301) 610 -9130 www. bioscicon. com 12/3/2020 26
- Slides: 26