Major Functions of the Kidneys 1 body fluid




Major Functions of the Kidneys 1. 조절기능 body fluid osmolarity and volume electrolyte balance acid-base balance blood pressure 2. 배설 기능 metabolic products foreign substances (pesticides, chemicals etc. ) excess substance (water, etc) 3. 내분비기능 erythropoitin 1, 25 -dihydroxy vitamin D 3 (vitamin D activation) renin prostaglandin 4. 대사 기능

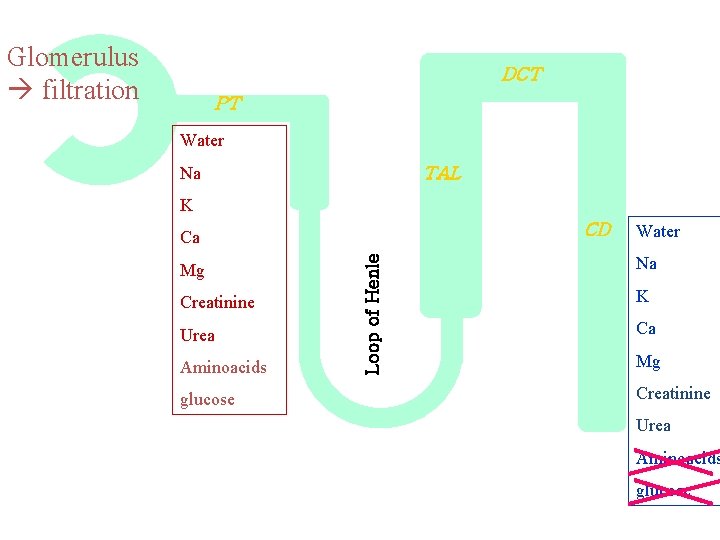
Glomerulus filtration DCT PT Water Na TAL K CD Mg Creatinine Urea Aminoacids glucose Loop of Henle Ca Water Na K Ca Mg Creatinine Urea Aminoacids glucose
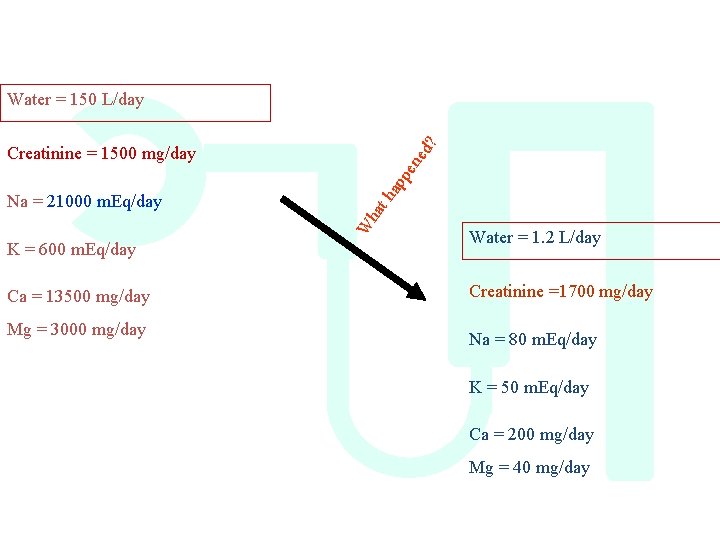
d? Water = 150 L/day K = 600 m. Eq/day Ca = 13500 mg/day Mg = 3000 mg/day th W ha Na = 21000 m. Eq/day ap pe ne Creatinine = 1500 mg/day Water = 1. 2 L/day Creatinine =1700 mg/day Na = 80 m. Eq/day K = 50 m. Eq/day Ca = 200 mg/day Mg = 40 mg/day
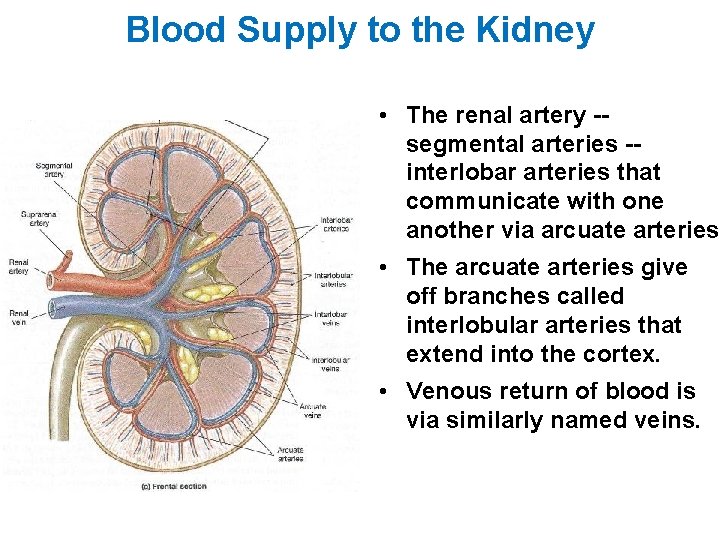
Blood Supply to the Kidney • The renal artery -segmental arteries -interlobar arteries that communicate with one another via arcuate arteries. • The arcuate arteries give off branches called interlobular arteries that extend into the cortex. • Venous return of blood is via similarly named veins.
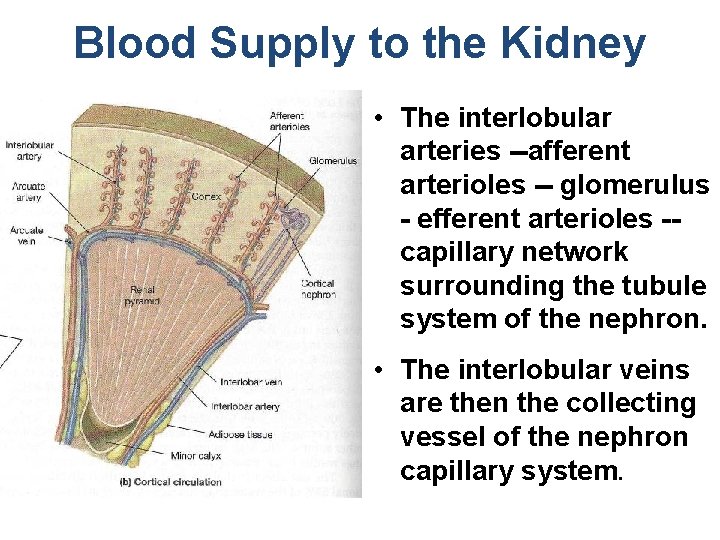
Blood Supply to the Kidney • The interlobular arteries --afferent arterioles -- glomerulus - efferent arterioles -capillary network surrounding the tubule system of the nephron. • The interlobular veins are then the collecting vessel of the nephron capillary system.
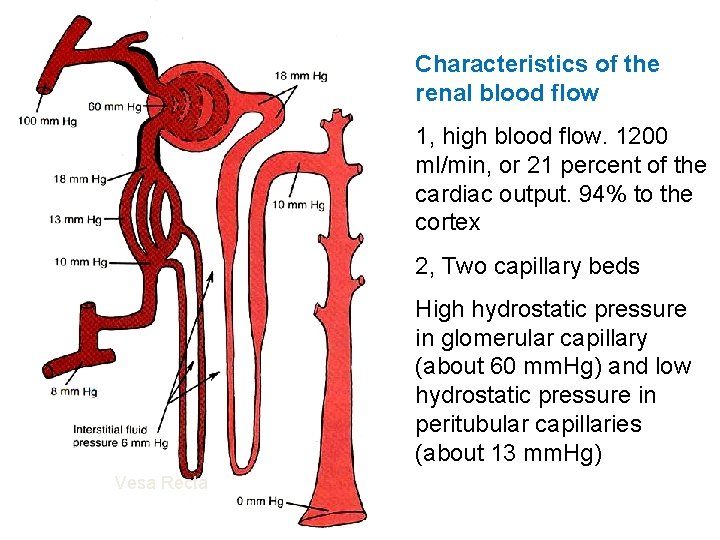
Characteristics of the renal blood flow 1, high blood flow. 1200 ml/min, or 21 percent of the cardiac output. 94% to the cortex 2, Two capillary beds High hydrostatic pressure in glomerular capillary (about 60 mm. Hg) and low hydrostatic pressure in peritubular capillaries (about 13 mm. Hg) Vesa Recta
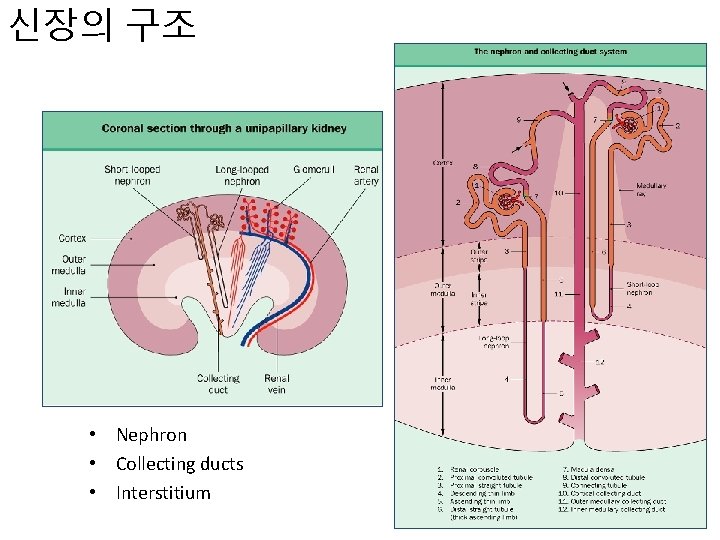
신장의 구조 • Nephron • Collecting ducts • Interstitium

Cortical and juxtamedullary nephron Cortical nephron – glomeruli in outer cortex & short loops of Henle that extend only short distance into medulla-blood flow through cortex is rapid – majority of nephrons are cortical – cortical interstitial fluid 300 m. Osmolar Juxtamedullary nephron – glomeruli in inner part of cortex & long loops of Henle which extend deeply into medulla. – blood flow through vasa recta in medulla is slow – medullary interstitial fluid is hyperosmotic – this nephron maintains osmolality in addition to filtering blood and maintaining acid-base balance
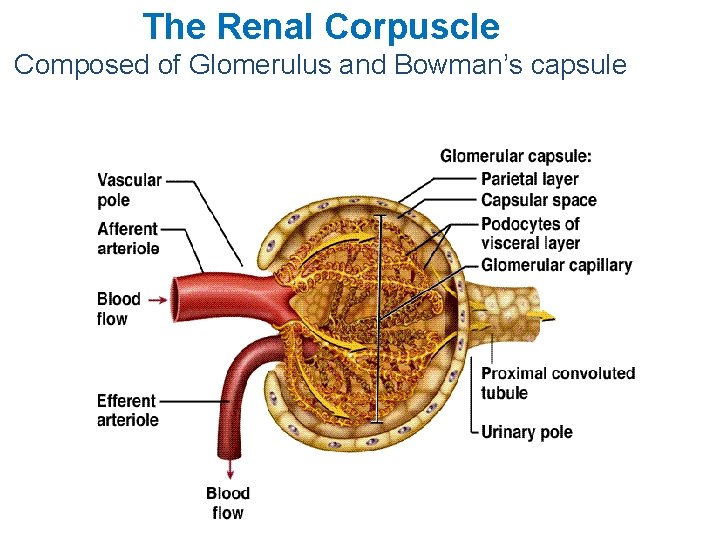
The Renal Corpuscle Composed of Glomerulus and Bowman’s capsule

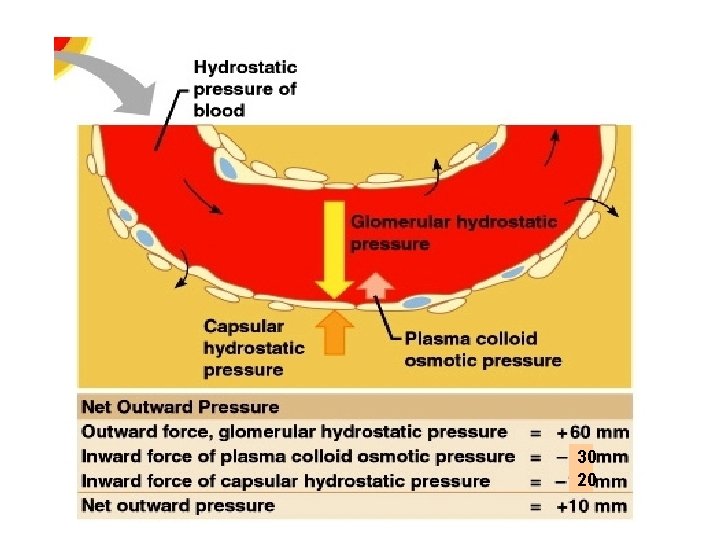
Glomerular filatration rate (GFR) • 신장에서 단위시간당 여과되는 여과액의 양 – The normal glomerular filtration = 125 ml/min in normal adults = 180 L/day • GFR = Kf x (PGC-PBS- GC) • Ultrafiltration coefficient (Kf) • Intraglomerular hydrostatic pressure (PGC) • Oncotic pressure ( GC) • Bowmann space pressure (PBS) • PGC-PBS = effective hydrostatic pressure gradient
30 20

Filtration fraction (FF) important expression of the extent of glomerular filtration Renal blood flow 1250 ml/min glomerulus FF = Glomerular filtration rate Renal plasma flow RPF 750 ml/min Efferent Arteriole 625 ml/min GFR 125 ml/min tubule = renal vein 124 ml/min Urine 1 ml/min 125 ≈ 0. 17 750 It is the fraction of renal plasma flow that is filtered at the glomerulus
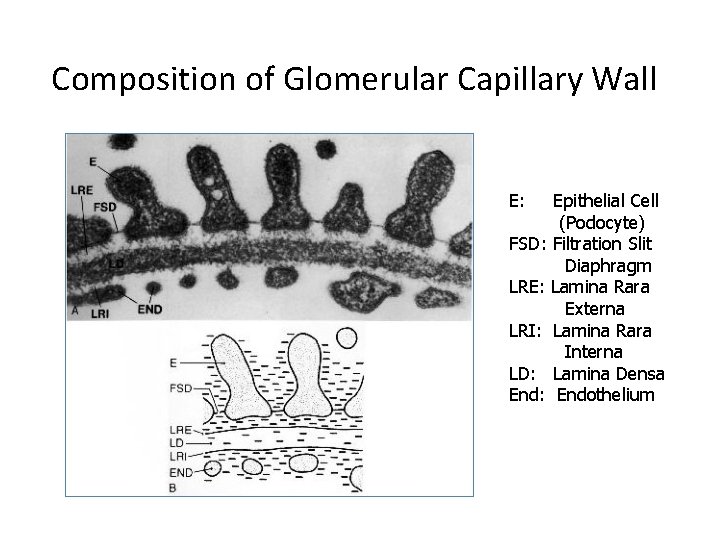
Composition of Glomerular Capillary Wall E: Epithelial Cell (Podocyte) FSD: Filtration Slit Diaphragm LRE: Lamina Rara Externa LRI: Lamina Rara Interna LD: Lamina Densa End: Endothelium
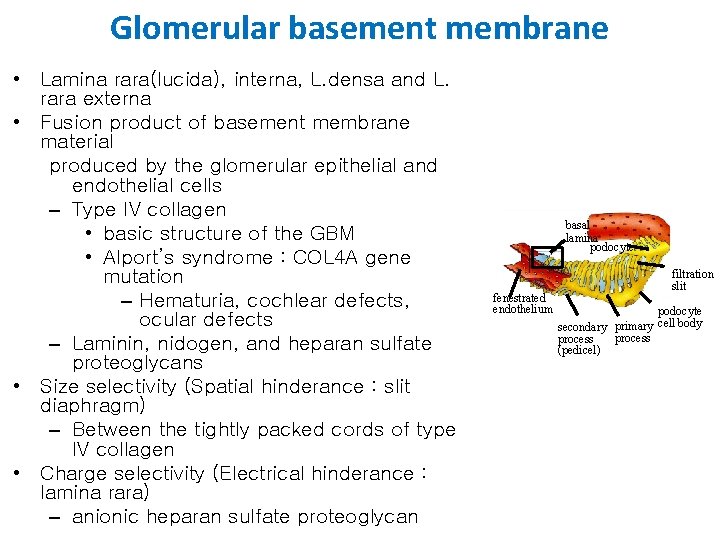
Glomerular basement membrane • Lamina rara(lucida), interna, L. densa and L. rara externa • Fusion product of basement membrane material produced by the glomerular epithelial and endothelial cells – Type IV collagen • basic structure of the GBM • Alport’s syndrome : COL 4 A gene mutation – Hematuria, cochlear defects, ocular defects – Laminin, nidogen, and heparan sulfate proteoglycans • Size selectivity (Spatial hinderance : slit diaphragm) – Between the tightly packed cords of type IV collagen • Charge selectivity (Electrical hinderance : lamina rara) – anionic heparan sulfate proteoglycan basal lamina podocyte fenestrated endothelium filtration slit podocyte secondary primary cell body process (pedicel)
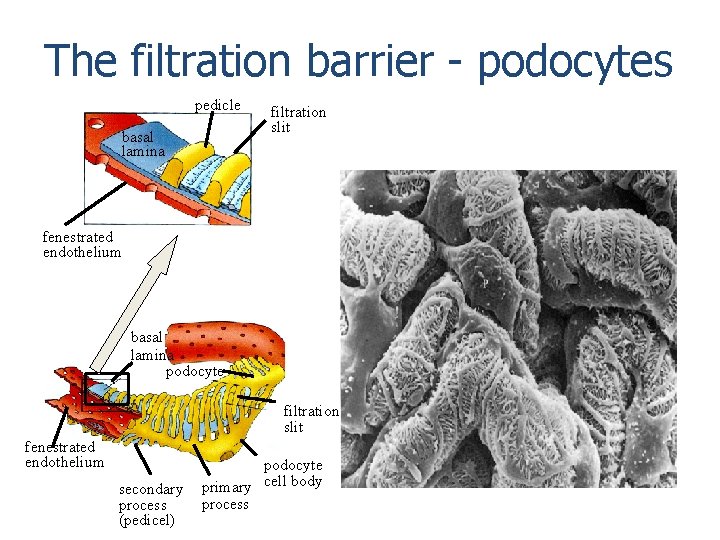
The filtration barrier - podocytes pedicle basal lamina filtration slit fenestrated endothelium basal lamina podocyte filtration slit fenestrated endothelium secondary process (pedicel) podocyte primary cell body process
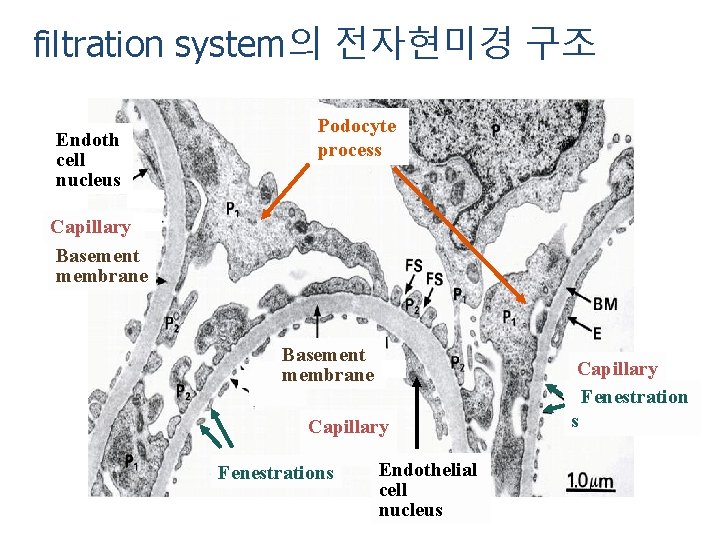
filtration system의 전자현미경 구조 Endoth cell nucleus Podocyte process Capillary Basement membrane Capillary Fenestrations Endothelial cell nucleus Capillary Fenestration s
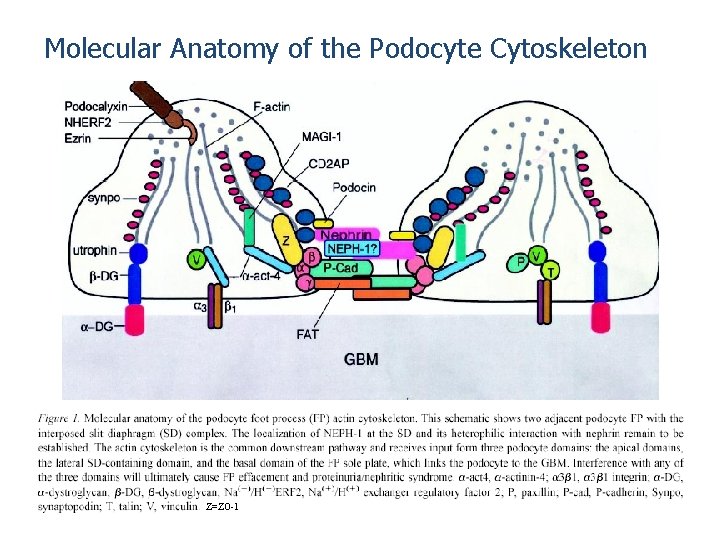
Molecular Anatomy of the Podocyte Cytoskeleton Z=ZO-1
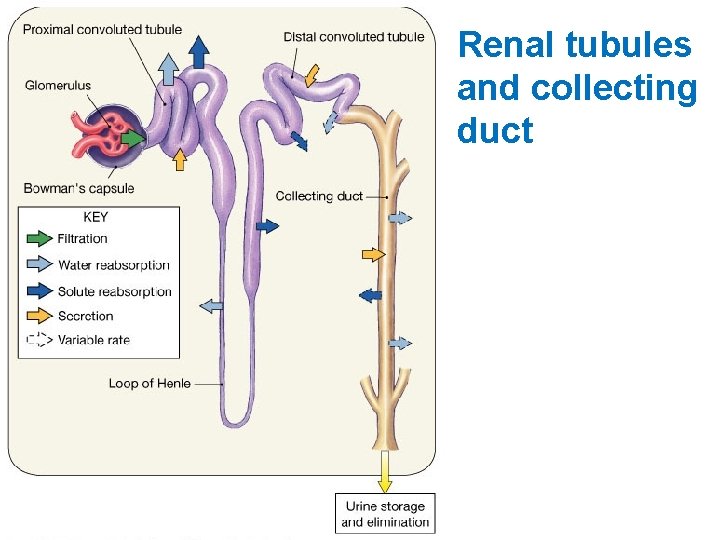
Renal tubules and collecting duct
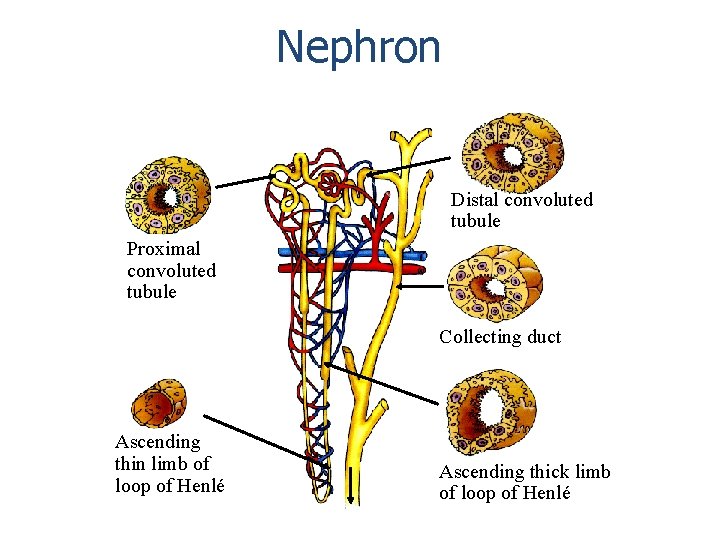
Nephron Distal convoluted tubule Proximal convoluted tubule Collecting duct Ascending thin limb of loop of Henlé Ascending thick limb of loop of Henlé
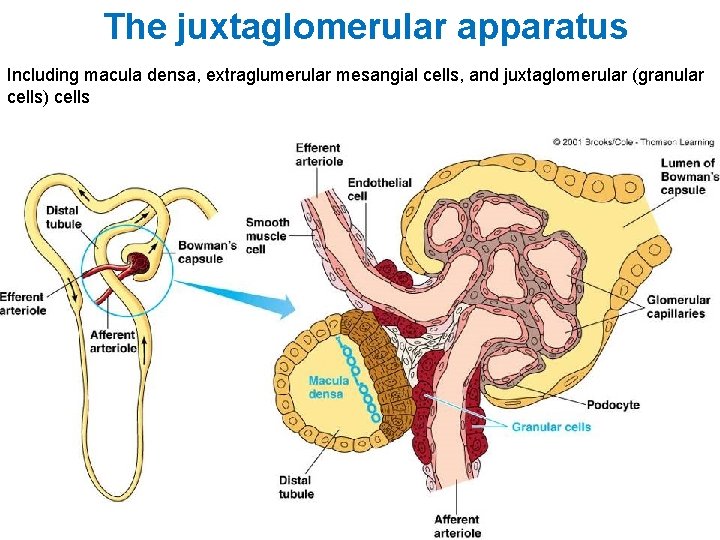
The juxtaglomerular apparatus Including macula densa, extraglumerular mesangial cells, and juxtaglomerular (granular cells) cells

GFR regulation : Adjusting blood flow • GFR is regulated using three mechanisms 1. Renal Autoregulation 2. Neural regulation 3. Hormonal regulation All three mechanism adjust renal blood pressure and resulting blood flow

1. Renal autoregulation RBF determines GFR Renal blood flow is autoregulated between 90 and 180 mm. Hg by varying renal vascular resistance (RVR) i. e. the resistances of the interlobular artery, afferent arteriole and efferent arteriole Autoregulation effectively uncouples renal function from arterial blood pressure and ensures that fluid and solute excretion is constant. Two hypotheses have been proposed to explain autoregulation. Flow, l/min 1. 5 Renal blood flow 1. 0 0. 5 0 GFR 0 100 200 Arterial blood pressure, mm Hg

Mechanism?
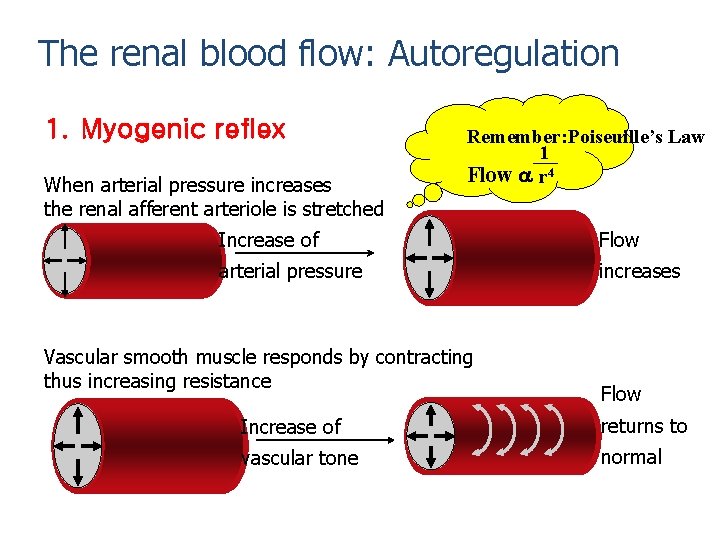
The renal blood flow: Autoregulation 1. Myogenic reflex When arterial pressure increases the renal afferent arteriole is stretched Remember: Poiseuille’s Law 1 Flow a r 4 Increase of Flow arterial pressure increases Vascular smooth muscle responds by contracting thus increasing resistance Flow Increase of returns to vascular tone normal
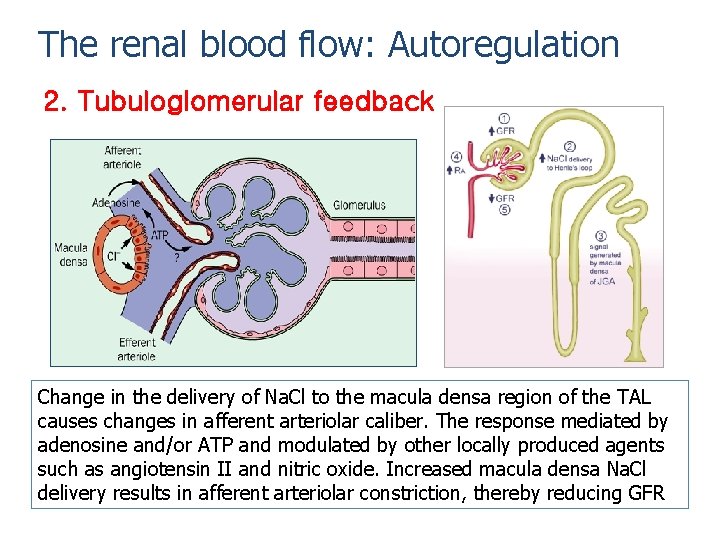
The renal blood flow: Autoregulation 2. Tubuloglomerular feedback Change in the delivery of Na. Cl to the macula densa region of the TAL causes changes in afferent arteriolar caliber. The response mediated by adenosine and/or ATP and modulated by other locally produced agents such as angiotensin II and nitric oxide. Increased macula densa Na. Cl delivery results in afferent arteriolar constriction, thereby reducing GFR

2. Neural regulation of GFR • Sympathetic nerve fibers innervate afferent and efferent arteriole • Normally sympathetic stimulation is low but can increase during hemorrhage and exercise • Vasoconstriction occurs as a result which conserves blood volume(hemorrhage)and permits greater blood flow to other body parts(exercise)

3. Hormonal regulation of GFR • Several hormones contribute to GFR regulation • Angiotensin II. Produced by Renin, released by JGA cells is a potent vasoconstrictor. Reduces GFR • ANP(released by atria when stretched) increases GFR by increasing capillary surface area available for filtration • NO • Endothelin • Prostaglandin E 2
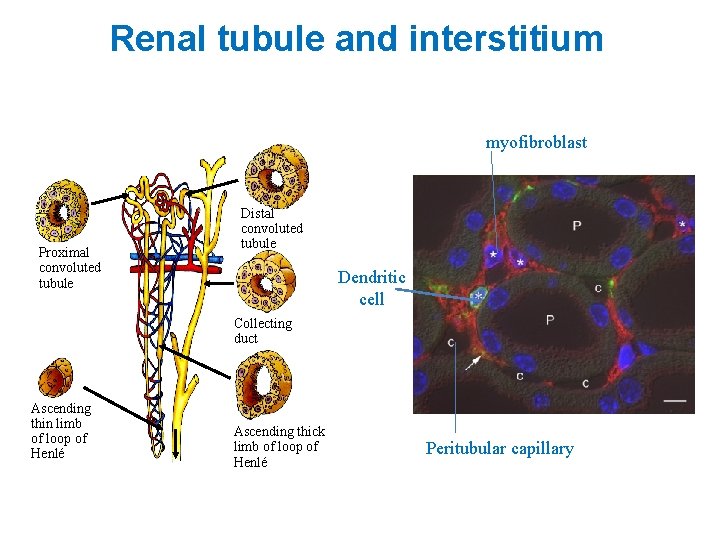
Renal tubule and interstitium myofibroblast Proximal convoluted tubule Distal convoluted tubule Dendritic cell Collecting duct Ascending thin limb of loop of Henlé Ascending thick limb of loop of Henlé Peritubular capillary
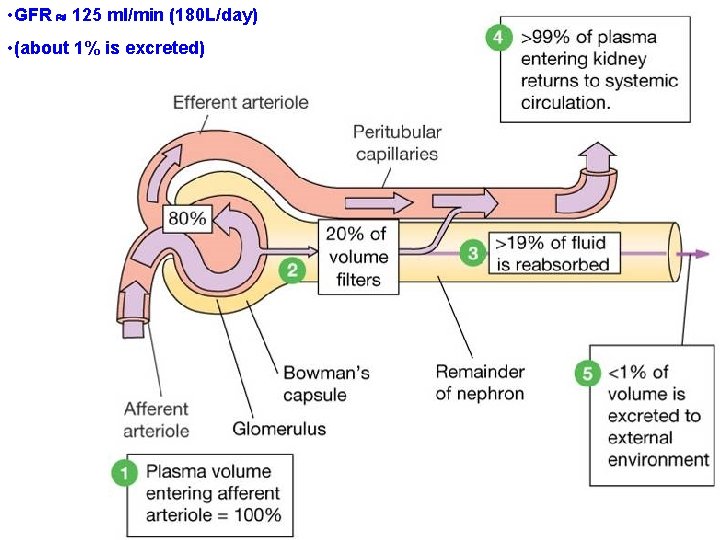
• GFR 125 ml/min (180 L/day) • (about 1% is excreted)
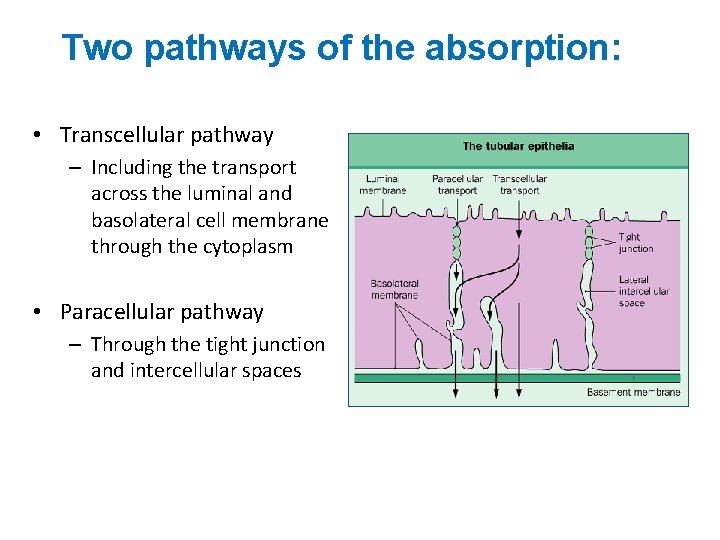
Two pathways of the absorption: • Transcellular pathway – Including the transport across the luminal and basolateral cell membrane through the cytoplasm • Paracellular pathway – Through the tight junction and intercellular spaces
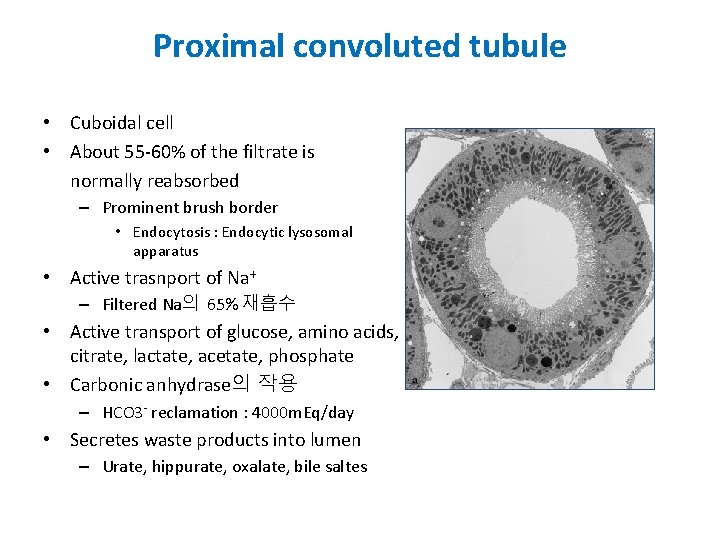
Proximal convoluted tubule • Cuboidal cell • About 55 -60% of the filtrate is normally reabsorbed – Prominent brush border • Endocytosis : Endocytic lysosomal apparatus • Active trasnport of Na+ – Filtered Na의 65% 재흡수 • Active transport of glucose, amino acids, citrate, lactate, acetate, phosphate • Carbonic anhydrase의 작용 – HCO 3 - reclamation : 4000 m. Eq/day • Secretes waste products into lumen – Urate, hippurate, oxalate, bile saltes

The loop of Henle • Hair pin shape – Squamous cell – Superficial cortical nephron에서 시작된 Henle’s loop의 tip은 outer zone과 inner zone of medulla 의 경계에서 u-turn 하고 juxta medullary nephron의 tip은 papilla 에서 u-turn – Superficial cortical nephron : Na+ looser – Juxtamedullary nephron : Na+ reservoir • Segment – Thin descending limb • Highly permeable to water • Important role for urinary concentration and dilution • Maximum tonicity of fluid in lumen : 1200 m. Osm/kg of H 2 O – Thin ascending limb • Impermeable to water • Permeable to salt which enters interstitium

The thick ascending limb of Henle’s loop • Thick ascending limb (TAL) – Active co-transporter : Na+-K+ -2 Cl- cotransporter by Na+-K+ -ATPase pumps • Reabsorb 25 -30% of the filtered Na. Cl (TAL) • Target diuretics : furosemide – Often called diluting segment • Water impermeable, but reabsorbs considerable amount of salt – Calcium, Magnesium의 재흡수 – Tamm-Horsefall protein 생성 – JG apparatus 존재 • Distal convoluted tubule – Active transporter : Na+-Cl- pumps by Na+-K+ -ATPase pumps – Na+-H- , Na+-K+ exchange • Target diuretics : Thiazide
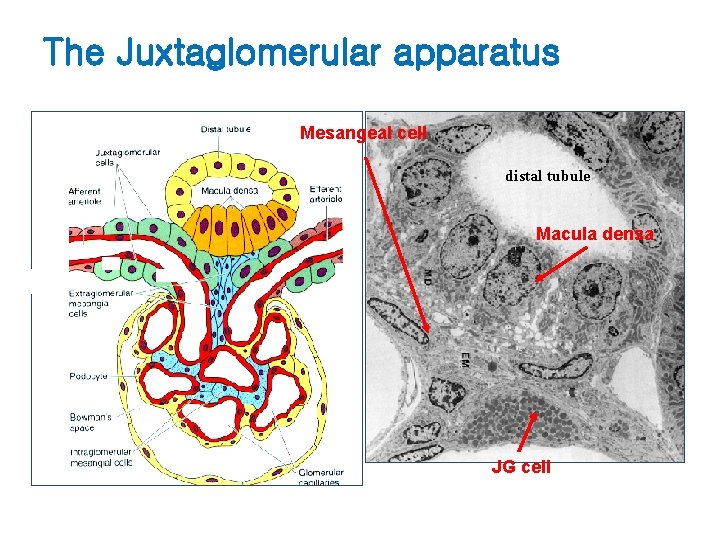
The Juxtaglomerular apparatus Mesangeal cell distal tubule Macula densa JG cell

The Juxtaglomerular apparatus • Macula densa – Plaque of specialized cells in the wall of the TAL – Distal urine is sensed by the macula densa, and this information is used to adjust the tone of the glomerular arteriole • Tubuloglmerular feedback • JG cells – – Primarly in afferent arteriole, specialized smooth cell No, basal lamina between JG cells & macular densa Secrete renin into circulation Contain ACE, Angiotensin I, II • Extramedullary mesangial cell (Lacis cell) – Interconnects all structures of the glomerular enterance
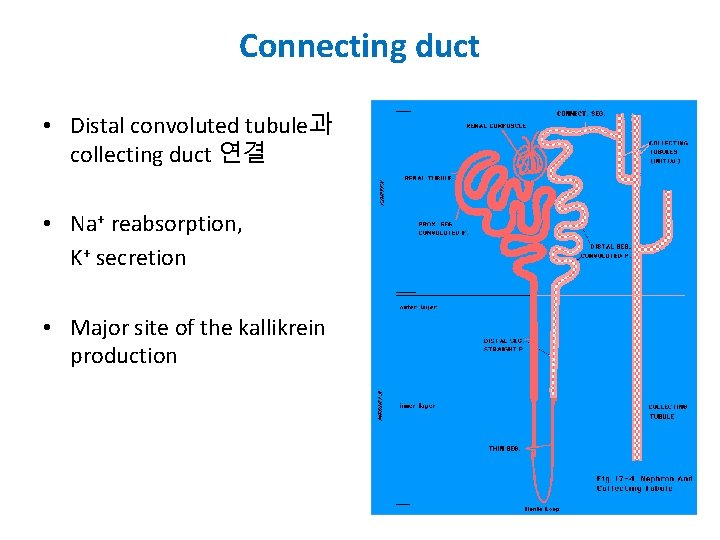
Connecting duct • Distal convoluted tubule과 collecting duct 연결 • Na+ reabsorption, K+ secretion • Major site of the kallikrein production

The Collecting duct • Part – Cortical collecting duct (CCD) – Outer-medullary CD (OMCD) – Inner medullary CD (IMCD) • Urea transport under the ADH • Function – Functioning site of ADH • Water reabsorption with aquaporin 2 -5 – Acid secretion – Sodium and K transport • Aldosterone • Epithelial Na channel (ENa. C) – Overexpression : Liddle’s disease
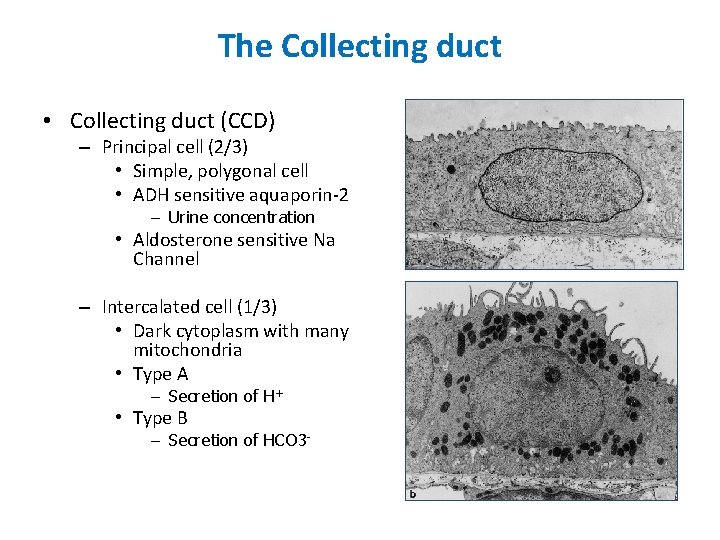
The Collecting duct • Collecting duct (CCD) – Principal cell (2/3) • Simple, polygonal cell • ADH sensitive aquaporin-2 – Urine concentration • Aldosterone sensitive Na Channel – Intercalated cell (1/3) • Dark cytoplasm with many mitochondria • Type A – Secretion of H+ • Type B – Secretion of HCO 3 - Figure 1 -11, 1 -12

Kidney development and renal progenitor cells
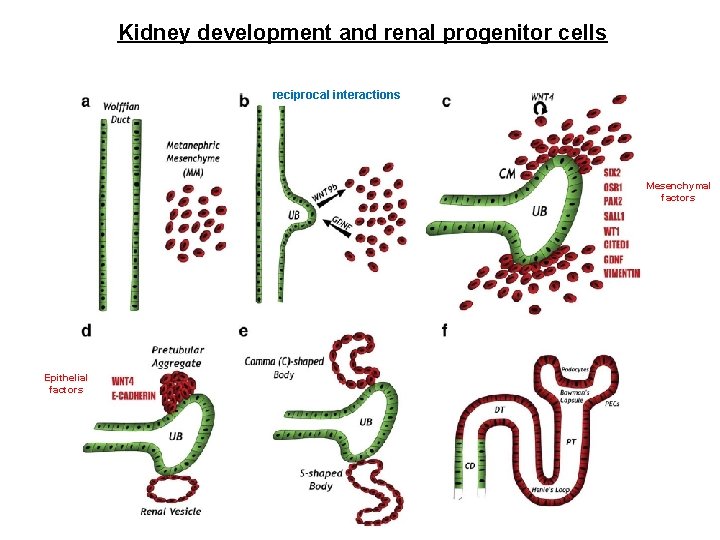
Kidney development and renal progenitor cells reciprocal interactions Mesenchymal factors Epithelial factors
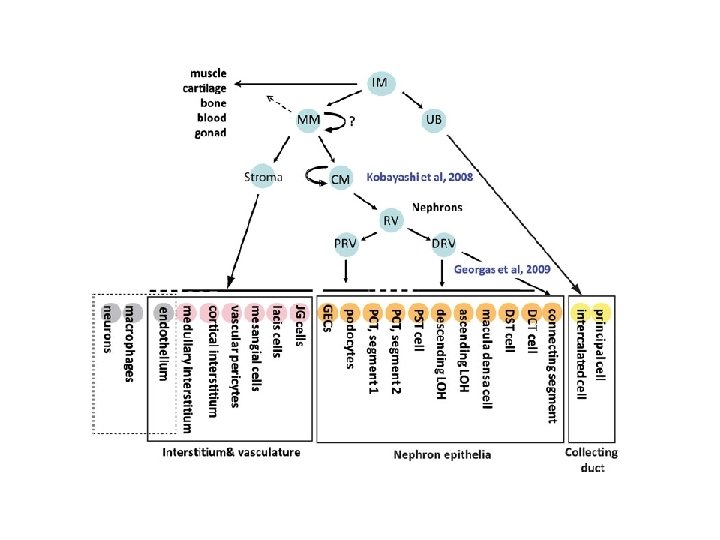

CM renal progenitor cells • Hox 11 paralogs, Osr 1, Pax 2, Eya 1, Wt 1, Six 2, Sall 1, and Cited 1 slicencing • Termination of nephrogenesis • human, 34 th gestational week • mice, 2 weeks postnatal

Nervous Regulation of Kidney

INNERVATION OF THE KIDNEY Nerves from the renal plexus (sympathetic nerve) of the autonomic nervous system enter kidney at the hilus innervate smooth muscle of afferent & efferent arterioles regulates blood pressure & distribution throughout kidney Effect (1) Reduce the GPF and GFR and through contracting the afferent and efferent artery (α receptor) (2) Increase the Na+ reabsorption in the proximal tubules (β receptor) (3) Increase the release of renin (β receptor)

Nerve reflex: 1. Cardiopulmonary reflex and Baroreceptor Reflex 2. Renorenal reflex Sensory nerves located in the renal pelvic wall are activated by stretch of the renal pelvic wall, which may occur during diuresis or ureteral spasm/occlusion. Activation of these nerves leads to an increase in afferent renal nerve activity, which causes a decrease in efferent renal nerve activity and an increase in urine flow rate and urinary sodium excretion.

Hormonal Regulation of Kidney
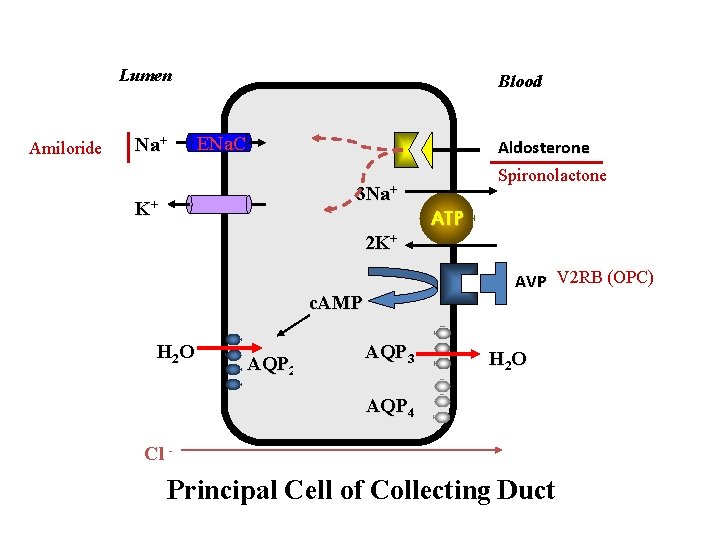
Lumen Amiloride Na+ Blood ENa. C Aldosterone Spironolactone 3 Na+ K+ 2 K+ AVP V 2 RB (OPC) c. AMP H 2 O AQP 2 ATP AQP 3 H 2 O AQP 4 Cl - Principal Cell of Collecting Duct

Antidiuretic Hormone (ADH) • Retention of Water is controlled by ADH: – Anti Diuretic Hormone – ADH Release Is Controlled By: • Decrease in Blood Volume • Decrease in Blood Pressure • Increase in extracellular fluid (ECF) Osmolarity
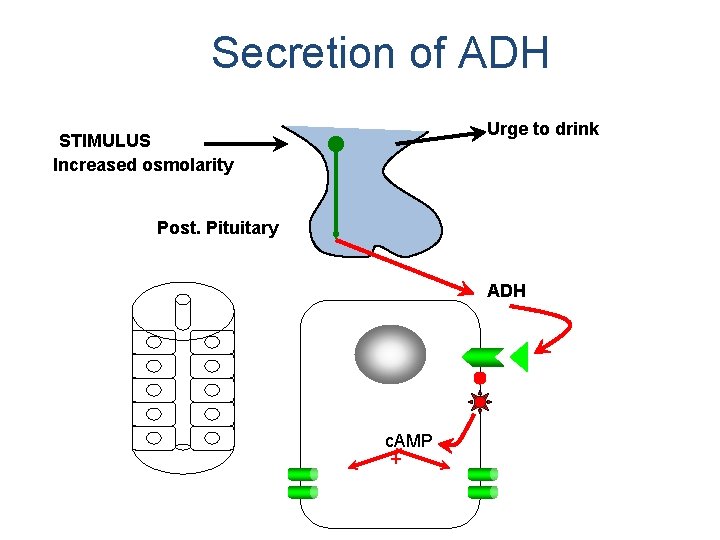
Secretion of ADH Urge to drink STIMULUS Increased osmolarity Post. Pituitary ADH c. AMP +
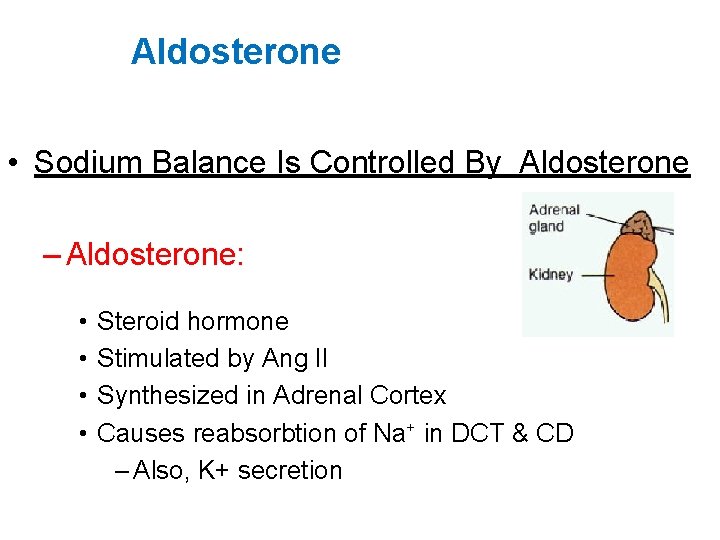
Aldosterone • Sodium Balance Is Controlled By Aldosterone – Aldosterone: • • Steroid hormone Stimulated by Ang II Synthesized in Adrenal Cortex Causes reabsorbtion of Na+ in DCT & CD – Also, K+ secretion

Atrial natriuretic peptide(ANP) • ANP is released by atrium in response to atrial stretching due to increased blood volume • ANP inhibits Na+ and water reabsorption, also inhibits ADH secretion • Thus promotes increased sodium excretion (natriuresis) and water excretion (diuresis) in urine

Endocrine & metabolic functions of Kidney

Endocrine functions of Kidney 1) Endocrine regulation of extracellular fluid volume and blood pressure – Renin - angiotensin system – Renal kallikrein - kinin - prostaglandins axis • kallikrein secretion by the cells in the distal tubule and connecting segment 2) Control of red cell mass – Erythropoietin are secreted by the stimulation of peripheral hypoxia (feedback mechanism) 3) Endocrine control of mineral metabolism: formation of 1, 25 dihydroxycholecalciferol or 24, 25 -dihydroxycholecalciferol
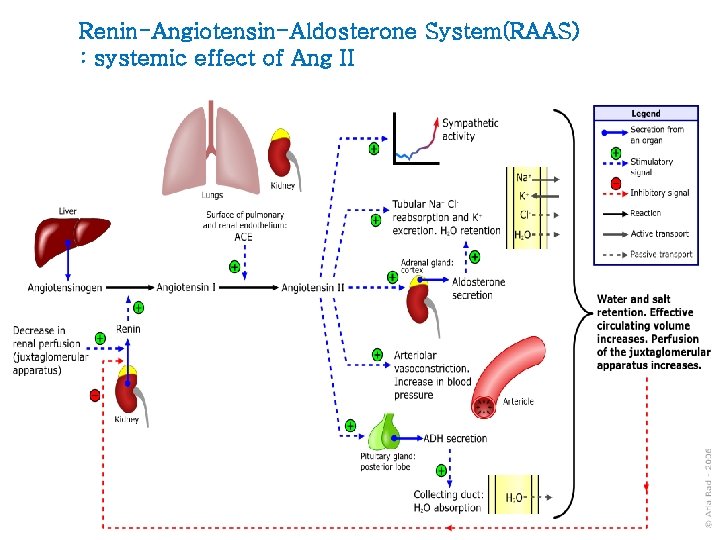
Renin-Angiotensin-Aldosterone System(RAAS) : systemic effect of Ang II
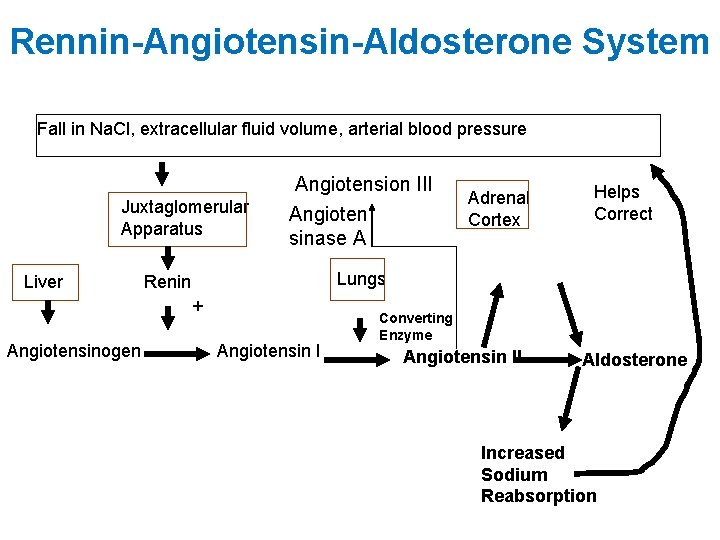
Rennin-Angiotensin-Aldosterone System Fall in Na. Cl, extracellular fluid volume, arterial blood pressure Juxtaglomerular Apparatus Liver Angiotension III Angioten sinase A Helps Correct Lungs Renin + Angiotensinogen Adrenal Cortex Angiotensin I Converting Enzyme Angiotensin II Aldosterone Increased Sodium Reabsorption

Regulation of the Renin Secretion: Renal Mechanism: • Tension of the afferent artery (stretch receptor) • Macula densa (content of the Na+ ion in the distal convoluted tubuyle) Nervous Mechanism: • Sympathetic nerve Humoral Mechanism: • E, NE, PGE 2, PGI 2

Renin-Angiotensin system Angiotensinogen Renin Angiotensin I Angiotensin converting enzyme Angiotensin II Function 1) Vasoconstriction 2) Stimulation of adrenal mineralocorticoid 3) Simulation of thirst center 4) Behave as like as a cytokine
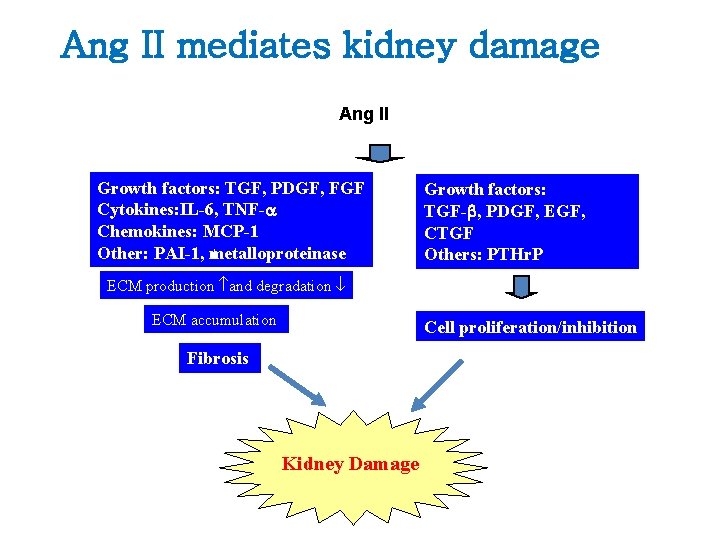
Ang II mediates kidney damage Ang II Growth factors: TGF, PDGF, FGF Cytokines: IL-6, TNF-a Chemokines: MCP-1 Other: PAI-1, metalloproteinase Growth factors: TGF-b, PDGF, EGF, CTGF Others: PTHr. P ECM production and degradation ECM accumulation Cell proliferation/inhibition Fibrosis Kidney Damage
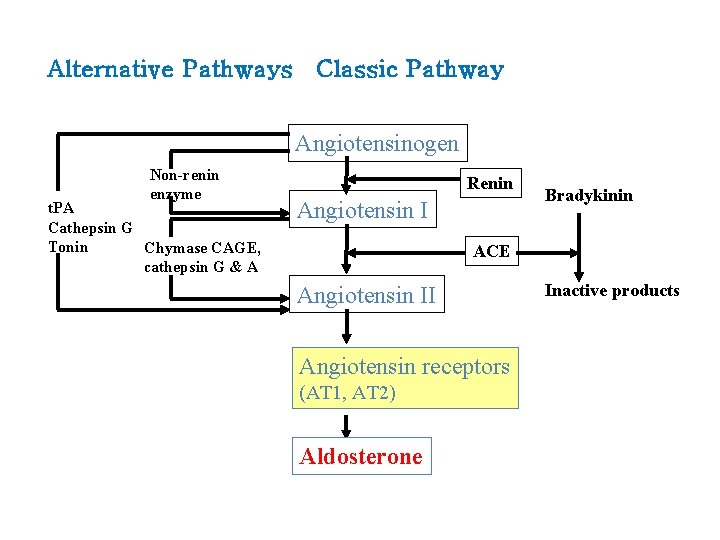
Alternative Pathways Classic Pathway Angiotensinogen Non-renin enzyme t. PA Cathepsin G Tonin Chymase CAGE, cathepsin G & A Renin Angiotensin I Bradykinin ACE Angiotensin II Angiotensin receptors (AT 1, AT 2) Aldosterone Inactive products
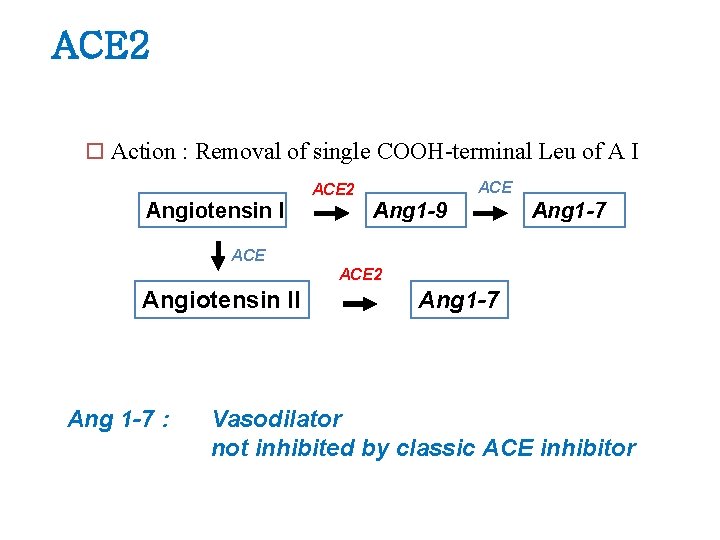
ACE 2 ¨ Action : Removal of single COOH-terminal Leu of A I ACE 2 Angiotensin I Ang 1 -9 Ang 1 -7 ACE 2 Angiotensin II Ang 1 -7 : Ang 1 -7 Vasodilator not inhibited by classic ACE inhibitor
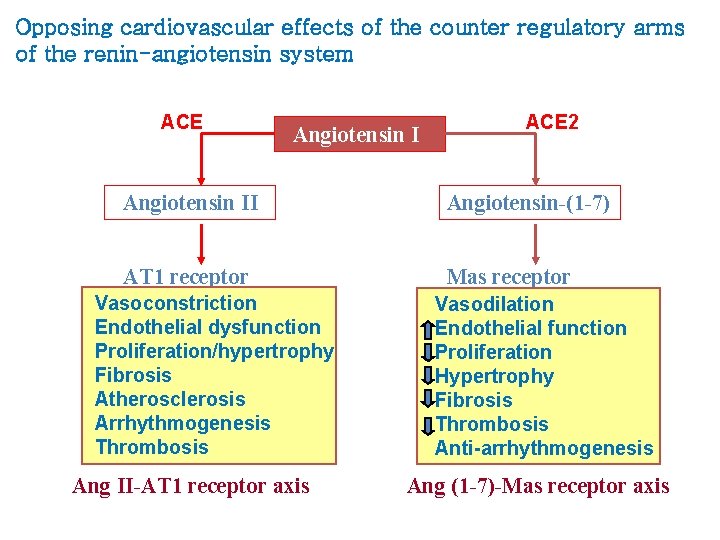
Opposing cardiovascular effects of the counter regulatory arms of the renin-angiotensin system ACE Angiotensin I ACE 2 Angiotensin II Angiotensin-(1 -7) AT 1 receptor Mas receptor Vasoconstriction Endothelial dysfunction Proliferation/hypertrophy Fibrosis Atherosclerosis Arrhythmogenesis Thrombosis Ang II-AT 1 receptor axis Vasodilation Endothelial function Proliferation Hypertrophy Fibrosis Thrombosis Anti-arrhythmogenesis Ang (1 -7)-Mas receptor axis
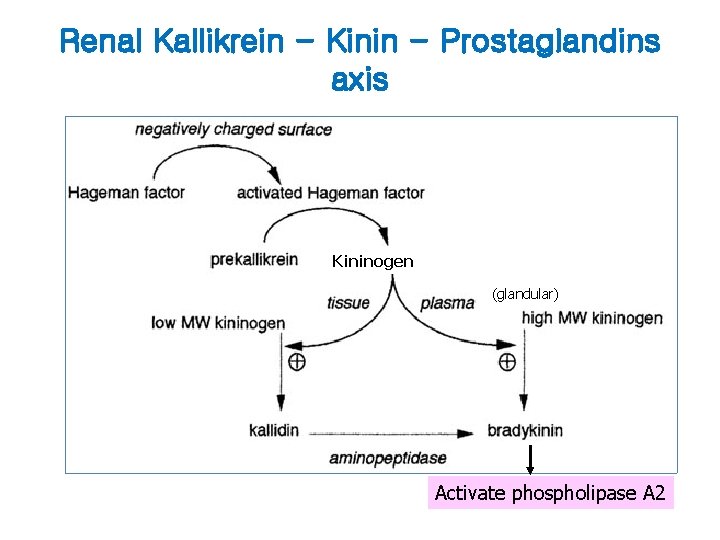
Renal Kallikrein - Kinin - Prostaglandins axis Kininogen (glandular) Activate phospholipase A 2
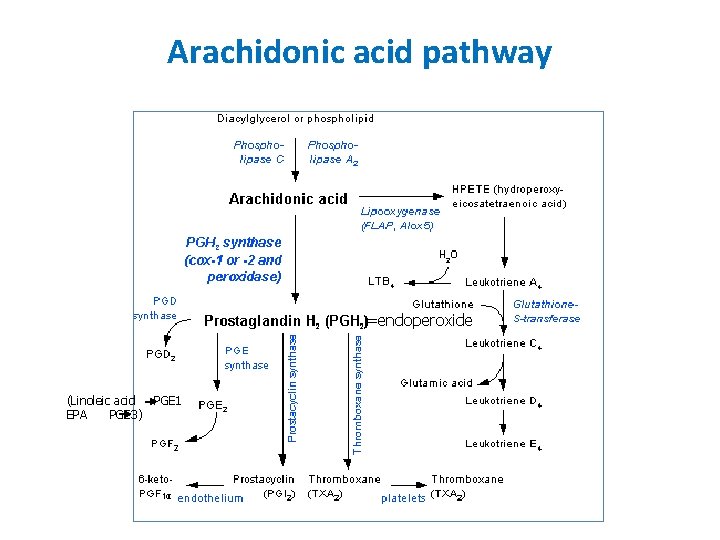
Arachidonic acid pathway =endoperoxide (Linoleic acid PGE 1 EPA PGE 3)
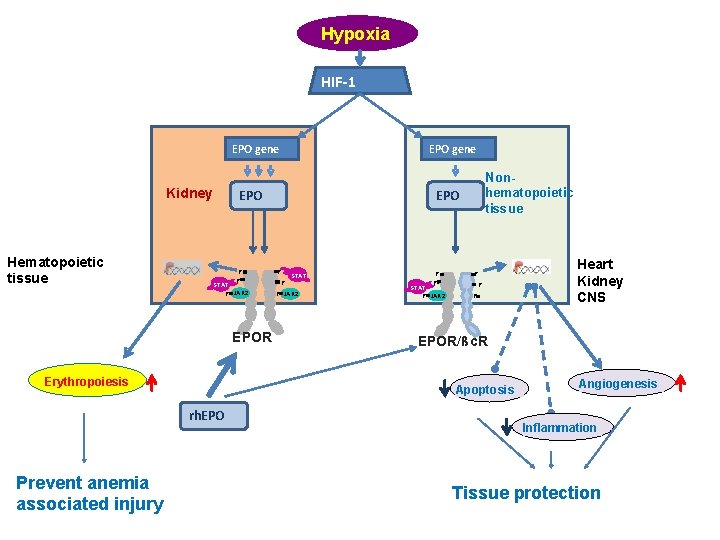
Hypoxia HIF-1 EPO gene Kidney Hematopoietic tissue EPO P P STAT P JAK 2 EPOR Erythropoiesis Nonhematopoietic tissue EPO P STAT P P JAK 2 P P STAT P JAK 2 P P P Heart Kidney CNS EPOR/ßc. R Apoptosis rh. EPO Prevent anemia associated injury EPO gene Angiogenesis Inflammation Tissue protection
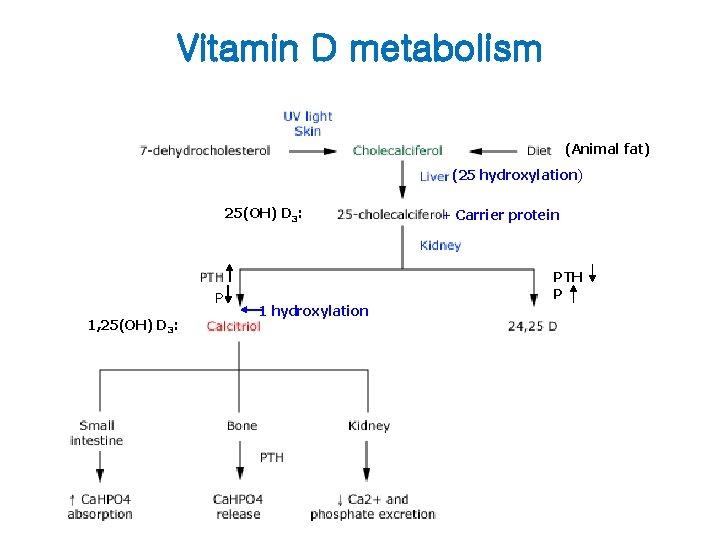
Vitamin D metabolism (Animal fat) (25 hydroxylation) 25(OH) D 3: P 1, 25(OH) D 3: + Carrier protein PTH P 1 hydroxylation

Actions of PTH • 1 -hydroxylation of Vit D 3 – Ca absorption in gut (Calcium binding protein, Ca. BP) • • Renal tubule Ca absorption Renal tubule P excretion Osteoclast activation Peripheral blood의 RBC로 부터 O 2 dissociation – Masking of peripheral hypoxia • Anemia 유발
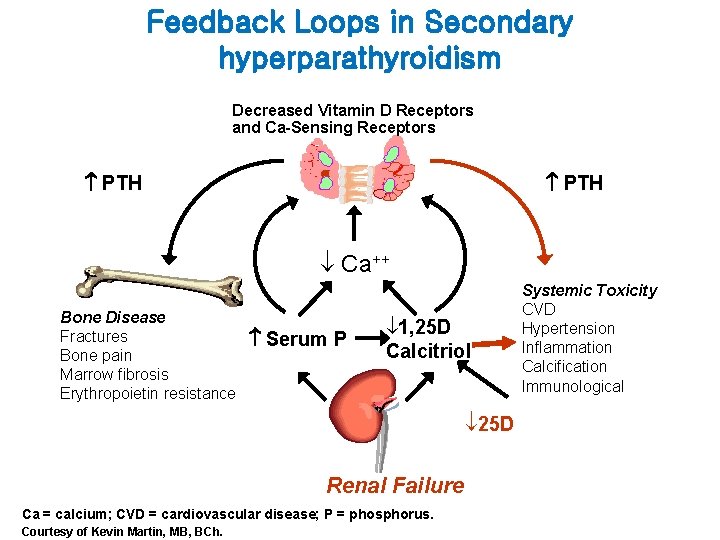
Feedback Loops in Secondary hyperparathyroidism Decreased Vitamin D Receptors and Ca-Sensing Receptors PTH Ca++ Bone Disease Fractures Bone pain Marrow fibrosis Erythropoietin resistance Serum P 1, 25 D Calcitriol Systemic Toxicity CVD Hypertension Inflammation Calcification Immunological 25 D Renal Failure Ca = calcium; CVD = cardiovascular disease; P = phosphorus. Courtesy of Kevin Martin, MB, BCh. © 2005 The Johns Hopkins University School of Medicine.

Metabolic function • Degradation and catabolism of peptide hormones – insulin, glucagon, parathyroid hormone • Catabolism of small molecular weight proteins – Endocytosis or pinocytosis of light chain or beta-2 microglobulin • Metabolic interconversions in gluconeogenesis – lipid metabolism
- Slides: 72