Maintenance therapies in Multiple Myeloma Saurabh Chhabra MD

Maintenance therapies in Multiple Myeloma Saurabh Chhabra, MD MS Associate Professor of Medicine Froedtert & the Medical College of Wisconsin

Definitions Transplant-Eligible • Induction • Maintenance • Consolidation Transplant-Ineligible • Continuous vs. fixed duration therapy

Cytogenetics Staging • High risk • Standard risk International Staging System • Stage I-III, based on • albumin • β 2 -microglobulin Testing by: • Fluorescent In Situ Hybridization (FISH) • Karyotyping Revised-International Staging System • Stage I-III, based on • ISS stage • lactate dehydrogenase • cytogenetics
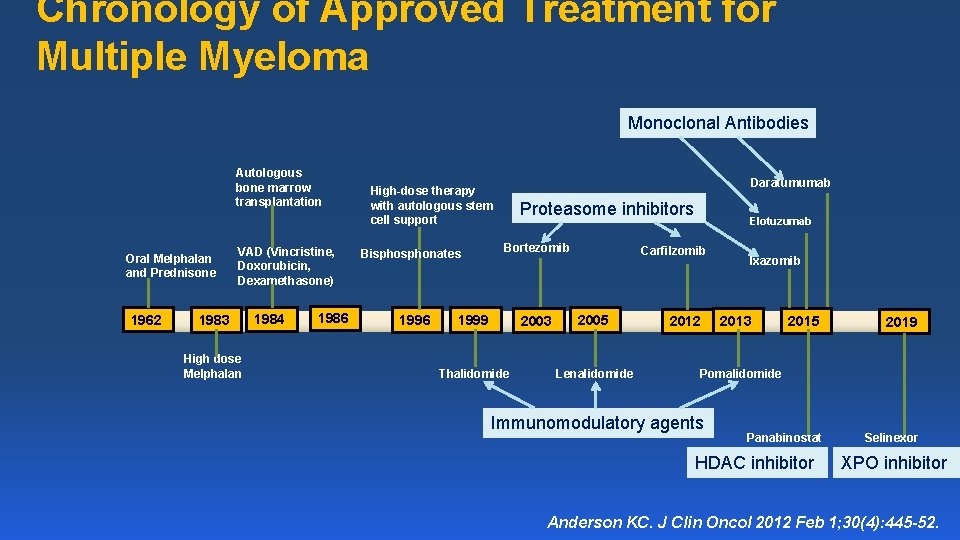
Chronology of Approved Treatment for Multiple Myeloma Monoclonal Antibodies Autologous bone marrow transplantation Oral Melphalan and Prednisone 1962 VAD (Vincristine, Doxorubicin, Dexamethasone) 1983 High dose Melphalan 1984 1986 Daratumumab High-dose therapy with autologous stem cell support Bisphonates 1996 Proteasome inhibitors Bortezomib 1999 2003 Thalidomide Elotuzumab Carfilzomib 2005 Lenalidomide 2012 Ixazomib 2015 2019 Panabinostat Selinexor 2013 Pomalidomide Immunomodulatory agents HDAC inhibitor XPO inhibitor Anderson KC. J Clin Oncol 2012 Feb 1; 30(4): 445 -52.

Attributes of maintenance therapy for Multiple Myeloma • Effective (in preventing return of multiple myeloma) • Safe (for long-term use) • Convenient (e. g. , oral >> intravenous) • Preserves quality of life

Drugs considered for maintenance therapy • Thalidomide (oral) Immunomodulatory Drugs • Lenalidomide (oral)(IMi. Ds) • Bortezomib (injectable) • Carfilzomib (injectable) Proteasome Inhibitors (PIs) • Ixazomib (oral) • Daratumumab (injectable) Monoclonal Antibodies • Elotuzumab (injectable) (with lenalidomide) (Mo. Abs)
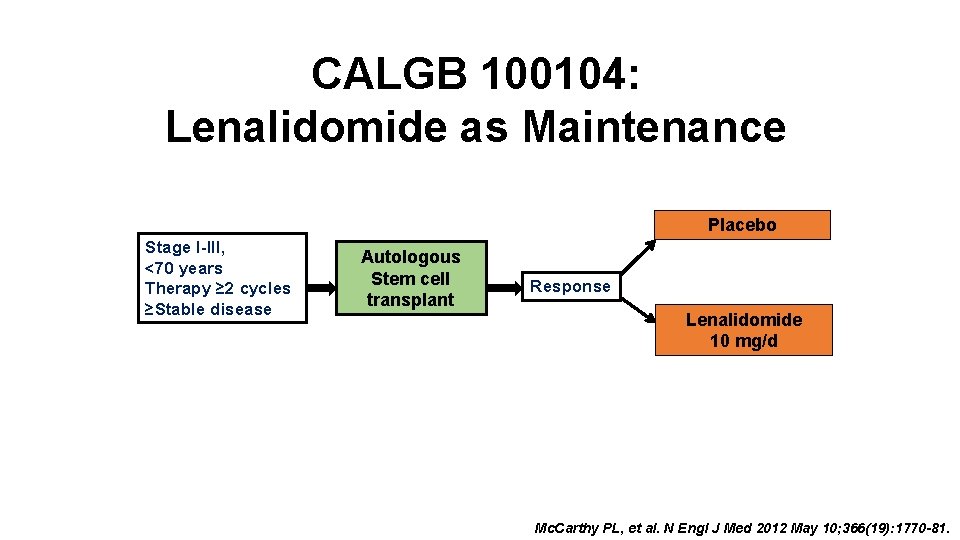
CALGB 100104: Lenalidomide as Maintenance Placebo Stage I-III, <70 years Therapy ≥ 2 cycles ≥Stable disease Autologous Stem cell transplant Response Lenalidomide 10 mg/d Mc. Carthy PL, et al. N Engl J Med 2012 May 10; 366(19): 1770 -81.
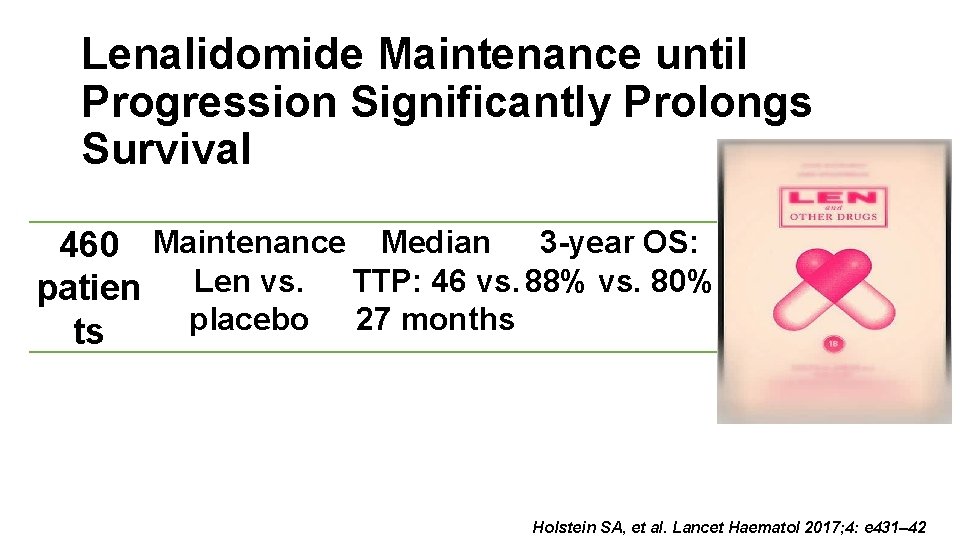
Lenalidomide Maintenance until Progression Significantly Prolongs Survival 460 Maintenance Median 3 -year OS: patien Len vs. TTP: 46 vs. 88% vs. 80% placebo 27 months ts Holstein SA, et al. Lancet Haematol 2017; 4: e 431– 42
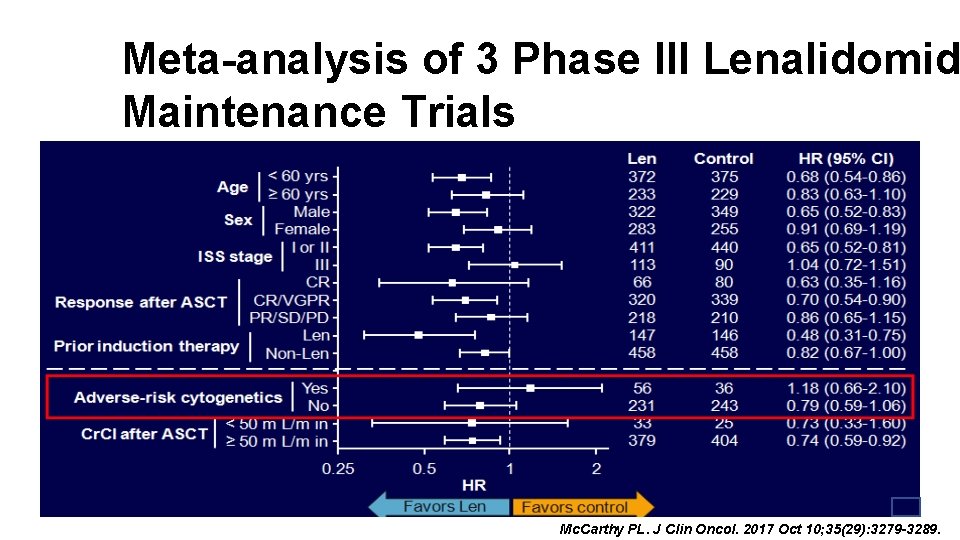
Meta-analysis of 3 Phase III Lenalidomid Maintenance Trials Mc. Carthy PL. J Clin Oncol. 2017 Oct 10; 35(29): 3279 -3289.
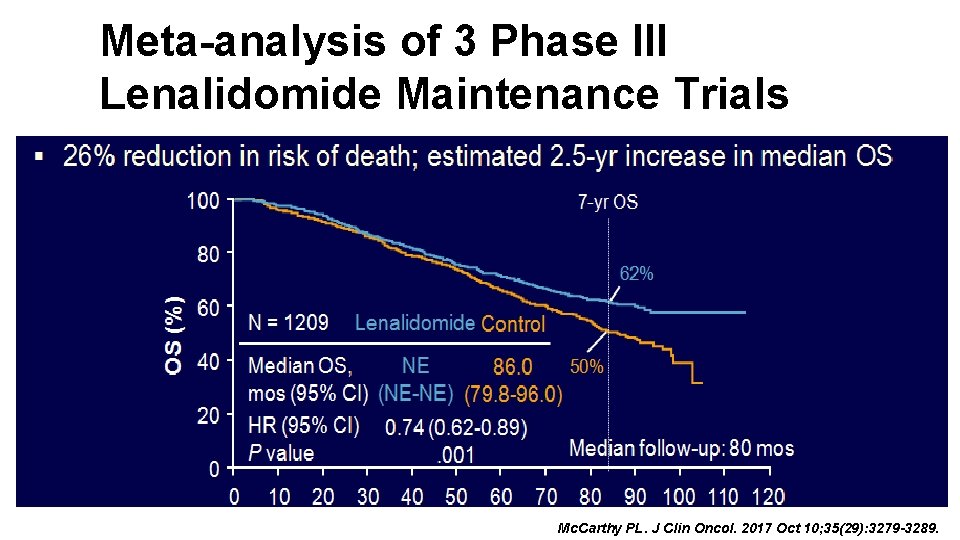
Meta-analysis of 3 Phase III Lenalidomide Maintenance Trials Mc. Carthy PL. J Clin Oncol. 2017 Oct 10; 35(29): 3279 -3289.
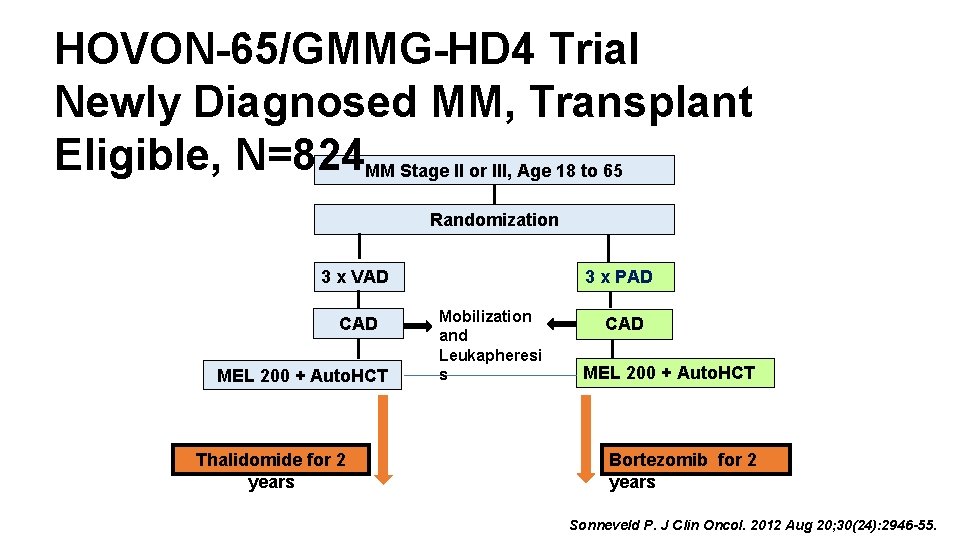
HOVON-65/GMMG-HD 4 Trial Newly Diagnosed MM, Transplant Eligible, N=824 MM Stage II or III, Age 18 to 65 Randomization 3 x VAD CAD MEL 200 + Auto. HCT Thalidomide for 2 years NDMM=Newly Diagnosed Multiple Myeloma 3 x PAD Mobilization and Leukapheresi s CAD MEL 200 + Auto. HCT Bortezomib for 2 years Sonneveld P. J Clin Oncol. 2012 Aug 20; 30(24): 2946 -55.
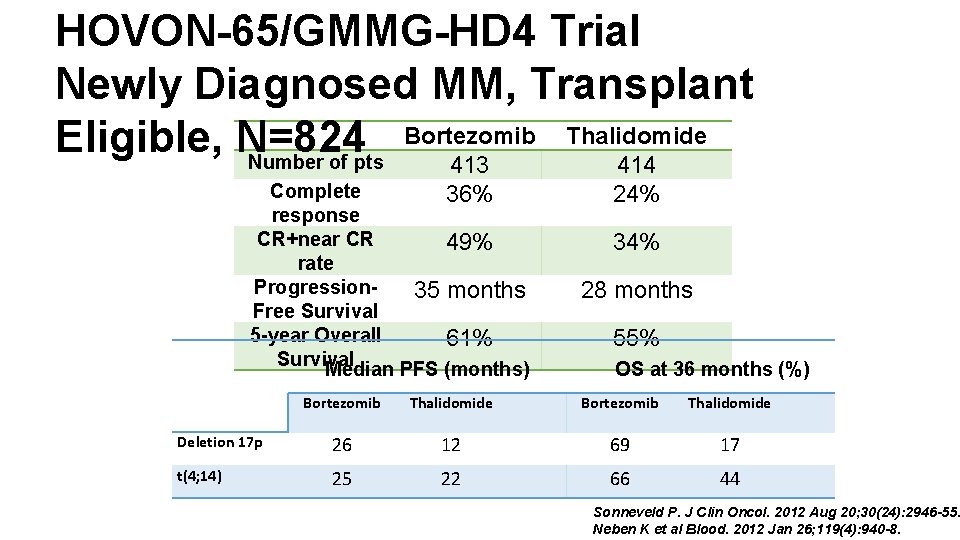
HOVON-65/GMMG-HD 4 Trial Newly Diagnosed MM, Transplant Bortezomib Thalidomide Eligible, N=824 Number of pts 413 414 Complete 36% response CR+near CR 49% rate Progression 35 months Free Survival 5 -year Overall 61% Survival Median PFS (months) 24% 34% 28 months 55% OS at 36 months (%) Bortezomib Thalidomide Deletion 17 p 26 12 69 17 t(4; 14) 25 22 66 44 Sonneveld P. J Clin Oncol. 2012 Aug 20; 30(24): 2946 -55. Neben K et al Blood. 2012 Jan 26; 119(4): 940 -8.
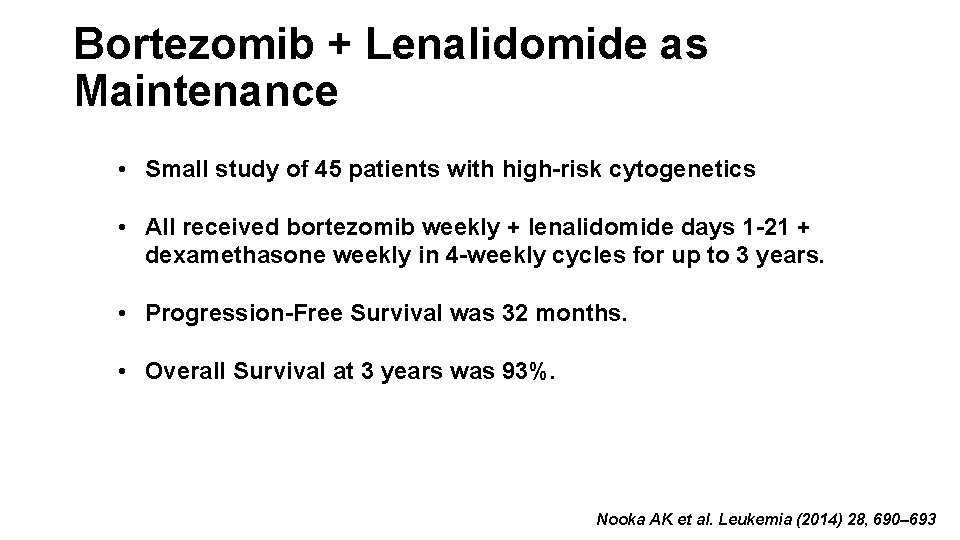
Bortezomib + Lenalidomide as Maintenance • Small study of 45 patients with high-risk cytogenetics • All received bortezomib weekly + lenalidomide days 1 -21 + dexamethasone weekly in 4 -weekly cycles for up to 3 years. • Progression-Free Survival was 32 months. • Overall Survival at 3 years was 93%. Nooka AK et al. Leukemia (2014) 28, 690– 693
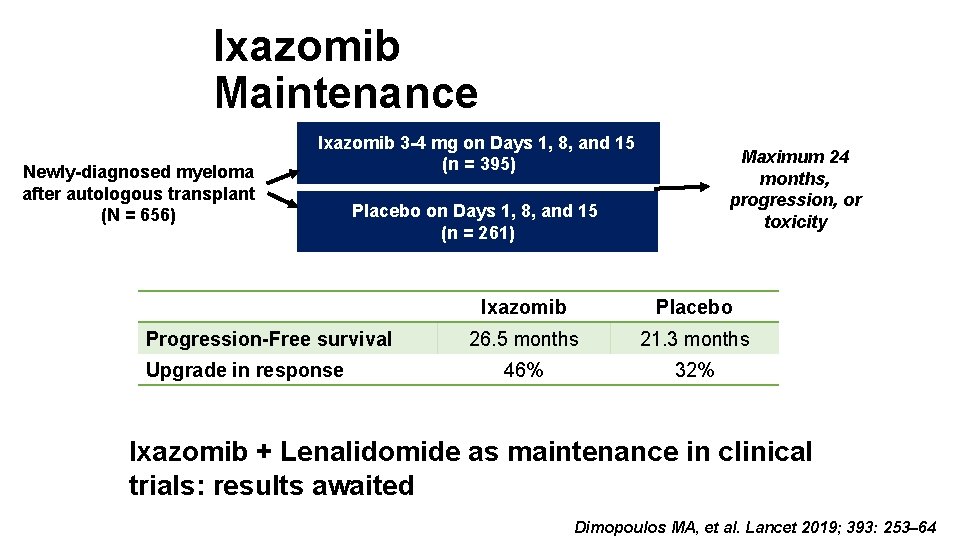
Ixazomib Maintenance Newly-diagnosed myeloma after autologous transplant (N = 656) Ixazomib 3 -4 mg on Days 1, 8, and 15 (n = 395) Placebo on Days 1, 8, and 15 (n = 261) Progression-Free survival Upgrade in response Maximum 24 months, progression, or toxicity Ixazomib Placebo 26. 5 months 21. 3 months 46% 32% Ixazomib + Lenalidomide as maintenance in clinical trials: results awaited Dimopoulos MA, et al. Lancet 2019; 393: 253– 64
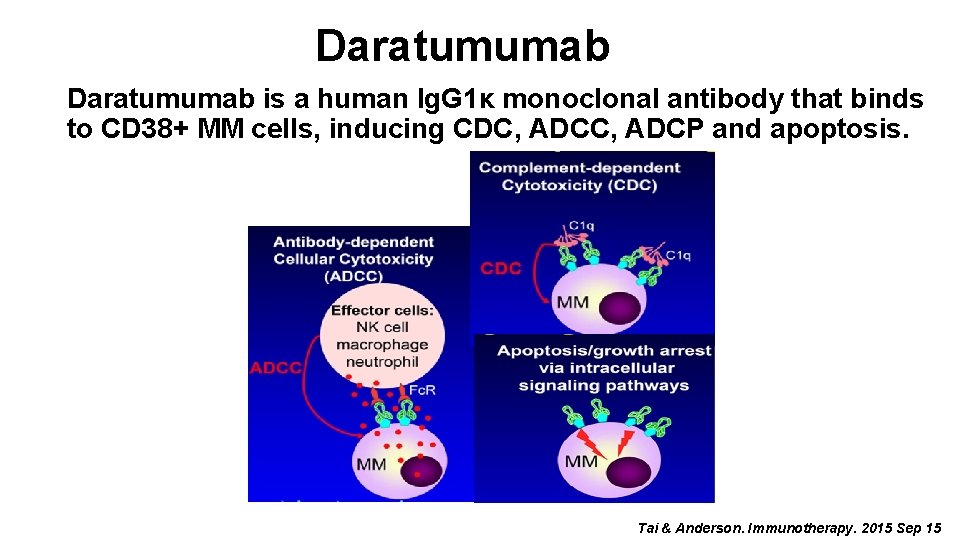
Daratumumab is a human Ig. G 1κ monoclonal antibody that binds to CD 38+ MM cells, inducing CDC, ADCP and apoptosis. Tai & Anderson. Immunotherapy. 2015 Sep 15
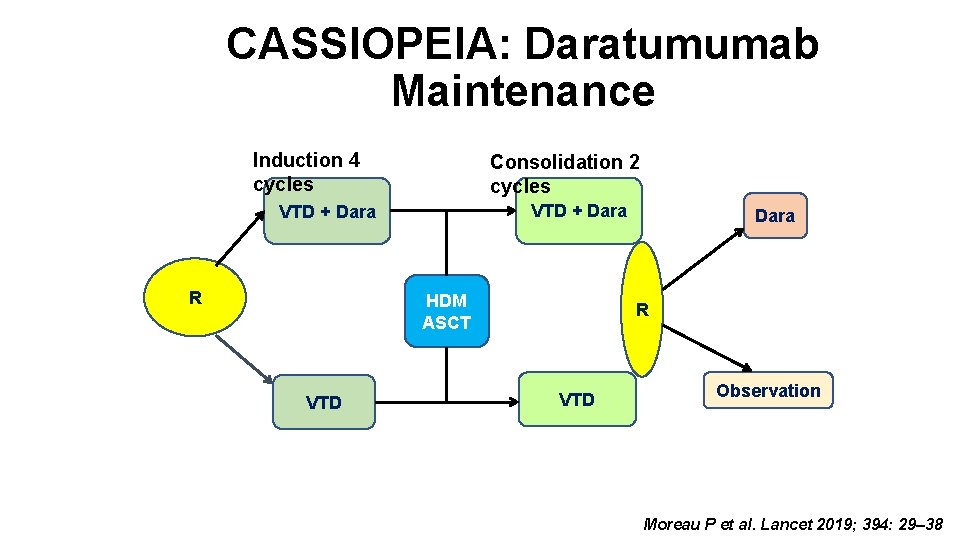
CASSIOPEIA: Daratumumab Maintenance Induction 4 cycles Consolidation 2 cycles VTD + Dara R HDM ASCT VTD Dara R VTD Observation Moreau P et al. Lancet 2019; 394: 29– 38

MAIA Phase III trial of daratumumab added to lenalidomide and dexamethasone in transplant-ineligible newly diagnosed MM Transplant-ineligible newly diagnosed MM (n=737) Daratumumab + Lenalidomide + Dex (n=368) ® Until progression Lenalidomide + Dex (n=369) Dara-Rd (n=368) Rd (n=369) 30 -months Progression. Free Survival 71% 56% Overall Response Rate Complete Response Rate 93% 47% 81% 24% Outcome Facon T et al. ASH 2018. Abstract LBA-2.

Summary • Maintenance is an integral part of myeloma treatment • Current recommendation is for all patients, regardless of stage, risk and response to receive maintenance. • Duration of maintenance – until progression or relapse of myeloma • Lenalidomide until progression is standard of care • High-risk patients: Bortezomib + lenalidomide • Role of Mo. Abs: results from trials are awaited
- Slides: 18