Management of Bleeding on Factor Xa Inhibitor Therapy

Management of Bleeding on Factor Xa Inhibitor Therapy – Does One Size Fit All? Nubriel Hernandez, Pharm. D PGY-1 Pharmacy Resident SUNY Downstate Medical Center Brooklyn, NY

Disclosure ▪ I have no conflict of interest to report and I intend to reference unlabeled/unapproved uses of drugs or products in my presentation 2

Objectives ▪ Recognize current management strategies for patients who present with major bleeding episodes while on oral anticoagulation ▪ Understand the limitations of current strategies of factor Xa inhibition reversal 3

Oral Anticoagulants ▪ Warfarin was the first anticoagulant approved in 1954 ▪ Due to limitations of warfarin, new oral anticoagulants have been approved ▪ Novel anticoagulants: direct thrombin inhibitors and factor Xa inhibitors Yang J, et. al. Eur J Med Chem. 2015; 101: 41 -51. . 4
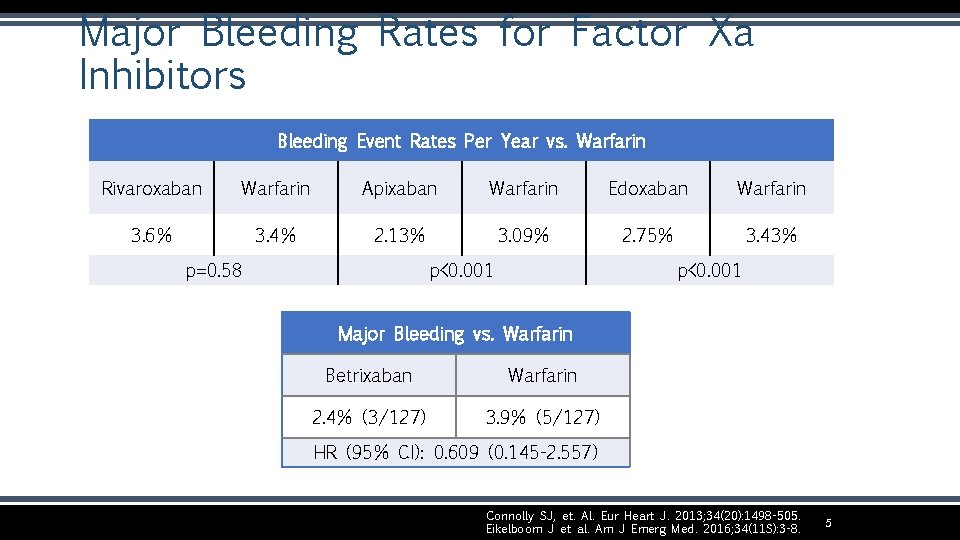
Major Bleeding Rates for Factor Xa Inhibitors Bleeding Event Rates Per Year vs. Warfarin Rivaroxaban Warfarin Apixaban Warfarin Edoxaban Warfarin 3. 6% 3. 4% 2. 13% 3. 09% 2. 75% 3. 43% p=0. 58 p<0. 001 Major Bleeding vs. Warfarin Betrixaban Warfarin 2. 4% (3/127) 3. 9% (5/127) HR (95% CI): 0. 609 (0. 145 -2. 557) Connolly SJ, et. Al. Eur Heart J. 2013; 34(20): 1498 -505. Eikelboom J et al. Am J Emerg Med. 2016; 34(11 S): 3 -8. 5
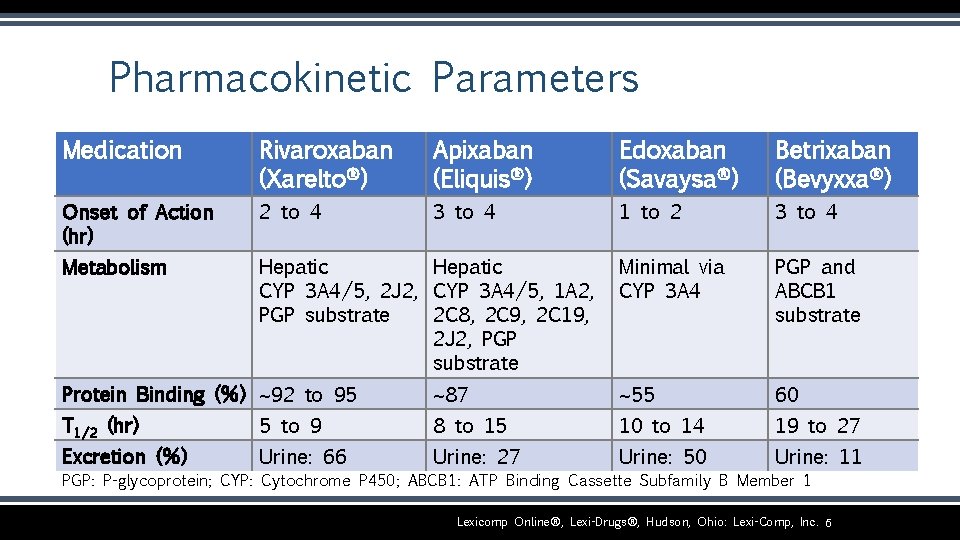
Pharmacokinetic Parameters Medication Rivaroxaban (Xarelto®) Apixaban (Eliquis®) Edoxaban (Savaysa®) Betrixaban (Bevyxxa®) Onset of Action (hr) 2 to 4 3 to 4 1 to 2 3 to 4 Metabolism Hepatic CYP 3 A 4/5, 2 J 2, CYP 3 A 4/5, 1 A 2, PGP substrate 2 C 8, 2 C 9, 2 C 19, 2 J 2, PGP substrate Minimal via CYP 3 A 4 PGP and ABCB 1 substrate Protein Binding (%) ~92 to 95 ~87 ~55 60 T 1/2 (hr) 5 to 9 8 to 15 10 to 14 19 to 27 Excretion (%) Urine: 66 Urine: 27 Urine: 50 Urine: 11 PGP: P-glycoprotein; CYP: Cytochrome P 450; ABCB 1: ATP Binding Cassette Subfamily B Member 1 Lexicomp Online®, Lexi-Drugs®, Hudson, Ohio: Lexi-Comp, Inc. 6

Management of Factor Xa Toxicity ▪ Activated Charcoal – Within 2 -4 hours of exposure – Dose: 50 g x 1 dose ▪ 4 Factor Prothrombin Complex Concentrates (4 FPCC) or activated Prothrombin Complex Concentrates (a. PCC) ▪ Assessment of laboratory markers and symptoms of bleeding ▪ Hemodialysis ineffective ▪ Fresh Frozen Plasma (FFP) last line option? Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 - 7
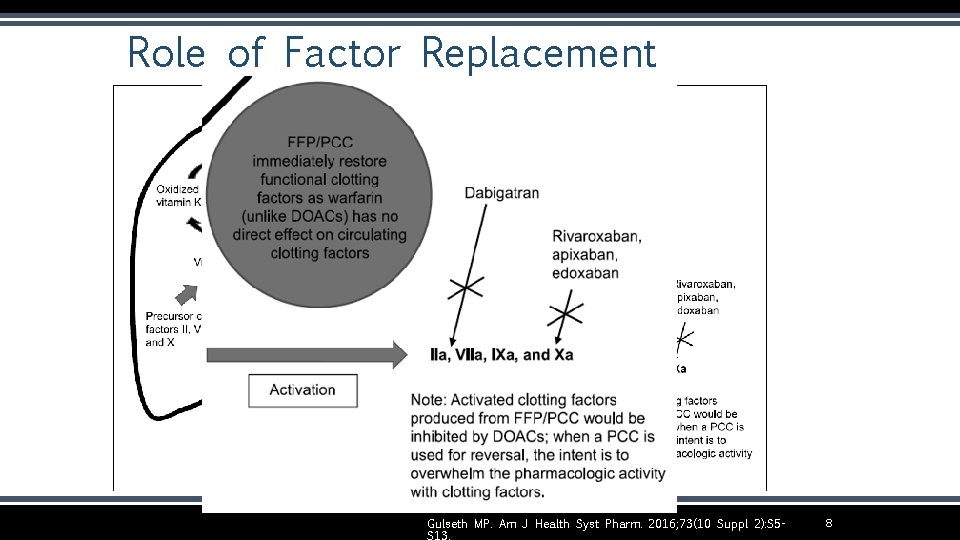
Role of Factor Replacement Gulseth MP. Am J Health Syst Pharm. 2016; 73(10 Suppl 2): S 5 - 8

Prothrombin Complex Concentrates (PCCs)* ▪ 3 factor PCCs (Bebulin® or Profilnine®) ▪ 4 factor PCCs (Kcentra®); 50 Units/kg IV (Max: 5, 000 Units) ▪ Activated PCCs (FEIBA®); 50 – 100 Units/kg IV ▪ Differ in presence of factor VII ▪ Contraindicated: – Active disseminated intravascular coagulopathy – Recent history of heparin-induced thrombocytopenia (HIT) * These products are not FDA-indicated for reversal of bleeding with exposure to Factor Xa Inhibitors Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 - 9

Fresh Frozen Plasma ▪ Obtained from whole blood donation ▪ 1 Unit = 200 -250 m. L ▪ Requires ABO blood type matching and thawing ▪ May take up to 90 minutes from time of order to time of administration Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 - 10
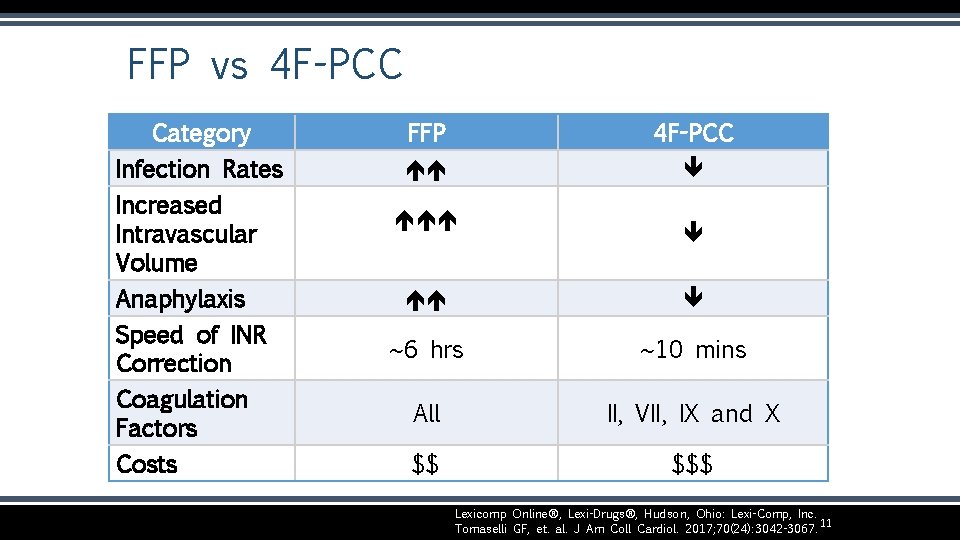
FFP vs 4 F-PCC Category Infection Rates Increased Intravascular Volume Anaphylaxis Speed of INR Correction Coagulation Factors Costs FFP 4 F-PCC ~6 hrs ~10 mins All II, VII, IX and X $$ $$$ Lexicomp Online®, Lexi-Drugs®, Hudson, Ohio: Lexi-Comp, Inc. 11 Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 -3067.

Guideline Recommendations: Warfarin 4 F-PCC based on patient’s weight and INR Fixed low dose regimen of 4 F-PCC INR 2 -4: INR 4 -6: INR 2 -4: 25 Units/kg 50 Units/kg 25 Units/kg 1, 000 Units (non-major bleed) 1, 500 Units (Intracranial Hemorrhage) * If 4 F-PCC is not available: FFP 10 -15 m. L/kg Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 - 12

Guideline Recommendations Factor Xa Inhibitors 4 F-PCC: 50 Units/kg a. PCC: 50 Units/kg Tomaselli GF, et. al. J Am Coll Cardiol. 2017; 70(24): 3042 - 13

Literature Review Klein L, Peters J, Miner J, Gorlin J. Evaluation of fixed dose 4 -factor prothrombin complex concentrate for emergent warfarin reversal. Am J Emerg Med. 2015; 33(9): 1213 -8. Study Design Retrospective cohort study Number 38 subjects on chronic oral anticoagulation with warfarin; 1 subject on chronic oral anticoagulation with rivaroxaban Population Patients admitted to urban level I trauma center in MN from March 2014 to January 2015 Intervention 4 F-PCC 1, 500 Units x 1 dose; additional dose if deemed necessary (median dose 1659 Units) Outcome • 36 patients (92. 3%) had a successful INR reversal with target less than 2. 0 and 28 patients (71. 8%) had a successful INR reversal with target of 1. 5 or less • No thrombotic events reported within 7 days 14
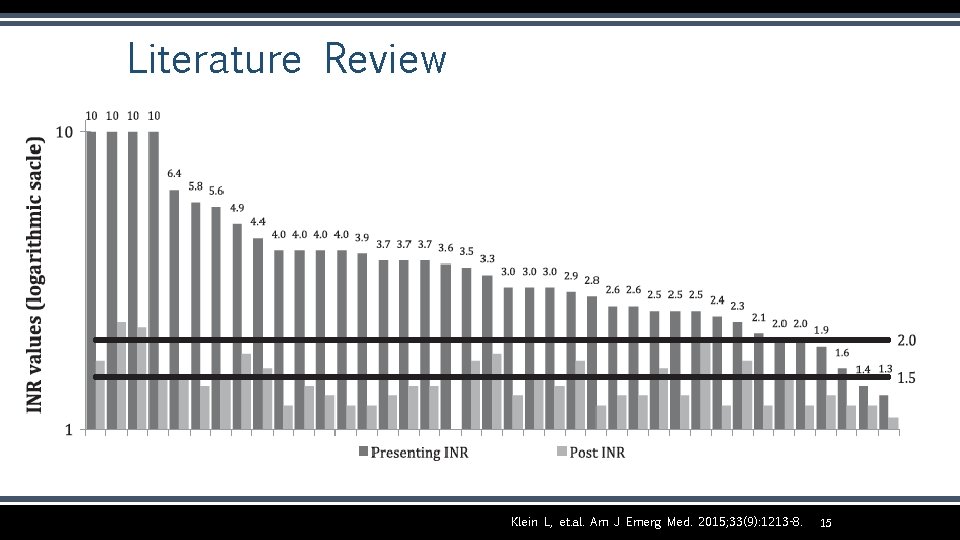
Literature Review Klein L, et. al. Am J Emerg Med. 2015; 33(9): 1213 -8. 15

Literature Review Zahir H, Brown KS, Vandell AG, et al. Edoxaban effects on bleeding following punch biopsy and reversal by a 4 -factor prothrombin complex concentrate. Circulation. 2015; 131(1): 82 -90. Study Design Phase I, double-blind, randomized, placebo-controlled, 2 -way crossover study Number 110 subjects; Part 1: 17 subjects and Part 2: 93 subjects Population Healthy men and women, aged 18 to 45 years, weighing ≤ 110 kg were eligible for study enrollment. All patients received edoxaban 60 mg by mouth Intervention 4 F-PCC in decreasing dosing increments (50, 25 and 10 Units/kg) or placebo Outcome • Average of 21% decrease in baseline normalized post-dose bleeding duration following administration of 50 Units/kg of 4 F-PCC (mean ratio 4 F-PCC versus placebo, 79. 2; 95% CI, 61. 5 – 101. 9; p= 0. 068) • Complete reversal was observed following 4 F-PCC dose of 50 Units/kg, partial reversal following 25 Units/kg and no reversal following 10 Units/kg • No deaths, serious adverse events, or thromboembolic events occurred 16
Literature Review Majeed A, Ågren A, Holmström M, et al. Management of rivaroxaban- or apixaban-associated major bleeding with prothrombin complex concentrates: a cohort study. Blood. 2017; 130(15): 1706 -1712. Study Design Prospective cohort study Number 84 subjects; 39 were on apixaban and 45 were on rivaroxaban Population Patients requiring treatment with 4 F-PCC who presented with a major bleeding event defined by the International Society of Thrombosis and Hemostasis (ISTH) Intervention 4 F-PCC given as fixed dose dependent on weight: < 65 kg = 1, 500 IU, > 65 kg = 2, 000 IU Outcome • Hemostatic effectiveness in 58 patients (69. 1%) and ineffective in 26 patients (30. 9%) • 27 patients (32%) died within 30 days of major bleeding event, of which 20 had an intracranial hemorrhage • PCCs were given at a median dose of 2000 Units, suggesting this dose may be adequate in patients who present with a major bleeding event 17

Summary ▪ One size does not fit all – limited data to suggest that similar benefits exist for factor Xa inhibitors using fixed dose 4 F-PCC ▪ FFP has greater disadvantages associated with its use as a reversal agent compared to 4 F-PCC ▪ Acute ingestion – Consider activated charcoal if time of ingestion is known, symptom management and watch for signs of bleeding 18

Test Your Knowledge

Test Your Knowledge ▪ Which strategies could be considered in a patient for Factor Xa reversal? a) b) c) d) e) Hemodialysis 4 F-PCC given at a dose of 50 IU/kg 4 F-PCC given at a dose of 10 IU/kg a. PCC given at a dose of 50 IU/kg B and D 20

Test Your Knowledge ▪ What are current limiting factors in managing patients who present with bleeding while on Factor Xa Inhibitor Therapy? a) b) c) d) Lack of specific reversal agent availability Limited data of 4 F-PCC use in bleeding patients Limited availability and/or utility of laboratory markers All of the above 21

References Yang J, Su G, Ren Y, Chen Y. Synthesis of 3, 4 -diaminobenzoyl derivatives as factor Xa inhibitors. Eur J Med Chem. 2015; 101: 41 -51. Lexicomp Online® , Lexi-Drugs® , Hudson, Ohio: Lexi-Comp, Inc. ; accessed January 21, 2018. Tomaselli GF, Mahaffey KW, Cuker A, et al. 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol. 2017; 70(24): 3042 -3067. Levi M, Moore KT, Castillejos CF, et al. Comparison of three-factor and four-factor prothrombin complex concentrates regarding reversal of the anticoagulant effects of rivaroxaban in healthy volunteers. J Thromb Haemost. 2014; 12(9): 1428 -36. Zahir H, Brown KS, Vandell AG, et al. Edoxaban effects on bleeding following punch biopsy and reversal by a 4 -factor prothrombin complex concentrate. Circulation. 2015; 131(1): 82 -90. Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011; 124(14): 1573 -9. Gulseth MP. Overview of direct oral anticoagulant therapy reversal. Am J Health Syst Pharm. 2016; 73(10 Suppl 2): S 5 -S 13. Eikelboom J, Merli G. Bleeding with direct oral anticoagulants vs warfarin: clinical experience. Am J Emerg Med. 2016; 34(11 S): 3 -8. 22

References, Continued Majeed A, Ågren A, Holmström M, et al. Management of rivaroxaban- or apixaban-associated major bleeding with prothrombin complex concentrates: a cohort study. Blood. 2017; 130(15): 17061712. Tellor KB, Barasch NS, Lee BM. Clinical experience reversing factor Xa inhibitors with four-factor prothrombin complex concentrate in a community hospital. Blood Transfus. 2017; 1 -5. Connolly SJ, Eikelboom J, Dorian P, et al. Betrixaban compared with warfarin in patients with atrial fibrillation: results of a phase 2, randomized, dose-ranging study (Explore-Xa). Eur Heart J. 2013; 34(20): 1498 -505. Klein L, Peters J, Miner J, Gorlin J. Evaluation of fixed dose 4 -factor prothrombin complex concentrate for emergent warfarin reversal. Am J Emerg Med. 2015; 33(9): 1213 -8. Khorsand N, Veeger NJ, Muller M, et al. Fixed versus variable dose of prothrombin complex concentrate for counteracting vitamin K antagonist therapy. Transfus Med. 2011; 21(2): 116 -23. Wang X, Mondal S, Wang J, et al. Effect of activated charcoal on apixaban pharmacokinetics in healthy subjects. Am J Cardiovasc Drugs. 2014; 14(2): 147 -54. Yeh CH, Fredenburgh JC, Weitz JI. Oral direct factor Xa inhibitors. Circ Res. 2012; 111(8): 1069 -78. Joseph R, Burner J, Yates S, Strickland A, Tharpe W, Sarode R. Thromboembolic outcomes after use of a four-factor prothrombin complex concentrate for vitamin K antagonist reversal in a realworld setting. Transfusion. 2016; 56(4): 799 -807. 23

Questions Available at: https: //infocus. dellemc. com/william_schmarzo/most-excellent-big-data-questions-top-down-or-bottom-up-use-cases/. 24
- Slides: 24