Management of Dabigatran and Rivaroxaban Xuan Wang July

Management of Dabigatran and Rivaroxaban Xuan Wang July 27, 2012

Outline • • • Introduction to dabigatran and rivaroxaban RELY trial ROCKET AF trial Monitoring Emergency management Prevalence at Vanderbilt

Conventional Anticoagulants • Traditionally heparin (UFH or LMWH) followed by long-term warfarin for prevention of thromboembolic events • 2004 – 25 million US received warfarin, nearly 31 million prescriptions • Problems with warfarin: slow onset/offset of action, a narrow therapeutic window, food and drug interactions, individualized dosing, and frequent lab monitoring • Rates of discontinuation high

New Generation of Anticoagulants • Dabigatran • Rivaroxaban • Approved for: – Stroke prophylaxis in Afib (D in 10/2010, R in 11/2011) – Prevention of DVT after elective hip and knee replacement surgery (R in 7/2011) • Advantages: both are safe and effective, require no monitoring, have a direct mode of action against only one clotting factor (thrombin or factor Xa), have limited drug interactions, and have rapid peak blood levels.
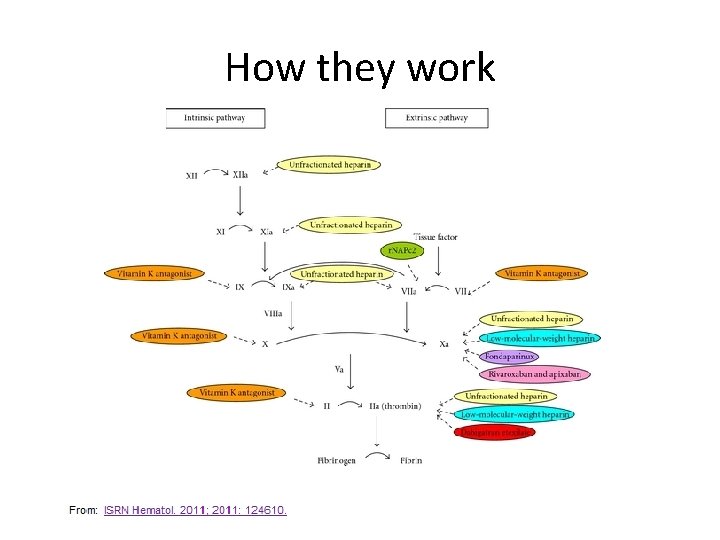
How they work

Dabigatran Site of action: Direct Thrombin inhibitor Bioavailability: 6% 80% renally excreted Absorption/Metabolism: Oral prodrug converted in the liver through esterases to the active compound • Time to peak serum concentration: 0. 5 – 2 h • Half-life: 11 h (14 -17 in elderly) • Duration of effect: ~22 h • • Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 113: 575 -580

Rivaroxaban • • • Site of action: Factor Xa Bioavailability: 80%-100% Time to peak serum concentration: 2 – 4 h Half-life: 5 -9 h (11 -13 h in elderly) Duration of effect: 10 -18 h Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 113: 575 -580
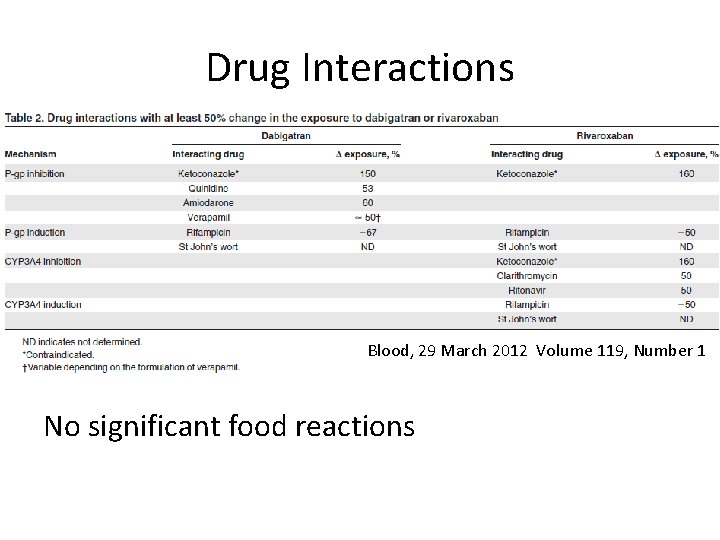
Drug Interactions Blood, 29 March 2012 Volume 119, Number 1 No significant food reactions

RE-LY Trial • Randomized Evaluation of Long-Term Anticoagulation Therapy • Compared two fixed doses of dabigatran (110 and 150 mg twice daily) with open label warfarin • Was either dose of dabigatran noninferior to warfarin? • Primary study outcome: stroke or systemic embolism • Primary safety outcome: Major bleeding • AF and ≥ 1 risk factor Connolly SJ, Ezekowitz MD, Yusuf S et al. N Engl J Med 2009; 361: 1139– 51

Patients • • 18, 113 patients Median age 71. 5 yrs, ~40% women Mean CHADS ₂ score = 2. 1 Control group had TTR of 64%
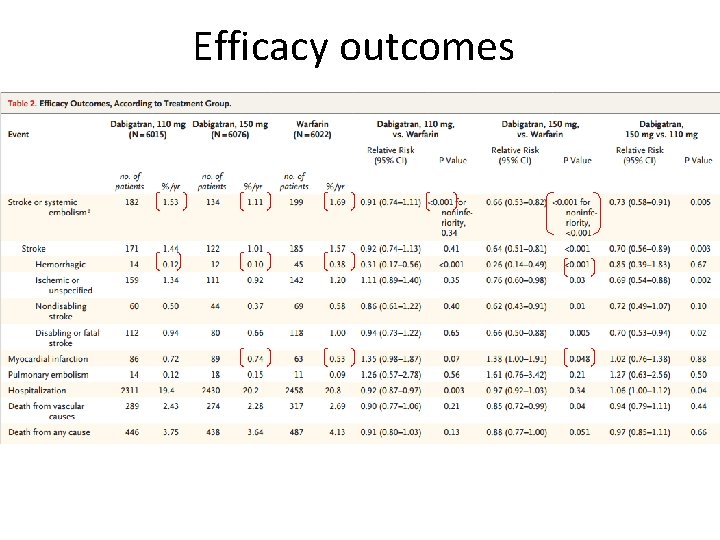
Efficacy outcomes
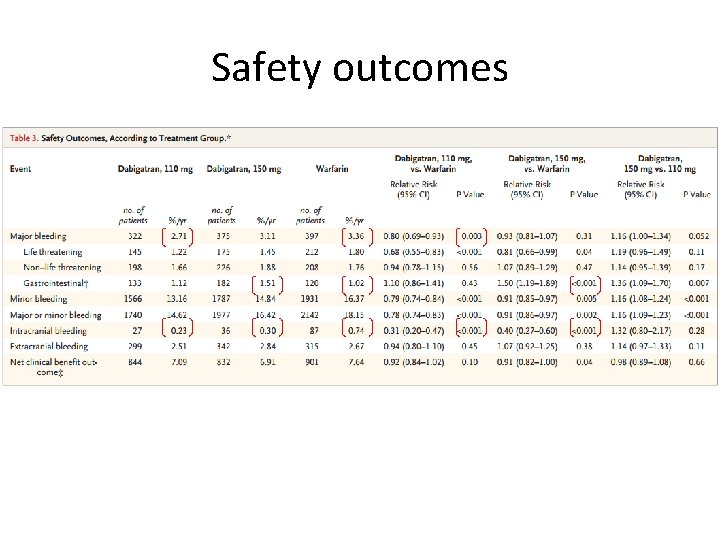
Safety outcomes

RE-LY Conclusions • Stroke or systemic embolism: Both dabigatran doses noninferior to warfarin. 150 mg dose superior to warfarin • 110 mg dabigatran superior to warfarin with respect to major bleeding (150 mg similar) • Hemorrhagic stroke was lower on both dabigatran 150 and 110 mg bid compared with warfarin, respectively (0. 10, 0. 12 and 0. 38%, respectively, P < 0. 001 for both). • Non-major bleeding (except GI) were higher in warfarin than both dabigatran doses • Only adverse effect that was sig. more common with dabigatran (both doses) was dyspepsia (~11. 5% vs. 5. 8%, p<0. 001)

ROCKET AF Trial • Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation • Compared once-daily oral rivaroxaban with doseadjusted warfarin for the prevention of stroke and systemic embolism • Multicenter, randomized, double-blind, doubledummy, noninferiority design • Patients with AF and ≥ 2 risk factors • Primary efficacy end point: stroke and systemic embolism Patel MR, Mahaffey KW, Garg J et al. N Engl J Med 2011; 365: 883– 91

Patients • • • 14, 264 patients Median age 73 yrs, ~40% women Mean CHADS ₂ score = 3. 5 Control group had TTR of 55% Higher risk patients compared to RE-LY
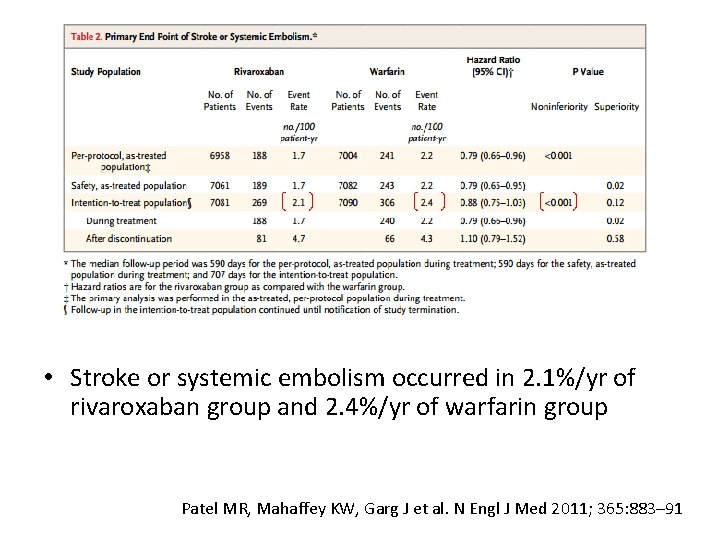
Results • Stroke or systemic embolism occurred in 2. 1%/yr of rivaroxaban group and 2. 4%/yr of warfarin group Patel MR, Mahaffey KW, Garg J et al. N Engl J Med 2011; 365: 883– 91
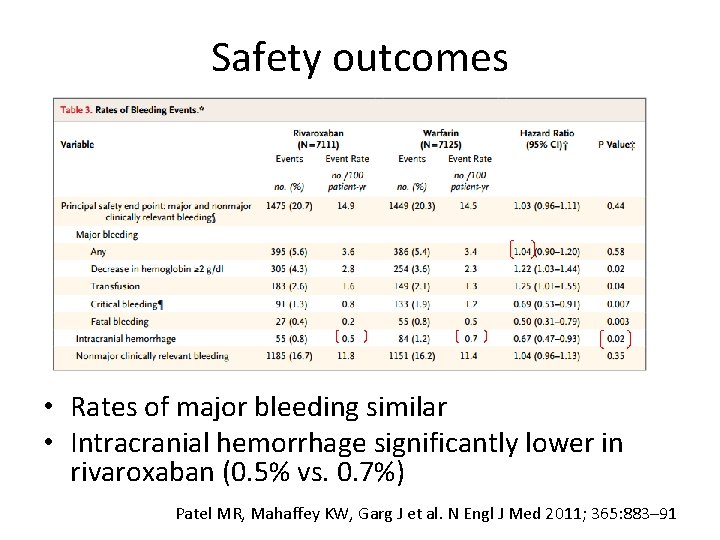
Safety outcomes • Rates of major bleeding similar • Intracranial hemorrhage significantly lower in rivaroxaban (0. 5% vs. 0. 7%) Patel MR, Mahaffey KW, Garg J et al. N Engl J Med 2011; 365: 883– 91

ROCKET AF Conclusions • Rivaroxaban was noninferior to warfarin in the prevention of subsequent stroke or systemic embolism. • No significant differences in rates of major and clinically relevant nonmajor bleeding between the two study groups, although intracranial and fatal bleeding occurred less frequently in the rivaroxaban group

Monitoring Warfarin - PT/INR Rivaroxaban - PT shows a linear dose-response - Antifactor Xa assay (must be specifically calibrated for rivaroxaban) Dabigatran - TT (linear dose-response) a. PTT (curvilinear response) ECT (linear dose-response) PT is insensitive and not useful Renal function should be assessed annually

Overdose/Bleeding complications • Risk stratified: minor, moderate, major/life-threatening Dabigatran - Currently no antidote - If detected soon after ingestion: gastric lavage and/or administration of charcoal - FFP or PCC (*activated PCC or r. FVIIa) - Extreme cases: acute hemodialysis Rivaroxaban - Not dialyzable - All measurable anticoagulant effects shown to be reversed by 4 factor PCC (in healthy volunteers) - No available 4 -factor PCC in the US Schulman S, Crowther MA. Blood 2012; 119(13): 3016 -3023.
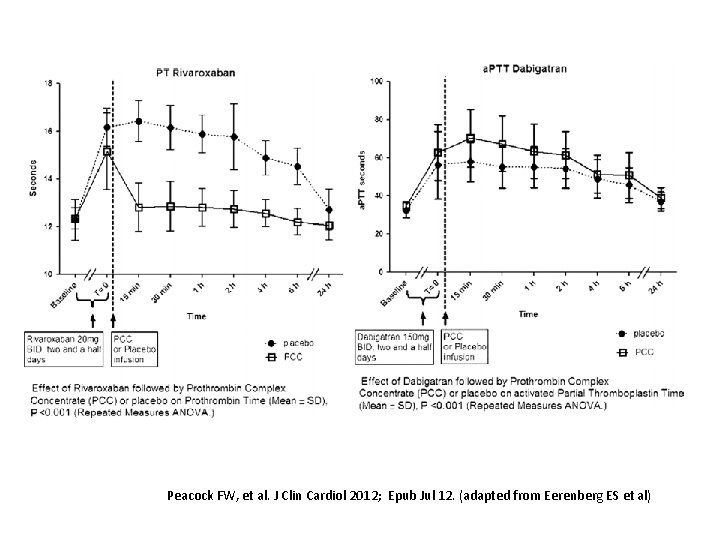
Peacock FW, et al. J Clin Cardiol 2012; Epub Jul 12. (adapted from Eerenberg ES et al)
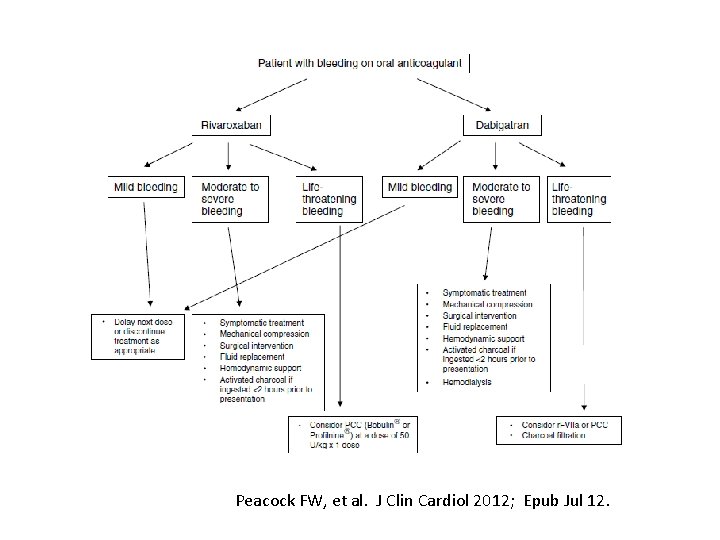
Peacock FW, et al. J Clin Cardiol 2012; Epub Jul 12.

Reasons for selecting warfarin over new anticoagulants • • • Good level of control Mechanical heart valve replacement GI disease Renal disease Elderly patients, poor compliance, drug cost Schulman S, Crowther MA. Blood 2012; 119(13): 3016 -3023.

Reasons for selecting new anticoagulants over warfarin • Unexplained poor warfarin control • Drug-drug interactions with warfarin – frequent need for antibiotic treatment, chemo, amiodarone, frequent use of acetaminophen, azathioprine • New anticoagulant patients on therapy for Afib

Dabigatran vs. Rivaroxaban • No direct head to head study • Indirect comparison of dabigatran, rivaroxaben, and apixaban in patients with a CHADS₂ score ≥ 3 resulted in statistically similar rates of stroke or systemic embolism • The selection may depend on other factors related to each agent’s adverse effect profile, dosing schedule and cost • Of all drugs marketed in US, dabigatran had the highest number of serious adverse events reported to the FDA in 2011 (warfarin was 2 nd) – 3781 (2367 hemorrhages; 542 deaths) versus 1, 106 events with warfarin (72 deaths)

Here at Vanderbilt • Outpatient pharmacy – 80 out of 3400 patients on dabigatran – No rivaroxaban • Inpatient pharmacy – 4 patients (3 in the last 3 months) on rivaroxaban – 6 or 7 pt/week on dabigatran ($260/month) – If bleeding: supportive measures first then FFP, p. RBCs, Novo. Seven ($10, 000/dose) – Usage of both will probably go up if reversal agent come on the market

Assessment of Dabigatran at VUMC • January 2011 – January 2012 • Evaluated 103 hospital admissions in 94 patients Jenna Faircloth, Dabitgatran MUE, VUMC
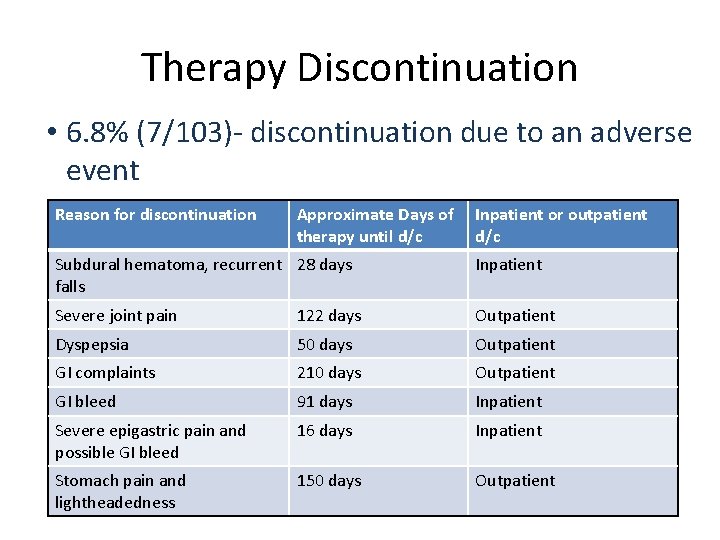
Therapy Discontinuation • 6. 8% (7/103)- discontinuation due to an adverse event Reason for discontinuation Approximate Days of therapy until d/c Inpatient or outpatient d/c Subdural hematoma, recurrent 28 days falls Inpatient Severe joint pain 122 days Outpatient Dyspepsia 50 days Outpatient GI complaints 210 days Outpatient GI bleed 91 days Inpatient Severe epigastric pain and possible GI bleed 16 days Inpatient Stomach pain and lightheadedness 150 days Outpatient

Here at Vanderbilt 2 • Stroke/Neuro ICU Division – Physician 1: “I personally have been more or less sticking with warfarin – mainly out of habit & concern for lack of antidote (I think I started at least one patient on dabigatran, but have not prescribed rivaroxaban” – Physician 2: Uses dabigatran a lot. Protocol given by company states that FFP, PCC, Factor VII all reverse dabigatran to some extent. Dialysis is always an option. Rivaroxaban has not been used as much.

Key Monitoring Parameters Adjustments for renal function 150 mg BID = est. Cr. Cl >30 m. L/min 75 mg BID = est. Cr. Cl 15 -30 ml/min Not Recommended = est Cr. Cl <15 ml/min and/or HD Conversions from Anticoagulation to/from Dabigatran LMWH/Fondaparinux to Dabigatran = Stop parenteral agent and administer dabigatran 0 -2 h before next parenteral dose due Continuous parenteral anticoagulant to Dabigatran = Administer first dose of dabigatran at the time of discontinuation of IV anticoagulant Dabigatran to parenteral anticoagulant = Cr. Cl >30 m. L/min, start parenteral anticoagulant 12 hours after the last dose of dabigatran; Cr. Cl <30 m. L/min: Start parenteral anticoagulant 24 hours after the last dose of dabigatran Warfarin to Dabigatran= initiate when INR < 2. 0 Dabigatran to warfarin = Cr. Cl >50 m. L/min, start warfarin 3 days before discontinuing dabigatran Cr. Cl 30 -50 m. L/min, start warfarin 2 days before discontinuing dabigatran Cr. Cl 15 -30 m. L/min, start warfarin 1 day before discontinuing dabigatran Cr. Cl <15 m. L/min, no recommendations can be made Adjustments for Drug Interactions Ketoconazole: Switch 150 mg BID to 75 mg BID if est. Cr. Cl=30 -50 m. L/min and D/C if est. Cr. Cl <30 m. L/min Dronedarone: Switch 150 mg BID to 75 mg BID if est. Cr. Cl=30 -50 m. L/min and D/C if est. Cr. Cl <30 m. L/min Rifampin: Avoid concomitant use NSAIDs: Use with caution; Increase risk of bleeding Duloxetine: Use with caution; Increase risk of bleeding Holding Prior to Surgical Procedures est. Cr. Cl >50 m. L/min = Hold dabigatran 1 to 2 days est. Cr. Cl <50 m. L/min = Hold dabigatran 3 to 5 days May require doses to be held longer in patients undergoing major surgery, spinal puncture, or placement

Summary • Dabigatran was superior to warfarin in preventing strokes and equal/less in bleeding – GI side effects • Rivaroxaban noninferior to warfarin • Lower rates of intracranial bleeding with rivaroxaban and dabigatran (110 mg) • No defined reversal agent • As a result, warfarin is still predominant anticoagulant used • New anticoagulants will probably be used more if an antidote comes on market

References Little, JW. Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 113: 575 -580 Connolly SJ, Ezekowitz MD, Yusuf S et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361: 1139– 51. Patel MR, Mahaffey KW, Garg J et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011; 365: 883– 91. Peacock FW, et al. Emergency management of bleeding complications associated with old and new oral anticoagulants. J Clin Cardiol 2012; Epub Jul 12. Fatemeh Moheimani and Denise E. Jackson. Venous Thromboembolism: Classification, Risk Factors, Diagnosis, and Management. ISRN Hematol. 2011; 2011: 124610. Schulman S, Crowther MA. How I treat with anticoagulants in 2012: new and old anticoagulants, and when and how to switch. Blood 2012; 119(13): 3016 -3023. Eerenberg ES, Kamphuisen PW, Sijpkens MK, et al. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011; 124: 1573– 1579
- Slides: 32