Managing Locally Advanced Oropharyngeal Cancer James A Bonner
Managing Locally Advanced Oropharyngeal Cancer James A. Bonner, MD The University of Alabama at Birmingham Department of Radiation Oncology Birmingham, Alabama, United States

Oropharynx Cancer • Historically primarily related to smoking and drinking • Human papillomavirus (HPV)-associated oropharyngeal cancers have been increasing rapidly • Should HPV status help determine treatment?

Guidelines Recommend That HPV Status Should Not Influence Treatment Decisions in SCCHN ‘The NCCN panel believes that HPV status should not be a routine consideration in treatment selection at this time’ 1 Standard of care is applicable to HPV+ and HPV– SCCHN until further evidence is available National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Head and Neck Cancers; version 2. 2017, 5/8/2017.
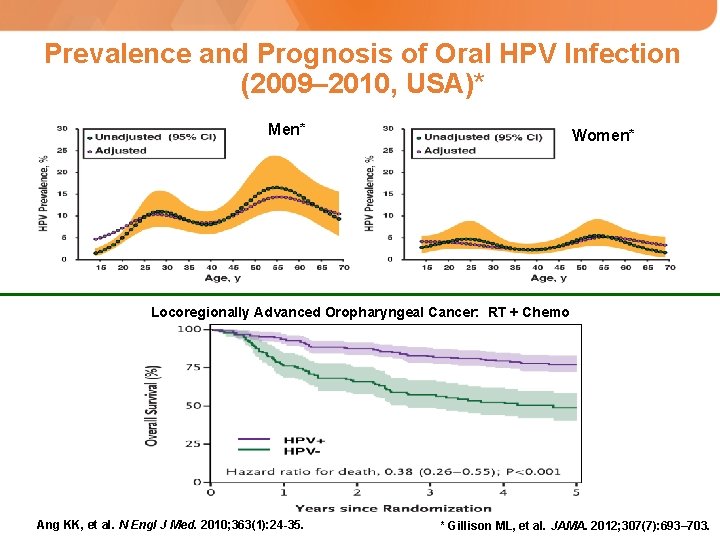
Prevalence and Prognosis of Oral HPV Infection (2009– 2010, USA)* Men* Women* Locoregionally Advanced Oropharyngeal Cancer: RT + Chemo Ang KK, et al. N Engl J Med. 2010; 363(1): 24 -35. * Gillison ML, et al. JAMA. 2012; 307(7): 693– 703.
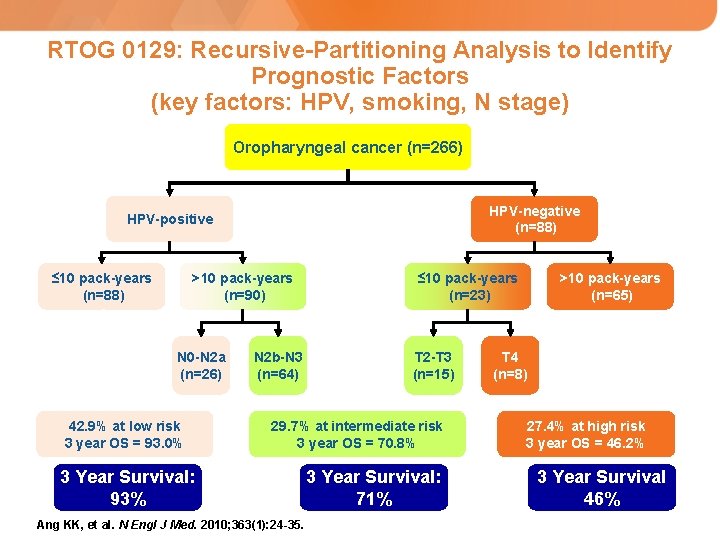
RTOG 0129: Recursive-Partitioning Analysis to Identify Prognostic Factors (key factors: HPV, smoking, N stage) Oropharyngeal cancer (n=266) HPV-negative (n=88) HPV-positive ≤ 10 pack-years (n=88) >10 pack-years (n=90) N 0 -N 2 a (n=26) 42. 9% at low risk 3 year OS = 93. 0% N 2 b-N 3 (n=64) ≤ 10 pack-years (n=23) T 2 -T 3 (n=15) 29. 7% at intermediate risk 3 year OS = 70. 8% 3 Year Survival: 93% Ang KK, et al. N Engl J Med. 2010; 363(1): 24 -35. 3 Year Survival: 71% >10 pack-years (n=65) T 4 (n=8) 27. 4% at high risk 3 year OS = 46. 2% 3 Year Survival 46%

Conclusions of Cleveland Clinic Experience • HPV-related oropharyngeal tumors present with larger nodal disease • With conventional treatments, HPV-positive patients do better than HPV-negative patients • HPV-positive patients are at risk for developing metastasis over an extended period of time and can live a prolonged time with metastatic disease Trossman SJ, et al. JAMA Otolaryngol Head Neck Surg. 2015; 141(5); 457 -462.
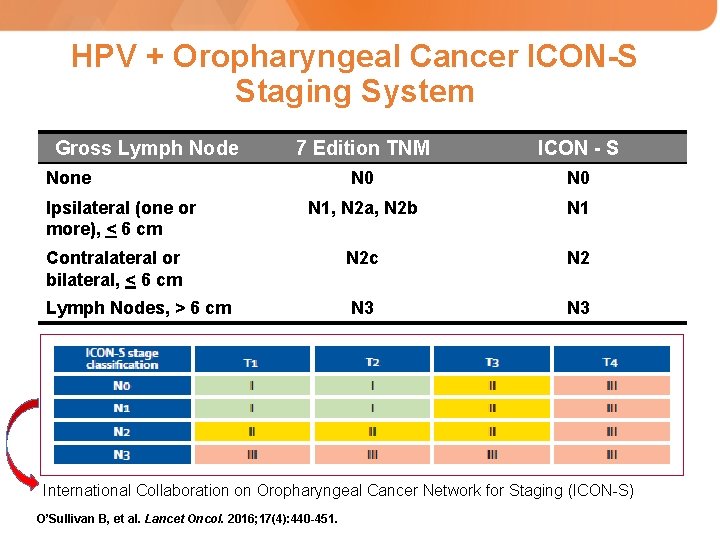
HPV + Oropharyngeal Cancer ICON-S Staging System Gross Lymph Node 7 Edition TNM ICON - S N 0 N 1, N 2 a, N 2 b N 1 Contralateral or bilateral, < 6 cm N 2 c N 2 Lymph Nodes, > 6 cm N 3 None Ipsilateral (one or more), < 6 cm International Collaboration on Oropharyngeal Cancer Network for Staging (ICON-S) O’Sullivan B, et al. Lancet Oncol. 2016; 17(4): 440 -451.
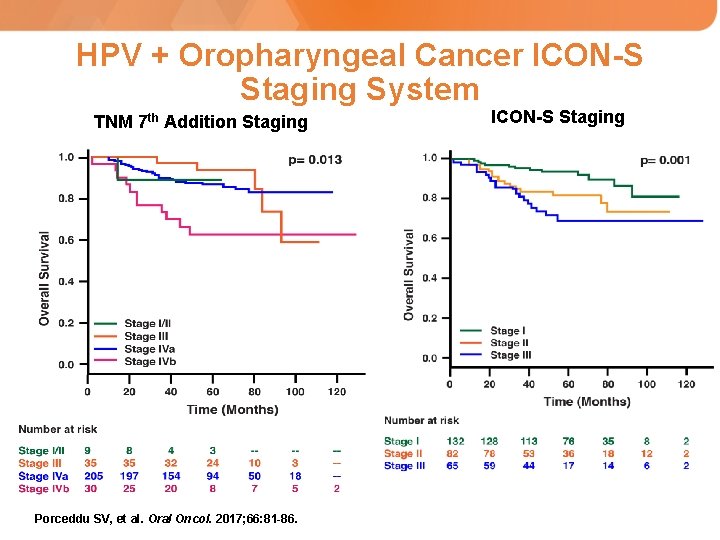
HPV + Oropharyngeal Cancer ICON-S Staging System TNM 7 th Addition Staging Porceddu SV, et al. Oral Oncol. 2017; 66: 81 -86. ICON-S Staging
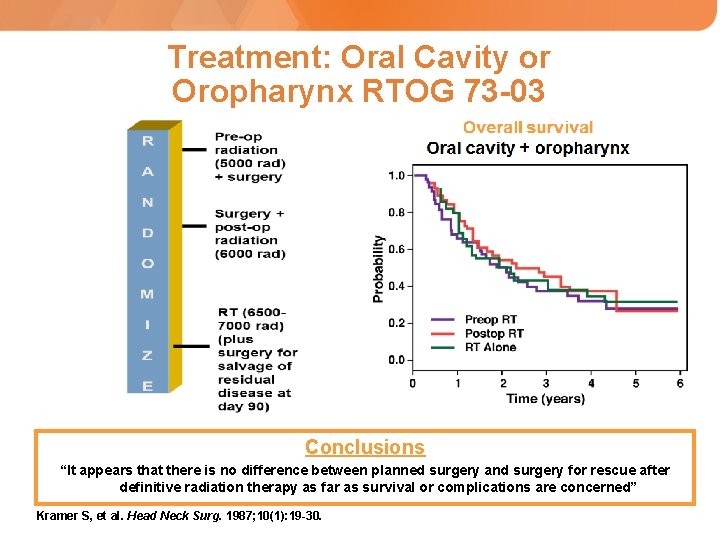
Treatment: Oral Cavity or Oropharynx RTOG 73 -03 Conclusions “It appears that there is no difference between planned surgery and surgery for rescue after definitive radiation therapy as far as survival or complications are concerned” Kramer S, et al. Head Neck Surg. 1987; 10(1): 19 -30.
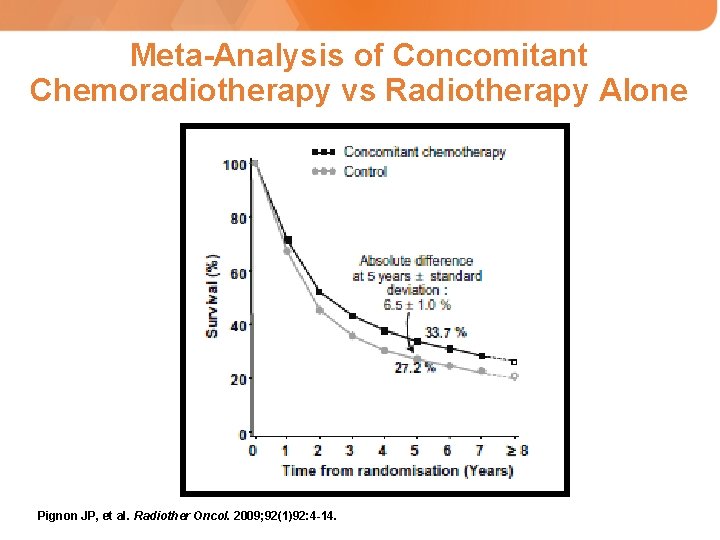
Meta-Analysis of Concomitant Chemoradiotherapy vs Radiotherapy Alone Pignon JP, et al. Radiother Oncol. 2009; 92(1)92: 4 -14.
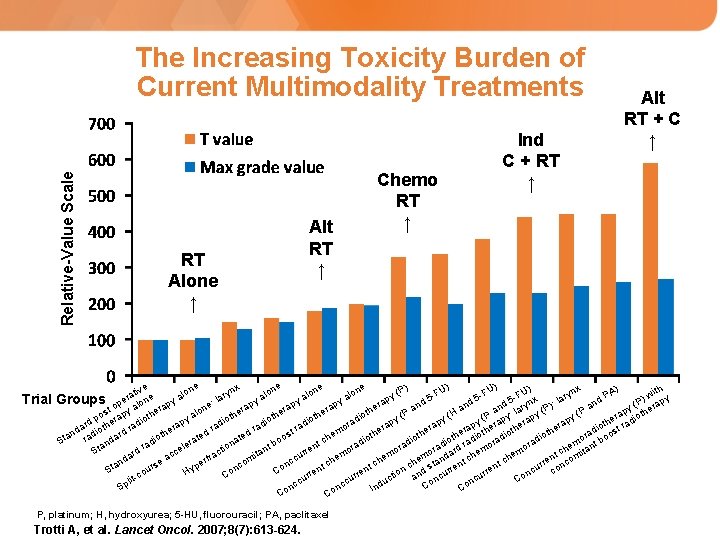
Relative-Value Scale The Increasing Toxicity Burden of Current Multimodality Treatments RT Alone ↑ Alt RT ↑ Chemo RT ↑ Ind C + RT ↑ Alt RT + C ↑ e e e ) e e x ) U) U) ith nx U) (P tiv e lon lon -F ryn -F F PA a y a a r 5 a 5 ) w py l ary x p d 5 l e lon y y : P d n d a n : p p ( p p d y ) a ra er a ne y an an ra rap to an : lar y (P (P H ap iothe he oth os rapy othe he he alo (P ( P r i t t y ( p p e d o y y p o p i i y ra p ra e p p di th ad di ra rd ra rad rap the iothe era da dioth rd ra era dio ost r he mo t ra t e d d h n h h a s e t t t o o r h e i e a o bo io ra nda io io di ch oo at at iot ad St rad em tant nt tb ler a rad rad ad rd r mo ora ion h e r n e t o o r d St c m c o c i a e r r a t em em nt com ac he cu fra da mi em tand nt ch r h h c e c n e o r h c c e t n r a p s n nc re nt nt urs nc cu con St Co Hy rre Co co tio and ncur rre on u c u t u c C i l u c c n Co nc nc Sp Ind Co Co Co Trial Groups P, platinum; H, hydroxyurea; 5 -HU, fluorouracil; PA, paclitaxel Trotti A, et al. Lancet Oncol. 2007; 8(7): 613 -624.
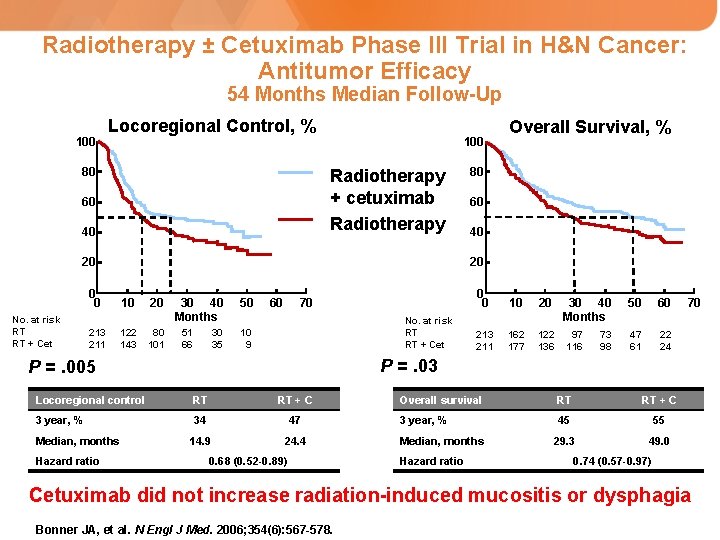
Radiotherapy ± Cetuximab Phase III Trial in H&N Cancer: Antitumor Efficacy 54 Months Median Follow-Up 100 Locoregional Control, % 80 100 Radiotherapy + cetuximab Radiotherapy 60 40 20 0 0 No. at risk RT RT + Cet 213 211 10 122 143 20 80 101 30 40 Months 51 66 30 35 50 60 70 No. at risk RT RT + Cet 10 9 60 40 0 0 10 20 213 211 162 177 122 136 30 40 Months 97 116 73 98 50 60 47 61 22 24 70 P =. 03 Locoregional control RT RT + C 3 year, % 34 47 14. 9 24. 4 Hazard ratio 80 20 P =. 005 Median, months Overall Survival, % 0. 68 (0. 52 -0. 89) Overall survival RT RT + C 3 year, % 45 55 29. 3 49. 0 Median, months Hazard ratio 0. 74 (0. 57 -0. 97) Cetuximab did not increase radiation-induced mucositis or dysphagia Bonner JA, et al. N Engl J Med. 2006; 354(6): 567 -578.
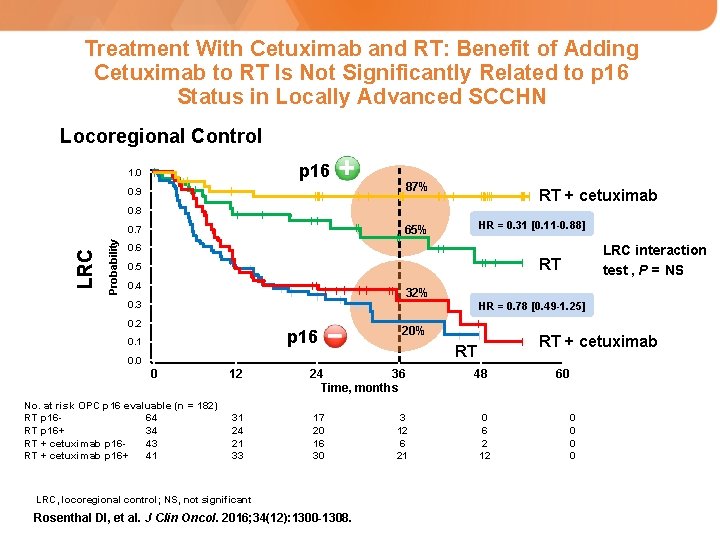
Treatment With Cetuximab and RT: Benefit of Adding Cetuximab to RT Is Not Significantly Related to p 16 Status in Locally Advanced SCCHN Locoregional Control p 16 1. 0 87% 0. 9 RT + cetuximab 0. 8 Probability LRC 0. 7 HR = 0. 31 [0. 11 -0. 88] 65% 0. 6 LRC interaction test , P = NS RT 0. 5 0. 4 32% 0. 3 HR = 0. 78 [0. 49 -1. 25] 0. 2 p 16 0. 1 20% 0. 0 0 No. at risk OPC p 16 evaluable (n = 182) RT p 1664 RT p 16+ 34 RT + cetuximab p 1643 RT + cetuximab p 16+ 41 12 31 24 21 33 24 36 Time, months 17 20 16 30 LRC, locoregional control; NS, not significant Rosenthal DI, et al. J Clin Oncol. 2016; 34(12): 1300 -1308. RT + cetuximab RT 3 12 6 21 48 0 6 2 12 60 0 0
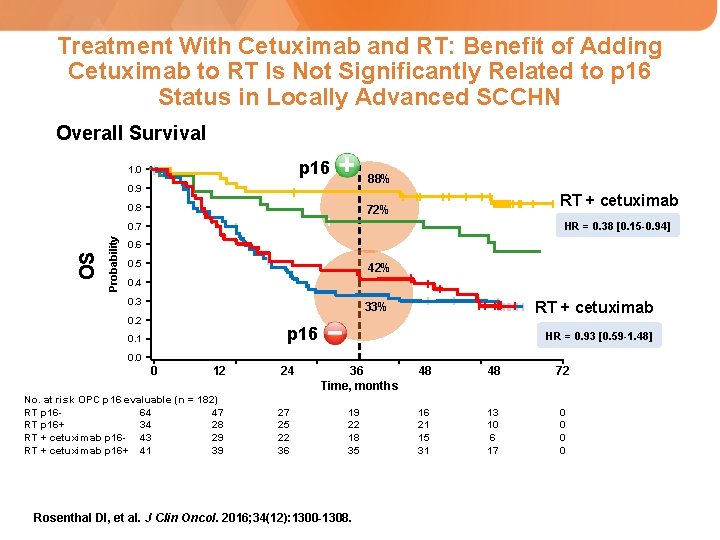
Treatment With Cetuximab and RT: Benefit of Adding Cetuximab to RT Is Not Significantly Related to p 16 Status in Locally Advanced SCCHN Overall Survival p 16 1. 0 88% 0. 9 0. 8 RT + cetuximab 72% Probability OS 0. 7 HR = 0. 38 [0. 15 -0. 94] 0. 6 RT 0. 5 42% 0. 4 0. 3 33% 0. 2 RT p 16 0. 1 RT + cetuximab HR = 0. 93 [0. 59 -1. 48] 0. 0 0 12 No. at risk OPC p 16 evaluable (n = 182) RT p 1664 47 RT p 16+ 34 28 RT + cetuximab p 16 - 43 29 RT + cetuximab p 16+ 41 39 24 27 25 22 36 36 Time, months 19 22 18 35 Rosenthal DI, et al. J Clin Oncol. 2016; 34(12): 1300 -1308. 48 48 72 16 21 15 31 13 10 6 17 0 0
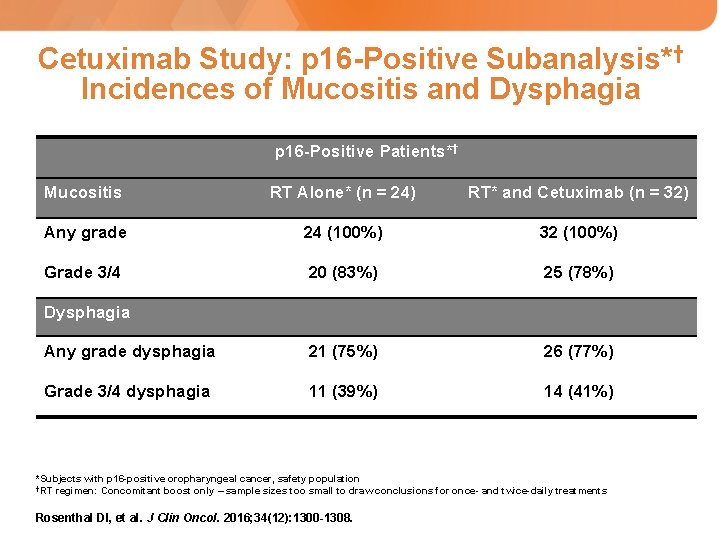
Cetuximab Study: p 16 -Positive Subanalysis*† Incidences of Mucositis and Dysphagia p 16 -Positive Patients*† Mucositis RT Alone* (n = 24) RT* and Cetuximab (n = 32) Any grade 24 (100%) 32 (100%) Grade 3/4 20 (83%) 25 (78%) Any grade dysphagia 21 (75%) 26 (77%) Grade 3/4 dysphagia 11 (39%) 14 (41%) Dysphagia *Subjects with p 16 -positive oropharyngeal cancer, safety population †RT regimen: Concomitant boost only – sample sizes too small to draw conclusions for once- and twice-daily treatments Rosenthal DI, et al. J Clin Oncol. 2016; 34(12): 1300 -1308.

Current Considerations in the Treatment of Oropharynx Cancer Deintensification of Treatment for HPV Positive Tumors • Less radiation dose • Less chemotherapy dose • Less concomitant therapy
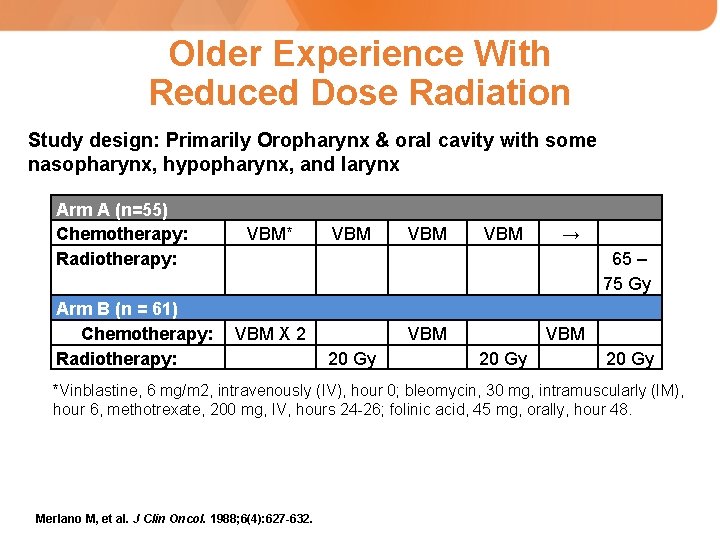
Older Experience With Reduced Dose Radiation Study design: Primarily Oropharynx & oral cavity with some nasopharynx, hypopharynx, and larynx Arm A (n=55) Chemotherapy: Radiotherapy: Arm B (n = 61) Chemotherapy: Radiotherapy: VBM* VBM VBM → 65 – 75 Gy VBM X 2 VBM 20 Gy *Vinblastine, 6 mg/m 2, intravenously (IV), hour 0; bleomycin, 30 mg, intramuscularly (IM), hour 6, methotrexate, 200 mg, IV, hours 24 -26; folinic acid, 45 mg, orally, hour 48. Merlano M, et al. J Clin Oncol. 1988; 6(4): 627 -632.
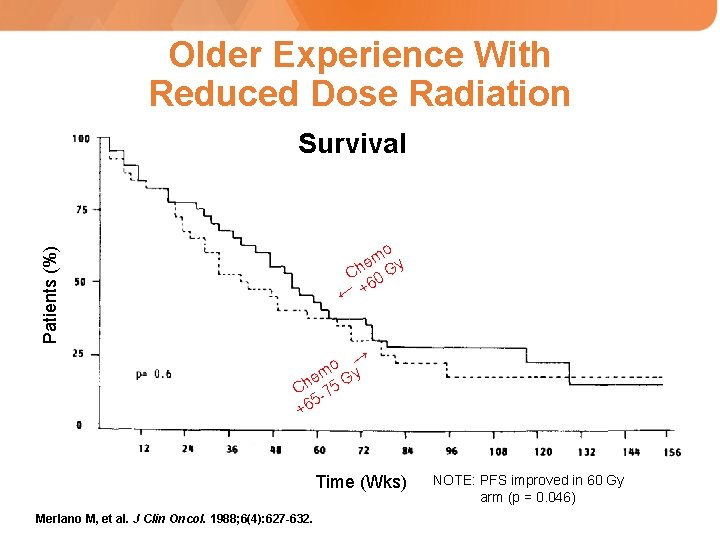
Older Experience With Reduced Dose Radiation Survival Patients (%) mo y e Ch 0 G +6 → → mo Gy e Ch -75 5 +6 Time (Wks) Merlano M, et al. J Clin Oncol. 1988; 6(4): 627 -632. NOTE: PFS improved in 60 Gy arm (p = 0. 046)
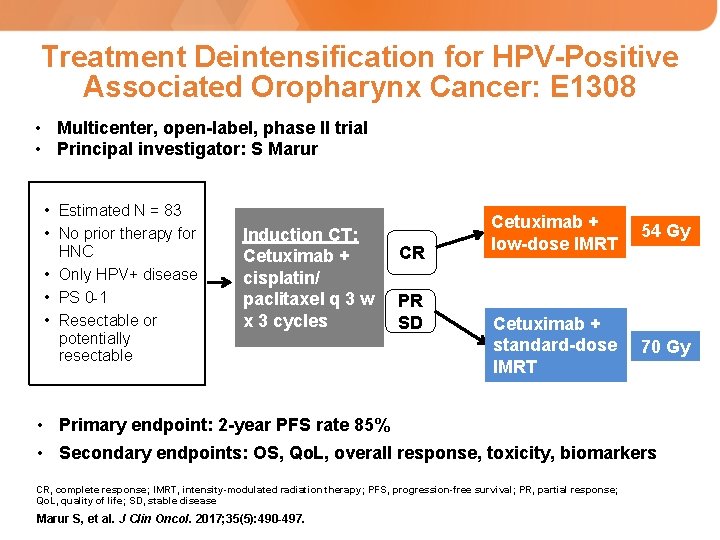
Treatment Deintensification for HPV-Positive Associated Oropharynx Cancer: E 1308 • Multicenter, open-label, phase II trial • Principal investigator: S Marur • Estimated N = 83 • No prior therapy for HNC • Only HPV+ disease • PS 0 -1 • Resectable or potentially resectable Induction CT: Cetuximab + cisplatin/ paclitaxel q 3 w x 3 cycles CR PR SD Cetuximab + low-dose IMRT 54 Gy Cetuximab + standard-dose IMRT 70 Gy • Primary endpoint: 2 -year PFS rate 85% • Secondary endpoints: OS, Qo. L, overall response, toxicity, biomarkers CR, complete response; IMRT, intensity-modulated radiation therapy; PFS, progression-free survival; PR, partial response; Qo. L, quality of life; SD, stable disease Marur S, et al. J Clin Oncol. 2017; 35(5): 490 -497.
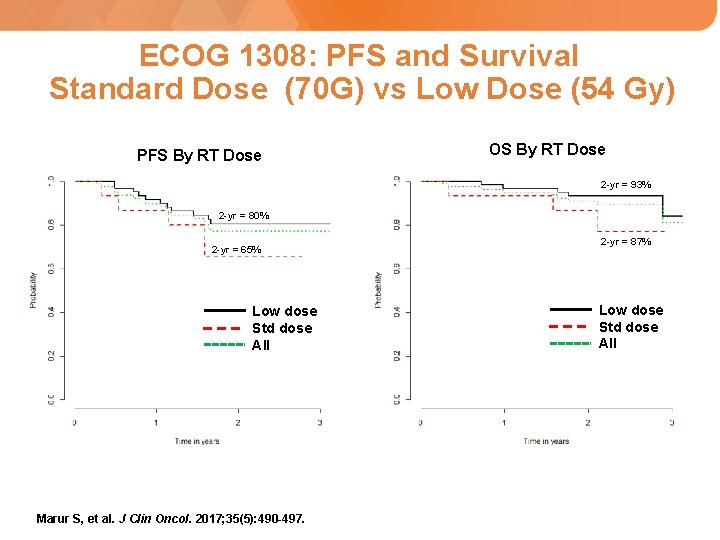
ECOG 1308: PFS and Survival Standard Dose (70 G) vs Low Dose (54 Gy) PFS By RT Dose OS By RT Dose 2 -yr = 93% 2 -yr = 80% 2 -yr = 65% Low dose Std dose All Marur S, et al. J Clin Oncol. 2017; 35(5): 490 -497. 2 -yr = 87% Low dose Std dose All
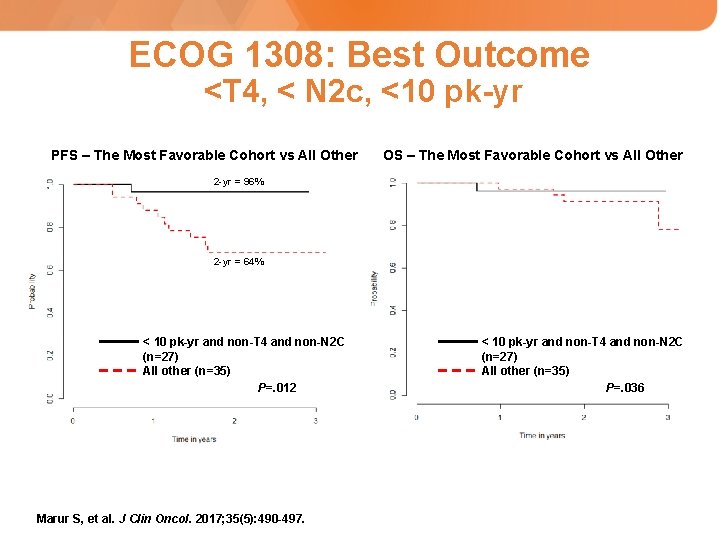
ECOG 1308: Best Outcome <T 4, < N 2 c, <10 pk-yr PFS – The Most Favorable Cohort vs All Other OS – The Most Favorable Cohort vs All Other 2 -yr = 96% 2 -yr = 64% < 10 pk-yr and non-T 4 and non-N 2 C (n=27) All other (n=35) P=. 012 Marur S, et al. J Clin Oncol. 2017; 35(5): 490 -497. < 10 pk-yr and non-T 4 and non-N 2 C (n=27) All other (n=35) P=. 036
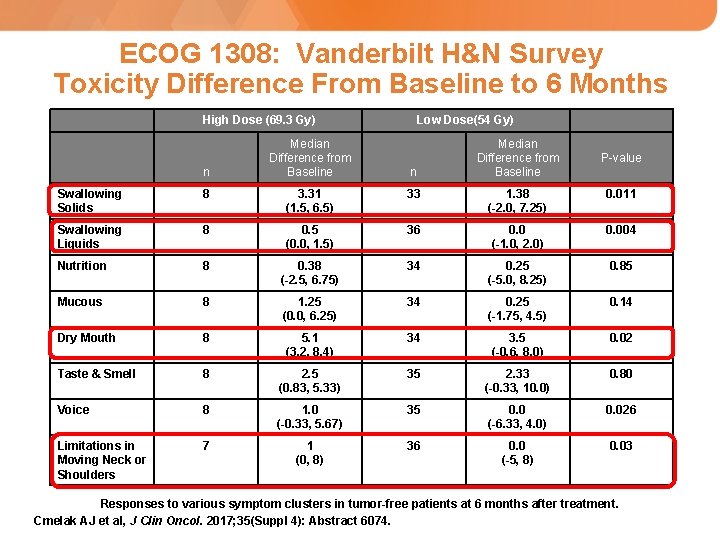
ECOG 1308: Vanderbilt H&N Survey Toxicity Difference From Baseline to 6 Months High Dose (69. 3 Gy) n Median Difference from Baseline Low Dose(54 Gy) n Median Difference from Baseline P-value Swallowing Solids 8 3. 31 (1. 5, 6. 5) 33 1. 38 (-2. 0, 7. 25) 0. 011 Swallowing Liquids 8 0. 5 (0. 0, 1. 5) 36 0. 0 (-1. 0, 2. 0) 0. 004 Nutrition 8 0. 38 (-2. 5, 6. 75) 34 0. 25 (-5. 0, 8. 25) 0. 85 Mucous 8 1. 25 (0. 0, 6. 25) 34 0. 25 (-1. 75, 4. 5) 0. 14 Dry Mouth 8 5. 1 (3. 2, 8. 4) 34 3. 5 (-0. 6, 8. 0) 0. 02 Taste & Smell 8 2. 5 (0. 83, 5. 33) 35 2. 33 (-0. 33, 10. 0) 0. 80 Voice 8 1. 0 (-0. 33, 5. 67) 35 0. 0 (-6. 33, 4. 0) 0. 026 Limitations in Moving Neck or Shoulders 7 1 (0, 8) 36 0. 0 (-5, 8) 0. 03 Responses to various symptom clusters in tumor-free patients at 6 months after treatment. Cmelak AJ et al, J Clin Oncol. 2017; 35(Suppl 4): Abstract 6074.
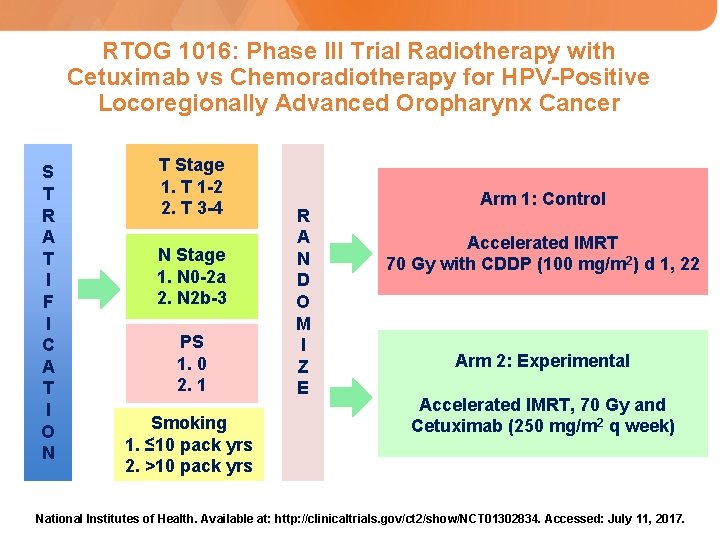
RTOG 1016: Phase III Trial Radiotherapy with Cetuximab vs Chemoradiotherapy for HPV-Positive Locoregionally Advanced Oropharynx Cancer S T R A T I F I C A T I O N T Stage 1. T 1 -2 2. T 3 -4 N Stage 1. N 0 -2 a 2. N 2 b-3 PS 1. 0 2. 1 Smoking 1. ≤ 10 pack yrs 2. >10 pack yrs R A N D O M I Z E Arm 1: Control Accelerated IMRT 70 Gy with CDDP (100 mg/m 2) d 1, 22 Arm 2: Experimental Accelerated IMRT, 70 Gy and Cetuximab (250 mg/m 2 q week) National Institutes of Health. Available at: http: //clinicaltrials. gov/ct 2/show/NCT 01302834. Accessed: July 11, 2017.

Princess Margaret Hospital: Evaluation of Chemotherapy Doses • Retrospective review of newly diagnosed HNSCC patients from 2000 -2012, who had been treated with cisplatin-based chemoradiotherapy, and in whom the HPV status had been ascertained. • 659 patients identified – 404 HPV+ (95% OPC) – 255 HPV- (38% OPC) • Predictors of overall survival were identified in the HPV+ and HPV- patients including total cisplatin dose given (>200 mg/m 2 vs. <200 mg/m 2) Spreafico A, et al. Eur J Cancer. 2016; 67: 174 -182.
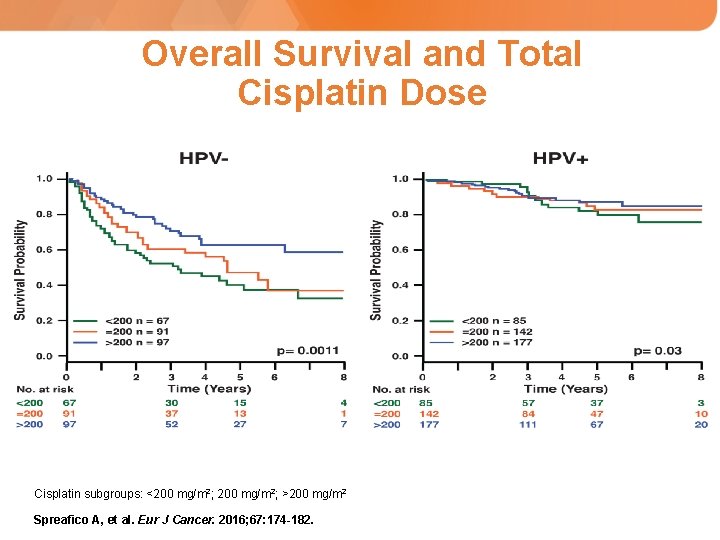
Overall Survival and Total Cisplatin Dose Cisplatin subgroups: <200 mg/m 2; >200 mg/m 2 Spreafico A, et al. Eur J Cancer. 2016; 67: 174 -182.
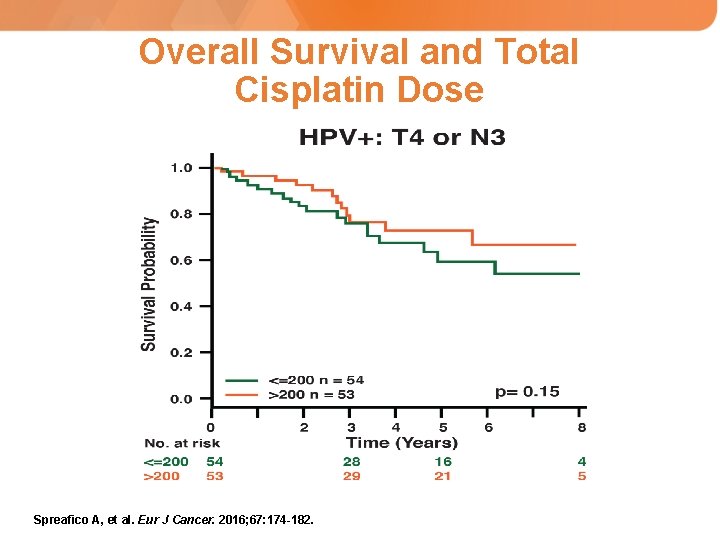
Overall Survival and Total Cisplatin Dose Spreafico A, et al. Eur J Cancer. 2016; 67: 174 -182.

HPV- Related Tumors • HPV-related tumors do well compared to HPV negative tumor • Induction chemotherapy may be appropriate for some patients – yet unknown • In the future, deintensification may be possible for some HPV+ tumors
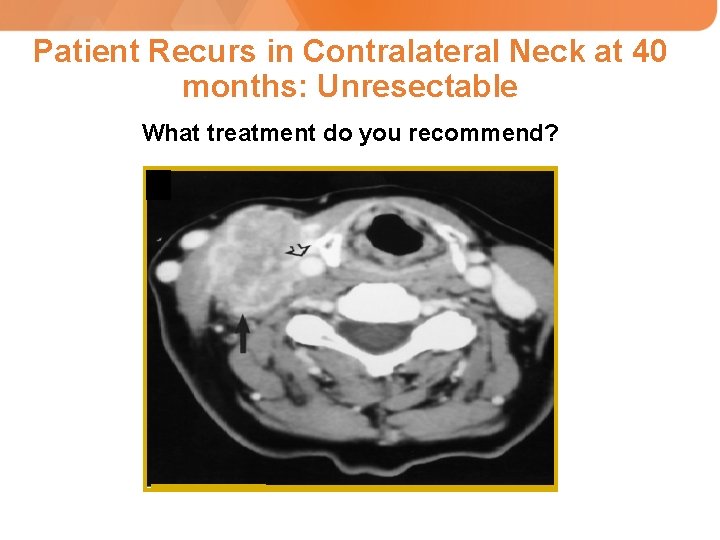
Patient Recurs in Contralateral Neck at 40 months: Unresectable What treatment do you recommend?
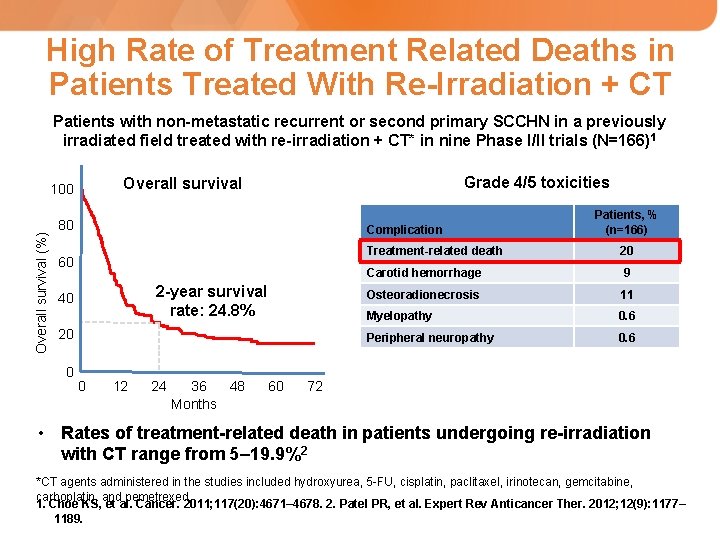
High Rate of Treatment Related Deaths in Patients Treated With Re-Irradiation + CT Patients with non-metastatic recurrent or second primary SCCHN in a previously irradiated field treated with re-irradiation + CT* in nine Phase I/II trials (N=166) 1 Overall survival (%) Grade 4/5 toxicities Overall survival 100 80 Complication 60 2 -year survival rate: 24. 8% 40 20 0 0 12 24 36 48 Months 60 Patients, % (n=166) Treatment-related death 20 Carotid hemorrhage 9 Osteoradionecrosis 11 Myelopathy 0. 6 Peripheral neuropathy 0. 6 72 • Rates of treatment-related death in patients undergoing re-irradiation with CT range from 5– 19. 9%2 *CT agents administered in the studies included hydroxyurea, 5 -FU, cisplatin, paclitaxel, irinotecan, gemcitabine, carboplatin, and pemetrexed 1. Choe KS, et al. Cancer. 2011; 117(20): 4671– 4678. 2. Patel PR, et al. Expert Rev Anticancer Ther. 2012; 12(9): 1177– 1189.
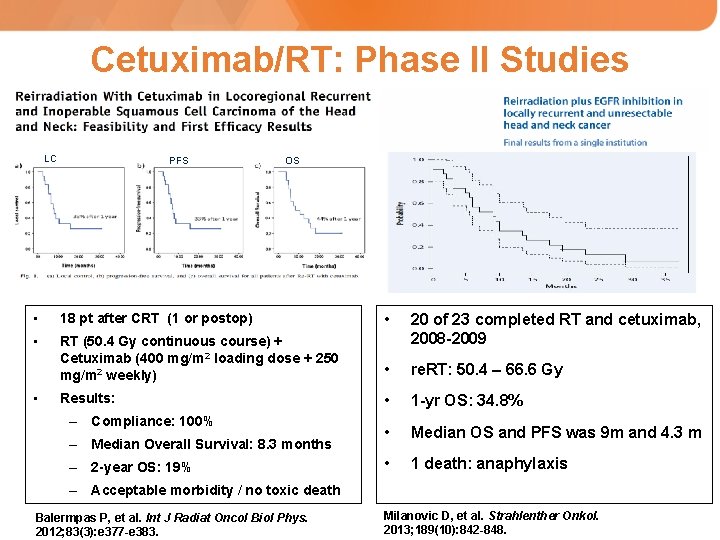
Cetuximab/RT: Phase II Studies LC PFS OS • 18 pt after CRT (1 or postop) • • RT (50. 4 Gy continuous course) + Cetuximab (400 mg/m 2 loading dose + 250 mg/m 2 weekly) 20 of 23 completed RT and cetuximab, 2008 -2009 • re. RT: 50. 4 – 66. 6 Gy Results: • 1 -yr OS: 34. 8% • Median OS and PFS was 9 m and 4. 3 m • 1 death: anaphylaxis • – Compliance: 100% – Median Overall Survival: 8. 3 months – 2 -year OS: 19% – Acceptable morbidity / no toxic death Balermpas P, et al. Int J Radiat Oncol Biol Phys. 2012; 83(3): e 377 -e 383. Milanovic D, et al. Strahlenther Onkol. 2013; 189(10): 842 -848.
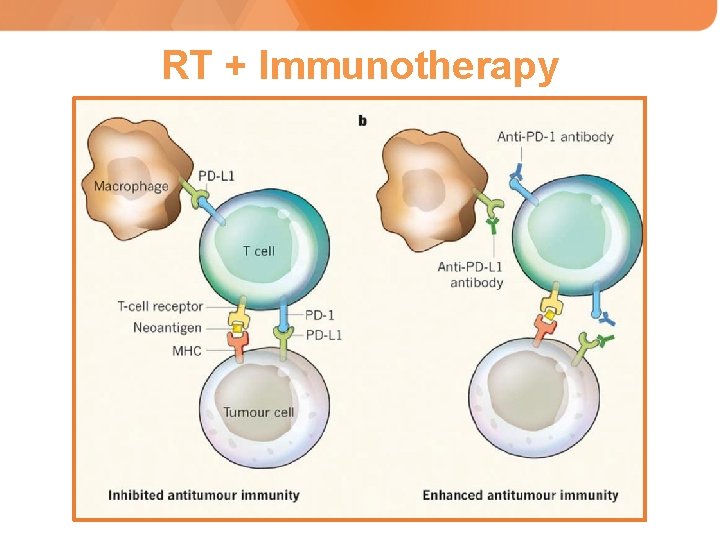
RT + Immunotherapy
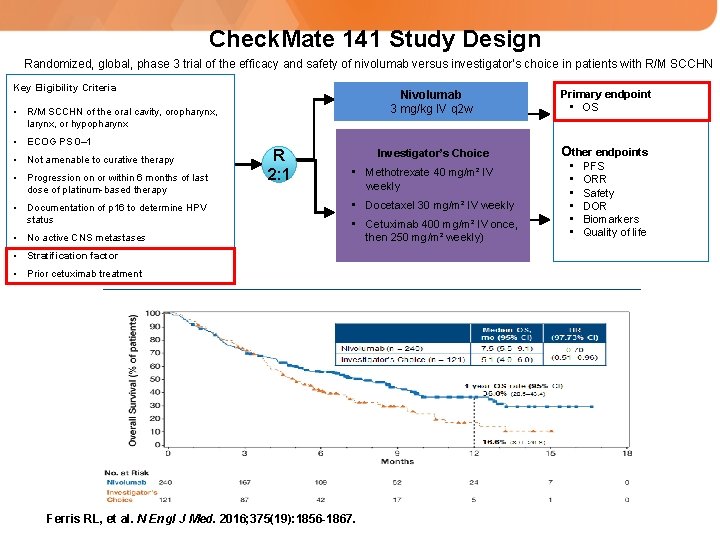
Check. Mate 141 Study Design Randomized, global, phase 3 trial of the efficacy and safety of nivolumab versus investigator’s choice in patients with R/M SCCHN Key Eligibility Criteria • R/M SCCHN of the oral cavity, oropharynx, larynx, or hypopharynx • ECOG PS 0– 1 • Not amenable to curative therapy • Progression on or within 6 months of last dose of platinum-based therapy • Documentation of p 16 to determine HPV status • No active CNS metastases R 2: 1 Nivolumab 3 mg/kg IV q 2 w Primary endpoint • OS Investigator’s Choice Other endpoints • Methotrexate 40 mg/m² IV weekly • Docetaxel 30 mg/m² IV weekly • Cetuximab 400 mg/m² IV once, then 250 mg/m² weekly) • Stratification factor • Prior cetuximab treatment Ferris RL, et al. N Engl J Med. 2016; 375(19): 1856 -1867. • • • PFS ORR Safety DOR Biomarkers Quality of life
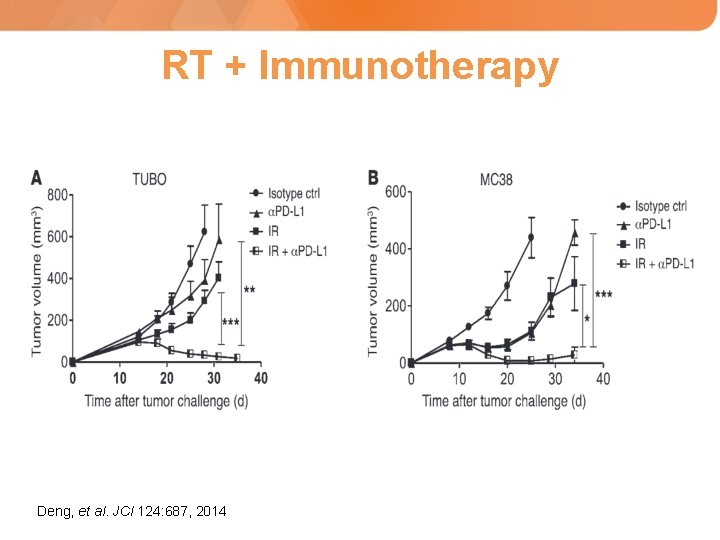
RT + Immunotherapy Deng, et al. JCI 124: 687, 2014
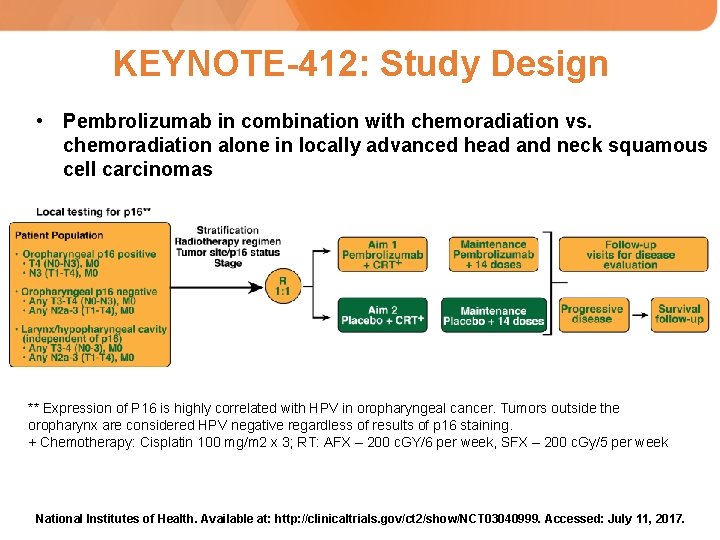
KEYNOTE-412: Study Design • Pembrolizumab in combination with chemoradiation vs. chemoradiation alone in locally advanced head and neck squamous cell carcinomas ** Expression of P 16 is highly correlated with HPV in oropharyngeal cancer. Tumors outside the oropharynx are considered HPV negative regardless of results of p 16 staining. + Chemotherapy: Cisplatin 100 mg/m 2 x 3; RT: AFX – 200 c. GY/6 per week, SFX – 200 c. Gy/5 per week National Institutes of Health. Available at: http: //clinicaltrials. gov/ct 2/show/NCT 03040999. Accessed: July 11, 2017.
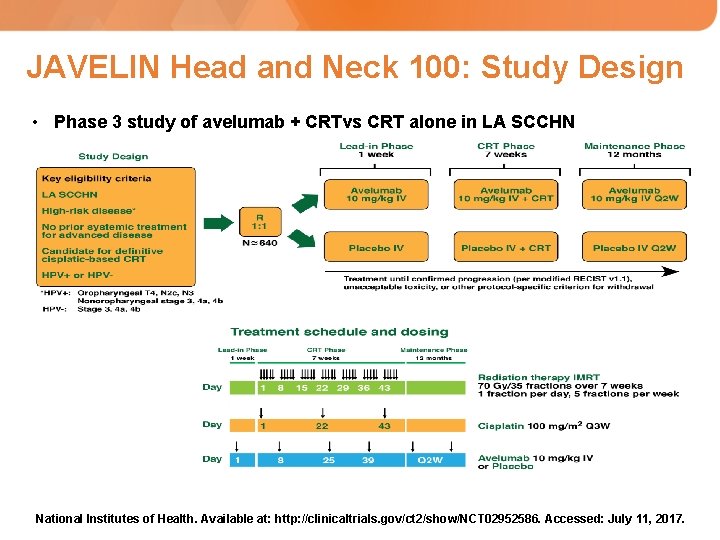
JAVELIN Head and Neck 100: Study Design • Phase 3 study of avelumab + CRTvs CRT alone in LA SCCHN National Institutes of Health. Available at: http: //clinicaltrials. gov/ct 2/show/NCT 02952586. Accessed: July 11, 2017.

Conclusions • Oropharyngeal cancers have greatly evolved over the last 20 years • HPV has changed prognosis and the direction of our studies • The appropriate integration of cisplatin, cetuximab, surgery, and RT remains the subject of investigation

Extra Slides
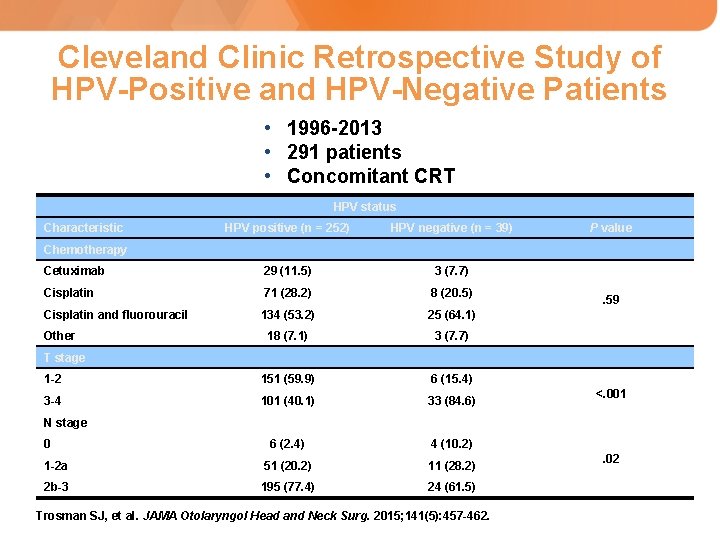
Cleveland Clinic Retrospective Study of HPV-Positive and HPV-Negative Patients • 1996 -2013 • 291 patients • Concomitant CRT HPV status Characteristic HPV positive (n = 252) HPV negative (n = 39) Cetuximab 29 (11. 5) 3 (7. 7) Cisplatin 71 (28. 2) 8 (20. 5) Cisplatin and fluorouracil 134 (53. 2) 25 (64. 1) 18 (7. 1) 3 (7. 7) 1 -2 151 (59. 9) 6 (15. 4) 3 -4 101 (40. 1) 33 (84. 6) 6 (2. 4) 4 (10. 2) 1 -2 a 51 (20. 2) 11 (28. 2) 2 b-3 195 (77. 4) 24 (61. 5) P value Chemotherapy Other . 59 T stage <. 001 N stage 0 Trosman SJ, et al. JAMA Otolaryngol Head and Neck Surg. 2015; 141(5): 457 -462. . 02
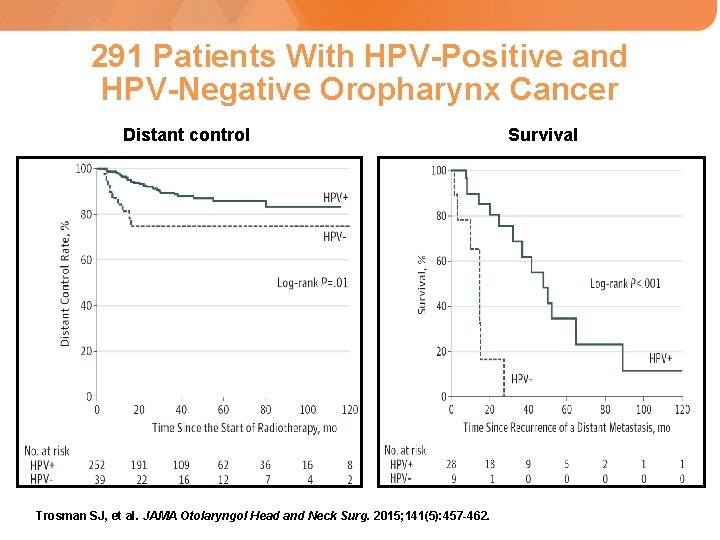
291 Patients With HPV-Positive and HPV-Negative Oropharynx Cancer Distant control Trosman SJ, et al. JAMA Otolaryngol Head and Neck Surg. 2015; 141(5): 457 -462. Survival
Cancer of the Oropharynx
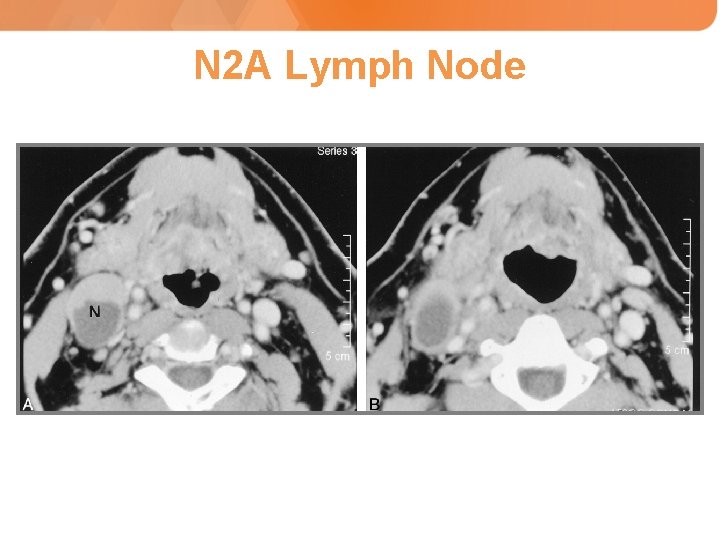
N 2 A Lymph Node
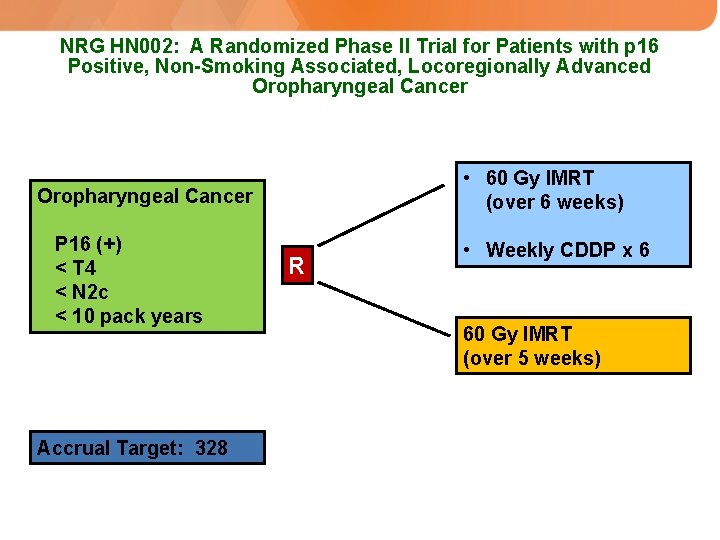
NRG HN 002: A Randomized Phase II Trial for Patients with p 16 Positive, Non-Smoking Associated, Locoregionally Advanced Oropharyngeal Cancer • 60 Gy IMRT (over 6 weeks) Oropharyngeal Cancer P 16 (+) < T 4 < N 2 c < 10 pack years Accrual Target: 328 R • Weekly CDDP x 6 60 Gy IMRT (over 5 weeks)
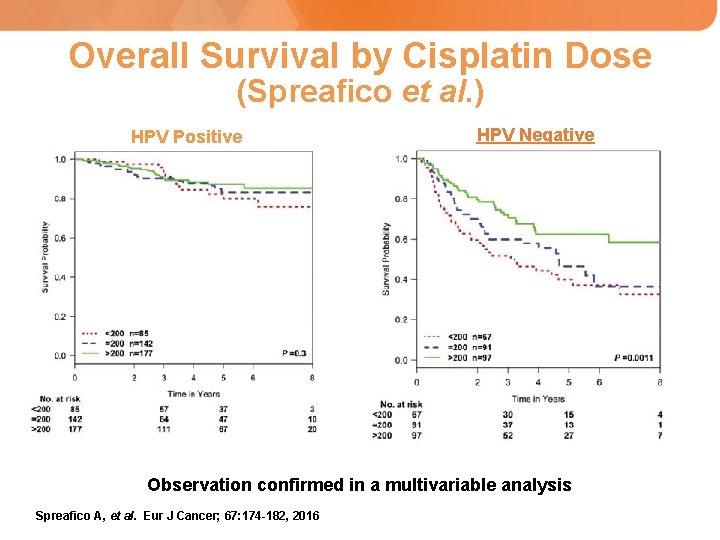
Overall Survival by Cisplatin Dose (Spreafico et al. ) HPV Positive HPV Negative Observation confirmed in a multivariable analysis Spreafico A, et al. Eur J Cancer; 67: 174 -182, 2016
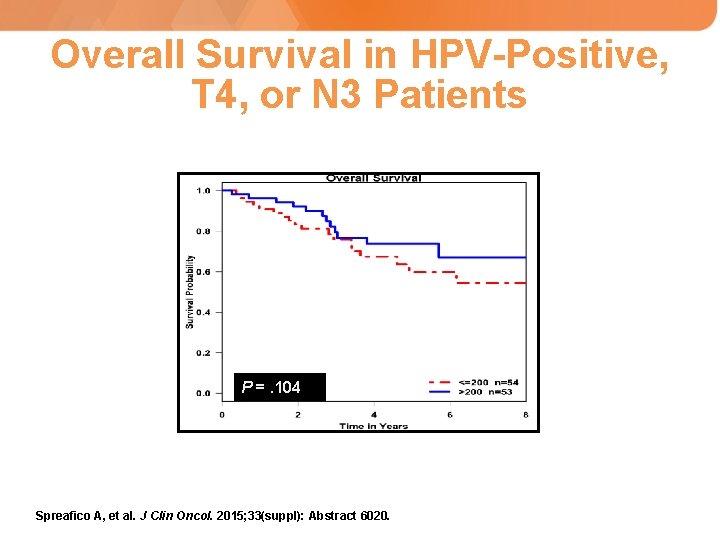
Overall Survival in HPV-Positive, T 4, or N 3 Patients P =. 104 Spreafico A, et al. J Clin Oncol. 2015; 33(suppl): Abstract 6020.
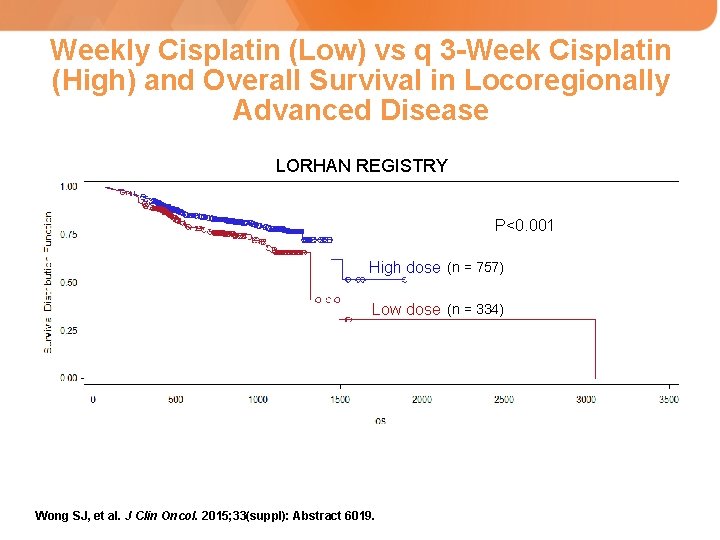
Weekly Cisplatin (Low) vs q 3 -Week Cisplatin (High) and Overall Survival in Locoregionally Advanced Disease LORHAN REGISTRY P<0. 001 High dose (n = 757) Low dose (n = 334) Wong SJ, et al. J Clin Oncol. 2015; 33(suppl): Abstract 6019.
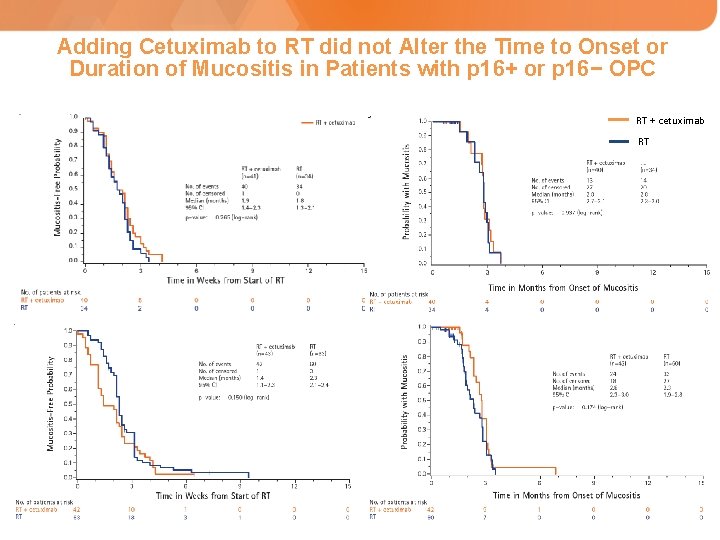
Adding Cetuximab to RT did not Alter the Time to Onset or Duration of Mucositis in Patients with p 16+ or p 16− OPC Time to Onset Mucositis Duration RT + cetuximab p 16− OPC p 16+ OPC RT a All grades of mucositis were considered.
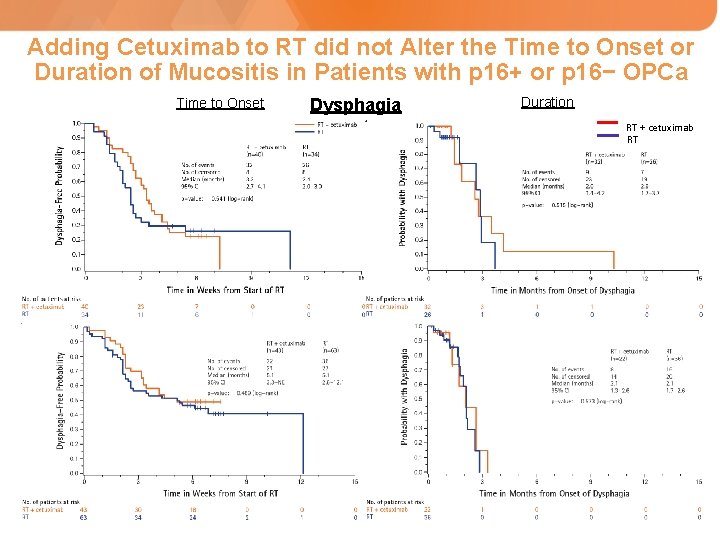
Adding Cetuximab to RT did not Alter the Time to Onset or Duration of Mucositis in Patients with p 16+ or p 16− OPCa Time to Onset Dysphagia Duration RT + cetuximab RT p 16− OPC p 16+ OPC Dysphagia a All grades of dysphagia were considered.
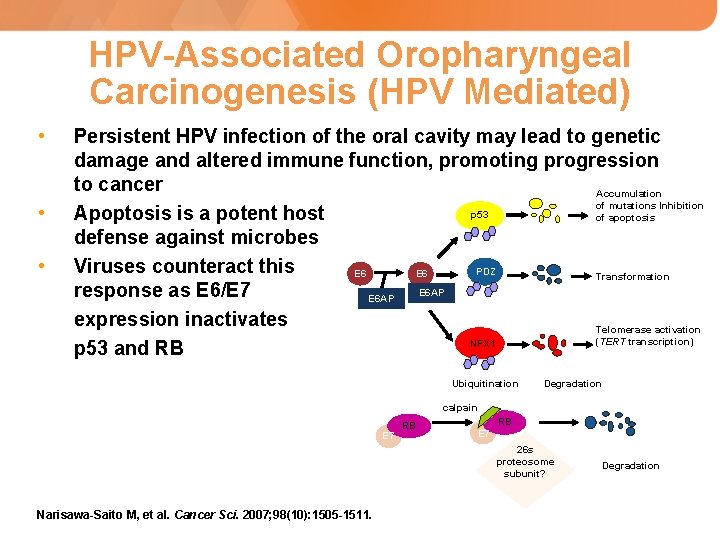
HPV-Associated Oropharyngeal Carcinogenesis (HPV Mediated) • • • Persistent HPV infection of the oral cavity may lead to genetic damage and altered immune function, promoting progression to cancer Accumulation of mutations Inhibition p 53 Apoptosis is a potent host of apoptosis defense against microbes Viruses counteract this PDZ E 6 Transformation E 6 AP response as E 6/E 7 E 6 AP expression inactivates Telomerase activation (TERT transcription) NFX 1 p 53 and RB Ubiquitination Degradation calpain E 7 RB RB E 7 26 s proteosome subunit? Narisawa-Saito M, et al. Cancer Sci. 2007; 98(10): 1505 -1511. Degradation
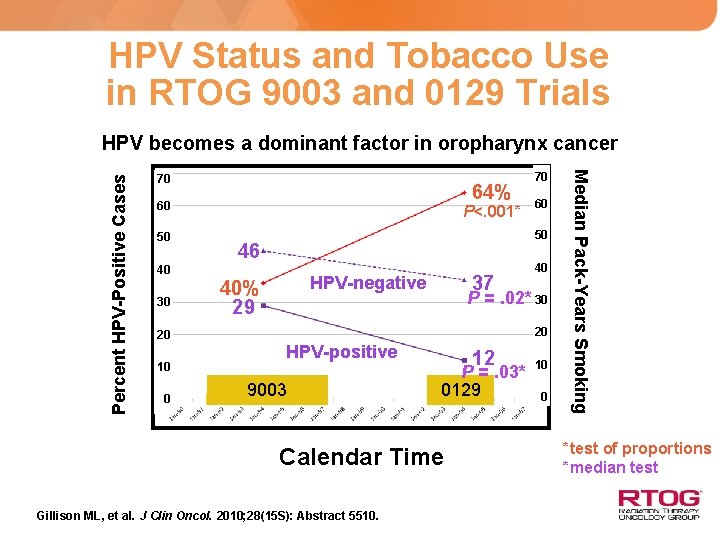
HPV Status and Tobacco Use in RTOG 9003 and 0129 Trials 70 64% 60 50 P<. 001* 46 37 HPV-negative 40% 29 0 40 P =. 02* 30 20 20 10 60 50 40 30 70 HPV-positive 9003 Calendar Time Gillison ML, et al. J Clin Oncol. 2010; 28(15 S): Abstract 5510. 12 P =. 03* 0129 10 0 Median Pack-Years Smoking Percent HPV-Positive Cases HPV becomes a dominant factor in oropharynx cancer *test of proportions *median test
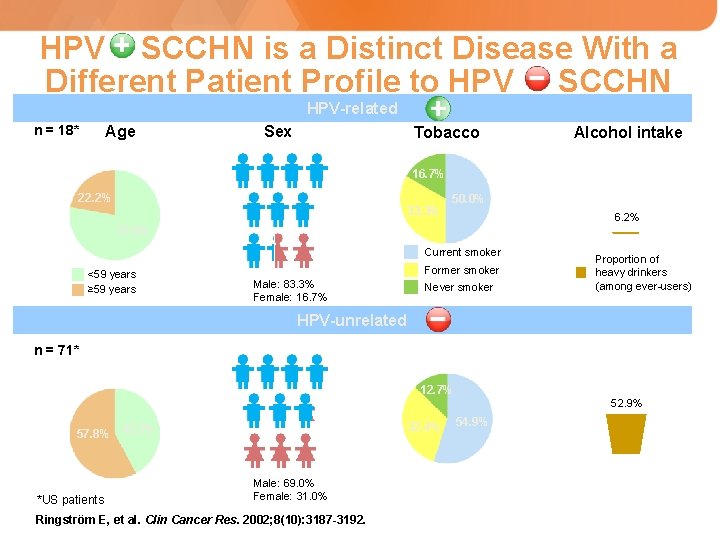
HPV SCCHN is a Distinct Disease With a Different Patient Profile to HPV SCCHN HPV-related n = 18* Age Sex Tobacco Alcohol intake 16. 7% 22. 2% 33. 3% 50. 0% 6. 2% 77. 8% Current smoker <59 years ≥ 59 years Former smoker Male: 83. 3% Female: 16. 7% Never smoker Proportion of heavy drinkers (among ever-users) HPV-unrelated n = 71* 12. 7% 52. 9% 32. 4% 57. 8% 42. 2% *US patients Male: 69. 0% Female: 31. 0% Ringström E, et al. Clin Cancer Res. 2002; 8(10): 3187 -3192. 54. 9%
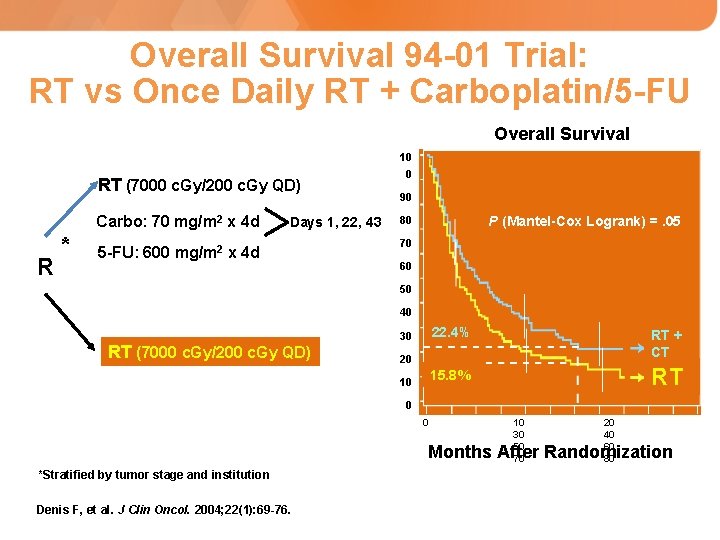
Overall Survival 94 -01 Trial: RT vs Once Daily RT + Carboplatin/5 -FU Overall Survival 10 RT (7000 c. Gy/200 c. Gy QD) R * Carbo: 70 mg/m 2 x 4 d Days 1, 22, 43 5 -FU: 600 mg/m 2 x 4 d 0 90 P (Mantel-Cox Logrank) =. 05 80 70 60 50 40 30 RT (7000 c. Gy/200 c. Gy QD) 22. 4% RT + CT 15. 8% RT 20 10 0 0 10 30 50 70 20 40 60 80 Months After Randomization *Stratified by tumor stage and institution Denis F, et al. J Clin Oncol. 2004; 22(1): 69 -76.
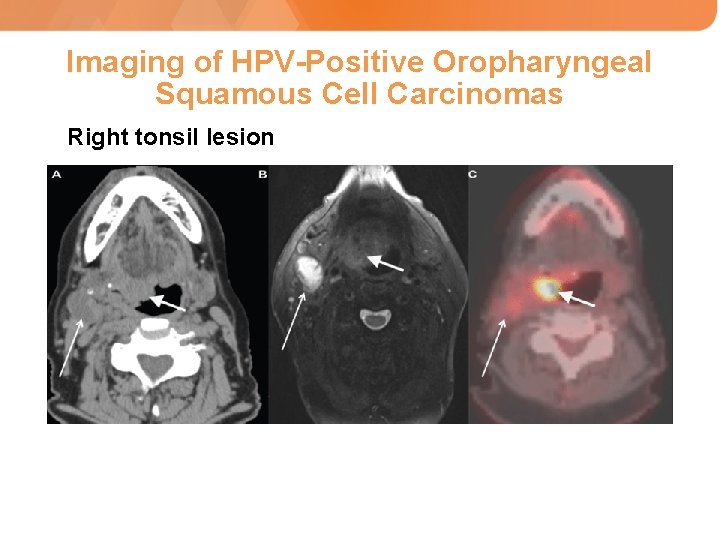
Imaging of HPV-Positive Oropharyngeal Squamous Cell Carcinomas Right tonsil lesion CT of the head and neck MRI (T 2 phase) *Note that cystic metastatic nodes may not be significantly PET-avid Lewis A, et al. Oncology (Williston Park). 2015; 29(9): pii: 213344. PET/CT*

The Use of PET/CT in HPV-Positive Tumors • 70 patients; 13 HPV positive • In HPV tumors total lesion glycolysis >121. 9 g and uniformity* ≤ 0. 138 – correlated with decreased PFS, disease-specific survival, and OS *Uniformity: Using a three dimensional gray-tone difference matrix (NGTDM) was applied Cheng NM, et al. J Nucl Med. 54(10): 1703 -1709, 2013

The Use of PET/CT in HPV-Positive Tumors for Follow-Up • PET/CT was less accurate than CT for assessing treatment response at 8 weeks 1 • PET/CT was more accurate than CT for assessing treatment response at 12 weeks 2 1. Moelleo BJ, et al. J Clin Onc. 2009; 27(15): 2509 -2515. 2. Chan JY, et al. Arch Oto Head Neck Surg. 2012; 138(11): 1040 -1046.
Title
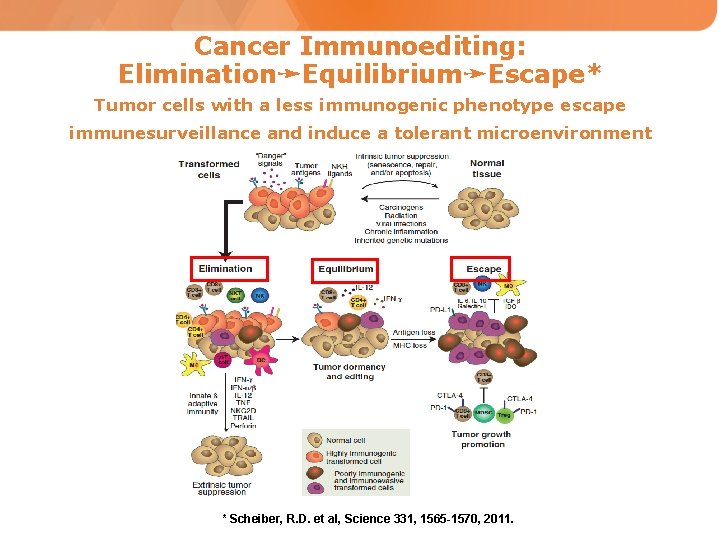
Cancer Immunoediting: Elimination➛Equilibrium➛Escape* Tumor cells with a less immunogenic phenotype escape immunesurveillance and induce a tolerant microenvironment * Scheiber, R. D. et al, Science 331, 1565 -1570, 2011. Schreiber, R. D. , Old, L. J. & Smyth, M. J. Cancer in cancer suppression and promotion. Science 331, 1565 -1570 (2011).

A 43 year-old otherwise healthy non-smoking male, presents with two-month history of sore throat, odynophagia and enlarging left-sided neck mass that did not respond to antibiotic therapy prescribed by his primary care physician. Physical Examination: - Intraoral examination and fiberoptic endoscopy: indurated left tonsillar mass extending to left base of the tongue - Neck palpation: firm 4 cm lymph node in the left lymph node level II Biopsy and Histology: - Poorly differentiated (grade 3) squamous cell carcinoma, p 16 positive Imaging: - A CT scan of the neck and subsequent PET/CT show a 3. 5 x 3. 2 cm mass involving left tonsil and extending to left base of tongue, and a left lymph node 4. 7 x 3. 8 cm in size. There are no distant sites of hypermetabolic activity seen on PET/CT. Stage: - T 2 N 2 a. M 0 (IVA) ECOG Performance Status: 0 Laboratory: - CBC, LFT, Renal test: normal
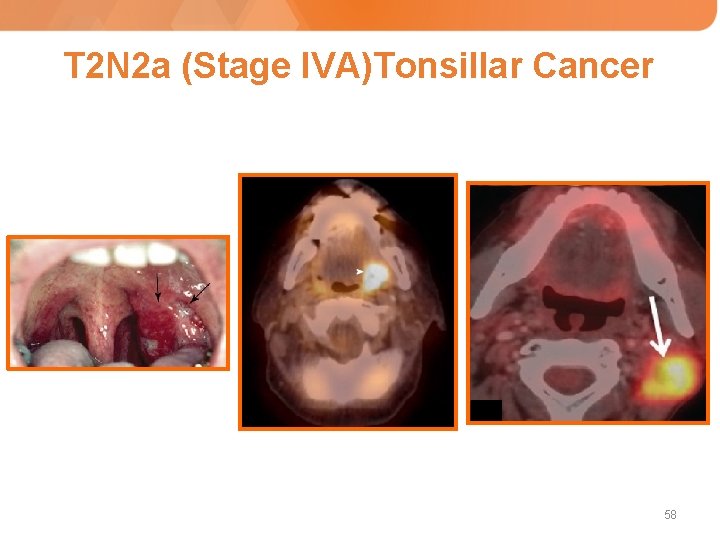
T 2 N 2 a (Stage IVA)Tonsillar Cancer 58

Best Treatments • Surgery • Chemoradiotherapy • Cetuximab-radiotherapy
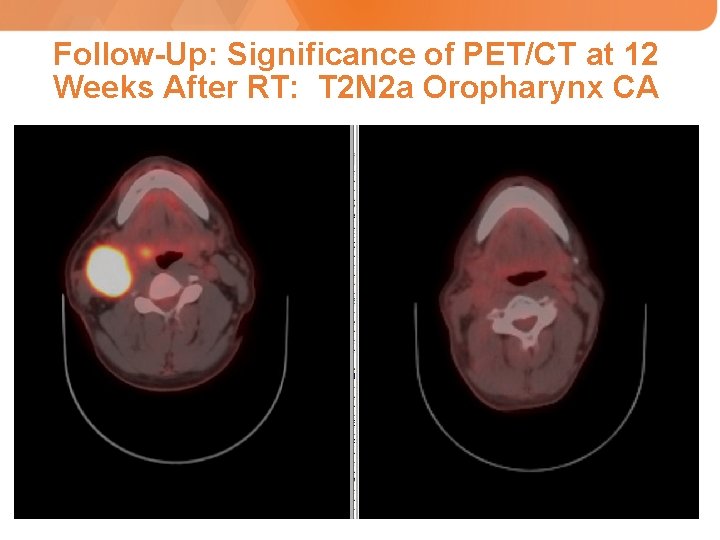
Follow-Up: Significance of PET/CT at 12 Weeks After RT: T 2 N 2 a Oropharynx CA

Is further imaging necessary if the patient shows no clinical evidence of recurrence?
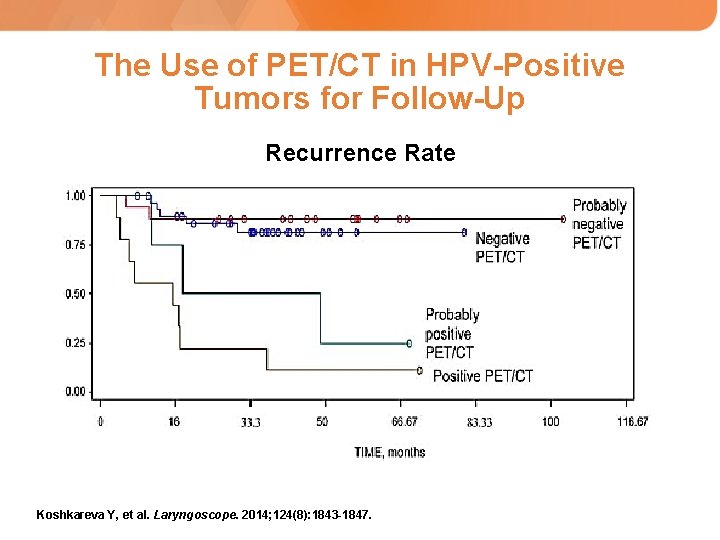
The Use of PET/CT in HPV-Positive Tumors for Follow-Up Recurrence Rate Koshkareva Y, et al. Laryngoscope. 2014; 124(8): 1843 -1847.

The Use of PET/CT in HPV-Positive Tumors for Follow-Up: HPV positive vs HPV negative • Post CRT treatment PET/CT’s provide useful information • A negative first post-treatment PET/CT at 12 weeks for HPV positive patients: Better prognosis and rare recurrence – therefore, further imaging can be directed by physical exam * • All accuracy measures for PET/CT vs. CT are higher in HPV+ patients: Eg, 93% negative predictive value Koshkareva Y, et al. Laryngoscope. 2014; 124(8): 1843 -1847. * Frakes J, et al. Presented at: Multidisciplinary Head and Neck Cancer Symposium; February 18 -20, 2016: Scottsdale, Arizona, United States. Abstract 6.

Key Prognostic Factors for Patients Undergoing Re-irradiation of SCCHN Compilation of 9 re-irradiation trials multivariate analysis (favorable factors) • No previous chemotherapy during original radiation • Surgery before re-irradiation • Re-irradiation dose of > 60 Gy • Treatment interval of > 36 months Choe KS, et al. Cancer. 2011; 117(20): 4671– 4678.
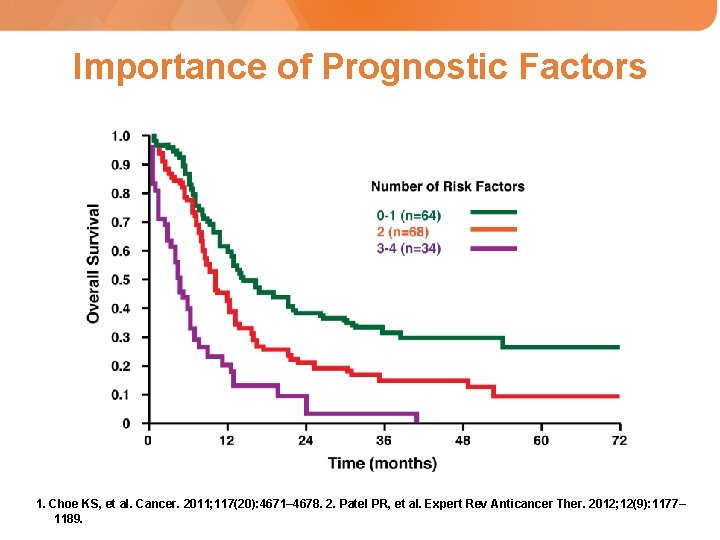
Importance of Prognostic Factors 1. Choe KS, et al. Cancer. 2011; 117(20): 4671– 4678. 2. Patel PR, et al. Expert Rev Anticancer Ther. 2012; 12(9): 1177– 1189.
- Slides: 65