Manajemen Nutrisi Pada Penyakit Ginjal Sumber Kuliah Gizi

Manajemen Nutrisi Pada Penyakit Ginjal Sumber : Kuliah Gizi Nutritional Management on Kidney Diseases by DR. dr. Haerani Rasyid, M. Kes, Sp. PD , K-GH, Sp. GK, Nutrition Department /Nephrology Div School of Medicine Hasanuddin University Makassar 2011
Tujuan Umum • Mahasiswa mampu menjelaskan manajemen nutris pada penyakit ginjal Tujuan khusus • Mahasiswa mampu menjelaskan manajemen nutrisi pada penyakit Sindrom Nefrotik, GGA, GGK, Batu Ginjal
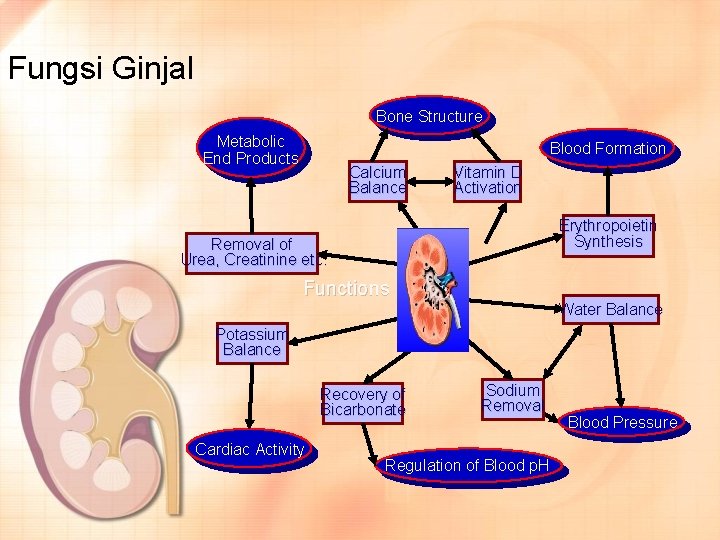
Fungsi Ginjal Bone Structure Metabolic End Products Blood Formation Calcium Balance Vitamin D Activation Erythropoietin Synthesis Removal of Urea, Creatinine etc. Functions Water Balance Potassium Balance Recovery of Bicarbonate Cardiac Activity Sodium Removal Regulation of Blood p. H Blood Pressure

Peran nutrisi pada penyakit ginjal • To prevent or reverse associated malnourished states • To minimize the adverse effect of substances that are inadequately excreted • Favorably affect the progression and outcome of kidney disease
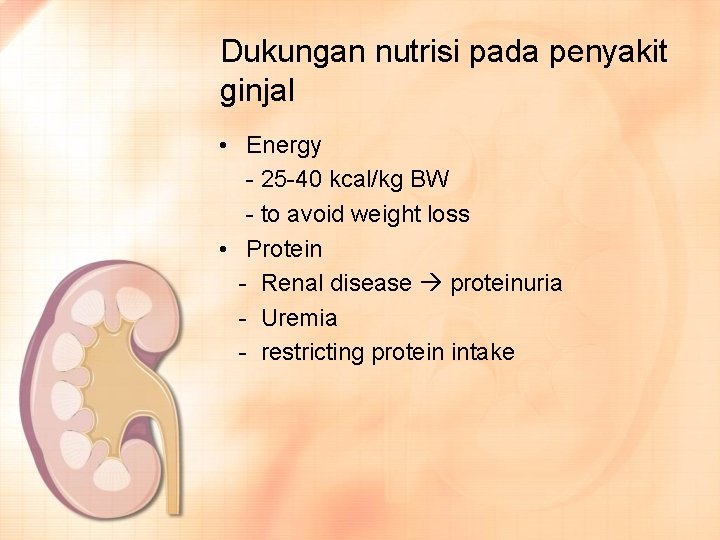
Dukungan nutrisi pada penyakit ginjal • Energy - 25 -40 kcal/kg BW - to avoid weight loss • Protein - Renal disease proteinuria - Uremia - restricting protein intake
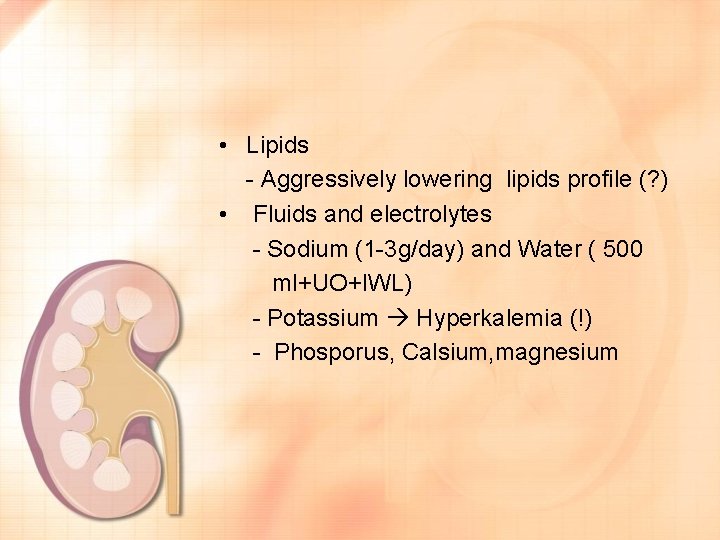
• Lipids - Aggressively lowering lipids profile (? ) • Fluids and electrolytes - Sodium (1 -3 g/day) and Water ( 500 ml+UO+IWL) - Potassium Hyperkalemia (!) - Phosporus, Calsium, magnesium
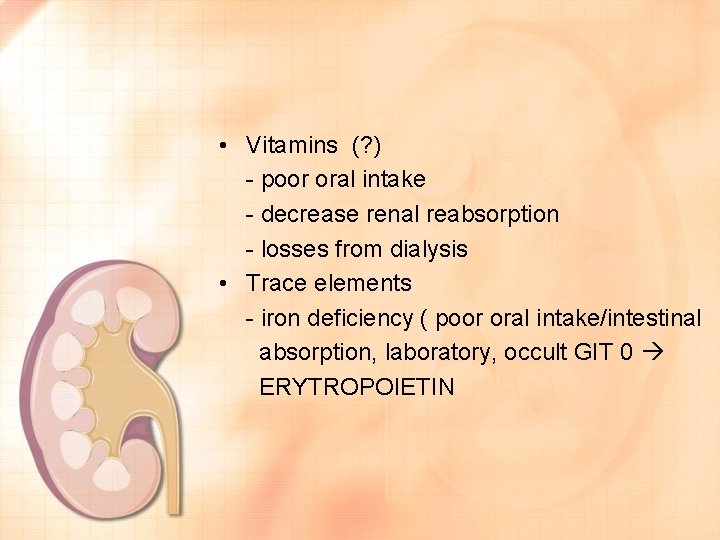
• Vitamins (? ) - poor oral intake - decrease renal reabsorption - losses from dialysis • Trace elements - iron deficiency ( poor oral intake/intestinal absorption, laboratory, occult GIT 0 ERYTROPOIETIN
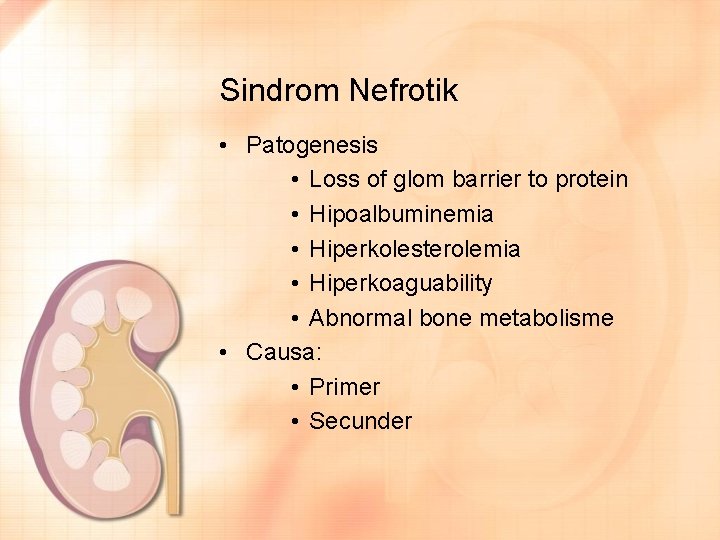
Sindrom Nefrotik • Patogenesis • Loss of glom barrier to protein • Hipoalbuminemia • Hiperkolesterolemia • Hiperkoaguability • Abnormal bone metabolisme • Causa: • Primer • Secunder
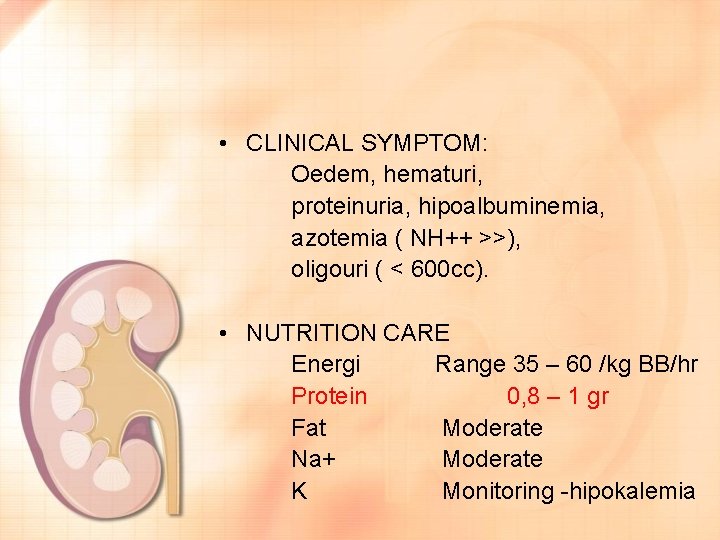
• CLINICAL SYMPTOM: Oedem, hematuri, proteinuria, hipoalbuminemia, azotemia ( NH++ >>), oligouri ( < 600 cc). • NUTRITION CARE Energi Range 35 – 60 /kg BB/hr Protein 0, 8 – 1 gr Fat Moderate Na+ Moderate K Monitoring -hipokalemia

Goal 1. MAINTAIN OPTIMAL NUTRITION 2. MAINTAIN NUTRITIONAL STORES 3. MINIMIZE DISEASE METABOLISM 4. PREVENT PROGRESSIVITAS OF DISEASE 5. SLOW DIALYSIS OCCURANCE
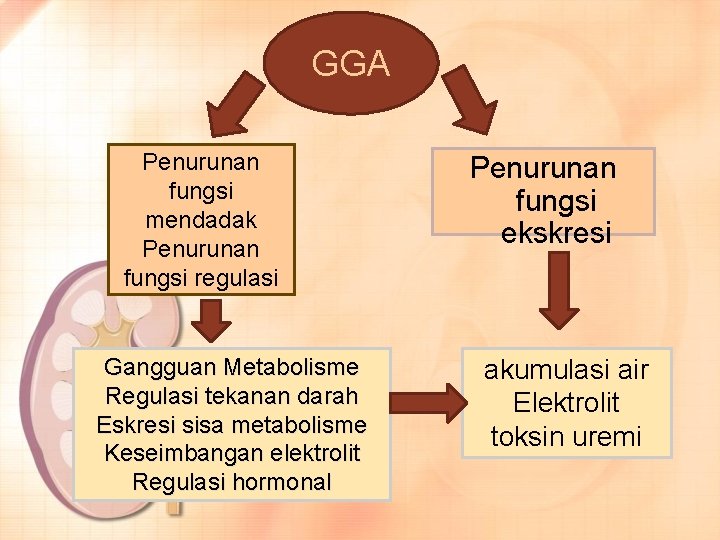
GGA Penurunan fungsi mendadak Penurunan fungsi regulasi Gangguan Metabolisme Regulasi tekanan darah Eskresi sisa metabolisme Keseimbangan elektrolit Regulasi hormonal Penurunan fungsi ekskresi akumulasi air Elektrolit toksin uremi
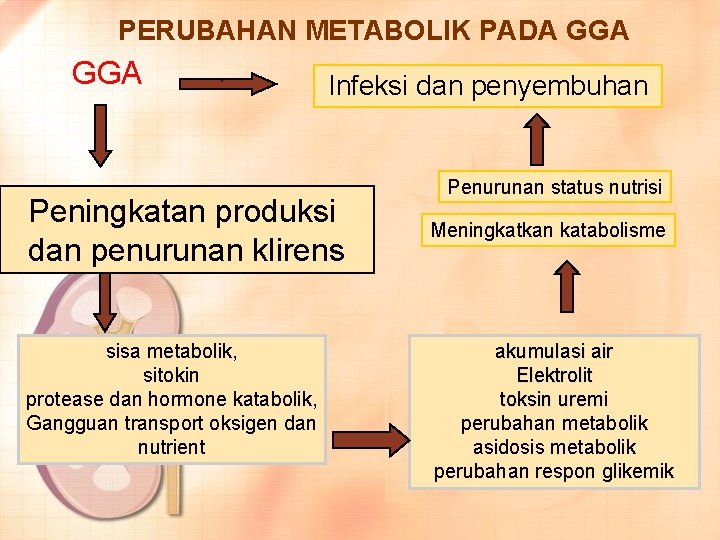
PERUBAHAN METABOLIK PADA GGA ` Penurunan ekskresi Infeksi dan fungsi penyembuhan penurunan fungsi mendadak Peningkatan produksi Penurunan fungsi regulasi dan penurunan klirens sisa metabolik, sitokin protease dan hormone katabolik, Gangguan transport oksigen dan nutrient Penurunan status nutrisi Meningkatkan katabolisme akumulasi air Elektrolit toksin uremi perubahan metabolik asidosis metabolik perubahan respon glikemik
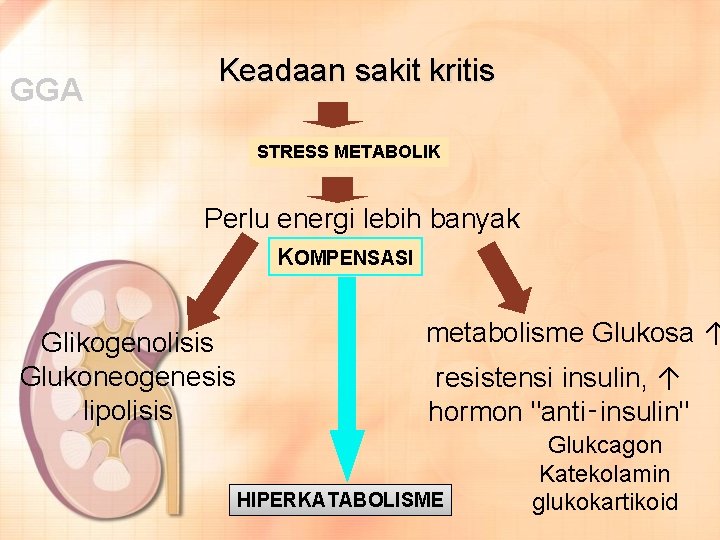
GGA Keadaan sakit kritis STRESS METABOLIK Perlu energi lebih banyak KOMPENSASI Glikogenolisis Glukoneogenesis lipolisis metabolisme Glukosa ↑ resistensi insulin, ↑ hormon "anti‑insulin" HIPERKATABOLISME Glukcagon Katekolamin glukokartikoid
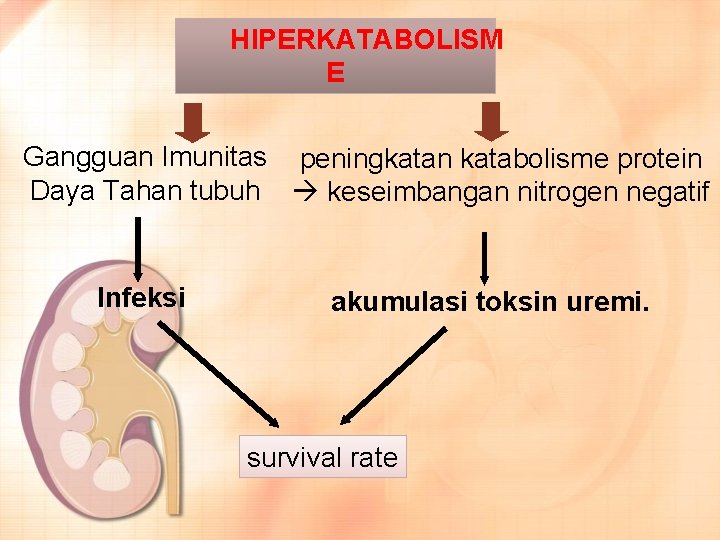
HIPERKATABOLISM E Gangguan Imunitas peningkatan katabolisme protein Daya Tahan tubuh keseimbangan nitrogen negatif Infeksi akumulasi toksin uremi. survival rate

Metabolisme energi dan kebutuhan energi • Metabolisme air, elektrolit dan asam basa, perubahan “milieu interieur” protein dan asam amino, karbohidrat serta lipid. • reaksi pro-inflamasi dan sistim antioksidasi. • merupakan komplikasi dari sepsis, trauma atau kegagalan multi organ. • Perubahan-perubahan metabolisme oleh penyakit yang mendasarinya dan/atau disertai komplikasi • adanya disfungsi organ lain • ditentukan oleh tipe dan intensitas terapi pengganti ginjal Chan, Curr Opin Clin Nutr Metab Care 2004: 207
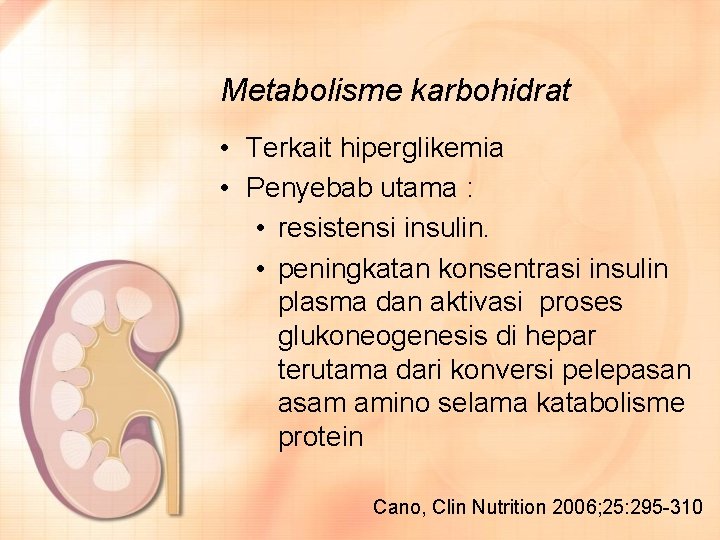
Metabolisme karbohidrat • Terkait hiperglikemia • Penyebab utama : • resistensi insulin. • peningkatan konsentrasi insulin plasma dan aktivasi proses glukoneogenesis di hepar terutama dari konversi pelepasan asam amino selama katabolisme protein Cano, Clin Nutrition 2006; 25: 295 -310
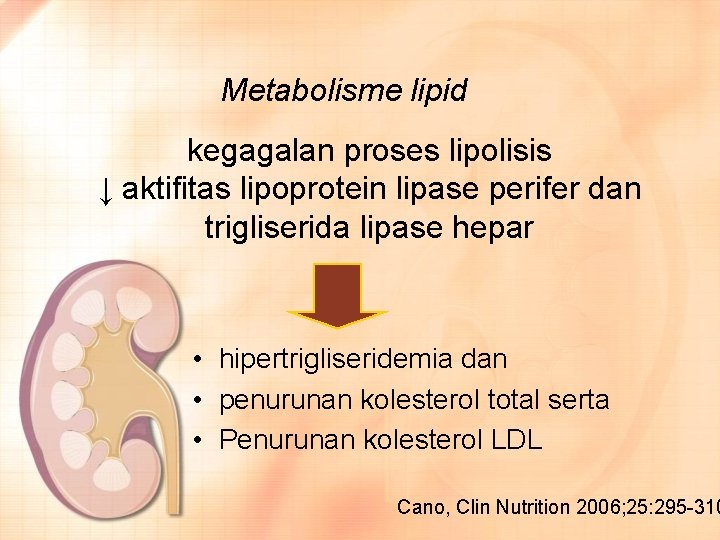
Metabolisme lipid kegagalan proses lipolisis ↓ aktifitas lipoprotein lipase perifer dan trigliserida lipase hepar • hipertrigliseridemia dan • penurunan kolesterol total serta • Penurunan kolesterol LDL Cano, Clin Nutrition 2006; 25: 295 -310

Metabolisme protein/asam amino • aktivasi katabolisme protein, • pelepasan asam amino yang berlebihan dari otot skelet dan terjadi keseimbangan nitrogen negatif, • peningkatan ekstraksi asam amino di hepar dari sirkulasi, • peningkatan glukoneogenesis dan ureagenesis. • Hati : sintesis protein perangsangan sekresi protein fase akut. plasma dan intra seluler : perubahan metabolisme asam amino nonesensial klirens asam amino akan meningkat Toigo, Clin Nutrition 2000; 281

Faktor yang terlibat di dalam patogenesis katabolisme protein pada GGA (1) • Suplai nutrient tidak adekuat • Toksin uremik • Faktor endokrin : • Resistensi insulin • Peningkatan sekresi hormon katabolisme • Resisten terhadap berkurangnya sekresi faktor anabolik Fiaccadori, J Nephrol 2008; 21: 645 -656
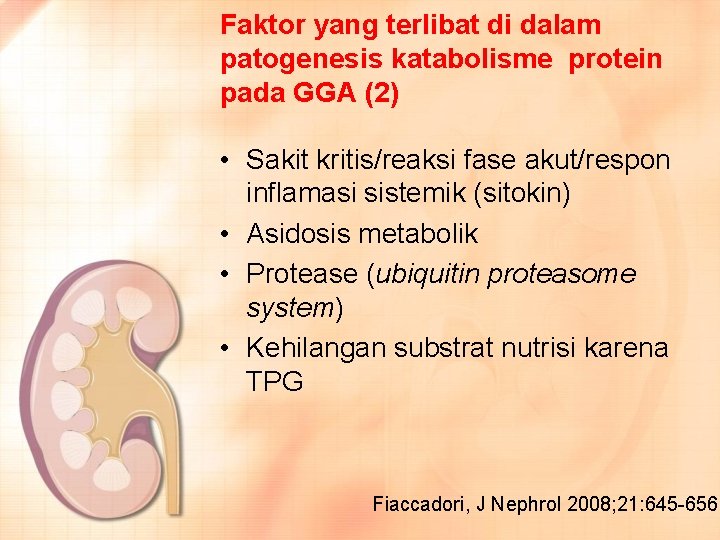
Faktor yang terlibat di dalam patogenesis katabolisme protein pada GGA (2) • Sakit kritis/reaksi fase akut/respon inflamasi sistemik (sitokin) • Asidosis metabolik • Protease (ubiquitin proteasome system) • Kehilangan substrat nutrisi karena TPG Fiaccadori, J Nephrol 2008; 21: 645 -656
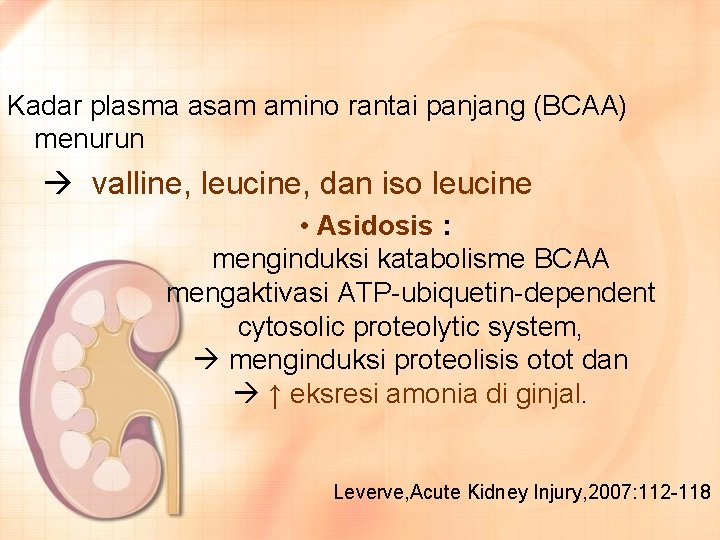
Kadar plasma asam amino rantai panjang (BCAA) menurun valline, leucine, dan iso leucine • Asidosis : menginduksi katabolisme BCAA mengaktivasi ATP-ubiquetin-dependent cytosolic proteolytic system, menginduksi proteolisis otot dan ↑ eksresi amonia di ginjal. Leverve, Acute Kidney Injury, 2007: 112 -118
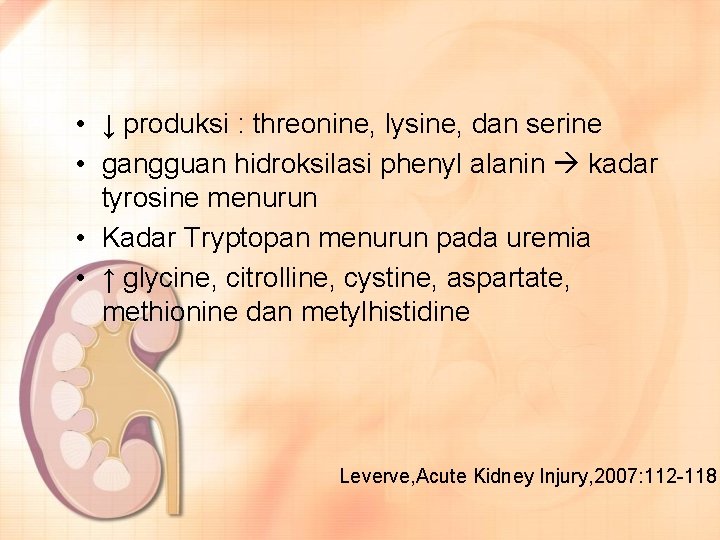
• ↓ produksi : threonine, lysine, dan serine • gangguan hidroksilasi phenyl alanin kadar tyrosine menurun • Kadar Tryptopan menurun pada uremia • ↑ glycine, citrolline, cystine, aspartate, methionine dan metylhistidine Leverve, Acute Kidney Injury, 2007: 112 -118
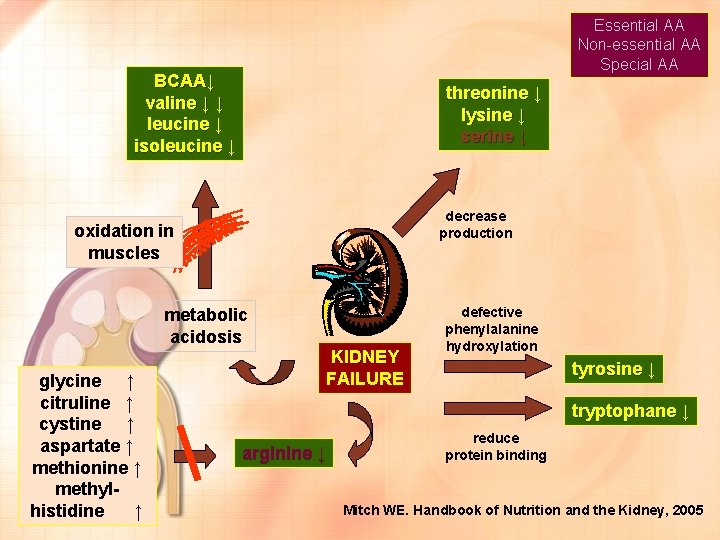
Essential AA Non-essential AA Special AA BCAA↓ valine ↓ ↓ leucine ↓ isoleucine ↓ threonine ↓ lysine ↓ serine ↓ decrease production oxidation in muscles metabolic acidosis glycine ↑ citruline ↑ cystine ↑ aspartate ↑ methionine ↑ methylhistidine ↑ KIDNEY FAILURE defective phenylalanine hydroxylation tyrosine ↓ tryptophane ↓ arginine ↓ reduce protein binding Mitch WE. Handbook of Nutrition and the Kidney, 2005
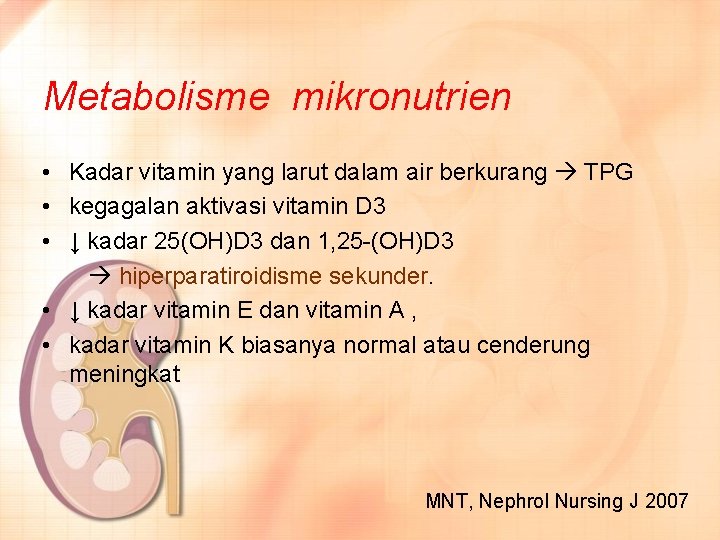
Metabolisme mikronutrien • Kadar vitamin yang larut dalam air berkurang TPG • kegagalan aktivasi vitamin D 3 • ↓ kadar 25(OH)D 3 dan 1, 25 -(OH)D 3 hiperparatiroidisme sekunder. • ↓ kadar vitamin E dan vitamin A , • kadar vitamin K biasanya normal atau cenderung meningkat MNT, Nephrol Nursing J 2007

Metabolisme trace elements • tidak mengalami perubahan spesifik pada Gg. GA , • penurunan kadar konsentrasi selenium di plasma dan eritrosit. • sakit kritis, pemberian selenium : • memperbaiki outcome • menurunkan insidensi kejadian Gg. GA yang memerlukan tindakan TPG MNT, Nephrol Nursing J 2007

PENGARUH TERAPI PENGGANTI GINJAL (TPG) TERHADAP METABOLISME Karena pemakaian yang berkesinambungan dan adanya pergantian cairan yang tinggi (fluid turnover), terapi ini memberikan : • pengaruh negatif terhadap keseimbangan elektrolit dan nutrient. • terdapat pembentukan reactive oxygen species Cano, Clin Nutrition 2006; 25: 295 -3

STATUS NUTRISI PADA AKI • Penderita sakit kritis dengan AKI potensi kehilangan nutrien • Evaluasi status nutrisi sulit perubahan di dalam komposisi tubuh • Protein Energy Wasting (PEW) Fiaccadori, J Nephrol 2008; 21: 645 -656

PENILAIAN STATUS NUTRISI • • Biokimia (albumin dan prealbumin) ↓ berat badan ↓ massa otot ↓ asupan energi dan protein Subjective Global Assessment (SGA) Fiaccadori, J Nephrol 2008; 21: 645 -656

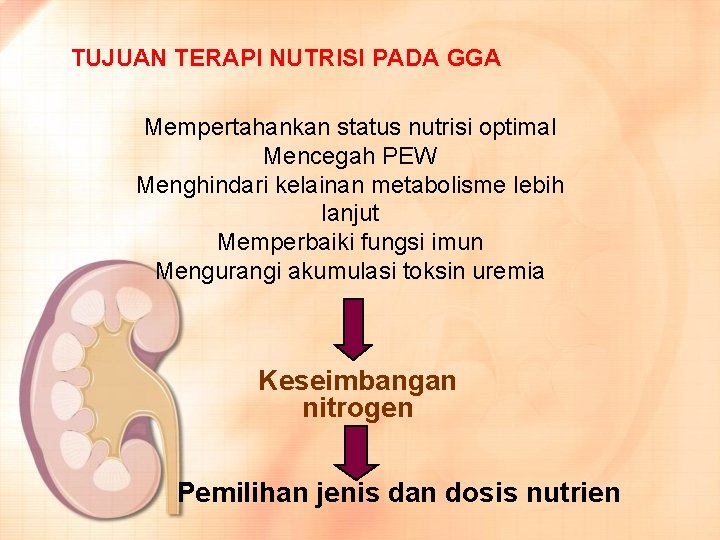
TUJUAN TERAPI NUTRISI PADA GGA Mempertahankan status nutrisi optimal Mencegah PEW Menghindari kelainan metabolisme lebih lanjut Memperbaiki fungsi imun Mengurangi akumulasi toksin uremia Keseimbangan nitrogen Pemilihan jenis dan dosis nutrien

Pemberian Nutrisi GGA tergantung: • • Ada / tidak adanya komplikasi pd Gg. GA Kelainan Metabolisme karbohidrat Kelainan Metabolisme Lipid Kelainan Metabolisme Asam amino Metabolisme mikronutrien Metabolisme trace elements TPG

Asupan nutrient yang optimal ditentukan oleh: - tingkat keparahan penyakit yang mendasarinya - komplikasi yang terjadi, - tingkat katabolisme, - status nutrisi - tipe dan frekuensi TPG Nutrisi yang tidak adekuat : Kontribusi dalam hilangnya massa tubuh pada GGA Penentu utama dari morbiditas dan mortalitas
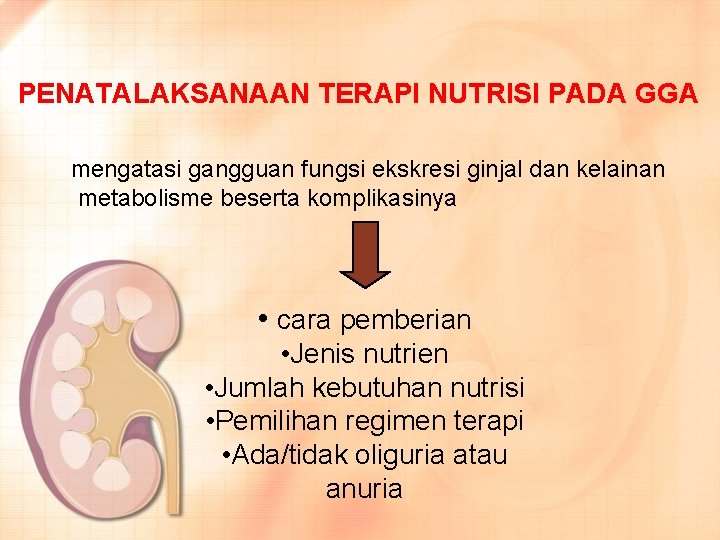
PENATALAKSANAAN TERAPI NUTRISI PADA GGA mengatasi gangguan fungsi ekskresi ginjal dan kelainan metabolisme beserta komplikasinya • cara pemberian • Jenis nutrien • Jumlah kebutuhan nutrisi • Pemilihan regimen terapi • Ada/tidak oliguria atau anuria

JENIS DAN JUMLAH KEBUTUHAN NUTRISI PADA Gg. GA

Kebutuhan energi / kalori • GGA tanpa komplikasi, konsumsi oksigen sama dengan subjek yang sehat, • adanya sepsis atau disfungsi multi organ terjadi peningkatan sekitar 25%. • Ekspenditur energi ditentukan oleh penyakit yang mendasarinya tidak oleh gagal ginjal • Pemberian kalori adekuat sangat penting keseimbangan nitrogen menjadi positif. MNT, Nephrol Nursing J 2007

• • Komplikasi : overfeeding dibandingkan underfeeding Sebaiknya : 20 sampai 30 kkal/kg BB/hari Gg. GA ringan : 35 kkal/kg BB/hari hipermetabolik : ekspenditur energi jarang melebihi 130% asupan energi tidak melebihi 30 kkal/kg BB/hari • gangguan respirasi ( respiratory distress) + ventilator : 25 kkal/kg BB/hari

• Pada keadaan Gg. GA : jumlah cairan perlu di batasi produksi urine yang rendah. • pemilihan sumber kalori harus hemat cairan misalnya : • glukosa hipertonis (70% dextrose) • infus lipid (20%) • asam amino (10 -15%)

Kebutuhan asam amino/protein • Gg. GA ringan (risk) tanpa katabolik : tidak kurang dari 0, 8 gr/kg BB/hari • Gg. GA + TPG intermiten (IHD) : minimum 1, 2 gr/kg BB/hari • Gg. GA + Sakit kritis + TPG (CRRT) diperkirakan : • protein catabolic rate (PCR) mencapai 1, 51, 7 gr/kg BB/hari, • asam amino/protein : 1, 4 -1, 7 gr/kg BB/hari • Dapat > tinggi : 2, 5 gr/kg BB/hari (kontroversi )
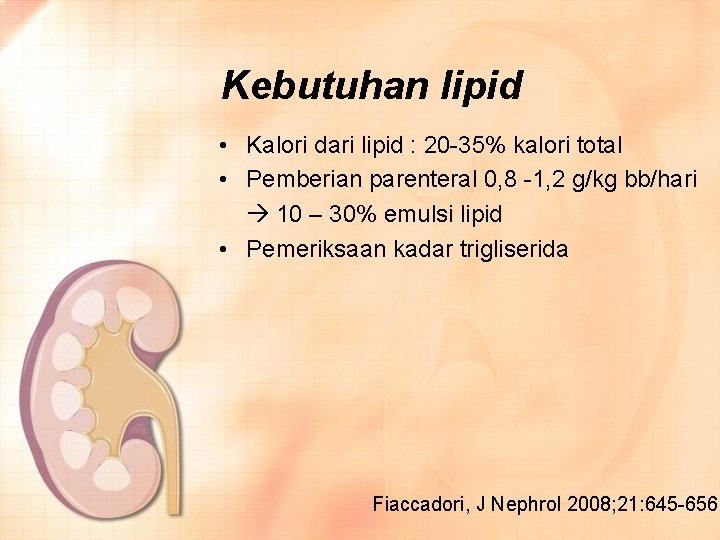
Kebutuhan lipid • Kalori dari lipid : 20 -35% kalori total • Pemberian parenteral 0, 8 -1, 2 g/kg bb/hari 10 – 30% emulsi lipid • Pemeriksaan kadar trigliserida Fiaccadori, J Nephrol 2008; 21: 645 -656
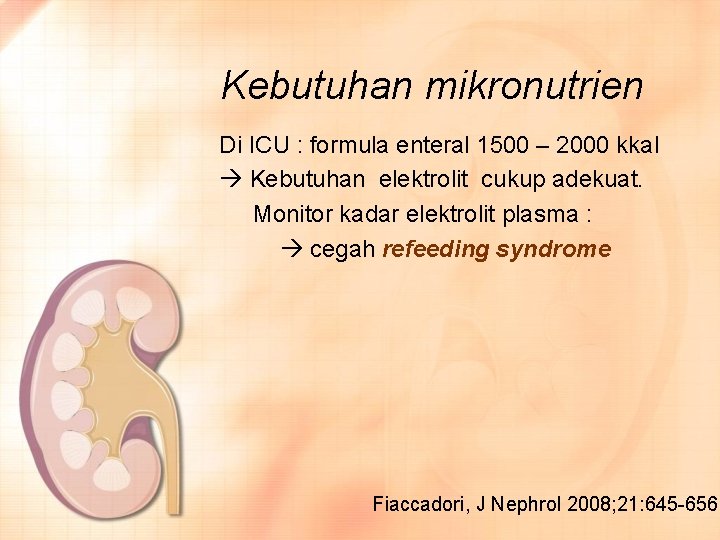
Kebutuhan mikronutrien Di ICU : formula enteral 1500 – 2000 kkal Kebutuhan elektrolit cukup adekuat. Monitor kadar elektrolit plasma : cegah refeeding syndrome Fiaccadori, J Nephrol 2008; 21: 645 -656
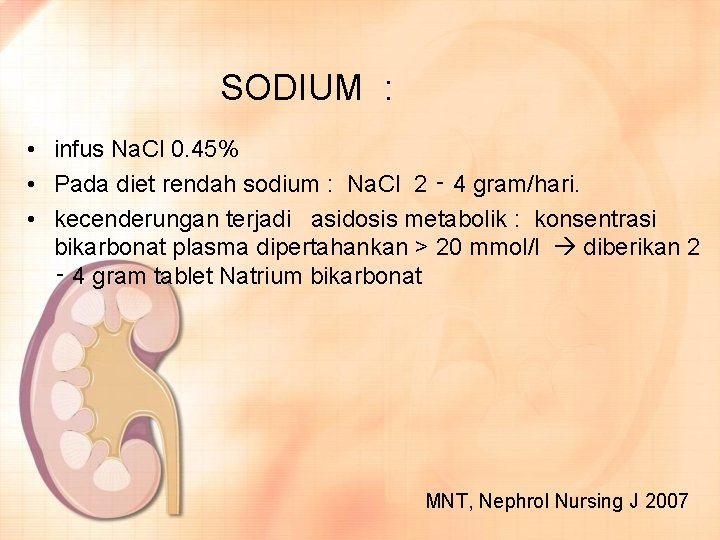
SODIUM : • infus Na. Cl 0. 45% • Pada diet rendah sodium : Na. Cl 2 ‑ 4 gram/hari. • kecenderungan terjadi asidosis metabolik : konsentrasi bikarbonat plasma dipertahankan > 20 mmol/l diberikan 2 ‑ 4 gram tablet Natrium bikarbonat MNT, Nephrol Nursing J 2007
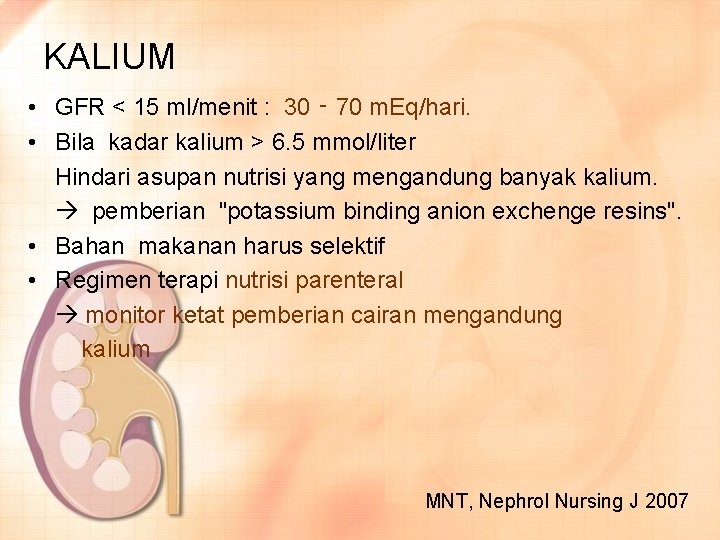
KALIUM • GFR < 15 ml/menit : 30 ‑ 70 m. Eq/hari. • Bila kadar kalium > 6. 5 mmol/liter Hindari asupan nutrisi yang mengandung banyak kalium. pemberian "potassium binding anion exchenge resins". • Bahan makanan harus selektif • Regimen terapi nutrisi parenteral monitor ketat pemberian cairan mengandung kalium MNT, Nephrol Nursing J 2007

VITAMIN • defisiensi vitamin dan zat besi. • Defisiensi asam folat, piridoksin, vitamin C , B complex paling sering terjadi • ↓kadar vit. D • Vitamin K tidak membutuhkan suplementasi. • ↑kadar vitamin A MNT, Nephrol Nursing J 2007

• VITAMIN DAN MINERAL 100% DARI RECOMMENDED DAILY ALLOWANCE (RDA) • IMMUNE MODULATING FORMULAE: • ARGININ, GLUTAMINE, NUKLEOTIDA • ANTIOKSIDAN, EPA, GLA

KEBUTUHAN NUTRISI PADA PENDERITA GGA (ESPEN) • Energi 20 -30 kkal/kg bb/hr • Karbohidrat 3 -5 (maks 7) gr/kg bb/hr • Lemak 0, 8 -1, 2 (maks 1, 5) gr/kg bb/hr • Protein (asam amino esensial dan nonesensial) Terapi konservatif 0, 6 -0, 8 (maks 1, 0) gr/kg bb/hr TPG (CRRT/SLED) 1, 0 -1, 5 gr/kg. BB/hari TPG (CRRT/SLED) Cano, Clin Nutrition 2006; 25: 295 -310 +hiperkatabolisme maks 1, 7 gr/kg
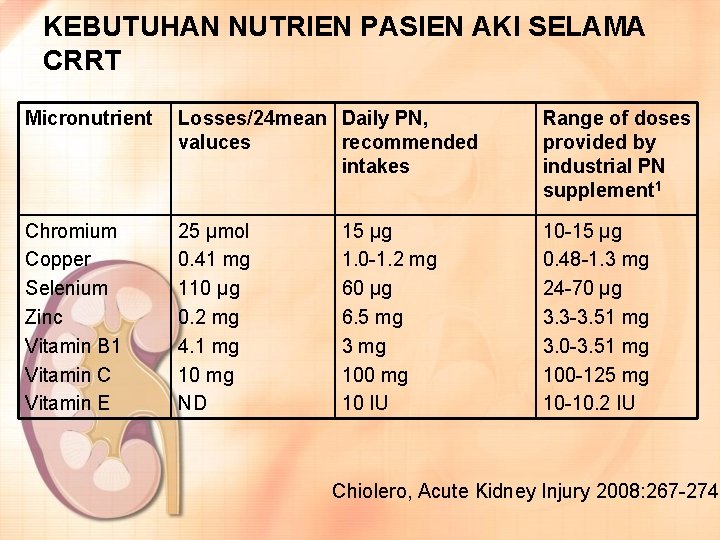
KEBUTUHAN NUTRIEN PASIEN AKI SELAMA CRRT Micronutrient Losses/24 mean Daily PN, valuces recommended intakes Range of doses provided by industrial PN supplement 1 Chromium Copper Selenium Zinc Vitamin B 1 Vitamin C Vitamin E 25 μmol 0. 41 mg 110 μg 0. 2 mg 4. 1 mg 10 mg ND 10 -15 μg 0. 48 -1. 3 mg 24 -70 μg 3. 3 -3. 51 mg 3. 0 -3. 51 mg 100 -125 mg 10 -10. 2 IU 15 μg 1. 0 -1. 2 mg 60 μg 6. 5 mg 3 mg 100 mg 10 IU Chiolero, Acute Kidney Injury 2008: 267 -274
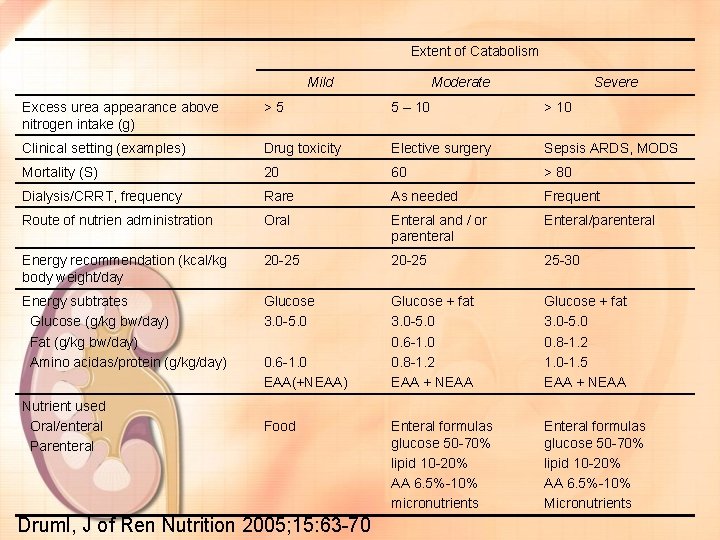
Extent of Catabolism Mild Moderate Severe Excess urea appearance above nitrogen intake (g) >5 5 – 10 > 10 Clinical setting (examples) Drug toxicity Elective surgery Sepsis ARDS, MODS Mortality (S) 20 60 > 80 Dialysis/CRRT, frequency Rare As needed Frequent Route of nutrien administration Oral Enteral and / or parenteral Enteral/parenteral Energy recommendation (kcal/kg body weight/day 20 -25 25 -30 Energy subtrates Glucose (g/kg bw/day) Fat (g/kg bw/day) Amino acidas/protein (g/kg/day) Glucose 3. 0 -5. 0 Glucose + fat 3. 0 -5. 0 0. 6 -1. 0 0. 8 -1. 2 EAA + NEAA Glucose + fat 3. 0 -5. 0 0. 8 -1. 2 1. 0 -1. 5 EAA + NEAA Enteral formulas glucose 50 -70% lipid 10 -20% AA 6. 5%-10% micronutrients Enteral formulas glucose 50 -70% lipid 10 -20% AA 6. 5%-10% Micronutrients Nutrient used Oral/enteral Parenteral 0. 6 -1. 0 EAA(+NEAA) Food Druml, J of Ren Nutrition 2005; 15: 63 -70

CARA PEMBERIAN TERAPI NUTRISI PADA GGA ¨Secara Oral ¨Nutrisi Enteral (tube feeding) ¨Nutrisi Parenteral (NPE)
TERAPI GIZI PARENTERAL NUTRITION FOOD FORTIFICATION ORAL NUTRITIONAL SUPPLEMENT ENTERAL NUTRITION
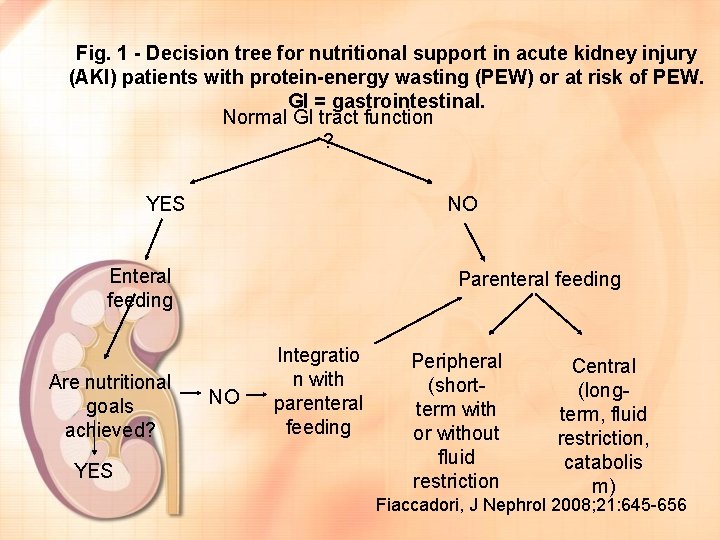
Fig. 1 - Decision tree for nutritional support in acute kidney injury (AKI) patients with protein-energy wasting (PEW) or at risk of PEW. GI = gastrointestinal. Normal GI tract function ? YES NO Enteral feeding Are nutritional goals achieved? YES Parenteral feeding NO Integratio n with parenteral feeding Peripheral (shortterm with or without fluid restriction Central (longterm, fluid restriction, catabolis m) Fiaccadori, J Nephrol 2008; 21: 645 -656

Terapi Nutrisi Secara Oral pilihan pertama, • penderita Gg. GA tanpa hiperkatabolik (kelompok 1). • Awal : 40 gram protein (0. 6 gr/kg BB/hari) • Secara bertahap dinaikkan menjadi 0. 8 gr / kg BB/hari jika kadar ureum < 100 mg/d. L. hemodialisis atau dialysis peritoneal : • protein sebaiknya dinaikkan menjadi 1, 0 -1, 4 gr/kg BB/hari Druml, J of Ren Nutrition 2005; 15: 63 -70

Nutrisi Enteral (tube feeding) • Sakit kritis Keuntungan : - mempertahankan fungsi gastro intestinal khususnya fungsi barier dari mukosa intestinal. - dapat menambah aliran plasma renal dan memperbaiki fungsi ginjal. • Memperbaiki prognosis Druml, J of Ren Nutrition 2005; 15: 63 -70

CARA PEMBERIAN EN • KONTINYU: • • 10 -25 m. L/jam untuk 12 jam pertama, dapat ditingkatkan menjadi 50 m. L/jam 12 jam kedua Hari pertama 1000 m. L selama 24 jam, hari kedua 1500 m. L selama 24 jam, hari ketiga sesuai kebutuhan • INTERMITEN: 250 -500 m. L SETIAP KALI PEMBERIAN, DIBERIKAN SELAMA 3060 MENIT, 5 -8 KALI SEHARI

Nutrisi Parenteral (NPE) • sakit kritis : sering disertai dengan keluhan gastrointestinal seperti mual dan muntah, perdarahan SMBA • kombinasi NPE dengan enteral atau oral • Komposisi larutan nutrisi parenteral : glukosa, emulsi lipid, asam amino Druml, J of Ren Nutrition 2005; 15: 63 -70

INDIKASI • • NUTRISI ORAL/ENTERAL INADEKUAT SELAMA 7 -10 HARI GIZI BURUK, NUTRISI ORAL/ENTERAL INADEKUAT SELAMA 3 -5 HARI HEMODINAMIK STABIL SALURAN CERNA TIDAK BERFUNGSI ATAU HARUS DIISTIRAHATKAN
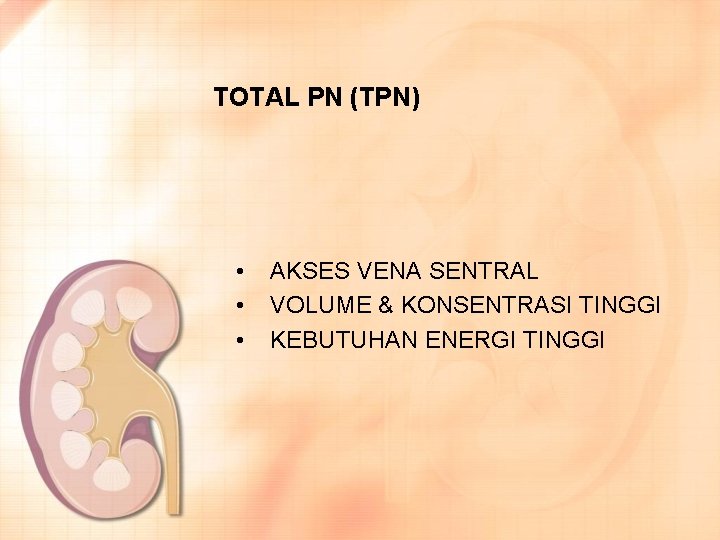
TOTAL PN (TPN) • • • AKSES VENA SENTRAL VOLUME & KONSENTRASI TINGGI KEBUTUHAN ENERGI TINGGI

PERIPHERAL PN (PPN) • • OSMOLARITAS ≤ 900 m. Osml PEMBERIAN JANGKA PENDEK: 7 -10 HARI KEBUTUHAN ENERGI TIDAK TINGGI TIDAK ADA RESTRIKSI CAIRAN

Ringkasan Terapi nutrisi rasional pd GGA bergantung kepada : Ada / tidak adanya komplikasi pd GGA Kelainan Metabolisme karbohidrat Kelainan Metabolisme Lipid Kelainan Metabolisme Asam amino Metabolisme mikronutrien dan trace elements TPG Seleksi penderita Cara pemberian dan komposisi nutrien Tujuan terapi nutrisi pada GGA adalah peningkatan survival
CHRONIC KIDNEY DISEASE (PENYAKIT GINJAL KRONIK) 59
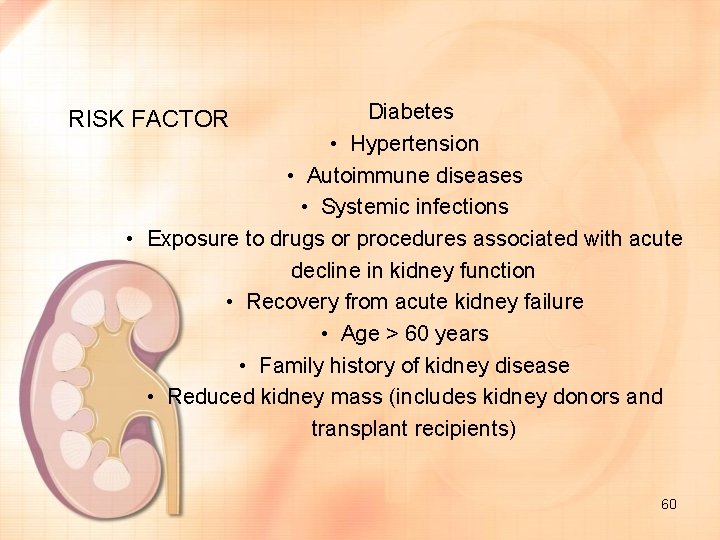
Diabetes • Hypertension • Autoimmune diseases • Systemic infections • Exposure to drugs or procedures associated with acute decline in kidney function • Recovery from acute kidney failure • Age > 60 years • Family history of kidney disease • Reduced kidney mass (includes kidney donors and transplant recipients) RISK FACTOR 60
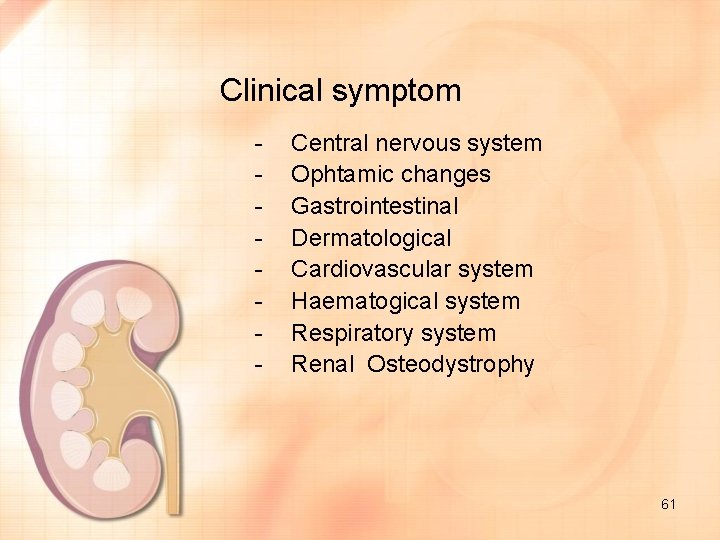
Clinical symptom - Central nervous system Ophtamic changes Gastrointestinal Dermatological Cardiovascular system Haematogical system Respiratory system Renal Osteodystrophy 61

Medical nutrition therapy GOAL 1. ADEQUATE FOOD, NOT MAKE HEAVIER RENAL FUNCTION 2. DECREASED OF UREUM & CREATININ LEVEL 3. MINIMIZED SALT RETENSION REQUIREMENT 1. Low protein High Biologi value of Protein 2. Limitation of Salt ( Heavy HT, >> K, edema, Oligo /anurie) 3. Limitation of K (Glom function or prod urine << 400 cc) 4. Adequate food 5. >> fluid 62

VERY LOW PROTEIN NEED KETO ACID ANALOGUES These agent are transaminated in the liver by non essential amino acids to the corresponding essential amino acids which are then use for protein synthesis 63

Keto acid supplemented protein restriction diet should play a principle role in the treatment of patients with CKD because such diets 1. Improve symptomes 2. Maintains a good nutritional state 3. Limits proteinuria 4. Can delay the time until renal replacement therapy is needed.

Specific International Guidelines & Recommendation s for keto acid therapy • Low protein diet (0. 6 -07 g/Kg b. w. /day Cr Cl 50 ml/min/1. 73 m 2) • Keto acid therapy indicated 20 -25 ml/min/1. 73 m 2) • Low protein diet +keto acid 0. 4 -0. 6 gr/kg b. w. /day • Dosage of keto acid 0. 1 gr/kg bw/day • Daily energy intake of 35 kcal/kgb. w. /day should be recommended • Protein calories must be replaced by complex CH calories-not by lipid Am J Nephrol 2005; 25(suppl 1): 1 -28
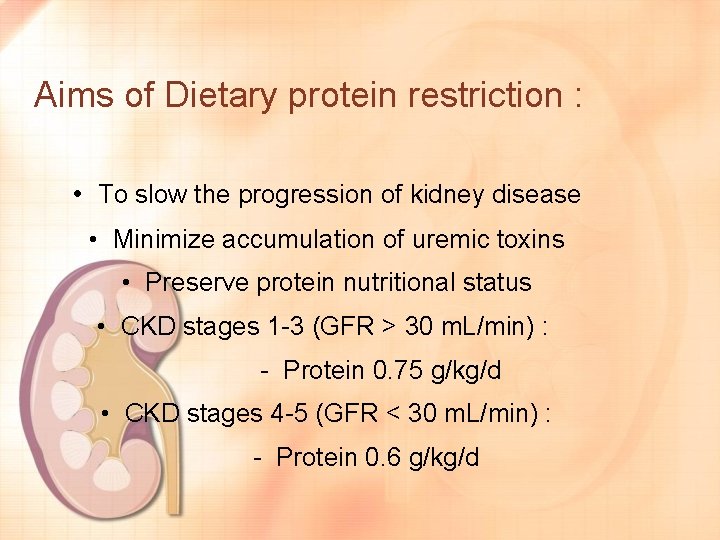
Aims of Dietary protein restriction : • To slow the progression of kidney disease • Minimize accumulation of uremic toxins • Preserve protein nutritional status • CKD stages 1 -3 (GFR > 30 m. L/min) : - Protein 0. 75 g/kg/d • CKD stages 4 -5 (GFR < 30 m. L/min) : - Protein 0. 6 g/kg/d
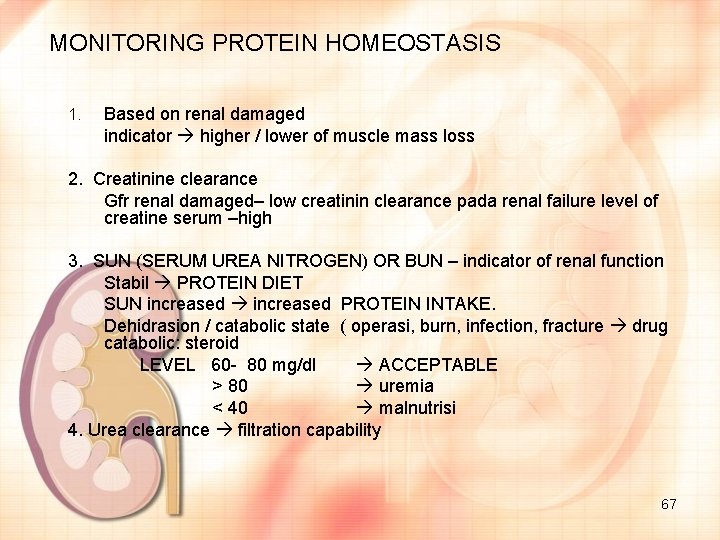
MONITORING PROTEIN HOMEOSTASIS 1. Based on renal damaged indicator higher / lower of muscle mass loss 2. Creatinine clearance Gfr renal damaged– low creatinin clearance pada renal failure level of creatine serum –high 3. SUN (SERUM UREA NITROGEN) OR BUN – indicator of renal function Stabil PROTEIN DIET SUN increased PROTEIN INTAKE. Dehidrasion / catabolic state ( operasi, burn, infection, fracture drug catabolic: steroid LEVEL 60 - 80 mg/dl ACCEPTABLE > 80 uremia < 40 malnutrisi 4. Urea clearance filtration capability 67
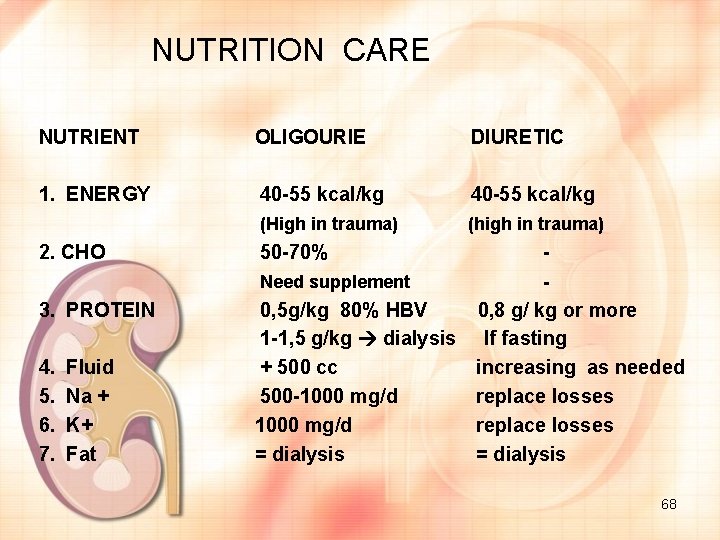
NUTRITION CARE NUTRIENT OLIGOURIE DIURETIC 1. ENERGY 40 -55 kcal/kg (High in trauma) (high in trauma) 2. CHO 3. PROTEIN 4. 5. 6. 7. Fluid Na + K+ Fat 50 -70% - Need supplement - 0, 5 g/kg 80% HBV 1 -1, 5 g/kg dialysis + 500 cc 500 -1000 mg/d = dialysis 0, 8 g/ kg or more If fasting increasing as needed replace losses = dialysis 68

OPTIONS- THERAPY OF ESRD 1. CONSERVATIF MANAGEMENT 2. DIALYSIS A. HEMODIALISIS B. PERITONEAL-DIALISIS 3. TRANSPLANT 69
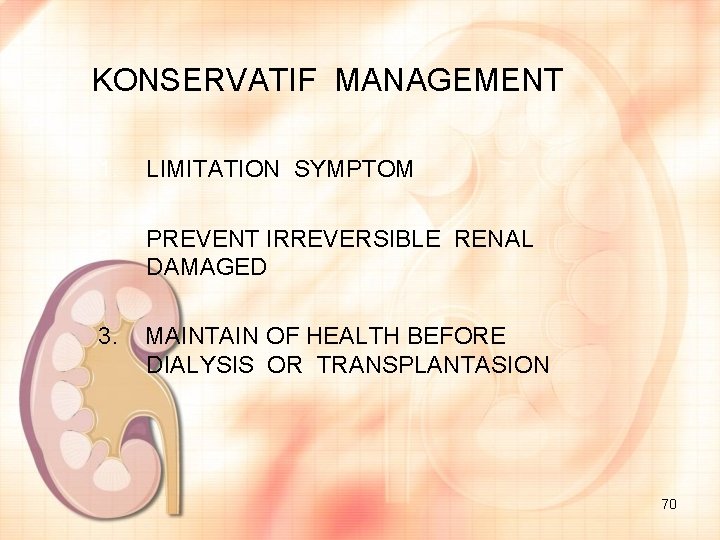
KONSERVATIF MANAGEMENT 1. LIMITATION SYMPTOM 2. PREVENT IRREVERSIBLE RENAL DAMAGED 3. MAINTAIN OF HEALTH BEFORE DIALYSIS OR TRANSPLANTASION 70

TYPE OF DIALYSIS A. HEMODIALYS BY MACHINE ( venous ) 3 -4 hours /d, 3 – 4 x week B. PERITONEAL DIALYSIS Intermittent ( IPD) Continous ambulatory ( CAPD) Continous Cyclic 71

NUTRITION MANAGEMENT ON RENAL TRANSPLANTASION 1. ADEQUATE FOOD 2. CHO 40 – 50 % FROM TOTAL CALORIES 3. PROTEIN 1. 2 - 1. 5 gr ADJUST TO NORMAL LEVEL (LAB AND ELECKTROLYT BALANCE) 4. LIMITATION OF Na+ 2 - 4 gr / day 5. K+ AS NEEDED 72

KIDNEY STONES q This disease is not transmittable. q Kidney stones can develop when certain chemicals in urine form crystals that stick together. q Stones may also develop from a persistent kidney infection. q Drinking small amounts of fluids. q More frequent in hot weather
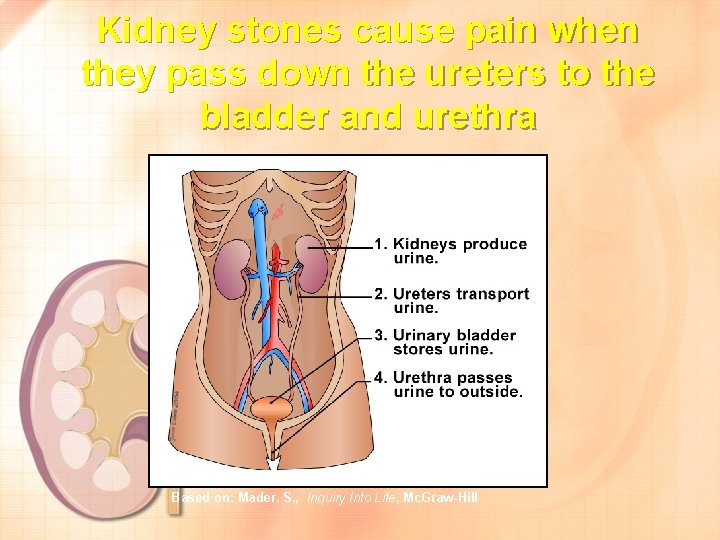
Kidney stones cause pain when they pass down the ureters to the bladder and urethra Based on: Mader, S. , Inquiry Into Life, Mc. Graw-Hill
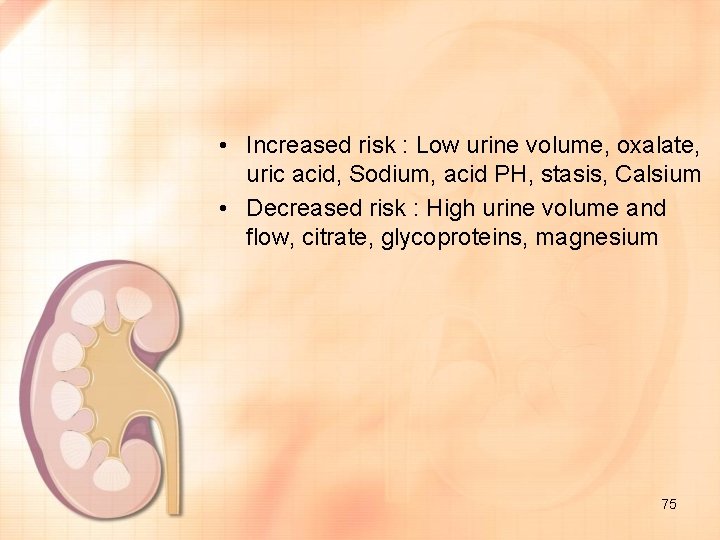
• Increased risk : Low urine volume, oxalate, uric acid, Sodium, acid PH, stasis, Calsium • Decreased risk : High urine volume and flow, citrate, glycoproteins, magnesium 75
Kidney Stones • Basic cause is unknown • Factors relating to urine or urinary tract environment contribute to formation • Present in 5% of U. S. women and 12% of U. S. men • Major stones are formed from one of three substances: • Calcium • Struvite • Uric acid (Cont’d…) Mosby items and derived items © 2006 by Slide 76
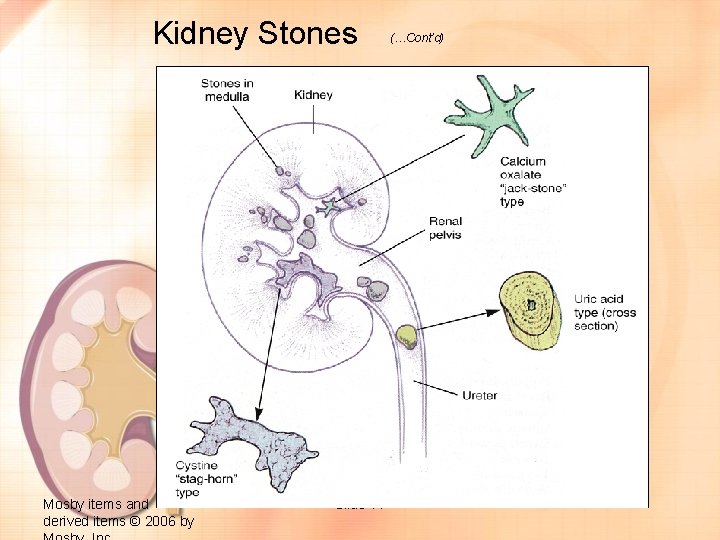
Kidney Stones Mosby items and derived items © 2006 by Slide 77 (…Cont’d)

A. ENVIRONMENTAL FACTOR 1. CALSIUM ( 96%) N eksresi 100 – 175 mg hipersecresion : high intake Ca, high Vit. D long imobilisasion, hiperparathyroid renal tubular asidosis, high calsiurie idiopatik Dietary factors associated with risk of calsium stones : Increased risk ( animal protein, oxalate, sodium ) Decreased risk ( calsium, potassium, Magnesium, fluid intake 2. CYSTEIN ( herediter ) homozygous cystinuria 78
Others : - Urid acid End product of purin metabolism - Struvite Magnesium ammonium phosphate, carbonate apatite Triple phosphate or Infection stones 79
B. TRACTUS UROGENITAL • CHANGED OF URINE PHYSICALLY • CHANGED OF URINE CONCENTRATION • CHANGED OF URINE BALANCED 80

C. MATRIX BATU ORGANIK • RECURRENT INFECTION • DEFICIENCY OF VITAMIN A ( DESQUAMATION OF CEL EPITHEL) • DOT CALCIFICATION RANDALL’S PLAQUE 81
Key Concepts • Renal disease interferes with the normal capacity of nephrons to filter waste products of body metabolism. • Short-term renal disease requires basic nutritional support for healing rather than dietary restriction. Mosby items and derived items © 2006 by Slide 82
Dual Role of the Kidneys • Kidneys make urine, through which they excrete most of the waste products of metabolism. • Kidneys control the concentrations of most constituents of body fluids, especially blood. Mosby items and derived items © 2006 by Slide 83

Treatments for Kidney Stones • • Small stones may pass with no pain Larger stones may pass but cause extreme of pain, requiring a lot pain medication • Stones that are too large to pass may require surgical treatment including: • using a ureteroscope to go up and snare the stone • using a nephroscope to crush the stone and retrieve it • using shock wave lithotripsy where a person is submerged in water containing shock waves to pulverize the stones

Risk Factors Mosby items and derived items © 2006 by Slide 85
Calcium Stones Mosby items and derived items © 2006 by • 70%-80% of kidney stones are composed of calcium oxalate • Almost half result from genetic predisposition • Other causes: • Excess calcium in blood (hypercalcemia) or urine (hypercalciuria) • Excess oxalate in urine (hyperoxaluria) • Low levels of citrate in urine (hypocitraturia) • Infection Slide 86

Examples of Food Sources of Oxalates Mosby items and derived items © 2006 by • Fruits: berries, Concord grapes, currants, figs, fruit cocktail, plums, rhubarb, tangerines • Vegetables: baked/green/wax beans, beet/collard greens, beets, celery, Swiss chard, chives, eggplant, endive, kale, okra, green peppers, spinach, sweet potatoes, tomatoes • Nuts: almonds, cashews, peanuts/peanut butter • Beverages: Slide 87 cocoa, draft beer, tea
Struvite Stones • Composed of magnesium ammonium phosphate • Mainly caused by urinary tract infections rather than specific nutrient • No diet therapy is involved • Usually removed surgically Mosby items and derived items © 2006 by Slide 88
Other Stones • Cystine stones • Caused by genetic metabolic defect • Occur rarely • Xanthine stones • Associated with treatment for gout and family history of gout • Occur rarely Mosby items and derived items © 2006 by Slide 89
Kidney Stones: Symptoms and Treatment • Clinical symptoms: severe pain, other urinary symptoms, general weakness, and fever • Several considerations for treatment • Fluid intake to prevent accumulation of materials • Dietary control of stone constituents • Achievement of desired p. H of urine via medication • Use of binding agents to prevent absorption of stone elements • Drug therapy in combination with diet therapy Mosby items and derived items © 2006 by Slide 90

VARIATION DIET 1. LOW CALCIUM HIGH ASH CAID 2. HIGH DIET ASH ALKALIS 3. LOW PURINE DIET 91

Acid ash and alkaline ash diet • Dietary intake can influence the acidity or alkalinity of the urine • The acid forming : chloride, phosphorus, sulfur ( high protein food, breads, cereal ) • The base forming : sodium, potassium, calsium, magnesium ( Fruit, vegetables ) MILK ? ? ? 92

Nutrition Therapy: Calcium Stones • Low-calcium diet (approx. 400 mg/day) recommended for those with supersaturation of calcium in the urine and who are not at risk for bone loss • If stone is calcium phosphate, sources of phosphorus (meats, legumes, nuts) are controlled • Fluid intake increased • Sodium intake decreased • Fiber foods high in phytates increased Mosby items and derived items © 2006 by Slide 93

LOW CALCIUM DIET HIGH ASH ACID • FLUID > 2500 cc/day • Low calcium • Limitation food intake contains: PROTEIN : milk, cheese, schrimp, crab, rilis, salt fish, sarden, animal brain, ren, liver, cor CHO : potatoes, sweet potatoes, cassava, biscuit, cake contain milk VEGETABLE : Spinach, mangkok leaf, melinjo leaf, papaya leaf, lamtoro leaf, cassava leaf, talas (taro) leaf, d. katuk leaf, kelor leaf, jtg pisang, melinjo, sawi, leunca FRUITS : All Fermented Fruits OTHERS : SOFT DRINK contains soda, alcohol, coclate, yeast 94
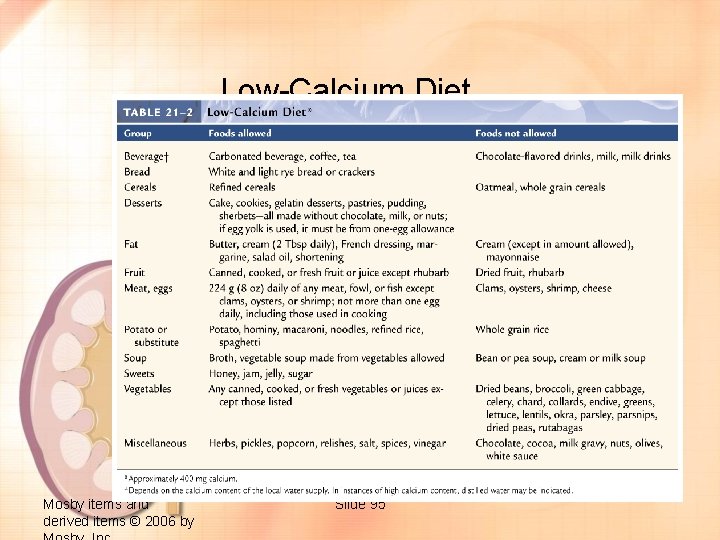
Low-Calcium Diet Mosby items and derived items © 2006 by Slide 95

Nutrition Therapy: Uric Acid Stones • Low-purine diet sometimes recommended • Avoid: • Organ meats • Alcohol • Anchovies, sardines • Yeast • Legumes, mushrooms, spinach, asparagus, cauliflower • Poultry Mosby items and derived items © 2006 by Slide 96

LOW PURINE DIET LIMIT FOOD SOURCES OF URIC ACID LOST WEIGHT TO IDEAL BODY WEIGHT 97
Nutrition Therapy: Cystine Stones • Low-methionine diet (essentially a lowprotein diet) sometimes recommended • In children, a regular diet to support growth is recommended • Medical drug therapy is used to control infection or produce more alkaline urine Mosby items and derived items © 2006 by Slide 98

HIGH DIET ASH ALKALIS Especially for Cysteine stone and Uric acid 1. Fluid > 2500 cc/day 2. Low AA (contain Sulfur) 3. Vegetables < 300 gr/day 4. Fruit < 300 gr/day 99
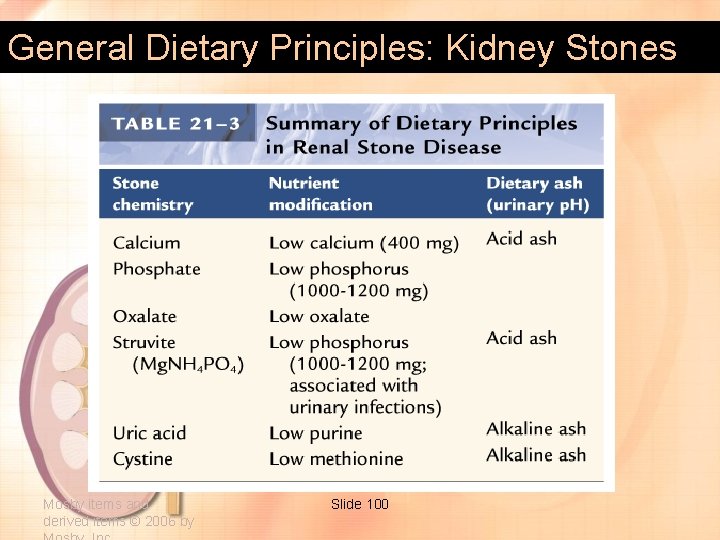
General Dietary Principles: Kidney Stones Mosby items and derived items © 2006 by Slide 100

Diet & Fluid Advice • High Fluid Intake • Restrict Salt (Na) • Oxalate Restrict • Avoid high intake of Purine food • Increased citrus fruits may help • If hypercalciuria restrict Ca intake Role of Potassium Citrate in preventing Cal Oxalate stone ds – KCit lowers urinary calcium whereas Na Citrate does not lower Calcium due to Sodium load

LIQUIDS Moderate Amounts : High Amounts : Apple Juice Cocoa Beer Fresh Tea Coffee Cola FOODS : Almonds, Asparagus, Cashew Nuts, Currants, Greens, Plums, Raspberries, Spinach



General measures to prevent recurrent stone formation • Increase fluid intake to maintain urine output of 2 -3 l/day: increase in urine sodium as a result of increased sodium intake. (Higher fluid intake alone will not prevent recurrent stones in patients with hypercalciuria) • Decrease intake of animal protein ( ≤ 52 g/day): Reduces production of metabolic acids, resulting in a lower level of acid induced calcium excretion; increases excretion of citrate that forms a soluble complex with calcium; and reduces supersaturation with respect to calcium oxalate and limits the excretion of uric acid

• Restrict salt intake ( ≤ 50 mmol/day of sodium chloride): Dietary and urinary sodium is directly correlated with urinary calcium excretion, and lower urinary excretion of sodium reduces urinary calcium excretion • Normal calcium intake ( ≥ 30 mmol/day): Low calcium diets increase urinary oxalate excretion, which may result in more stone formation and possibly a negative calcium balance • Decrease dietary oxalate: Reduce the intake of foods rich in oxalate—spinach, rhubarb, chocolate, and nuts • Cranberry juice: Decreases oxalate and phosphate excretion and increases citrate excretion
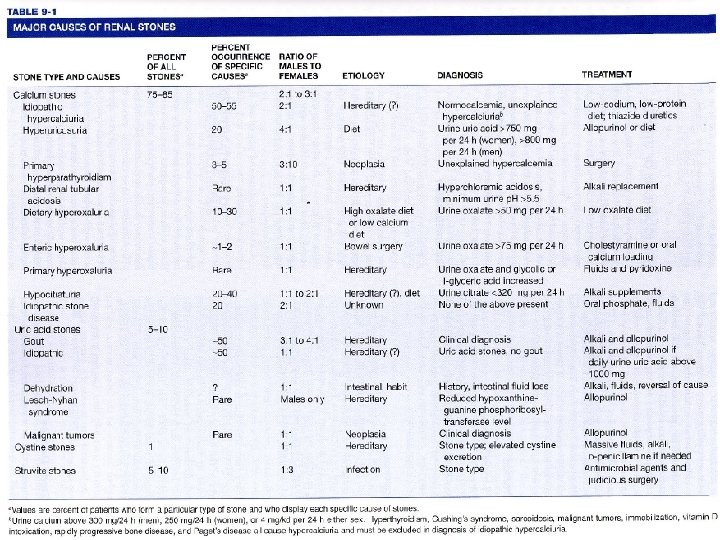

Low Purin Diet * Goal : 1. Eliminate uric acid development 2. Lost weight ----obese or maintain ideal body weight or normal weight 108

Requirement 1. 2. 3. 4. 5. Low purin contain 120 -150 mg Adequate calorie, protein, mineral and vitamin High carbohydrate Mild fat High fluid 109

IMPORTANT !! 110
Penilaian Kebutuhan Kalori • Sangat sulit • BEE bisa meningkat • Estimasi BEE: a. Indirect calorimetri b. Harris Benedict equation c. REE 111

Penilaian Kebutuhan Protein • • Ekskresi nitrogen Anjuran 1. 5 – 2. 2 g/kg. BB/hari secara bertahap Pemantauan: NUU dan kreatinin urin Monitor: fungsi ginjal (ureum & kreatinin); fungsi hepar. • BCAA dapat dipertimbangan • Serum albumin dipertahankan diatas 2. 2 g/d. L. 112

Cara menghitung kebutuhan nitrogen 1. Berdasarkan sekresi urea pada urine [urinary urea nitrogen = UUN] Untuk ini dibutuhkan urine tampung 24 jam. Langkah 2 yang harus dilakukan: • • Ukur UUN 24 jam Hitung total UUN dengan menggunakan rumus: • • Hitung asupan protein penderita/hari Hitung nitrogen balans dengan menggunakan rumus: Keterangan: asupan protein yang dikonversi ke Nitrogen = 6. 25 UUN = 4 gr [rata-rata nitrogen yang dikeluarkan melalui urine] 113
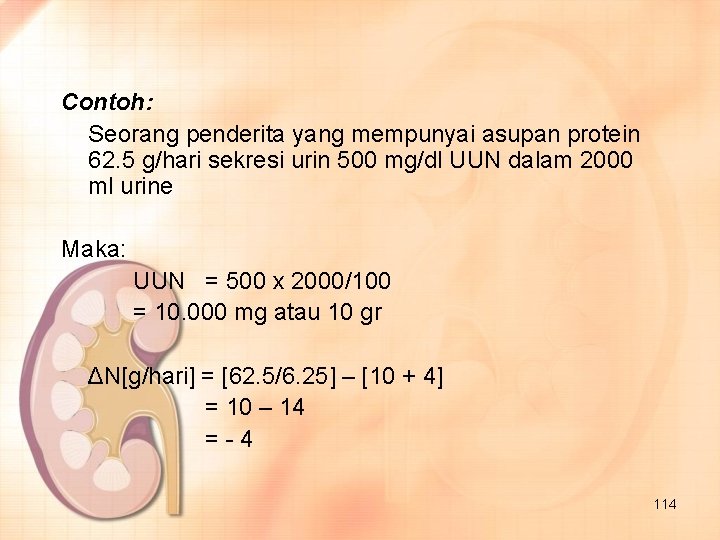
Contoh: Seorang penderita yang mempunyai asupan protein 62. 5 g/hari sekresi urin 500 mg/dl UUN dalam 2000 ml urine Maka: UUN = 500 x 2000/100 = 10. 000 mg atau 10 gr ΔN[g/hari] = [62. 5/6. 25] – [10 + 4] = 10 – 14 =-4 114
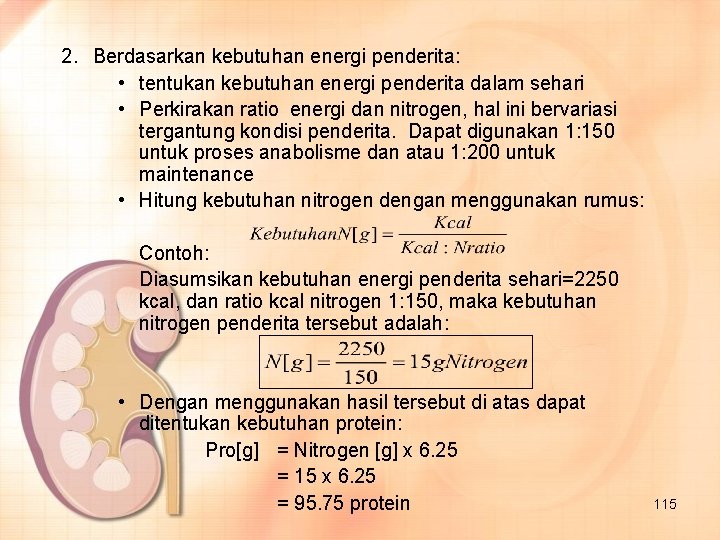
2. Berdasarkan kebutuhan energi penderita: • tentukan kebutuhan energi penderita dalam sehari • Perkirakan ratio energi dan nitrogen, hal ini bervariasi tergantung kondisi penderita. Dapat digunakan 1: 150 untuk proses anabolisme dan atau 1: 200 untuk maintenance • Hitung kebutuhan nitrogen dengan menggunakan rumus: Contoh: Diasumsikan kebutuhan energi penderita sehari=2250 kcal, dan ratio kcal nitrogen 1: 150, maka kebutuhan nitrogen penderita tersebut adalah: • Dengan menggunakan hasil tersebut di atas dapat ditentukan kebutuhan protein: Pro[g] = Nitrogen [g] x 6. 25 = 15 x 6. 25 = 95. 75 protein 115
Penilaian Kebutuhan Elektrolit • Monitor kadar elektrolit dalam darah Na, K, Cl , HCO 3, Ca • Monitor Blood gas 116

Terimakasih
- Slides: 117