Malaria Diagnostics and Medicines WHOUNICEF Technical Briefing Seminar

Malaria Diagnostics and Medicines WHO/UNICEF Technical Briefing Seminar Essential Medicines Policies 1 November 2011 - Geneva Silvia Schwarte Diagnosis, Treatment and Vaccines Global Malaria Programme

Diagnosis: RDTs 2 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
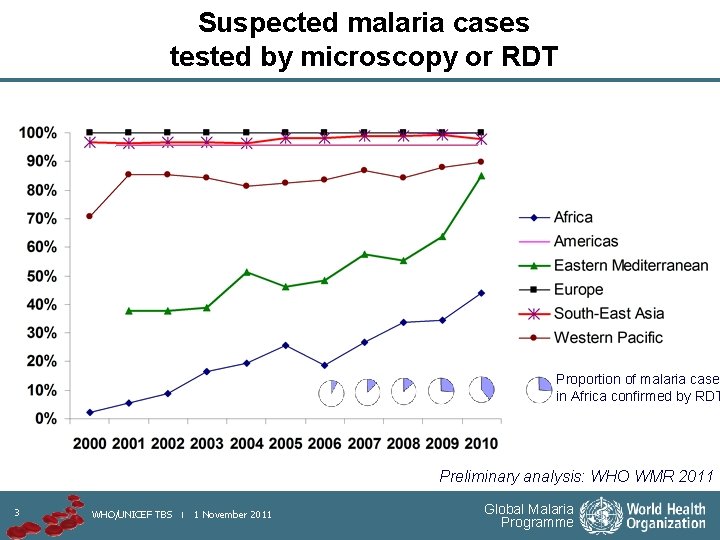
Suspected malaria cases tested by microscopy or RDT Proportion of malaria cases in Africa confirmed by RDT Preliminary analysis: WHO WMR 2011 3 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
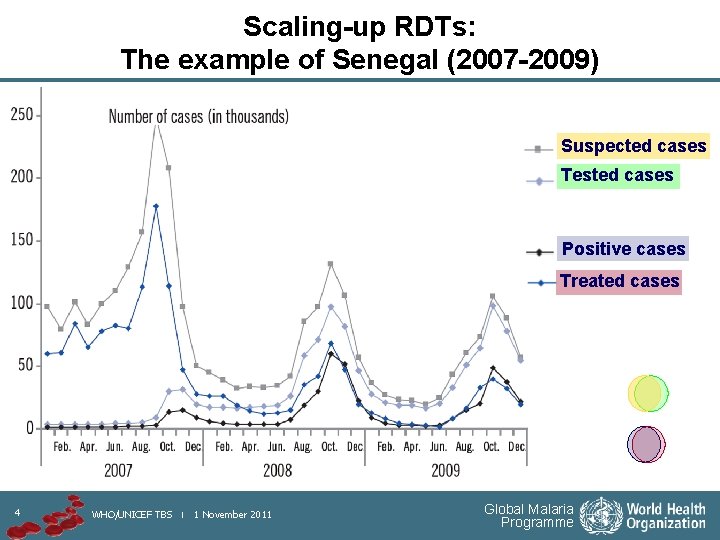
Scaling-up RDTs: The example of Senegal (2007 -2009) Suspected cases Tested cases Positive cases Treated cases 4 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
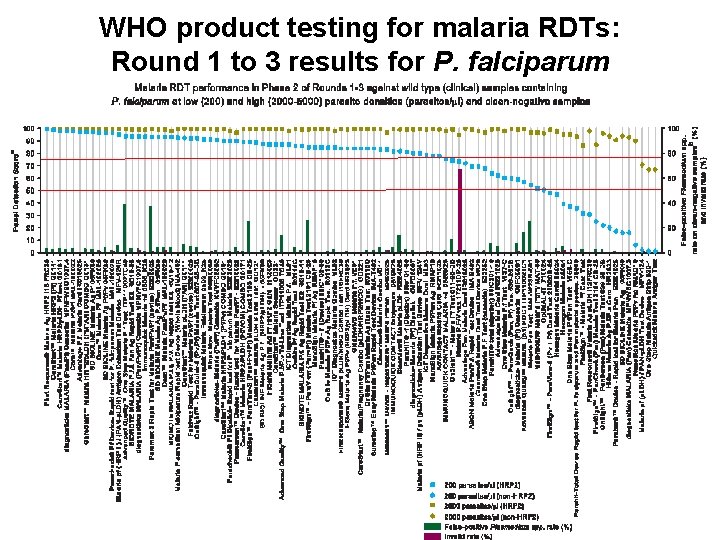
WHO product testing for malaria RDTs: Round 1 to 3 results for P. falciparum 5 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
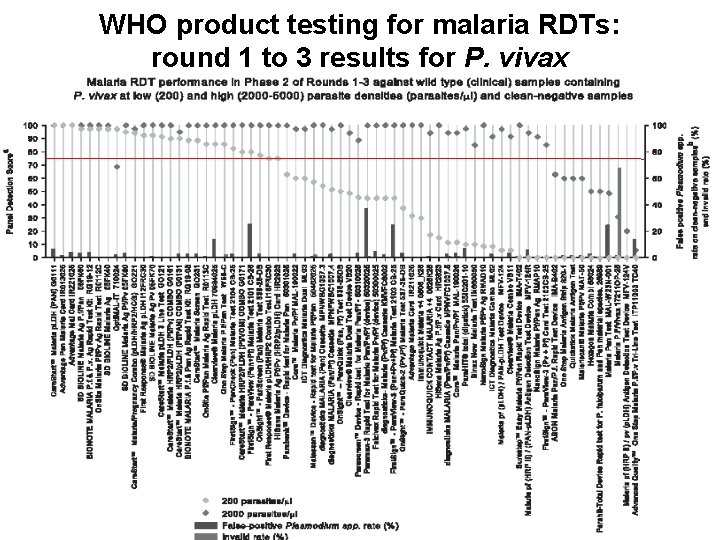
WHO product testing for malaria RDTs: round 1 to 3 results for P. vivax 6 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
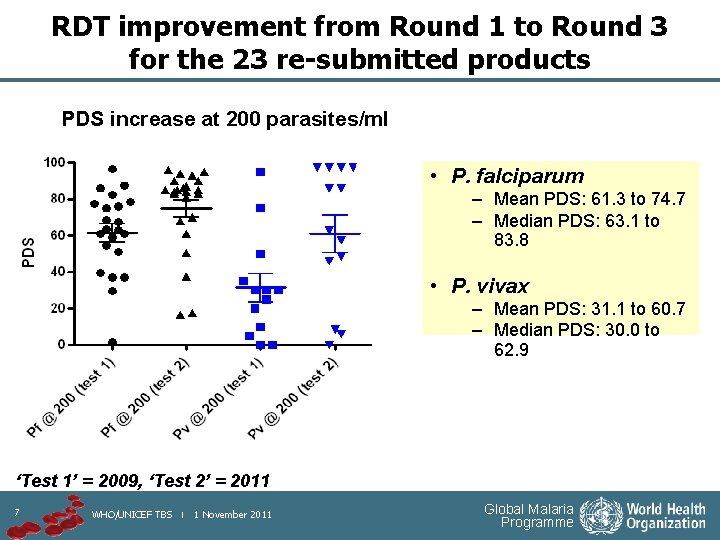
RDT improvement from Round 1 to Round 3 for the 23 re-submitted products PDS increase at 200 parasites/ml • P. falciparum – Mean PDS: 61. 3 to 74. 7 – Median PDS: 63. 1 to 83. 8 • P. vivax – Mean PDS: 31. 1 to 60. 7 – Median PDS: 30. 0 to 62. 9 ‘Test 1’ = 2009, ‘Test 2’ = 2011 7 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
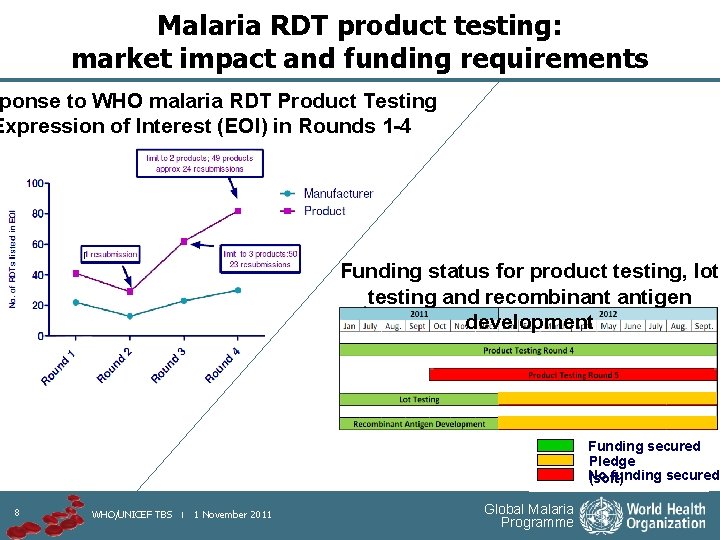
Malaria RDT product testing: market impact and funding requirements ponse to WHO malaria RDT Product Testing Expression of Interest (EOI) in Rounds 1 -4 Funding status for product testing, lot testing and recombinant antigen development Funding secured Pledge No funding secured (soft) 8 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme

Treatment: ACTs 9 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
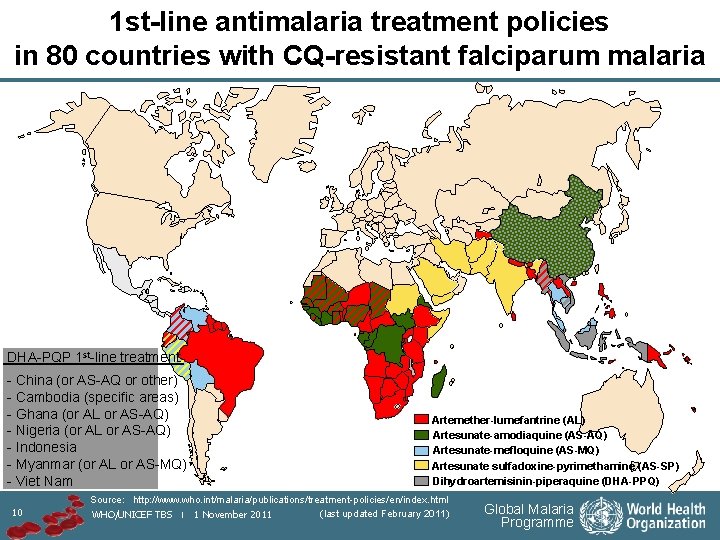
1 st-line antimalaria treatment policies in 80 countries with CQ-resistant falciparum malaria DHA-PQP 1 st-line treatment: - China (or AS-AQ or other) - Cambodia (specific areas) - Ghana (or AL or AS-AQ) - Nigeria (or AL or AS-AQ) - Indonesia - Myanmar (or AL or AS-MQ) - Viet Nam 10 Artemether-lumefantrine (AL) Artesunate-amodiaquine (AS-AQ) Artesunate-mefloquine (AS-MQ) Artesunate sulfadoxine-pyrimethamine (AS-SP) Dihydroartemisinin-piperaquine (DHA-PPQ) Source: http: //www. who. int/malaria/publications/treatment-policies/en/index. html (last updated February 2011) WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
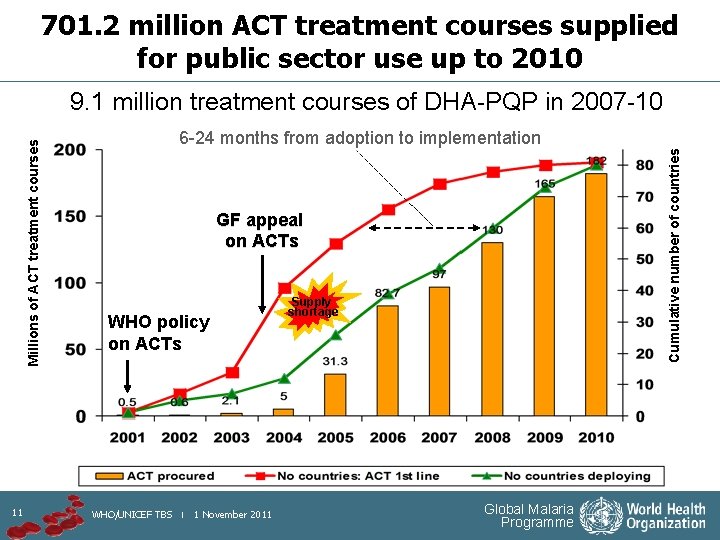
701. 2 million ACT treatment courses supplied for public sector use up to 2010 11 6 -24 months from adoption to implementation Cumulative number of countries Millions of ACT treatment courses 9. 1 million treatment courses of DHA-PQP in 2007 -10 GF appeal on ACTs WHO policy on ACTs WHO/UNICEF TBS | 1 November 2011 Supply shortage Global Malaria Programme

Tightening relation between global ACT supply and demand Warning signs since July 2011: • Longer lead times for ACT orders, i. e. >> 2 -4 months • Limited capacity of major ACT manufacturers to accept additional large orders for delivery in 2011 • Increased price of artemisinin on the “spot” market Contributing factors: • Increased volume and number of ACT orders from First-Line-Buyers to AMFm in May - June 2011 ("levers" recently introduced) Total orders confirmed: 135. 7 million (as of 29. 6 12 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme

Vaccines 13 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme

Malaria Vaccine (I) RTS, S/AS 01 Phase 3 Trial ongoing enrolling over 15, 000 African infants RTS, S/AS 01 – RT: Malaria protein (Segment of P. falciparum sporozoite surface protein) – S, S: Hepatitis B protein – AS 01: Adjuvant system 01 (most potent of many adjuvants tested) • Developed by GSK with funding from BMGF through PATH Malaria Vaccine Initiative (MVI) • Phase 2 data shows 40 -60% efficacy over up to 18 months against clinical malaria in 2 recent field trials • Currently in Phase 3 trials, at least 5 years ahead of other candidates • WHO Malaria Vaccine Technical Expert Group constituted in 2009, joint between Vaccine and Malaria Departments (JTEG) 14 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme

Malaria Vaccine (II) Timing for WHO policy recommendations • First interim data became publicly available on 18 Oct 2011 (NEJM), in a non-target population (5 -17 months olds without co-administration of other EPI vaccines): these data will not lead to WHO policy recommendation. • JTEG has advised WHO to base policy recommendation on the review of: – full Phase 3 data in the target population (infants 6 weeks old at first vaccination in co-administration with EPI vaccines) with 30 months follow-up, – site-specific clinical malaria efficacy, – severe malaria efficacy, and – impact of booster dose at 18 months. • WHO policy recommendation may occur in 2015, if Phase 3 data supports it. Policy-makers should be aware that WHO has defined its policy process and does not expect a policy recommendation before 2015 for RTS, S/AS 01. • For specific queries contact Dr Vasee Moorthy (e-mail: moorthyv@who. int) 15 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme

Thank you 16 WHO/UNICEF TBS | 1 November 2011 Global Malaria Programme
- Slides: 16