Major types of nutrients and digestion Lect 2





















- Slides: 28

Major types of nutrients and digestion Lect 2 Dr. Jehad Al-Shuneigat

Fats • Functions: 1 - Cell structure, 2 - fuel storage, 3 - hormone • essential fatty acids and nonessential fatty acids • nonessential fatty acids can be synthesized in our body • essential fatty acids we get them from food =The essential fatty acids α-linoleic and α-linolenic acid are supplied by dietary plant oils.

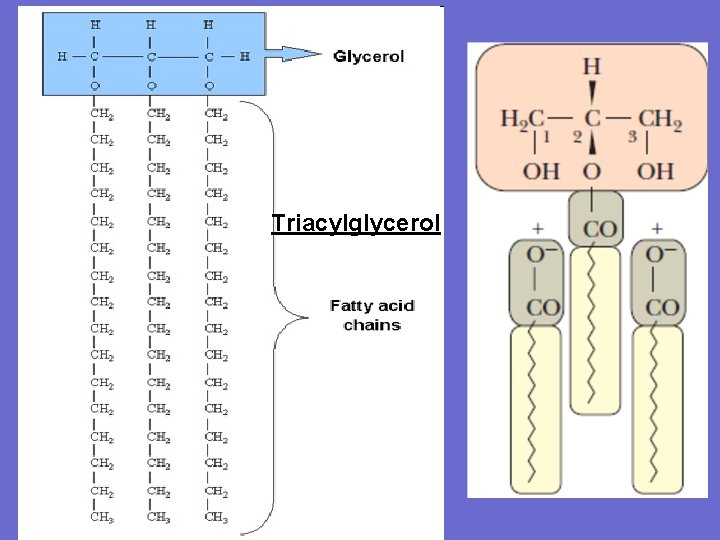
Digestion of fat • Triacylglycerols are the major fat in the human diet because they are the major storage lipid in the plants and animals that constitute our food supply. • The remainder of the dietary lipids consists primarily of cholesterol, phospholipids, and free fatty acids. • Triacylglycerols contain a glycerol backbone to which three fatty acids are esterified. • The main route for digestion of triacylglycerols involves hydrolysis to fatty acids and 2 -monoacylglycerols in the lumen of the intestine. However, the route depends to some extent on the chain length of the fatty acids.

Triacylglycerol

Digestion of fat A. Mouth and stomach Limited digestion of lipids occurs in the mouth and stomach because of the low solubility. 1. Lingual lipase produced by cells at the back of the tongue 2. Gastric lipase produced by stomach • Same function for both enzymes: to hydrolyze short- and medium-chain fatty acids (containing 12 or fewer carbon atoms).

• B. In small intestine • 1. Action of Bile: • Emulsification (suspended in small particles in the aqueous environment) by bile salts. • The biles are amphipathic compounds • The contraction of the gallbladder and secretion of pancreatic enzymes are stimulated by the gut hormone cholecystokinin, which is secreted by the intestinal cells when stomach contents enter the intestine. • Bile act as detergents, binding to dietary fat as they are broken up by the action of the intestinal muscle. This emulsified fat, which has an increased surface area as compared with unemulsified fat, is attacked by digestive enzymes from the pancreas.

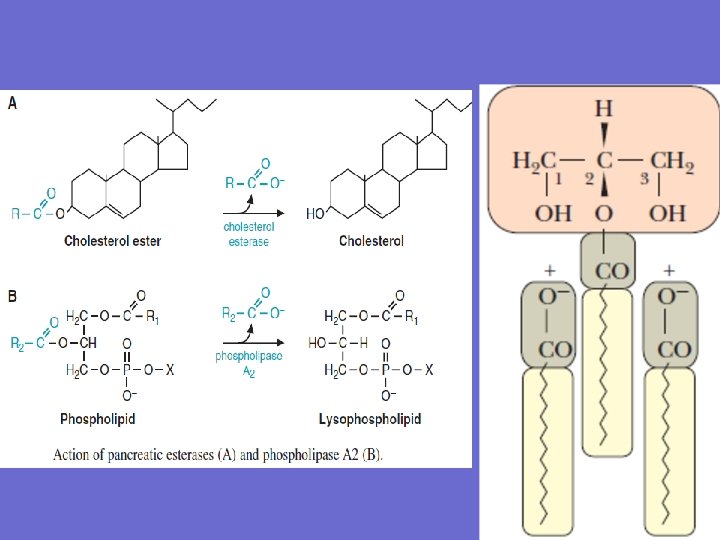
2. Action of Pancreatic secretion A- Bicarbonate (hormone secretin released from the intestine when acid enters the duodenum) raises the p. H of the contents of the intestinal lumen into a range (p. H ~ 6. 5). B-Lipase digests dietary triacylglycerols producing 2 free fatty acids and 2 -monoacylglycerol C-Colipase it binds to the dietary fat and to the lipase, thereby increasing lipase activity. D- Esterases remove fatty acids from compounds such as cholesterol esters (The ester bond is formed between the carboxylate group of a fatty acid and the hydroxyl group of cholesterol. ) E- phospholipase A 2 digests phospholipids to a free fatty acid and a lysophospholipid


• Absorption of digested fats • Short- and medium-chain fatty acids (C 4 to C 12) are absorbed directly into intestinal epithelial cells, they enter the portal blood and are transported to the liver bound to serum albumin. • The digested fatty acids and 2 -monoacylglycerols are resynthesized into triacylglycerols in intestinal epithelial cells, then packaged in lipoprotein particles chylomicrons because they are insoluble in water, and secreted by way of the lymph into the blood. • If triacylglycerols directly entered the blood, they would come together, impeding blood flow. • The fatty acids of the chylomicron triacylglycerols are stored mainly as triacylglycerols in adipose cells.

Protein digestion • Function: • Cytoskeleton, movement (actin and myosin ), transport (Hb), immune protection (antibodies), receptors and as catalysts enzymes • High and low quality protein • Amino acids are the building blocks of proteins. • The sequence of amino acids in a protein is determined by the genetic code. Four levels of protein structure are commonly defined: Primary structure, Secondary structure, Tertiary structure, and Quaternary structure • There are 20 different amino acids 8 essential and 12 nonessential amino acids that form human proteins.

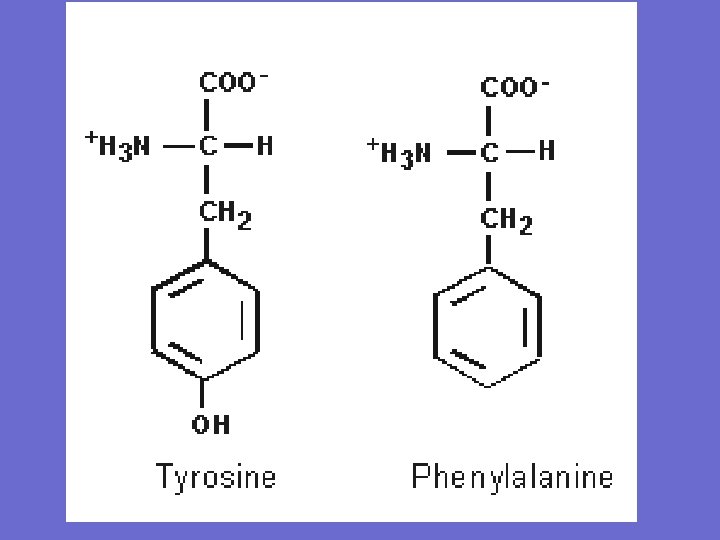
Conditionally essential amino acids • Children and pregnant women have a high rate of protein synthesis to support growth, and require more of arginine and histidine than their body synthesise. • Tyrosine is also considered conditionally essential. Tyrosine is synthesized from phenylalanine (by hydroxylation of phenylalanine), and it is required in the diet if phenylalanine intake is inadequate, or if an individual is congenitally deficient in an enzyme required to convert phenylalanine to tyrosine (the congenital disease phenylketonuria).


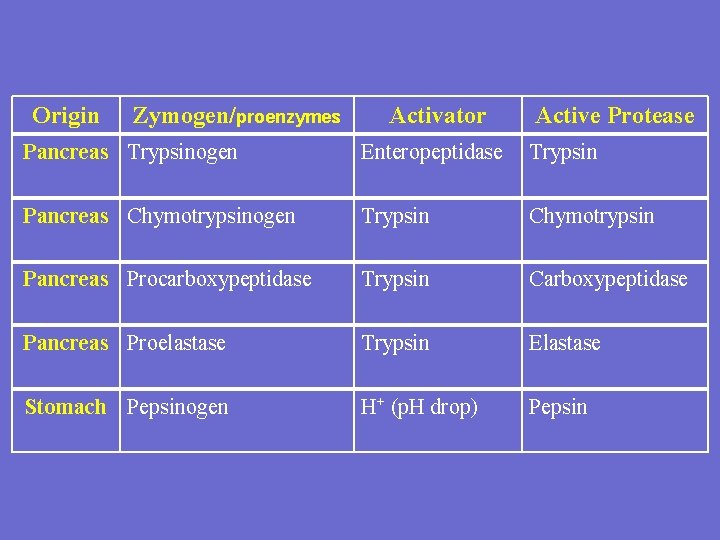
• Zymogens • Inactive enzymes precursors are called zymogens or proenzymes, • It only acquire full activity upon specific proteolytic cleavage of one or several of their peptide bonds. • Zymogen activation by specific proteolysis is an irreversible process. • Important to switch on processes at the appropriate time and place • The synthesis of zymogens as inactive precursors prevents them from cleaving proteins prematurely at their sites of synthesis or secretion.

Origin Zymogen/proenzymes Activator Active Protease Pancreas Trypsinogen Enteropeptidase Trypsin Pancreas Chymotrypsinogen Trypsin Chymotrypsin Pancreas Procarboxypeptidase Trypsin Carboxypeptidase Pancreas Proelastase Trypsin Elastase Stomach Pepsinogen H+ (p. H drop) Pepsin

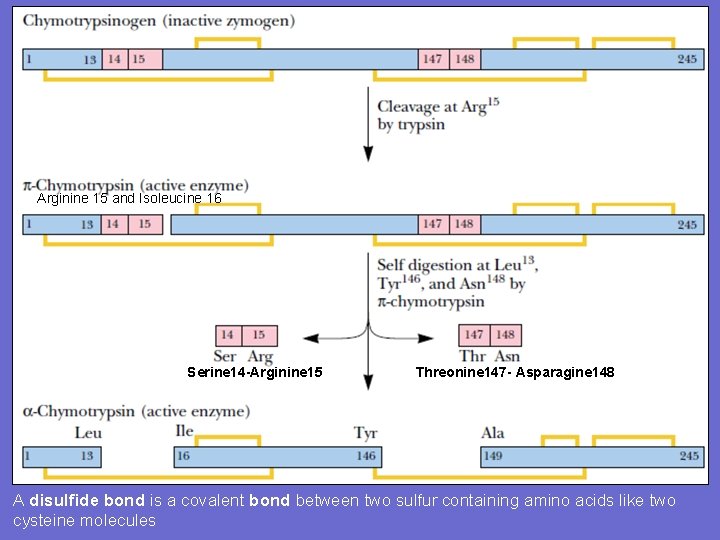
• Activation of chymotrypsinogen • Chymotrypsinogen is a 245 -aa cross-linked by five disulfide bonds. • Chymotrypsinogen is converted to an enzymatically active form called π-chymotrypsin when trypsin cleaves the peptide bond joining Arginine 15 and Isoleucine 16. • The enzymatically active π-chymotrypsin acts upon other π-chymotrypsin molecules, removing two dipeptides, Serine 14 -Arginine 15 and Threonine 147 - Asparagine 148. • The end product of this processing pathway is the mature protease α-chymotrypsin, in which the three peptide chains. • A (residues 1 through 13), B (residues 16 through 146), and C (residues 149 through 245), remain together because they are linked by two disulfide bonds, one from A to B, and one from B to C.

Arginine 15 and Isoleucine 16 Serine 14 -Arginine 15 Threonine 147 - Asparagine 148 A disulfide bond is a covalent bond between two sulfur containing amino acids like two cysteine molecules

• Digestion of Proteins in the Stomach # Gastrin hormone (gastric mucosa) stimulates the secretion A- Pepsinogen by the chief cells of the gastric glands. B- Hydrochloric acid by the parietal cells A- Pepsinogen is activated to its active form pepsin by acidic gastric juice (p. H 1. 0 to 2. 5) that alters the conformation of so that it can cleave itself, producing the active pepsin. • Pepsin acts as an endopeptidase, cleaving peptide bonds at various points within the protein chain. B- Stomach acidity: causes dietary proteins denaturation, this serves to inactivate the proteins and partially unfolds them such that they are better substrates for proteases. • Smaller peptides and some free amino acids are produced.

• Digestion of Proteins by Enzymes from the Pancreas 1 - Secretin hormone: stimulates bicarbonate secretion 2 - Bicarbonate causes raises the p. H such that the pancreatic proteases (the hormone cholecystokinin stimulate its release), which are also present in pancreatic secretions, can be active. A- Trypsinogen is cleaved to form trypsin by enteropeptidase (a protease) secreted by the brush-border cells of the small intestine. • Trypsin is most specific that cleaves peptide bonds between lysine or arginine (cleaves peptide bonds of basic amino acids (+ve)). B- Trypsin catalyzes conversion of = chymotrypsinogen to chymotrypsin: that favors residues that contain hydrophobic or acidic amino acids. = proelastase to elastase: that cleaves elastin and proteins with small side chains (alanine, glycine, or serine). = procarboxypeptidases to carboxypeptidases.

• Carboxypeptidase A preferentially releases hydrophobic amino acids, whereas • Carboxypeptidase B releases basic amino acids (arginine and lysine). • These exopeptidases remove amino acids from the carboxyl ends of peptide chains. • Sequences of amino acids in a peptide are read from the amino terminal end to the carboxy-terminal end.

• Digestion of Proteins by Enzymes from Intestinal Cells • Exopeptidases produced by intestinal epithelial cells act within the brush border and also within the cell. • Aminopeptidases, located on the brush border, cleave one amino acid at a time from the amino end of peptides. • Amino acids are absorbed from the intestinal lumen through Na+- dependent transport systems and through facilitated diffusion.

Vitamins • Vitamins are organic molecules • Most vitamins are used for the 1 -Synthesis of coenzymes 2 - hormones 3 - antioxidants • Vitamins, cannot be synthesized in the body or are synthesized from a very specific dietary precursor in insufficient amounts. • For example, we can synthesize the vitamin niacin (B 3) from the essential amino acid tryptophan, but not in sufficient quantities to meet our needs. Niacin is therefore still classified as a vitamin. The vitamins are often divided into two classes, 1 - water-soluble vitamins and 2 - fat-soluble vitamins.

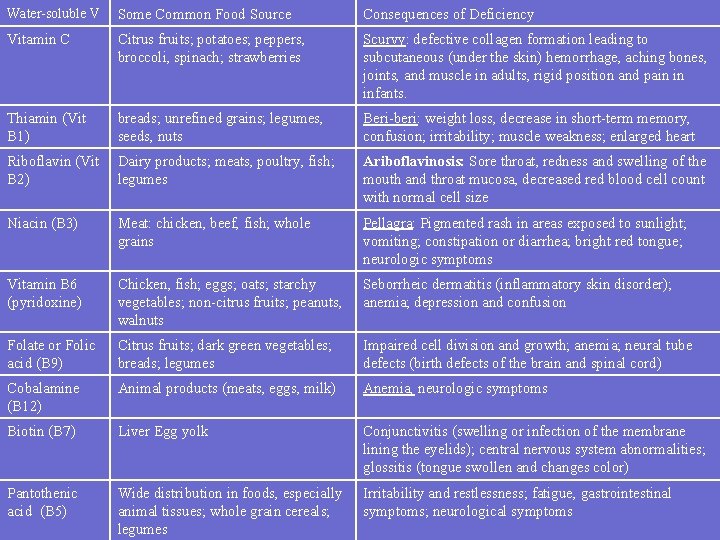
Water-soluble V Some Common Food Source Consequences of Deficiency Vitamin C Citrus fruits; potatoes; peppers, broccoli, spinach; strawberries Scurvy: defective collagen formation leading to subcutaneous (under the skin) hemorrhage, aching bones, joints, and muscle in adults, rigid position and pain in infants. Thiamin (Vit B 1) breads; unrefined grains; legumes, seeds, nuts Beri-beri: weight loss, decrease in short-term memory, confusion; irritability; muscle weakness; enlarged heart Riboflavin (Vit B 2) Dairy products; meats, poultry, fish; legumes Ariboflavinosis: Sore throat, redness and swelling of the mouth and throat mucosa, decreased red blood cell count with normal cell size Niacin (B 3) Meat: chicken, beef, fish; whole grains Pellagra: Pigmented rash in areas exposed to sunlight; vomiting; constipation or diarrhea; bright red tongue; neurologic symptoms Vitamin B 6 (pyridoxine) Chicken, fish; eggs; oats; starchy vegetables; non-citrus fruits; peanuts, walnuts Seborrheic dermatitis (inflammatory skin disorder); anemia; depression and confusion Folate or Folic acid (B 9) Citrus fruits; dark green vegetables; breads; legumes Impaired cell division and growth; anemia; neural tube defects (birth defects of the brain and spinal cord) Cobalamine (B 12) Animal products (meats, eggs, milk) Anemia, neurologic symptoms Biotin (B 7) Liver Egg yolk Conjunctivitis (swelling or infection of the membrane lining the eyelids); central nervous system abnormalities; glossitis (tongue swollen and changes color) Pantothenic acid (B 5) Wide distribution in foods, especially animal tissues; whole grain cereals; legumes Irritability and restlessness; fatigue, gastrointestinal symptoms; neurological symptoms

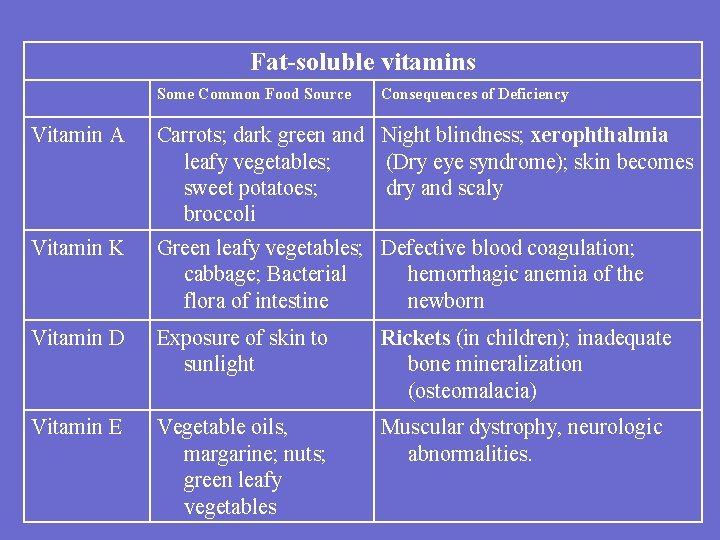
Fat-soluble vitamins Some Common Food Source Consequences of Deficiency Vitamin A Carrots; dark green and Night blindness; xerophthalmia leafy vegetables; (Dry eye syndrome); skin becomes sweet potatoes; dry and scaly broccoli Vitamin K Green leafy vegetables; Defective blood coagulation; cabbage; Bacterial hemorrhagic anemia of the flora of intestine newborn Vitamin D Exposure of skin to sunlight Rickets (in children); inadequate bone mineralization (osteomalacia) Vitamin E Vegetable oils, margarine; nuts; green leafy vegetables Muscular dystrophy, neurologic abnormalities.

Water (H 2 O) • Water constitutes about 70% of the weight of human body. • The intake of water required per day depends on the balance between the amount produced an lost by body. properties of water 1 - It is a polar molecule that makes it a highly interacting molecule. 2 - Rapid formation, dissociation and reformation of the hydrogen bonds maintain fluidity of water. 3 - These hydrogen bonds give water molecules the property of cohesion and adhesion. Cohesion is the hydrogen bonding of water molecules while adhesion is the bonding of water molecules to other compounds. 4 - Water functions as a universal solvent in which almost all polar and ionic substances can dissolve. 5 - It provides a matrix for macromolecules to assume their biologically active conformations. 6 - Water is a medium for diffusion, transport and solvents.

Minerals form about 1 to 3% of the cells composition. Function of minerals 1 - Minerals are component of certain organic compounds for example iron in haemoglobin. 2 - Minerals serve as the components of various tissues, like calcium phosphate in the bones. 3 - Minerals serve as cofactors and activators of several enzymes. Copper is a cofactor of the enzyme cytochrome oxidase. 4 - Minerals take part in several biological processes including: = Calcium plays a very important role in the clotting of blood. = Some mineral salts and ions function as buffers, maintaining the p. H of body fluids. Some minerals form the raw materials for the synthesis of metabolically important hormones. Iodine is involved in the synthesis of thyroid hormone.

• Xenobiotics • Have no nutritional value, are of no use in the body, and can be harmful if consumed in excessive amounts • They include drugs, food additives, pollutants, etc. • Pharmacologic and recreational drugs are also xenobiotic compounds. • The liver is the principal site in the body for the degradation of these compounds.

• Body fuel stores • A. Fat • Fat is our major fuel store, which is located in adipose tissue as adipose triacylglycerol (triglyceride), a lipid more commonly known as fat. Although fat is distributed throughout our bodies, it tends to increase in quantity in our hips and thighs and in our abdomens as we advance into middle age. • The average 70 -kg man has approximately 15 kg stored triacylglycerol, which accounts for approximately 85% of his total stored calories. • Two characteristics make adipose triacylglycerol a very efficient fuel store: 1 - Triacylglycerol contains more calories per gram than carbohydrate or protein (9 kcal/g versus 4 kcal/g) 2 - Adipose tissue does not contain much water. Adipose tissue contains only about 15% water, compared to tissues such as muscle that contain about 80%.

• B. Glycogen • Our stores of glycogen in liver, muscle, and other cells are relatively small in quantity but are nevertheless important. Liver glycogen is used to maintain blood glucose levels between meals. • The size of glycogen store in an average 70 -kg man might have 200 g or more of liver glycogen after a meal but only 80 g after an overnight fast. • Muscle glycogen supplies energy for muscle contraction during exercise. At rest, the 70 -kg man has approximately 500 g of muscle glycogen. Almost all cells, including neurons, maintain a small emergency supply of glucose as glycogen. • C. Protein • Protein is not solely a fuel store. Muscle protein is essential for body movement. • Only a limited amount of body protein can be degraded for energy, approximately 6 kg in the average 70 -kg man, before our body functions are compromised.