Manyelectron effects Experiments showing Coulomb Blockade in Molecular
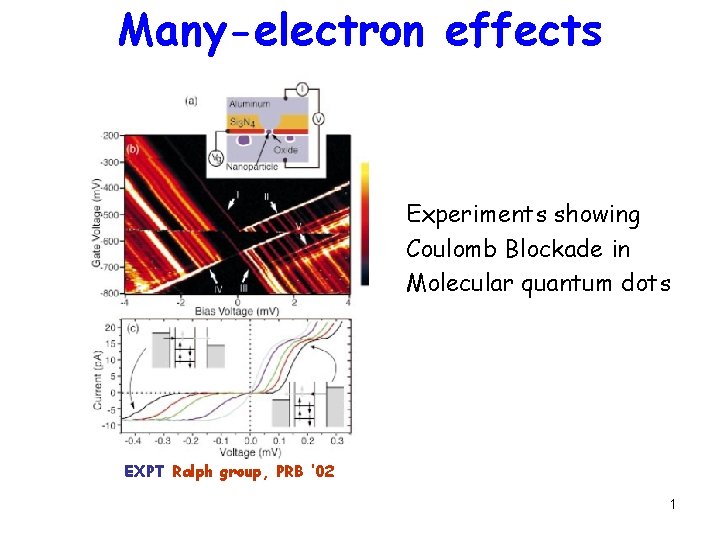
Many-electron effects Experiments showing Coulomb Blockade in Molecular quantum dots EXPT Ralph group, PRB ‘ 02 1

So far we’ve seen Ø Transport is determined by a few levels near EF Ø These levels are broadened by the contacts and shifted by the local potential Ø The levels come from solving Schrodinger Equation for the channel. For individual atoms, this is done selfconsistently with an approximate el-el interaction Ø Atomic levels combine to form molecular levels stabilized by long-ranged electrostatic forces (ionic bonds) or short-ranged quantum forces (covalent bonds) 2
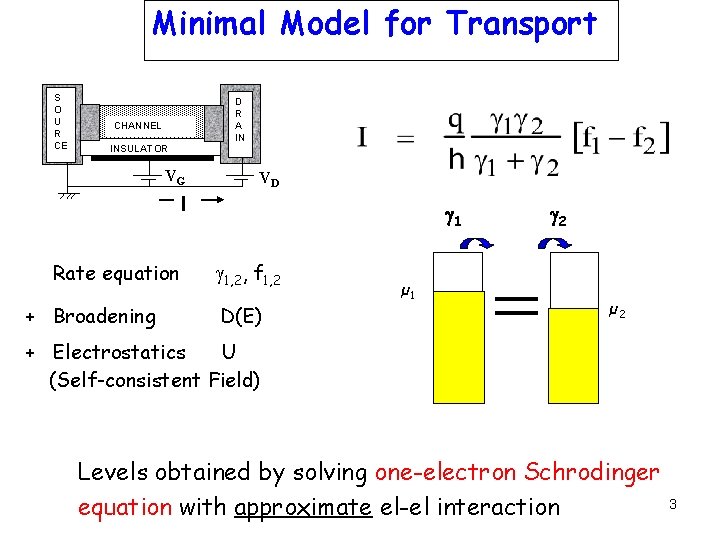
Minimal Model for Transport S O U R CE D R A IN CHANNEL INSULATOR VG VD I Rate equation + Broadening g 1, 2, f 1, 2 D(E) µ 1 g 2 µ 2 + Electrostatics U (Self-consistent Field) Levels obtained by solving one-electron Schrodinger equation with approximate el-el interaction 3

Can we do better than that? Suppose we have a computer big enough to solve the many-electron Schrodinger equation. How would we use those results? 4
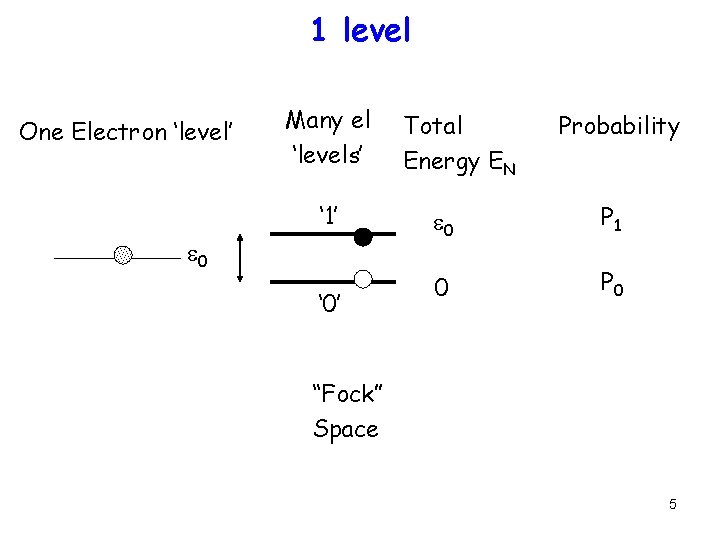
1 level One Electron ‘level’ Many el ‘levels’ ‘ 1’ e 0 ‘ 0’ Total Energy EN Probability e 0 P 1 0 P 0 “Fock” Space 5
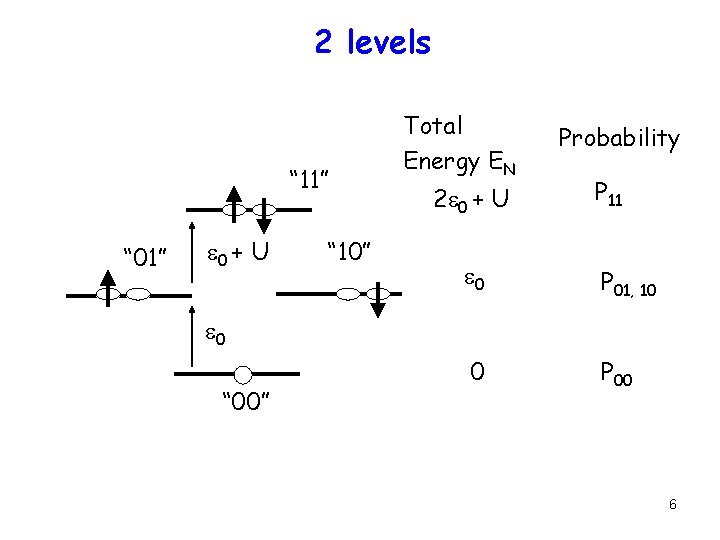
2 levels “ 11” “ 01” e 0 + U “ 10” Total Energy EN 2 e 0 + U Probability P 11 e 0 P 01, 10 0 P 00 e 0 “ 00” 6
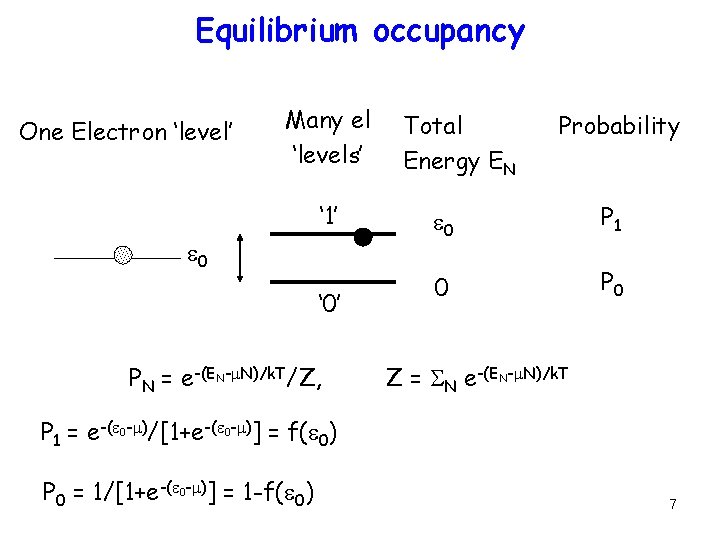
Equilibrium occupancy One Electron ‘level’ Many el ‘levels’ ‘ 1’ e 0 ‘ 0’ PN = e-(EN-m. N)/k. T/Z, Total Energy EN Probability e 0 P 1 0 P 0 Z = SN e-(EN-m. N)/k. T P 1 = e-(e 0 -m)/[1+e-(e 0 -m)] = f(e 0) P 0 = 1/[1+e-(e 0 -m)] = 1 -f(e 0) 7
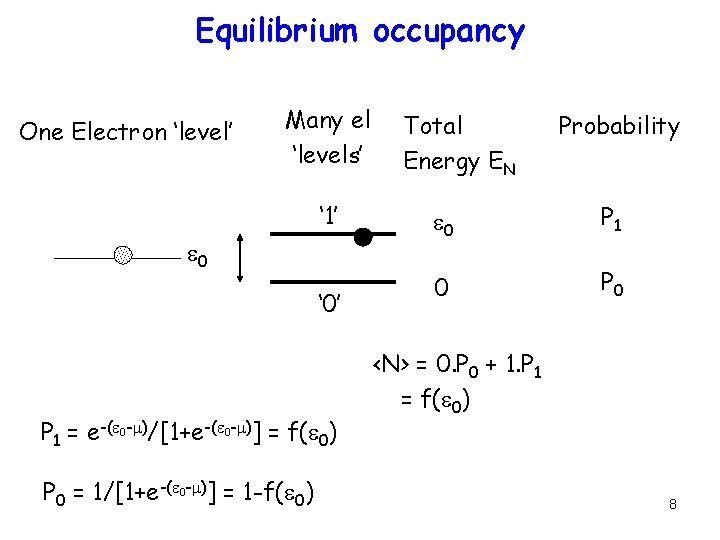
Equilibrium occupancy One Electron ‘level’ Many el ‘levels’ ‘ 1’ e 0 ‘ 0’ P 1 = e-(e 0 -m)/[1+e-(e 0 -m)] = f(e 0) P 0 = 1/[1+e-(e 0 -m)] = 1 -f(e 0) Total Energy EN Probability e 0 P 1 0 P 0 <N> = 0. P 0 + 1. P 1 = f(e 0) 8
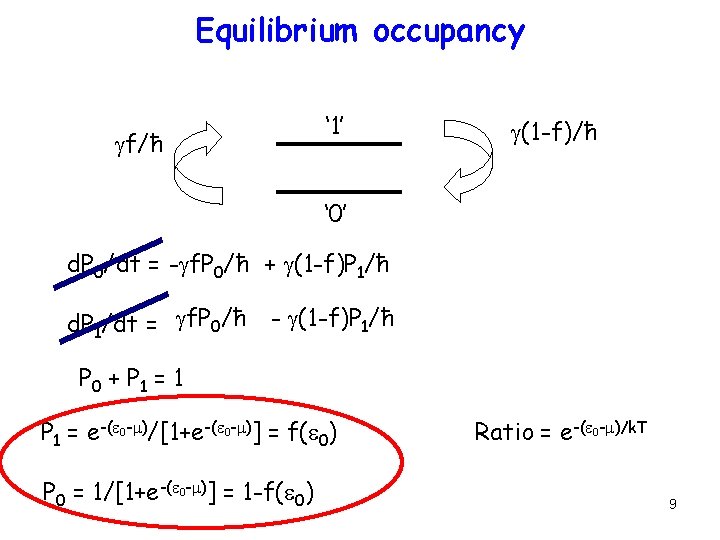
Equilibrium occupancy ‘ 1’ gf/ħ g(1 -f)/ħ ‘ 0’ d. P 0/dt = -gf. P 0/ħ + g(1 -f)P 1/ħ d. P 1/dt = gf. P 0/ħ - g(1 -f)P 1/ħ P 0 + P 1 = 1 P 1 = e-(e 0 -m)/[1+e-(e 0 -m)] = f(e 0) P 0 = 1/[1+e-(e 0 -m)] = 1 -f(e 0) Ratio = e-(e 0 -m)/k. T 9
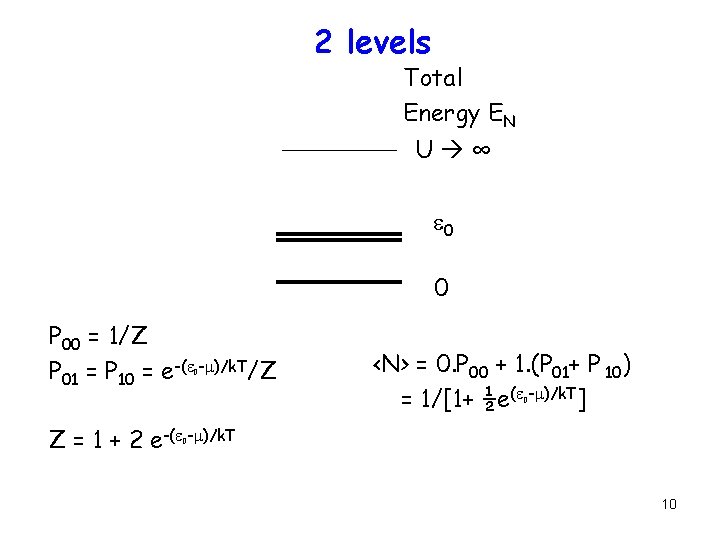
2 levels Total Energy EN U ∞ e 0 0 P 00 = 1/Z P 01 = P 10 = e-(e -m)/k. T/Z 0 <N> = 0. P 00 + 1. (P 01+ P 10) = 1/[1+ ½e(e -m)/k. T] 0 Z = 1 + 2 e-(e -m)/k. T 0 10

Now what happens under Nonequilibrium? Contacts create transitions between these many-electron levels 11
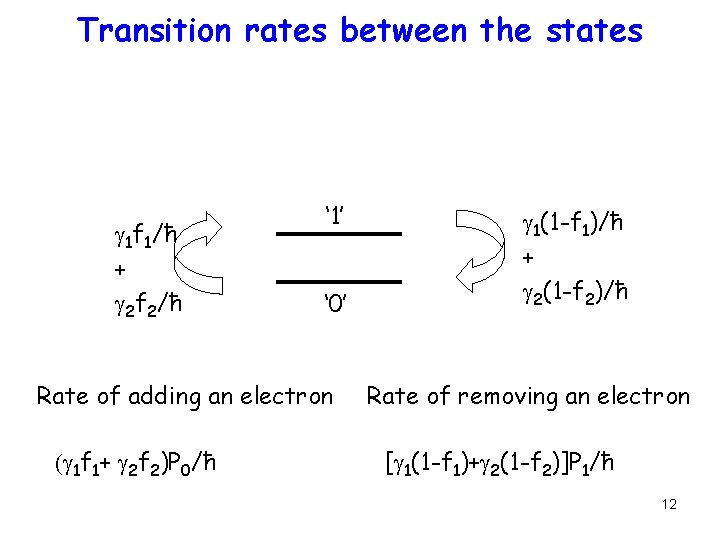
Transition rates between the states g 1 f 1/ħ + g 2 f 2/ħ ‘ 1’ ‘ 0’ Rate of adding an electron (g 1 f 1+ g 2 f 2)P 0/ħ g 1(1 -f 1)/ħ + g 2(1 -f 2)/ħ Rate of removing an electron [g 1(1 -f 1)+g 2(1 -f 2)]P 1/ħ 12

Rate equations in many-el space g 1 f 1/ħ + g 2 f 2/ħ ‘ 1’ ‘ 0’ g 1(1 -f 1)/ħ + g 2(1 -f 2)/ħ d. P 0/dt = -(g 1 f 1+ g 2 f 2)P 0/ħ + [g 1(1 -f 1) + g 2(1 -f 2)]P 1/ħ d. P 1/dt = (g 1 f 1+ g 2 f 2)P 0/ħ - [g 1(1 -f 1) + g 2(1 -f 2)]P 1/ħ P 0 + P 1 = 1 13

Solution P 1 = g 1 f 1+ g 2 f 2 g 1+ g 2 [g 1(1 -f 1)+ g 2(1 -f 2)] P 0 = g 1+ g 2 <N> = P 0. 0 + P 1. 1 = I 1 = q g 1 f 1 P 0 – g 1(1 -f 1)P 1 ħ g 1 f 1+ g 2 f 2 g 1+ g 2 g g (f -f ) = q 1 2 ħ (g 1 + g 2) 14
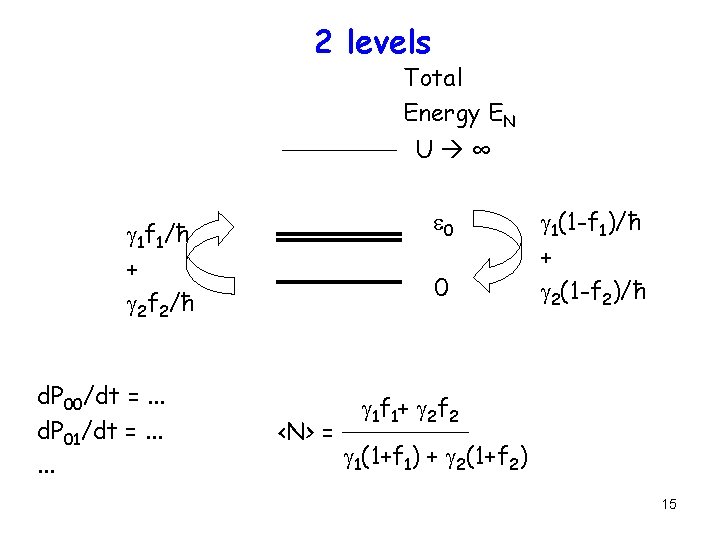
2 levels Total Energy EN U ∞ e 0 g 1 f 1/ħ + g 2 f 2/ħ d. P 00/dt =. . . d. P 01/dt =. . . 0 <N> = g 1(1 -f 1)/ħ + g 2(1 -f 2)/ħ g 1 f 1+ g 2 f 2 g 1(1+f 1) + g 2(1+f 2) 15

What did we achieve? We can now write down rate equations in one electron space with approximate el-el interactions (SCF) OR rate equations directly in many-electron configuration space with exact el-el interactions (a) What is the relation between the two descriptions? (b) When do we use which? 16

Relation between many-el and one-el levels Filled levels represent Ionization energy: Energy to take electron from the single N electron Ground state to one of the multiple (N-1) electron states IP(n) = EN(0) – EN-1(n) 17
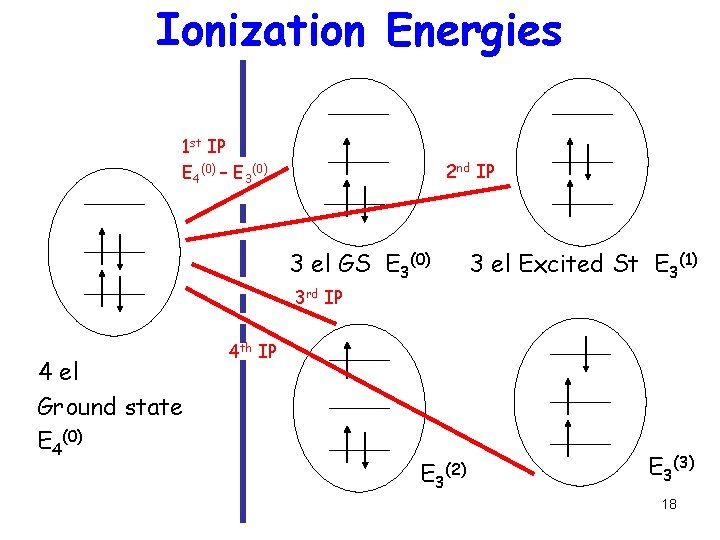
Ionization Energies 1 st IP E 4(0) – E 3(0) 2 nd IP 3 el GS E 3(0) 3 el Excited St E 3(1) 3 rd IP 4 el Ground state E 4(0) 4 th IP E 3 (2) E 3(3) 18
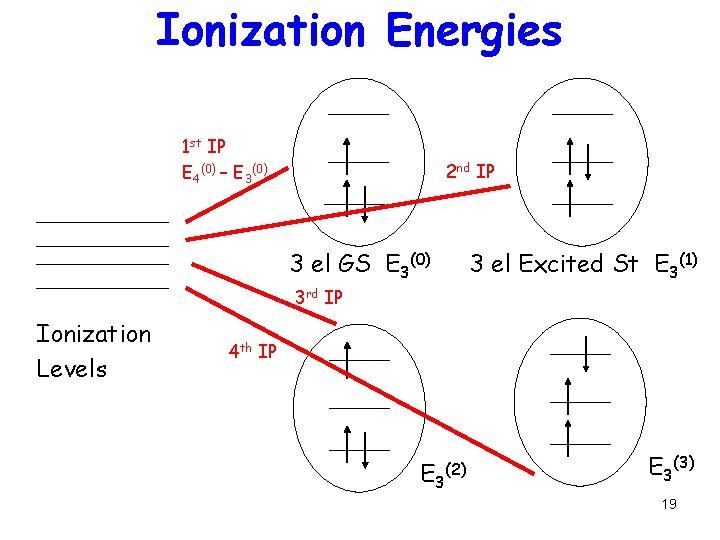
Ionization Energies 1 st IP E 4(0) – E 3(0) 2 nd IP 3 el GS E 3(0) 3 el Excited St E 3(1) 3 rd IP Ionization Levels 4 th IP E 3 (2) E 3(3) 19

What do empty energies mean? Empty levels represent Electron Affinity: Energy to add an electron to N electron ground state to create one of the multiple (N+1) electron states EA(n) = EN+1(n) – EN(0) 20

When do we need many-el viewpoint? When charging is important U 0 >> (g 1 + g 2), k. BT In the opposite limit, got by with U = UL + U 0 DN Many-el generalization, U 0 Hartree term But this doesn’t suffice if U 0 large 21

What does charging do? Uee(N) = U 0 N(N-1)/2 (U 0 times # pairs) IP(n) = EN(0) – EN-1(n) = e. HOMO + U 0(N-1) EA(n) = EN+1(n) – EN(0) = e. LUMO + U 0 N (extra charge due to extra el repulsion) Charging opens gap U 0 between filled and empty levels But here we add charging contribution differently to different levels 22
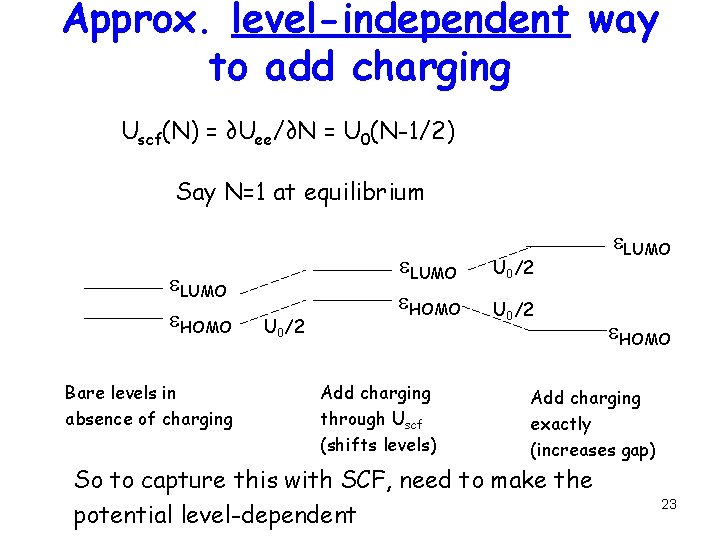
Approx. level-independent way to add charging Uscf(N) = ∂Uee/∂N = U 0(N-1/2) Say N=1 at equilibrium e. LUMO e. HOMO Bare levels in absence of charging U 0/2 e. LUMO e. HOMO Add charging through Uscf (shifts levels) U 0/2 e. LUMO e. HOMO Add charging exactly (increases gap) So to capture this with SCF, need to make the potential level-dependent 23
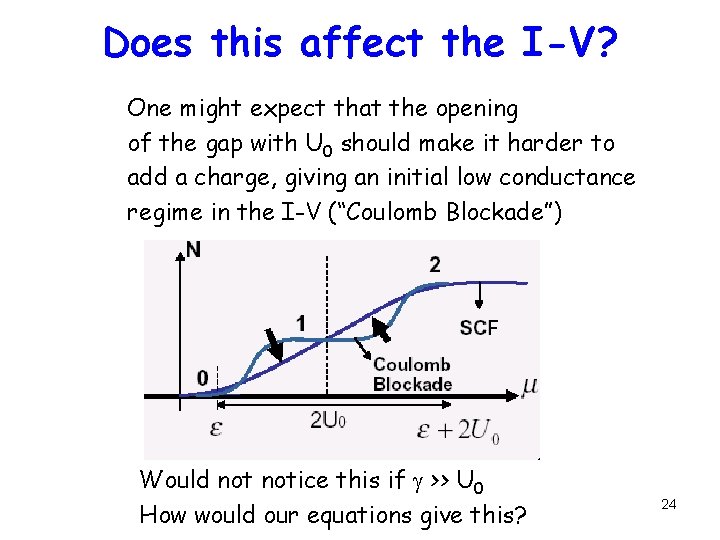
Does this affect the I-V? One might expect that the opening of the gap with U 0 should make it harder to add a charge, giving an initial low conductance regime in the I-V (“Coulomb Blockade”) Would notice this if g >> U 0 How would our equations give this? 24

Configuration space for 2 spins Write down rate equations between all states The resulting I-V should give the correct answer (Homework) 25
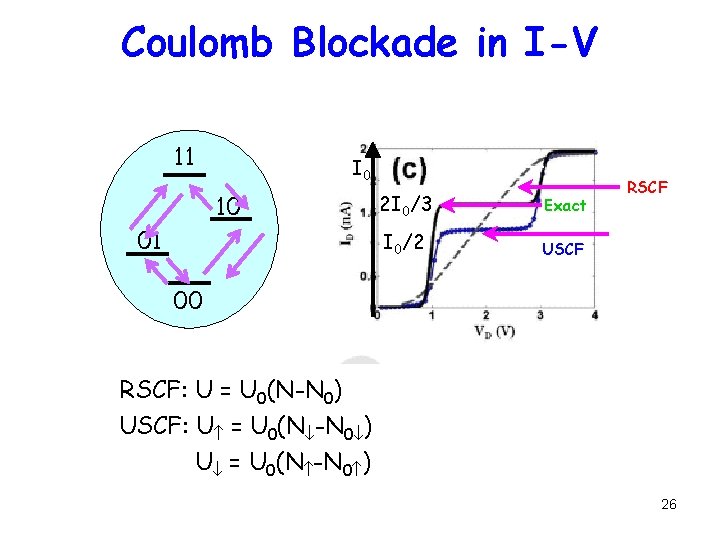
Coulomb Blockade in I-V 11 I 0 10 01 2 I 0/3 Exact I 0/2 USCF RSCF 00 RSCF: U = U 0(N-N 0) USCF: U = U 0(N -N 0 ) 26
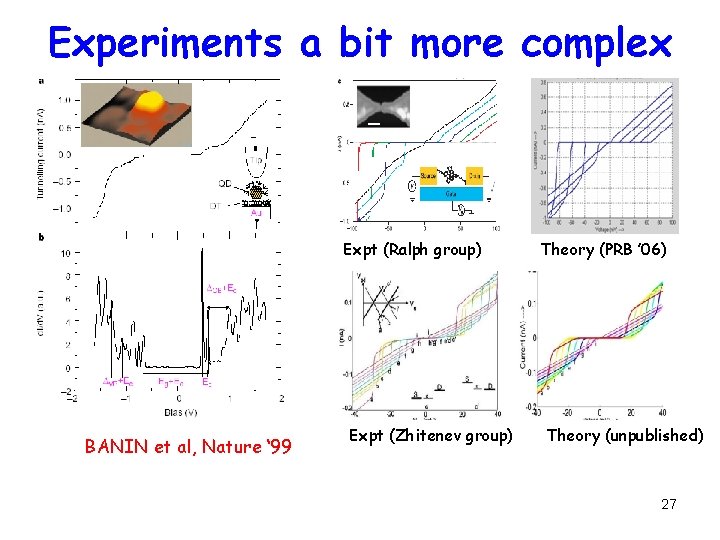
Experiments a bit more complex Expt (Ralph group) BANIN et al, Nature ‘ 99 Expt (Zhitenev group) Theory (PRB ’ 06) Theory (unpublished) 27
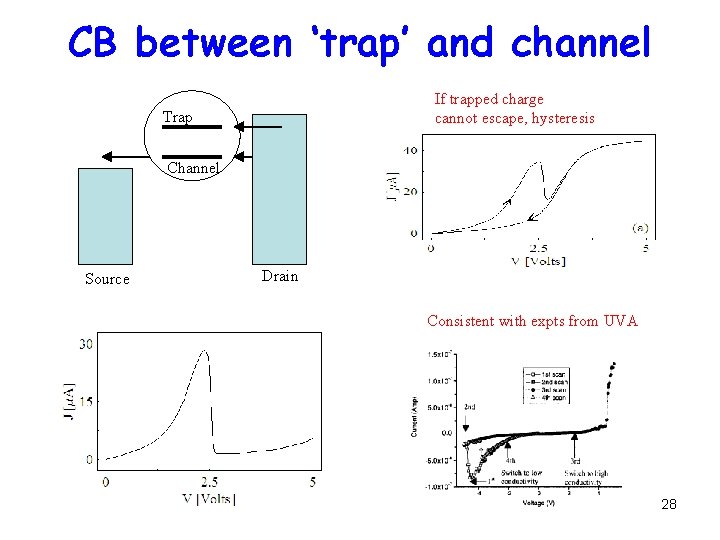
CB between ‘trap’ and channel If trapped charge cannot escape, hysteresis Trap Channel Source Drain Consistent with expts from UVA 28
When Electrons Cooperate (“Correlation”) Molecular Coulomb Blockade (Zhitenev) Magnets, superconductors ( A + B) Molecular Kondo (Ralph/Mc. Euen) ( A B - A B) 29

Summary For small charging, usual method of writing rate equations in real space work. Use Schrodinger equation with approximate SCF potential U and broadening to get currents. For large charging, SCF is not enough. Would need to work with rate equations in full many-electron configuration space with exact el-el interactions included. This would give Coulomb Blockade. Trouble with the latter (a) Size of conf. space increases fast (2 N for N orbitals) (b) Don’t know how to broaden the levels or the transitions 30

What next? Let’s go back now to the SCF approach to transport, keeping in mind that the approach need to be modified for large charging and Coulomb Blockade. We now understand what the SCF ‘levels’ represent, ie, ionization energies/electron affinities that represent transitions between many-electron levels. We know how to calculate the SCF levels for atoms and molecules, the latter forming covalent bonds on occasion. We’ll next see how these bonds can extend in a solid to 31 form bands
- Slides: 31