Major Clay Minerals Kaolinite Al 2 Si 2

![Phyllosilicates Si. O 4 tetrahedra polymerized into 2 -D sheets: [Si 2 O 5] Phyllosilicates Si. O 4 tetrahedra polymerized into 2 -D sheets: [Si 2 O 5]](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-2.jpg)


![Phyllosilicates Yellow = (OH) Kaolinite: Al 2 [Si 2 O 5] (OH)4 T-layers and Phyllosilicates Yellow = (OH) Kaolinite: Al 2 [Si 2 O 5] (OH)4 T-layers and](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-7.jpg)
![Phyllosilicates Yellow = (OH) Serpentine: Mg 3 [Si 2 O 5] (OH)4 T-layers and Phyllosilicates Yellow = (OH) Serpentine: Mg 3 [Si 2 O 5] (OH)4 T-layers and](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-8.jpg)


















- Slides: 37

Major Clay Minerals • Kaolinite – Al 2 Si 2 O 5(OH)4 • Illite – K 1 -1. 5 Al 4(Si, Al)8 O 20(OH)4 • Smectites: – Montmorillonite – (Ca, Na)0. 20. 4(Al, Mg, Fe)2(Si, Al)4 O 10(OH)2*n. H 2 O – Vermicullite - (Ca, Mg)0. 30. 4(Al, Mg, Fe)3(Si, Al)4 O 10(OH)2*n. H 2 O – Swelling clays – can take up extra water in their interlayers and are the major components of bentonite (NOT a mineral, but a mix of different clay minerals)
![Phyllosilicates Si O 4 tetrahedra polymerized into 2 D sheets Si 2 O 5 Phyllosilicates Si. O 4 tetrahedra polymerized into 2 -D sheets: [Si 2 O 5]](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-2.jpg)
Phyllosilicates Si. O 4 tetrahedra polymerized into 2 -D sheets: [Si 2 O 5] Apical O’s are unpolymerized and are bonded to other constituents

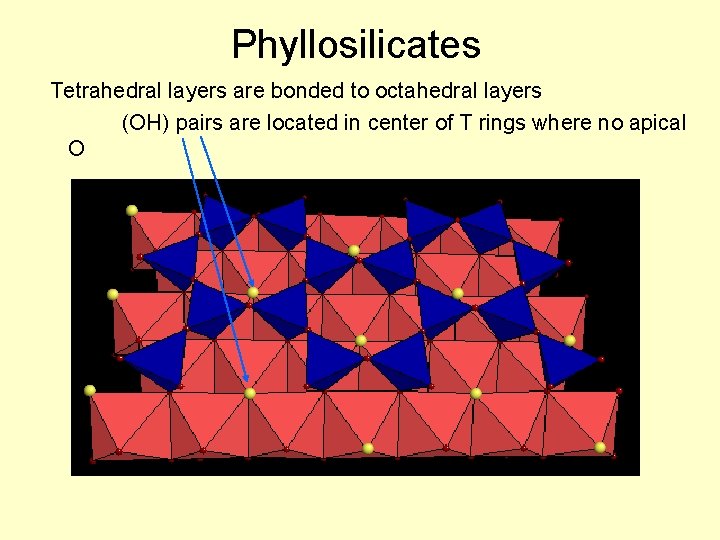
Phyllosilicates Tetrahedral layers are bonded to octahedral layers (OH) pairs are located in center of T rings where no apical O

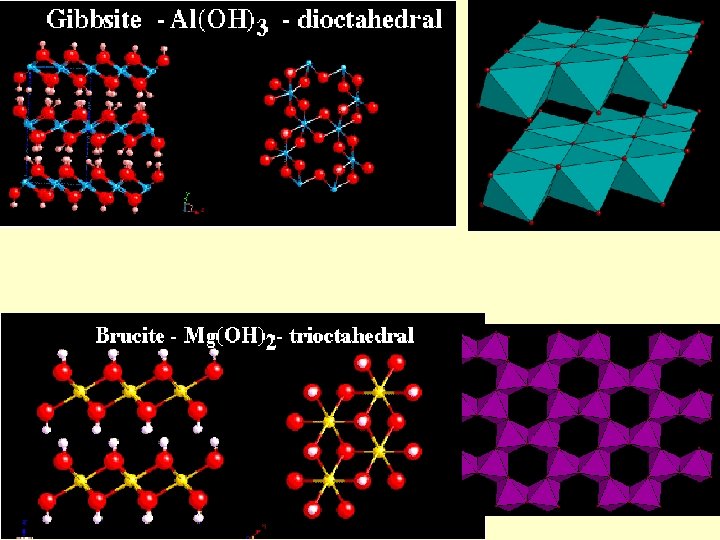
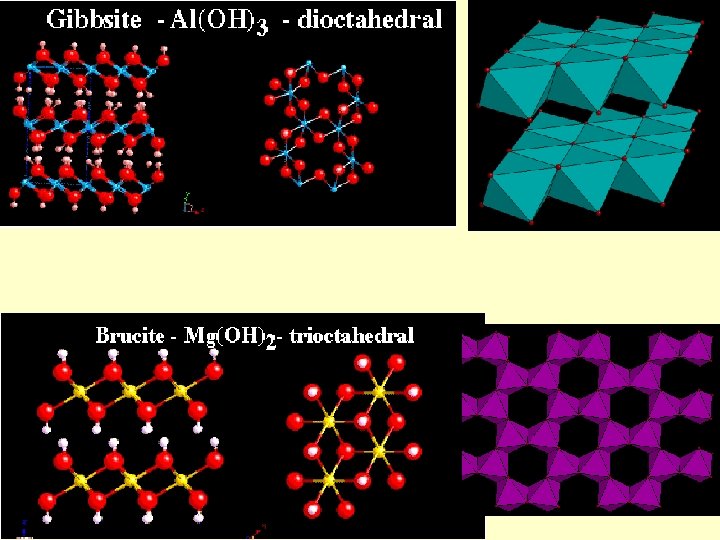
Phyllosilicates Octahedral layers can be understood by analogy with hydroxides Brucite: Mg(OH)2 c Layers of octahedral Mg in coordination with (OH) Large spacing along c due to weak van der waals bonds

Phyllosilicates a 2 a 1 Gibbsite: Al(OH)3 Layers of octahedral Al in coordination with (OH) Al 3+ means that only 2/3 of the VI sites may be occupied for charge-balance reasons Brucite-type layers may be called trioctahedral and gibbsite-type dioctahedral

![Phyllosilicates Yellow OH Kaolinite Al 2 Si 2 O 5 OH4 Tlayers and Phyllosilicates Yellow = (OH) Kaolinite: Al 2 [Si 2 O 5] (OH)4 T-layers and](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-7.jpg)
Phyllosilicates Yellow = (OH) Kaolinite: Al 2 [Si 2 O 5] (OH)4 T-layers and diocathedral (Al 3+) layers (OH) at center of T-rings and fill base of VI layer weak van der Waals bonds between T-O groups T O T O vdw
![Phyllosilicates Yellow OH Serpentine Mg 3 Si 2 O 5 OH4 Tlayers and Phyllosilicates Yellow = (OH) Serpentine: Mg 3 [Si 2 O 5] (OH)4 T-layers and](https://slidetodoc.com/presentation_image_h/dcf077cfcf40a5b763b69c954bdb473f/image-8.jpg)
Phyllosilicates Yellow = (OH) Serpentine: Mg 3 [Si 2 O 5] (OH)4 T-layers and triocathedral (Mg 2+) layers (OH) at center of T-rings and fill base of VI layer weak van der Waals bonds between T-O groups T O T O vdw

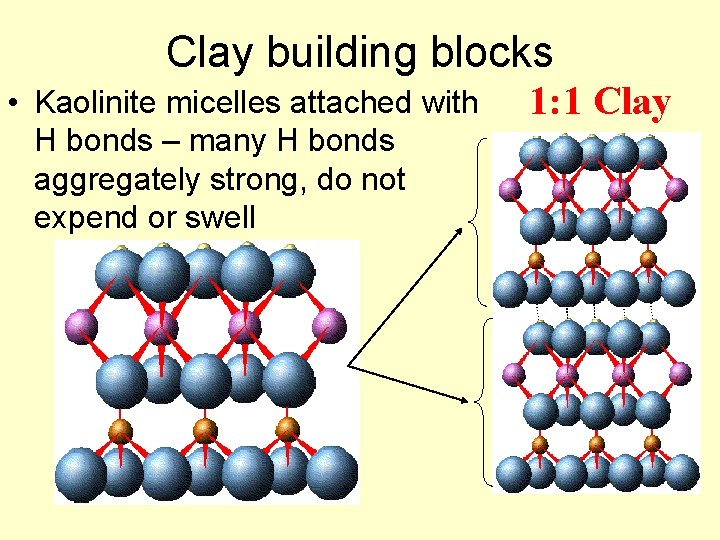
Clay building blocks • Kaolinite micelles attached with H bonds – many H bonds aggregately strong, do not expend or swell 1: 1 Clay

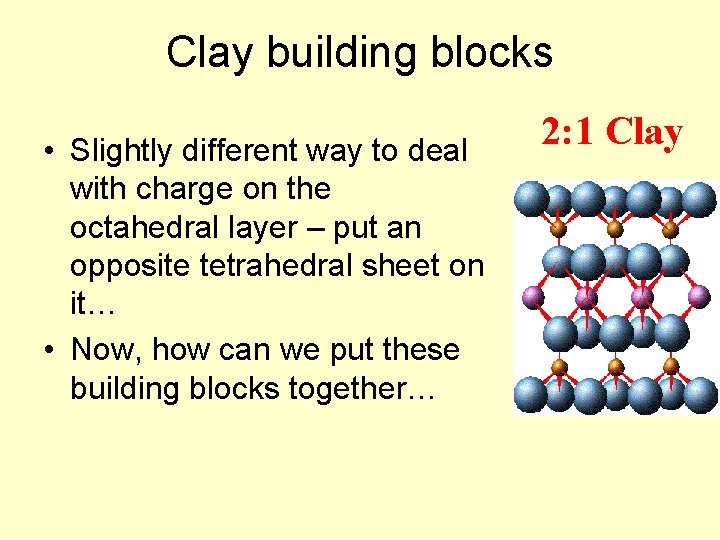
Clay building blocks • Slightly different way to deal with charge on the octahedral layer – put an opposite tetrahedral sheet on it… • Now, how can we put these building blocks together… 2: 1 Clay

Calcite vs. Dolomite • dolomite less reactive with HCl calcite has lower indices of refraction • calcite more commonly twinned • dolomite more commonly euhedral • calcite commonly colourless • dolomite may be cloudy or stained by iron oxide • Mg spectroscopic techniques! • Different symmetry cleavage same, but easily distinguished by XRD

Calcite Group • Variety of minerals varying by cation • Ca Calcite • Fe Siderite • Mn Rhodochrosite • Zn Smithsonite • Mg Magnesite

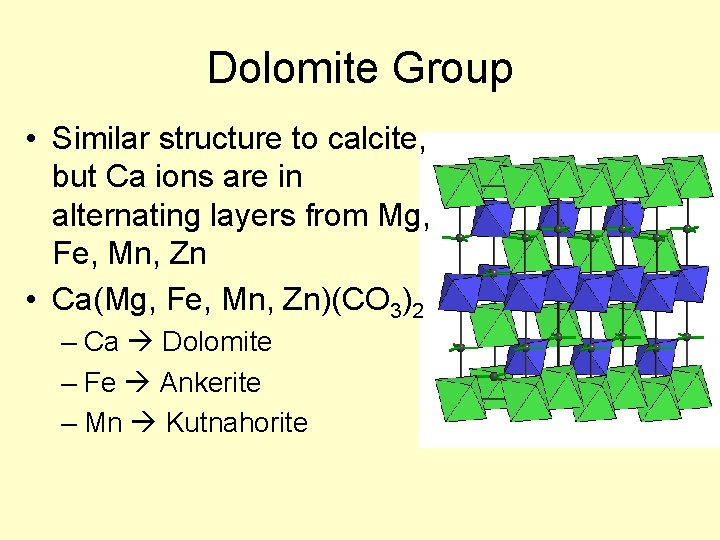
Dolomite Group • Similar structure to calcite, but Ca ions are in alternating layers from Mg, Fe, Mn, Zn • Ca(Mg, Fe, Mn, Zn)(CO 3)2 – Ca Dolomite – Fe Ankerite – Mn Kutnahorite

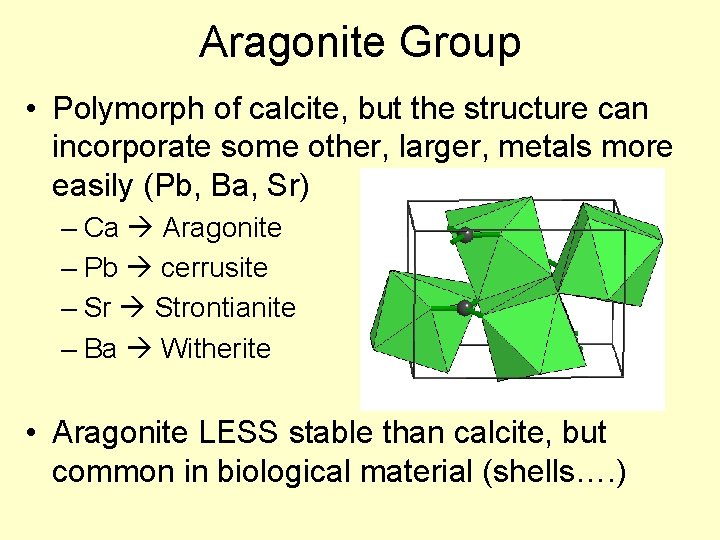
Aragonite Group • Polymorph of calcite, but the structure can incorporate some other, larger, metals more easily (Pb, Ba, Sr) – Ca Aragonite – Pb cerrusite – Sr Strontianite – Ba Witherite • Aragonite LESS stable than calcite, but common in biological material (shells…. )

Carbonate Minerals Calcite Group (hexagonal) Dolomite Group (hexagonal) Aragonite. Group (orthorhombic) mineral formula Calcite Ca. CO 3 Dolomite Ca. Mg(C O 3 )2 Aragonit e Ca. CO 3 Magnesi Mg. CO 3 te Ankerite Ca(Mg, F Witherite Ba. CO 3 e)(CO 3)2 Siderite, Fe. CO 3 Kutnoho Ca. Mn(C rite O 3 )2 Rhodoc hrosite Mn. CO 3 Strontia nite Sr. CO 3

Carbonate Minerals Ca Calcite, Ca. CO 3 Dolomite Ca. Mg(CO 3)2 Magnesite, Mg. CO 3 Mg Ankerite Ca. Fe(CO 3)2 Siderite, Fe. CO 3 Fe

Sulfate Minerals • More than 100 different minerals, separated into hydrous (with H 2 O) or anhydrous (without H 2 O) groups • Gypsum (Ca. SO 4*2 H 2 O) and anhydrite (Ca. SO 4) are the most common of the sulfate minerals • Gypsum typically forms in evaporitic basins – a polymorph of anhydrite (g-Ca. SO 4) forms when the gypsum is later dehydrated)

Gypsum

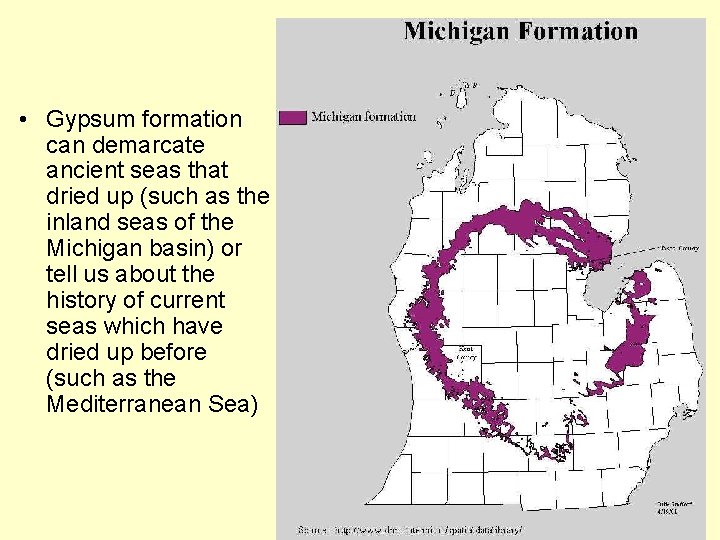
• Gypsum formation can demarcate ancient seas that dried up (such as the inland seas of the Michigan basin) or tell us about the history of current seas which have dried up before (such as the Mediterranean Sea)

Halide Minerals • Minerals contianing halogen elements as dominant anion (Cl- or F- typically) • Halite (Na. Cl) and Sylvite (KCl) form in VERY concentrated evaporitic waters – they are extremely soluble in water, indicate more complete evaporation than does gypsum • Fluorite (Ca. F 2) more typically occurs in veins associated with hydrothermal waters (F- in hydrothermal solutions is typically much higher – leached out of parent minerals such as biotites, pyroxenes, hornblendes or apatite)

Halite Structure • Na. Cl Na+ (gray) arranged in CCP with Cl- (red) at edges and center (in octahedral cavities)

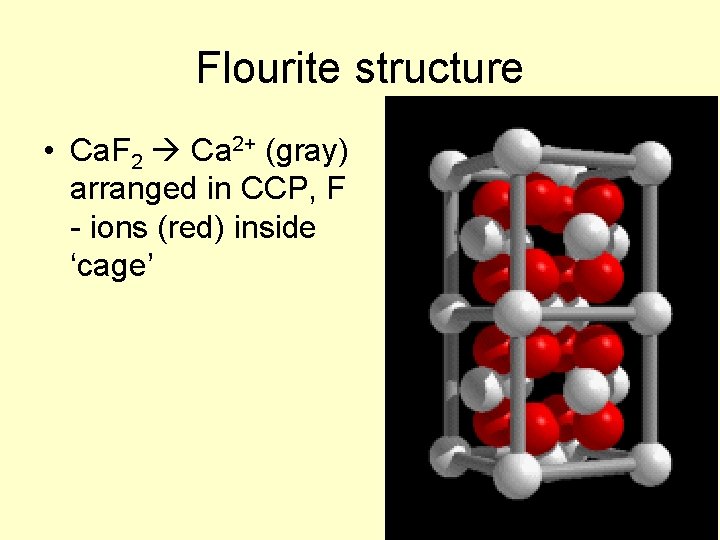
Flourite structure • Ca. F 2 Ca 2+ (gray) arranged in CCP, F - ions (red) inside ‘cage’

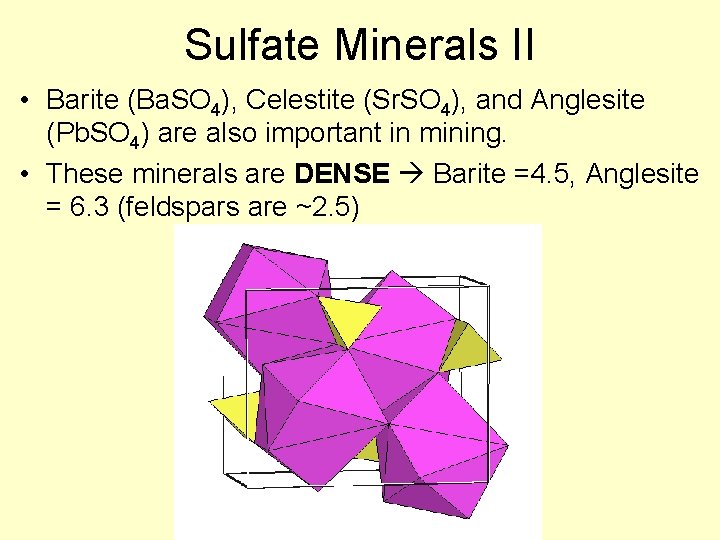
Sulfate Minerals II • Barite (Ba. SO 4), Celestite (Sr. SO 4), and Anglesite (Pb. SO 4) are also important in mining. • These minerals are DENSE Barite =4. 5, Anglesite = 6. 3 (feldspars are ~2. 5)

Barite, Celestite, Anglesite • Metals bond with sulfate much more easily, and thus are generally more insoluble – they do not require formation in evaporitic basins • What do they indicate then? Ba, Pb, Sr – very low SO 4 2 - Lots of SO 42 Not very much Ba, Sr, Pb

Just silica… • Chert – extremely fine grained quartz – Forms as nodules in limestone, recrystallization of siliceous fossils – Jasper – variety with hematite inclusions red – Flint – variety containing organic matter darker color • Chalcedony – microcrystaliine silica (very similar to low quartz, but different – it is yet uncertain how different…) typically shows banding, often colored to form an agate (rock formed of multiple bands of colored chalcedony) • Jasper – variety colored with inclusion of microcrystsalline oxides (often iron oxides = red) • Opal – a hydrogel (a solid solution of water in silica) – forms initially as water + silica colloids, then slowly the water diffuses into the silica making it amorphous (no XRD pattern!) – Some evidence opal slowly recrystallizes to chalcedony

Opal - Gemstone

Agates

Oxides - Oxyhydroxides • Fe. OOH minerals Goethite or Limonite (Fe. OOH) important alteration products of weathering Fe-bearing minerals • Hematite (Fe 2 O 3) primary iron oxide in Banded Iron Formations • Boehmite (Al. OOH) primary mineral in bauxite ores (principle Al ore) which forms in tropical soils • Mn oxides form Mn nodules in the oceans (estimated they cover 10 -30% of the deep Pacific floor) • Many other oxides important in metamorphic rocks…


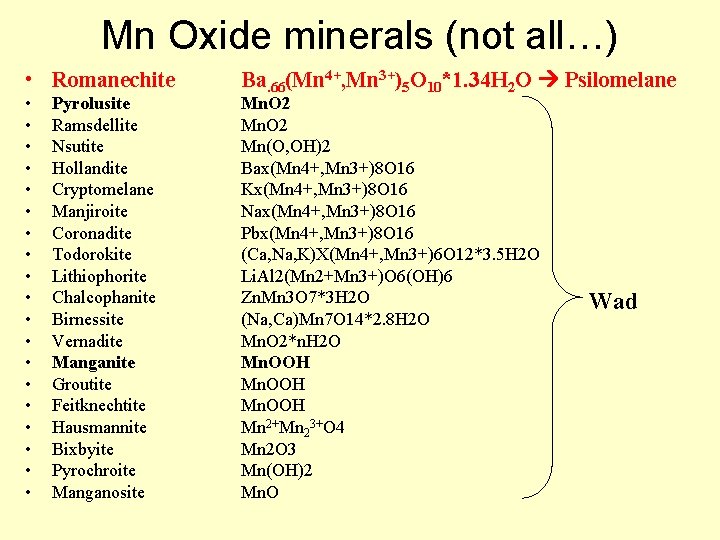
Mn oxides - oxyhydroxides • Mn exists as 2+, 3+, and 4+; oxide minerals are varied, complex, and hard to ID – ‘Wad’ soft (i. e. blackens your fingers), brown-black fine-grained Mn oxides – ‘Psilomelane’ hard (does not blacked fingers) grayblack botroyoidal, massive Mn oxides • XRD analyses do not easily distinguish different minerals, must combine with TEM, SEM, IR spectroscopy, and microprobe work

Mn Oxide minerals (not all…) • Romanechite • • • • • Pyrolusite Ramsdellite Nsutite Hollandite Cryptomelane Manjiroite Coronadite Todorokite Lithiophorite Chalcophanite Birnessite Vernadite Manganite Groutite Feitknechtite Hausmannite Bixbyite Pyrochroite Manganosite Ba. 66(Mn 4+, Mn 3+)5 O 10*1. 34 H 2 O Psilomelane Mn. O 2 Mn(O, OH)2 Bax(Mn 4+, Mn 3+)8 O 16 Kx(Mn 4+, Mn 3+)8 O 16 Nax(Mn 4+, Mn 3+)8 O 16 Pbx(Mn 4+, Mn 3+)8 O 16 (Ca, Na, K)X(Mn 4+, Mn 3+)6 O 12*3. 5 H 2 O Li. Al 2(Mn 2+Mn 3+)O 6(OH)6 Zn. Mn 3 O 7*3 H 2 O (Na, Ca)Mn 7 O 14*2. 8 H 2 O Mn. O 2*n. H 2 O Mn. OOH Mn 2+Mn 23+O 4 Mn 2 O 3 Mn(OH)2 Mn. O Wad

Iron Oxides • Interaction of dissolved iron with oxygen yields iron oxide and iron oxyhyroxide minerals • 1 st thing precipitated amorphous or extremely fine grained (nanocrystaliine) iron oxides called ferrihydrite Fe 2+ O 2

Ferrihydrite • Ferrihydrite (Fe 5 O 7 OH*H 2 O; Fe 10 O 15*9 H 2 O some argument about exact formula) – a mixed valence iron oxide with OH and water

Goethite • Ferrihydrite recrystallizes into Goethite (a. Fe. OOH) • There are other polymorphs of iron oxyhydroxides: – Lepidocrocite g-Fe. OOH – Akaganeite b-Fe. OOH

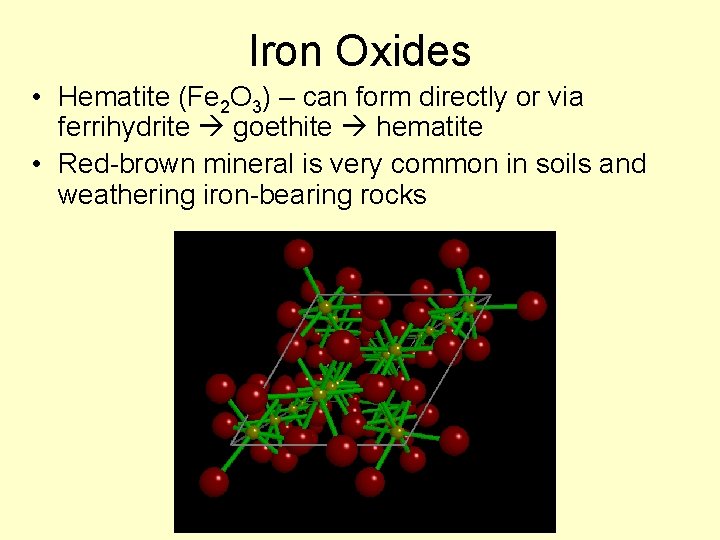
Iron Oxides • Hematite (Fe 2 O 3) – can form directly or via ferrihydrite goethite hematite • Red-brown mineral is very common in soils and weathering iron-bearing rocks

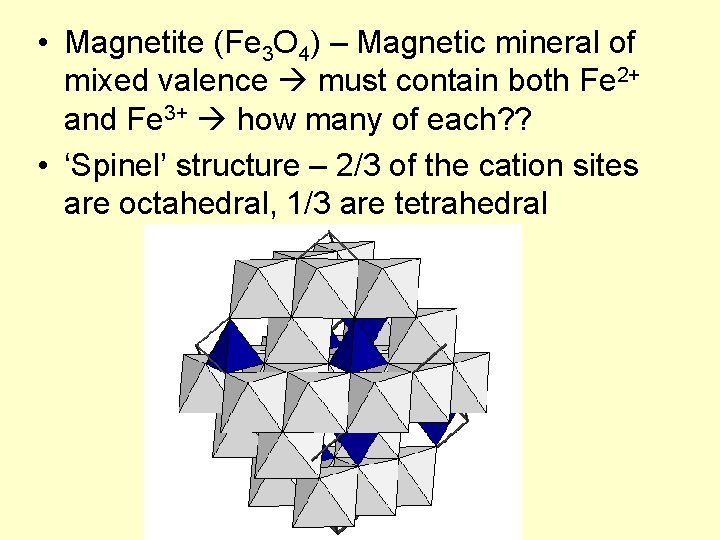
• Magnetite (Fe 3 O 4) – Magnetic mineral of mixed valence must contain both Fe 2+ and Fe 3+ how many of each? ? • ‘Spinel’ structure – 2/3 of the cation sites are octahedral, 1/3 are tetrahedral

Banded Iron Formations (BIFs) • HUGE Pre. Cambrian formations composed of hematite-jasper-chalcedony bands • Account for ~90% of the world’s iron supply • Occur only between 1. 9 and 3. 8 Ga many sites around the world Hammersley in Australia, Ishpeming in Michigan, Isua in Greenland, Carajas in Brazil, many other sites around the world…