MALARIA VACCINES Disease burden World and India Plasmodium
MALARIA -VACCINES

§ Disease burden- World and India § Plasmodium life cycle § Control strategies being used § Immunology of malaria § Different types of vaccines § Clinical trials till date § Problems addressed and remaining? ? § THE FUTURE !!!!!

“GLOBAL BURDEN OF MALARIA”

“ The malaria epidemic is like loading seven Boeing 747 airliners with people everyday, and then deliberately crashing them into Mt. Kilimanjaro. ” Dr. Wen Kilama African Malaria Network(AMANET)
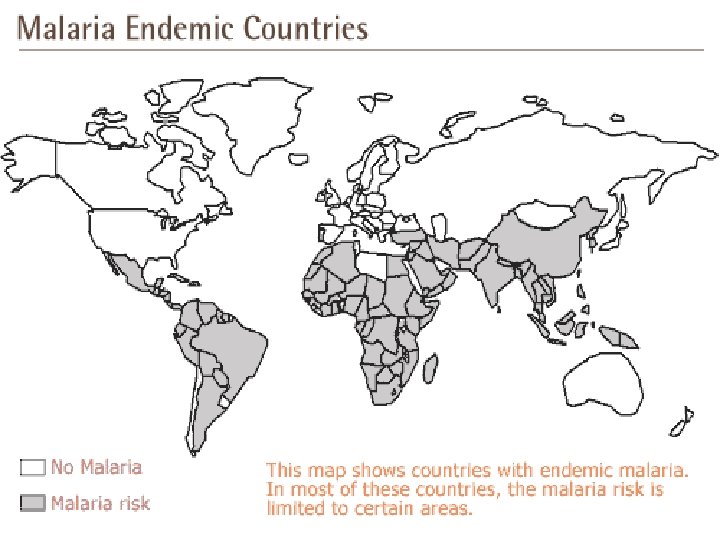

§ ONE THIRD HUMAN MANKIND at risk § World’s MOST DANGEROUS TROPICAL disease § Annual cases – 500 MILLION § Mortality – 3 MILLION DEATHS § Afflicted are children(1 million) aged < 5 YEARS, particularly in AFRICA § MULTI-SYSTEM INVOLVEMENT in c/o P. falciparum malaria( e. g. cerebral malaria)

“MALARIA BURDEN IN INDIA”

§ Outside Africa, 2/3 rd cases are concentrated in INDIA, Brazil, Sri Lanka, Vietnam, Columbia, Solomon Islands § 60 -65% infections caused by P. vivax 35 -40% by P. falciparum Few by P. malariae [from Orissa and Tumkur & Hassan districts of Karnataka] § Estimated 10. 6 million malaria cases reported from India in 2006, accounting for 60% cases of the WHO-SEAR region

§ Maximum burden in states of Uttar Pradesh, Bihar, Karnataka, Orissa, Rajasthan, Madhya Pradesh, Pondicherry § 80% of malaria cases derived from forestrelated areas and along the borders with Myanmar & India where malaria is endemic § Anopheles culicifacies- rural malaria Anopheles stephensi- urban malaria

“LIFE-CYCLE OF MALARIA PARASITE”


“PREVENTION STRATEGIES”

Improved diagnosis (e. g. blood film examination, rapid diagnostic methods) Prophylactic and therapeutic chemotherapy (e. g. Chloroquine, Artemesinin derivatives, Primaquine, Doxycycline) Integrated vectorcontrol - insecticide-treated bed-nets - residual house spraying(with DDT) VACCINATIO N

A PROPHYLACTIC VACCINE FOR HUMANS IS POSSIBLE!!!! (EVIDENCE FROM THE PAST)
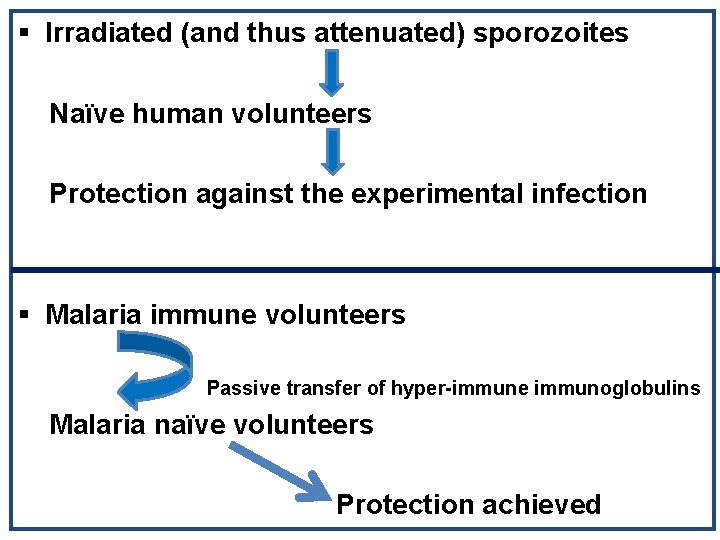
§ Irradiated (and thus attenuated) sporozoites Naïve human volunteers Protection against the experimental infection § Malaria immune volunteers Passive transfer of hyper-immune immunoglobulins Malaria naïve volunteers Protection achieved

§ Continuous antigenic stimulation in endemic areas Build-up of naturally acquired immunity, which affects - the severity of clinical disease - less incidence of parasitemia - significant protection from death

“DIFFICULTIES IN VACCINE DEVELOPMENT”
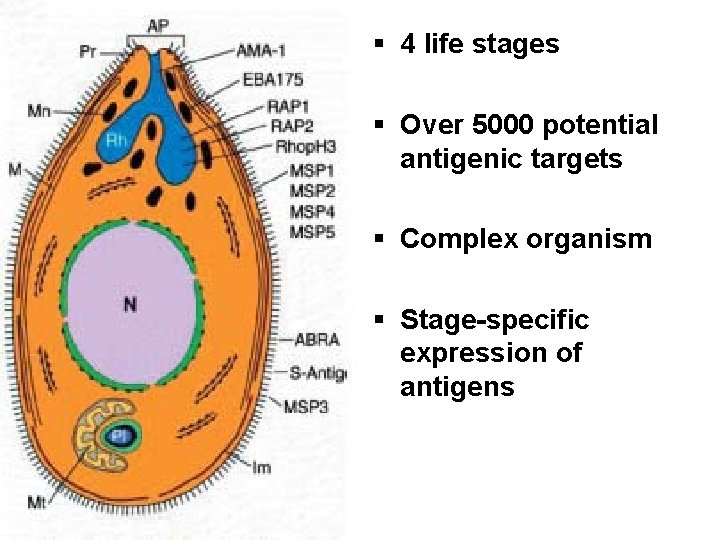
§ 4 life stages § Over 5000 potential antigenic targets § Complex organism § Stage-specific expression of antigens

§ Ability to adapt to its environment and confound the immune system by i. Antigenic variation between strains ii. Sequence polymorphisms of critical larger epitopes § Poor understanding of protective immunity in malaria § Lack of reliable and predictive animal models
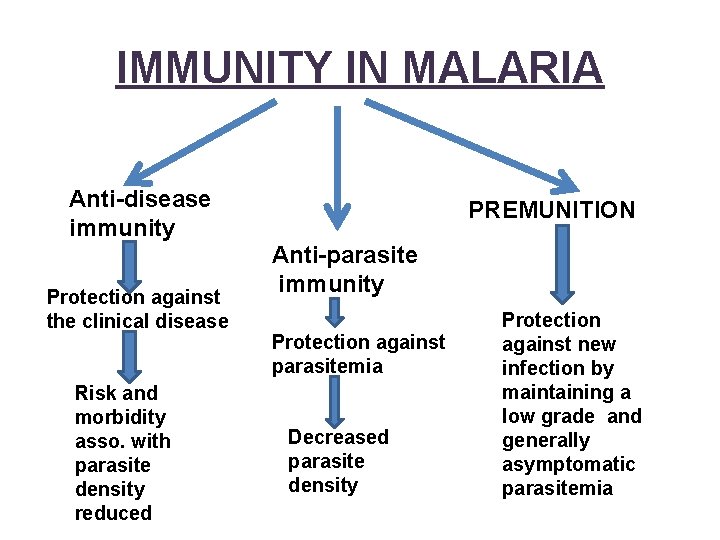
IMMUNITY IN MALARIA Anti-disease immunity Protection against the clinical disease Risk and morbidity asso. with parasite density reduced PREMUNITION Anti-parasite immunity Protection against parasitemia Decreased parasite density Protection against new infection by maintaining a low grade and generally asymptomatic parasitemia
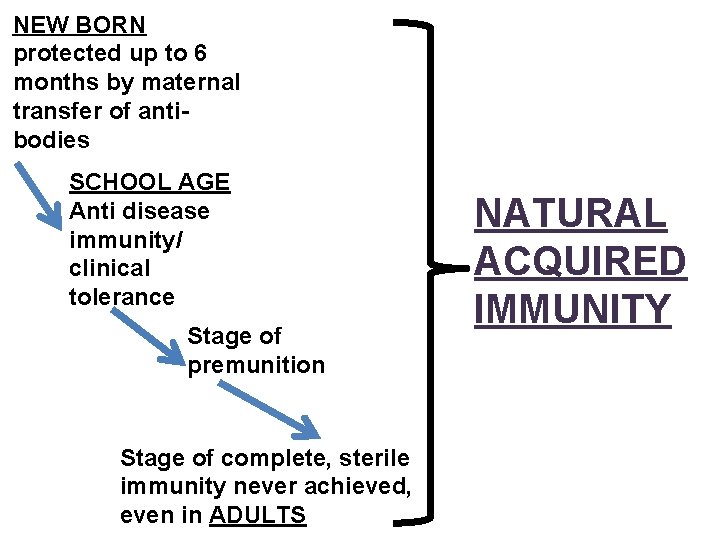
NEW BORN protected up to 6 months by maternal transfer of antibodies SCHOOL AGE Anti disease immunity/ clinical tolerance Stage of premunition Stage of complete, sterile immunity never achieved, even in ADULTS NATURAL ACQUIRED IMMUNITY

“POTENTIAL VACCINE CANDIDATES”
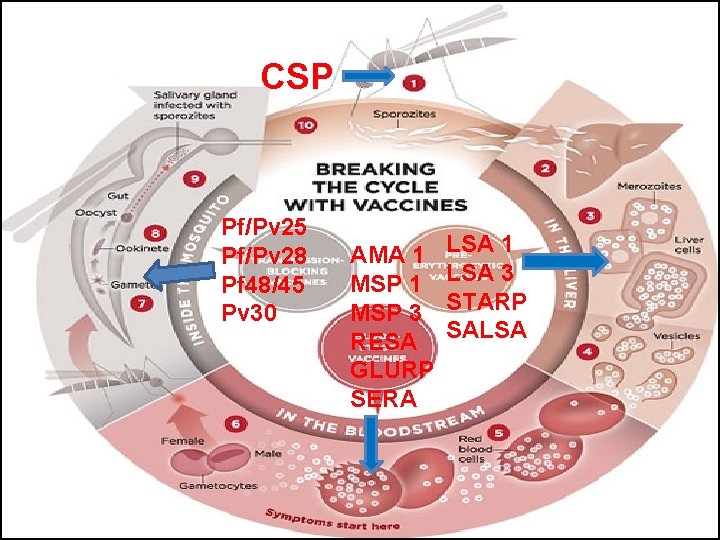
CSP Pf/Pv 25 Pf/Pv 28 Pf 48/45 Pv 30 AMA 1 MSP 3 RESA GLURP SERA LSA 1 LSA 3 STARP SALSA
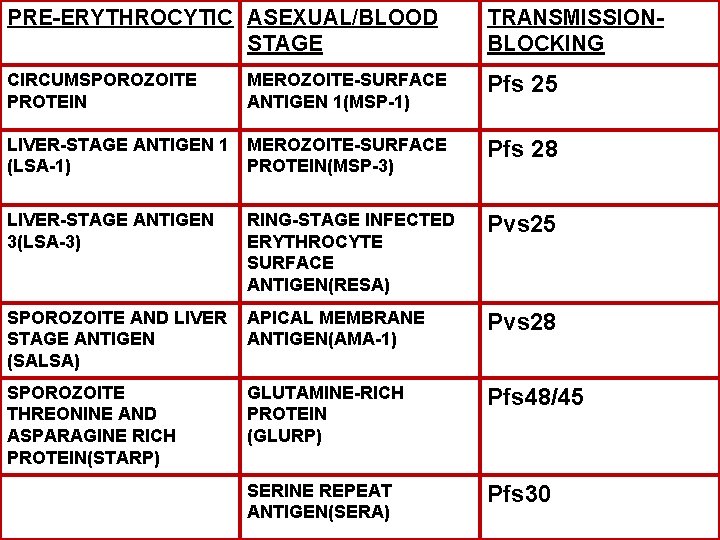
PRE-ERYTHROCYTIC ASEXUAL/BLOOD STAGE TRANSMISSIONBLOCKING CIRCUMSPOROZOITE PROTEIN MEROZOITE-SURFACE ANTIGEN 1(MSP-1) Pfs 25 LIVER-STAGE ANTIGEN 1 (LSA-1) MEROZOITE-SURFACE PROTEIN(MSP-3) Pfs 28 LIVER-STAGE ANTIGEN 3(LSA-3) RING-STAGE INFECTED ERYTHROCYTE SURFACE ANTIGEN(RESA) Pvs 25 SPOROZOITE AND LIVER STAGE ANTIGEN (SALSA) APICAL MEMBRANE ANTIGEN(AMA-1) Pvs 28 SPOROZOITE THREONINE AND ASPARAGINE RICH PROTEIN(STARP) GLUTAMINE-RICH PROTEIN (GLURP) Pfs 48/45 SERINE REPEAT ANTIGEN(SERA) Pfs 30

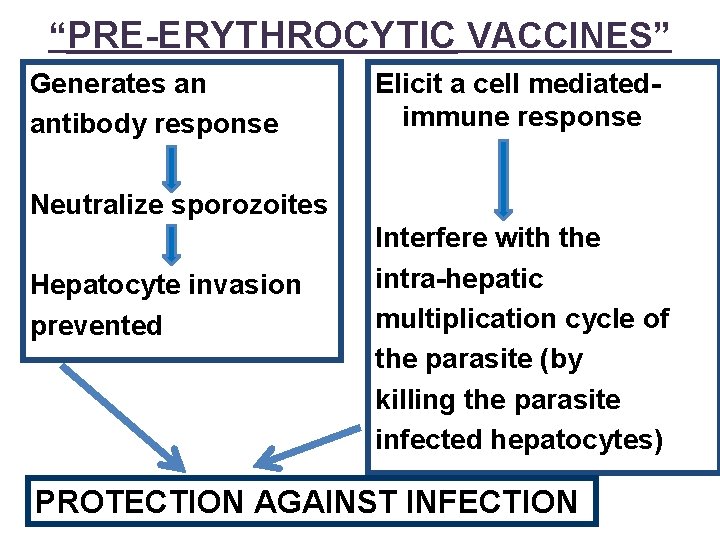
“PRE-ERYTHROCYTIC VACCINES” Generates an antibody response Elicit a cell mediatedimmune response Neutralize sporozoites Hepatocyte invasion prevented Interfere with the intra-hepatic multiplication cycle of the parasite (by killing the parasite infected hepatocytes) PROTECTION AGAINST INFECTION
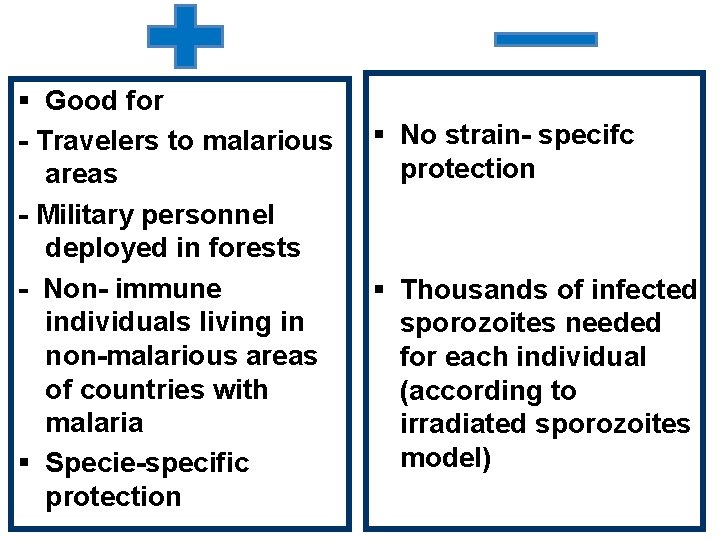
§ Good for - Travelers to malarious areas - Military personnel deployed in forests - Non- immune individuals living in non-malarious areas of countries with malaria § Specie-specific protection § No strain- specifc protection § Thousands of infected sporozoites needed for each individual (according to irradiated sporozoites model)
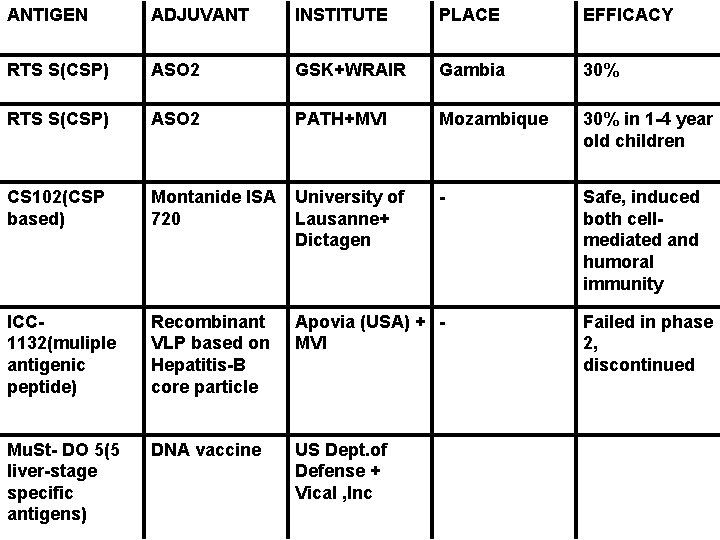
ANTIGEN ADJUVANT INSTITUTE PLACE EFFICACY RTS S(CSP) ASO 2 GSK+WRAIR Gambia 30% RTS S(CSP) ASO 2 PATH+MVI Mozambique 30% in 1 -4 year old children CS 102(CSP based) Montanide ISA 720 University of Lausanne+ Dictagen - Safe, induced both cellmediated and humoral immunity ICC 1132(muliple antigenic peptide) Recombinant VLP based on Hepatitis-B core particle Apovia (USA) + MVI Mu. St- DO 5(5 liver-stage specific antigens) DNA vaccine US Dept. of Defense + Vical , Inc Failed in phase 2, discontinued

CIRCUMSPOROZOITE PROTEIN § Expressed in large amounts on the surface of the sporozoite and of the infected hepatocyte § Central area of repeated amino-acid sequences NANP that are highly immunogenic and present in all but varies among all Plasmodium species § Candidate vaccine developed by GSK and WRAIR RTS, S

RTS, S § Expressed in Saccharomyces cerevisiae § RTS- corresponds to amino acids 207 to 395 of P. falciparum CSP § S- hepatocyte B virus antigen(HBs. Ag) § ASO 2 A- Adjuvant mixture based on monophosphoryl lipid A (oil-in-water emulsion) and QS 21 (a saponin derivative) § Field trials in - Gambia (65% efficacy, for 2 months) (Stoute JA et al. 1998. J Infect Dis) - Mozambique (35. 3% to first episode & 48. 6% against severe disease) (Alonso PL et al. 2005. Lancet))

INITIATIVES BY MVI AND PATH • Trials in Gabon, Ghana, Kenya, Senegal, Tanzania • Focus on infants and young children • Defining appropriate dosage schedule, incorporating it in Expanded Program on Immunization, and the best adjuvant RECENT ADVANCES § Different adjuvants (ASO 1 B, Montanide ISO 720) § No immune interference with concurrent administration of LSA 1/AS 01 B and RTS, S/AS 01 B at separate sites (Pichyangkul et al. 2008. Infection and Immunity)
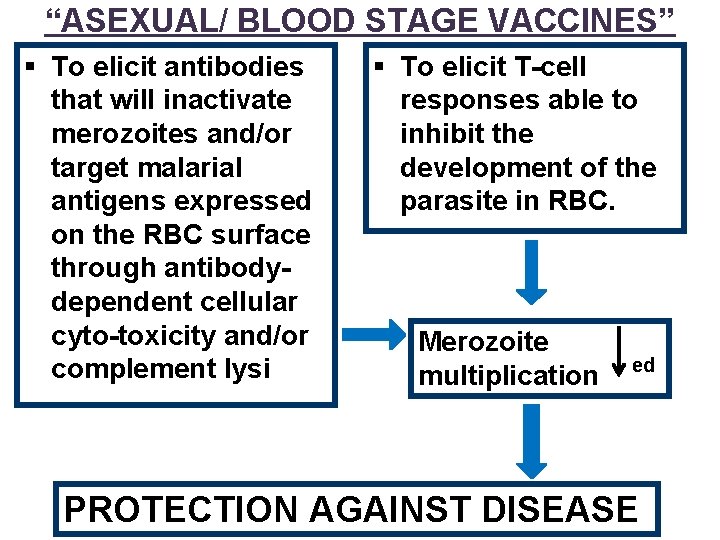
“ASEXUAL/ BLOOD STAGE VACCINES” § To elicit antibodies that will inactivate merozoites and/or target malarial antigens expressed on the RBC surface through antibodydependent cellular cyto-toxicity and/or complement lysis § To elicit T-cell responses able to inhibit the development of the parasite in RBC. Merozoite multiplication ed PROTECTION AGAINST DISEASE
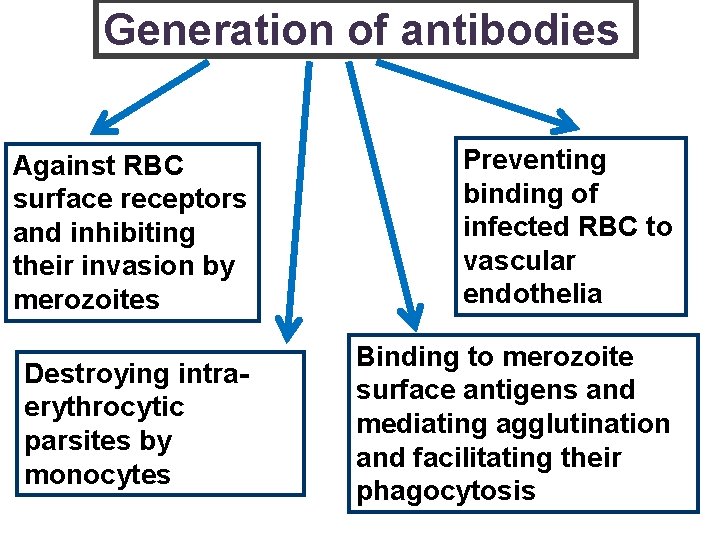
Generation of antibodies Against RBC surface receptors and inhibiting their invasion by merozoites Preventing binding of infected RBC to vascular endothelia Destroying intraerythrocytic parsites by monocytes Binding to merozoite surface antigens and mediating agglutination and facilitating their phagocytosis
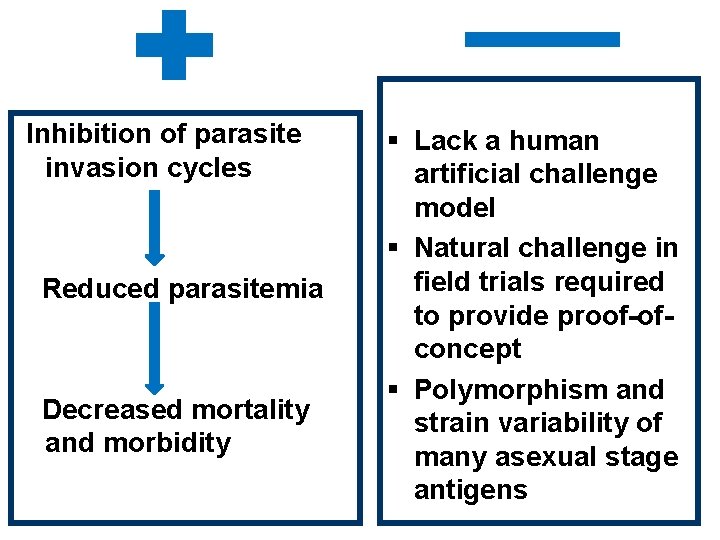
Inhibition of parasite invasion cycles Reduced parasitemia Decreased mortality and morbidity § Lack a human artificial challenge model § Natural challenge in field trials required to provide proof-ofconcept § Polymorphism and strain variability of many asexual stage antigens
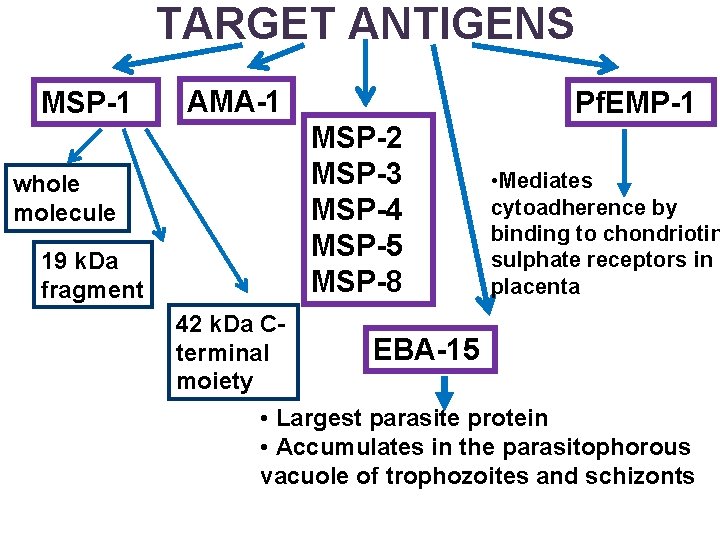
TARGET ANTIGENS MSP-1 AMA-1 Pf. EMP-1 MSP-2 MSP-3 MSP-4 MSP-5 MSP-8 whole molecule 19 k. Da fragment 42 k. Da Cterminal moiety • Mediates cytoadherence by binding to chondriotin sulphate receptors in placenta EBA-15 • Largest parasite protein • Accumulates in the parasitophorous vacuole of trophozoites and schizonts
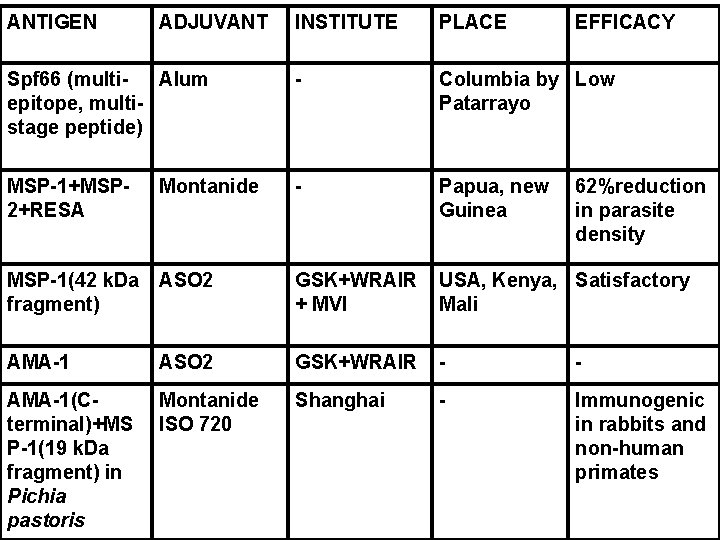
ANTIGEN ADJUVANT INSTITUTE PLACE Spf 66 (multi. Alum epitope, multistage peptide) - Columbia by Low Patarrayo MSP-1+MSP 2+RESA - Papua, new Guinea MSP-1(42 k. Da ASO 2 fragment) GSK+WRAIR + MVI USA, Kenya, Satisfactory Mali AMA-1 ASO 2 GSK+WRAIR - - AMA-1(Cterminal)+MS P-1(19 k. Da fragment) in Pichia pastoris Montanide ISO 720 Shanghai - Immunogenic in rabbits and non-human primates Montanide EFFICACY 62%reduction in parasite density
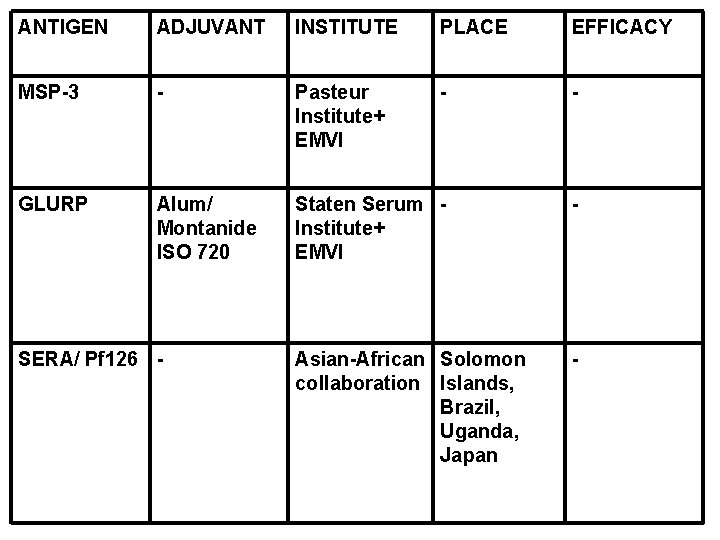
ANTIGEN ADJUVANT INSTITUTE PLACE EFFICACY MSP-3 - Pasteur Institute+ EMVI - - GLURP Alum/ Montanide ISO 720 Staten Serum Institute+ EMVI - Asian-African Solomon collaboration Islands, Brazil, Uganda, Japan - SERA/ Pf 126 -

§ A Bicistronic DNA Vaccine Containing Apical Membrane Antigen 1 and Merozoite Surface Protein 4/5 Can Prime Humoral and Cellular Immune Responses and Partially Protect Mice against Virulent Plasmodium chabaudi adami DS Malaria (Rainczuk et al. 2004. Infection and Immunity) § Genetic diversity of vaccine candidate antigens in Plasmodium falciparum isolates from the Amazon basin of Peru (Chenet et al. 2008. Malaria Journal) § Sequence diversity and natural selection at domain 1 of AMA-1 among P. falciparum in Indian population (More diversity in Assam, Orissa, Andaman & Nicobar islands as compared to UP & Goa)

“TRANSMISSION-BLOCKING VACCINES” § To prevent or decrease transmission of the parasite host in the community, not to prevent illness or infection in the vaccinated individual § Altruistic vaccine (does not protect the vaccinee) § Assessing their impact by the field trials in Phase 3 not possible
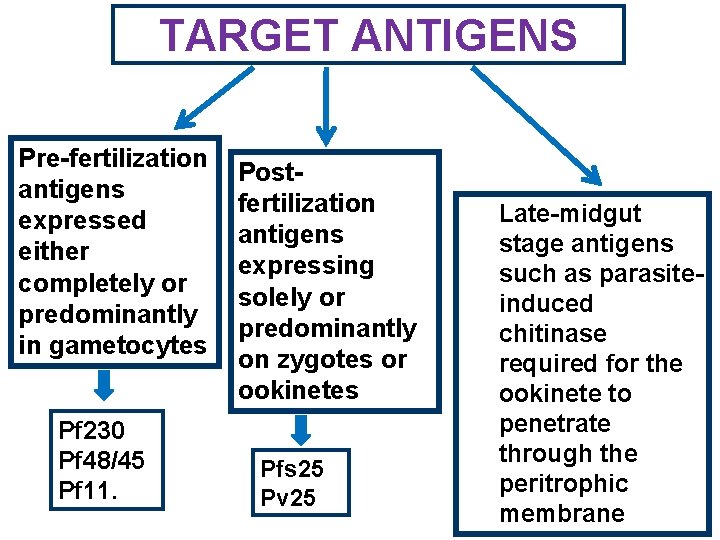
TARGET ANTIGENS Pre-fertilization antigens expressed either completely or predominantly in gametocytes Pf 230 Pf 48/45 Pf 11. 1 Postfertilization antigens expressing solely or predominantly on zygotes or ookinetes Pfs 25 Pv 25 Late-midgut stage antigens such as parasiteinduced chitinase required for the ookinete to penetrate through the peritrophic membrane

§ Nasal Immunization with a Malaria Transmission Blocking Vaccine Candidate, Pfs 25, Induces Complete Protective Immunity in Mice against Field Isolates of Plasmodium falciparum (Arakawa et al. 2005. Infection and Immunity)
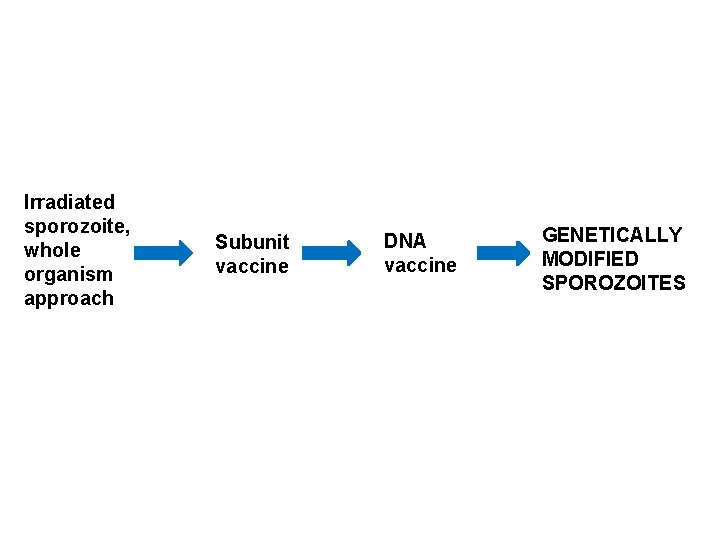
Irradiated sporozoite, whole organism approach Subunit vaccine DNA vaccine GENETICALLY MODIFIED SPOROZOITES

DNA VACCINES § SAFE as they don’t contain any pathogenic organism that may revert in virulence § PLASMID DNA- A STABLE MOLECULE as compared to recombinant or live-attenuated vaccines, making storage and distribution convenient in tropical countries, where cold chain is difficult to maintain § Simple administration (IM or ID) § Multiple plasmids can be incorporated to multivalent vaccine

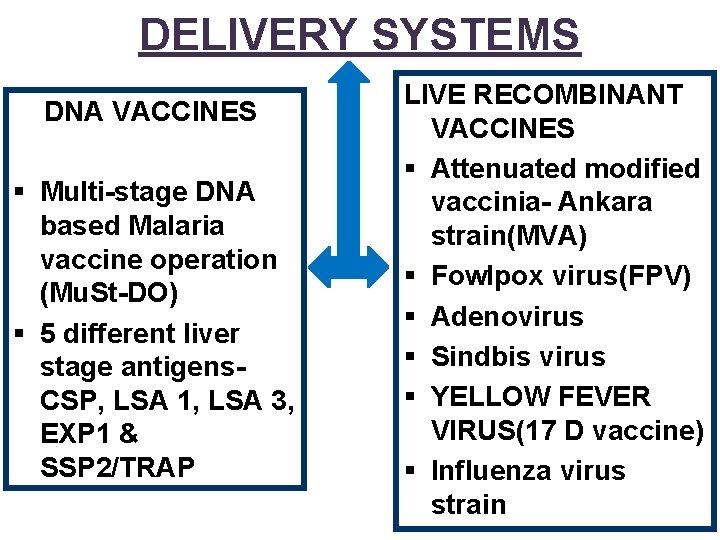
DELIVERY SYSTEMS DNA VACCINES § Multi-stage DNA based Malaria vaccine operation (Mu. St-DO) § 5 different liver stage antigens. CSP, LSA 1, LSA 3, EXP 1 & SSP 2/TRAP LIVE RECOMBINANT VACCINES § Attenuated modified vaccinia- Ankara strain(MVA) § Fowlpox virus(FPV) § Adenovirus § Sindbis virus § YELLOW FEVER VIRUS(17 D vaccine) § Influenza virus strain

§ Conjugating recombinant proteins to Pseudomonas aeruginosa exo. Protein A: a strategy for enhancing immunogenicity of malaria vaccine candidates (Feng Qian et al. 2007. Vaccine)

“FUTURE INSIGHTS” • Genetically-modified sporozoite vaccines • An anti-parasite toxin vaccine that targets the parasite toxins which contribute to the disease, such as the glycosyl-phosphatidyl-inositol (GPI) anchor

GENETICALLY MODIFIED SPOROZOITES § Safe § Genetically stable § Cryo-preservation possible(upto 10 years) § Large scale production feasible § Prioritizing protective antigens and tool for subunit vaccine development § Genes targetted- UIS 3

“PROBLEMS TO BE ADDRESSED”

§ Designing a malaria vaccine (identification of the appropriate antigen and formulation of the adjuvant) § Obscurity about different immune mechanisms in malaria § Choice of clinical-case definitions and end-points in malaria vaccine trials

§ Lack of funding agencies (with no commercial intentions) § Lack of immunological memory § Lack of definite biological assays and animal models to correlate with vaccine strategy


BIBLIOGRAPHY - Girard et al. 2007. A review of human vaccine research and development: Malaria. Vaccine. 25: 5671580 - Matuschewski K. 2006. Vaccine development against malaria. Current Opinion in Immunology. 18: 449 -457 - Jones et al. 1994. Malaria vaccine development. Clinical Microbiology Review. 7: 303 -310 - Phillips R S. 2001. Current status of malaria and potential for control. Clinical Microbiology Review. 14: 208 -226 - www. malariavaccine. org - www. who. int
- Slides: 53