Management of anemia in cancer patients By Alfred

Management of anemia in cancer patients By Alfred Elias Namour; M. D. NCI, Cairo University

Objectives To know the definition and different grades of anemia in cancer patients. To highlight the various causes of anemia. To correlate between different causes and the effective treatment used. To highlight the importance of treating anemia.

Anemia is defined as a reduction of the hemoglobin (Hb) concentration, red-cell count or packed cell volume below normal levels. Mild anemia is defined as an Hb concentration of 11. 9 g/dl and 10 g/dl, moderate anemia as Hb of 9. 9 and 8. 0 g/dl and severe anemia as Hb <8. 0 g/dl.

Treatment-related anemia is graded according to the National Cancer Institute-Common Toxicity Criteria of Adverse Events (CTCAE v 4. 02) grade 1: lower normal limit to 10. 0 g/dl; grade 2: 8. 0 to <10. 0 g/dl; grade 3: 6. 5 to <8. 0 g/dl; grade 4: <6. 5 g/dl). Anemia of cancer is present in 40% of patients with non-myeloid malignancies. Overall incidence of anemia during chemo- or radiotherapy is 54% (mild 39%, moderate 14% and severe 1%). The incidence increases with the number of chemotherapy cycles.

What are the causes of anemia in cancer patients?

Anemia in patients with cancer may be caused by: Blood loss or iron deficiency Nutritional deficiencies Anemia of chronic diseases (ACD) Parvovirus B 19 Pure red cell aplasia (PRCA) Warm antibody immune hemolysis Cold antibody immune hemolysis Disease or therapy induced anemia

1. Anemia caused by blood loss or iron deficiency Pathogenesis - ulcerating tumors - extensive surgery - benign GIT diseases - gastrectomy Blood studies microcytosis and hypochromia, elevated soluble transferrin receptor.

Stools should be tested for occult blood. Treatment of the cause & try to minimize or stop the blood loss. Therapeutic trials: Ferrous sulfate, 325 mg PO given 3 times daily usually elevate the hemoglobin concentration in patients with iron deficiency

2. Anemia caused by nutritional deficiencies - - Folic acid deficiency is the most common cause of megaloblastic anemia in cancer patients & is due to: Decreased intake of folate Increased requirements with : * autoimmune hemolytic anemia * postoperative state * prolonged IV therapy * excess use by proliferating tumor cells Folate antagonist drugs (e. g. , methotrexate)

Vitamin B 12 deficiency is usually seen after gastrectomy or in patients having malabsorption secondary to lymphoma that involves the ileum. Blood studies megaloblastic anemia, neutrophil hypersegmentation.

3. Anemia of chronic disease (ACD) - Pathogenesis immune activation with the production of cytokines that inhibit both the action and production of EPO ACD is more severe with widespread metastases but may occur with localized tumors There is increased levels of tumor necrosis factor (TNF) and IL-1. TNF stimulates marrow stromal cells to produce IFN-β, while IL-1 acts on T lymphocytes to produce IFN-gamma. Both IFN-β IFN-gamma inhibit erythropoiesis directly Neopterin levels, which indicate the activation of macrophages by IFN-gamma, are also increased in malignancies.

- The hemoglobin concentrations are inversely proportional to blood concentrations of neopterin and IFN-gamma. Blood studies normocytic and normochromic anemia, decreased levels of both serum iron and transferrin, the levels of soluble transferrin receptor are normal Bone marrow studies ineffective erythropoiesis with shortened RBC life spans and decreased sideroblasts. Treatment Recombinant human EPO can correct ACD and rarely transfusion may be needed in severe conditions.

4. Anemia caused by parvovirus B 19 In Immunocompromised patients who are unable to produce neutralizing antibodies against the virus, an infection can persist & cause chronic bone marrow failure Bone marrow studies erythroid hypoplasia Treatment hyperimmune gamma globulins

5. Pure red cell aplasia (PRCA) PRCA is the isolated severe hypoplasia of erythroid elements in the marrow Associated with thymoma in 10% of the cases, with lymphoprolepherative disorders and various carcinomas Blood studies normocytic, normochromic anemia Bone marrow studies markedly decreased-toabsent erythroid precursors and normal megakryocytes and myeloid elements
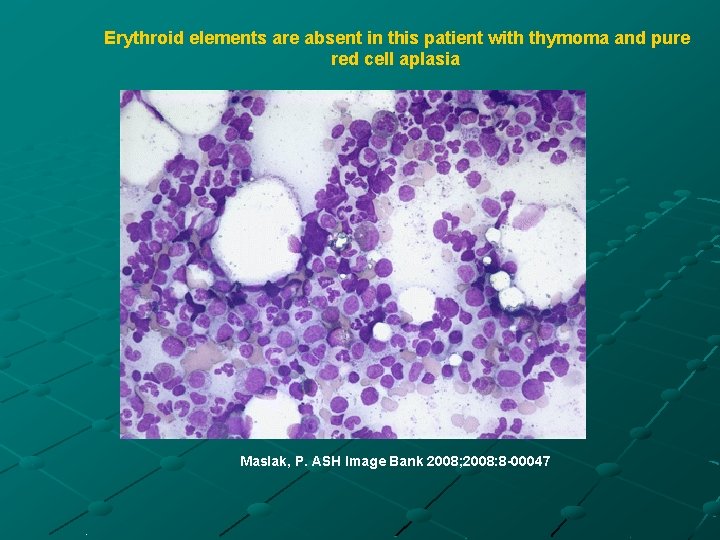
Erythroid elements are absent in this patient with thymoma and pure red cell aplasia Maslak, P. ASH Image Bank 2008; 2008: 8 -00047 .

Chest radiographs mediastinal mass if associated with thymoma Treatment removal of thymoma results in remission in 20% of cases. Patients with or without thymoma responded to therapy with cyclophosphamide, cyclosporine or antithymocyte globulin

6. Warm antibody immune hemolysis Pathogenesis Autoimmune hemolysis because of Ig. G antibodies most commonly in patients with lymphoproliferative neoplasms, only 2% of cases are associated with solid tumors Also reported after treatment with cytostatic drugs (e. g. , fludarabine) The Ig. G coated erythrocytes are removed by the reticuloendothelial system, predominantly the spleen (extravascular hemolysis)

Diagnosis insidious onset of severe anemia, mild jaundice, and splenomegaly Blood studies spherocytosis, nucleated RBCs, increased reticulocytes Positive Coombs’test or the direct antiglobulin test (DAT) with anti-Ig. G or anticomplement antisera Treatment Prednisone and treatment of the tumor are necessary, patients with unsatisfactory response to corticosteroids require spleenectomy Treatment with rituximab may be considered

7. Cold antibody immune hemolysis Pathogenesis Cold agglutinins are Ig. M molecules that attach to RBC membranes at cold temperature & fix complement. At 370 C, the Ig. M molecule dissociate from the cell, but the complement remains fixed. Most common with lymphoma. Diagnosis patients with high titers of cold agglutinins may have acrocyanosis or Raynaud phenomenon. RBC agglutination may be observed on blood smears.

The DAT is strongly positive when performed at 40 C but is positive only with anticomplement antisera at 370 C Treatment Rituximab 375 mg/m 2 weekly for 4 weeks is often effective chlorambucil or cyclophosphamide may be helpful for symptomatic patients

ESMO recommendations

The use of erythropoiesisstimulating agents (ESAs) All causes of anemia should be taken into account and, if possible, corrected before the use of erythropoiesis-stimulating agents (ESAs) [A]. The indication of ESAs is the treatment of symptomatic chemotherapy-induced anemia in adult patients with non-myeloid malignancies. The aim is to prevent transfusions and their possible complications and possibly to improve Qo. L by increasing the Hb level.

In patients treated with chemotherapy and an Hb level of ≤ 10 g/dl, treatment with ESAs might be considered to increase Hb to < 2 g/dl or to prevent further decline in Hb [II, A]. In patients not treated with chemotherapy, there is no indication for the use of ESAs and there might be an increased risk of death when ESAs are administered to a target Hb of 12– 14 g/dl [I, A]. In patients treated with curative intent, ESAs should be used with caution [D].

Conclusion In patients with anemia, it is necessary to take a complete history with emphasis on drug exposure; to review the peripheral blood smear, do a reticulocyte count and if necessary to perform a bone marrow examination; to evaluate iron, folate and vitamin B 12 status; to assess occult blood loss and renal insufficiency. Coombs testing should be considered in patients with chronic lymphocytic leukemia, non-Hodgkin lymphoma and in patients with a history of autoimmune disease.

WHY do you want to treat anemia in cancer patients?
![Anemia has a negative impact on the quality of life (Qo. L) [I] and Anemia has a negative impact on the quality of life (Qo. L) [I] and](http://slidetodoc.com/presentation_image_h2/56bd85f4e54bd980ad4da0533b9dab8e/image-26.jpg)
Anemia has a negative impact on the quality of life (Qo. L) [I] and is an important factor in cancer-related fatigue [II]. It also constitutes a negative prognostic factor for overall survival in most types of cancer [I].

References http: //evs. nci. nih. gov/ftp 1/CTCAE_ 4. 03_2010 -0614_Quick. Reference_8. 5 x 11. pdf http: //www. esmo. org/Guidelines/Supportiv e-Care/Erythropoiesis-Stimulating-Agentsin-the-Treatment-of-Anaemia-in-Cancer. Patients

- Slides: 28