MAHARASHTRA COSMOPOLITAN EDUCATION SOCIETYS AZAM CAMPUS PUNE 411001














- Slides: 18

MAHARASHTRA COSMOPOLITAN EDUCATION SOCIETY’S AZAM CAMPUS PUNE 411001 E-Content Description Name of School / College: M A Rangoonwala Institute of Hotel Management and Research M A Rangoonwala IHMRP

Name and Designation of content creator /Producer Title of E content Theory/practical Title and No of Module Title and code of Paper Assoc. Prof Imran Sayyed Broad Subject Course Class Semester University /Board Date of Content Creation Name of Reviewer HOD/Principal Engineering BSc. HS SY Third SPPU 2 Jan 2020 Imran Sayyed Vapor-compression cycle Theory Vapor-compression cycle The Science of Hotel Engineering Subject Code : HS 206 M A Rangoonwala IHMRP

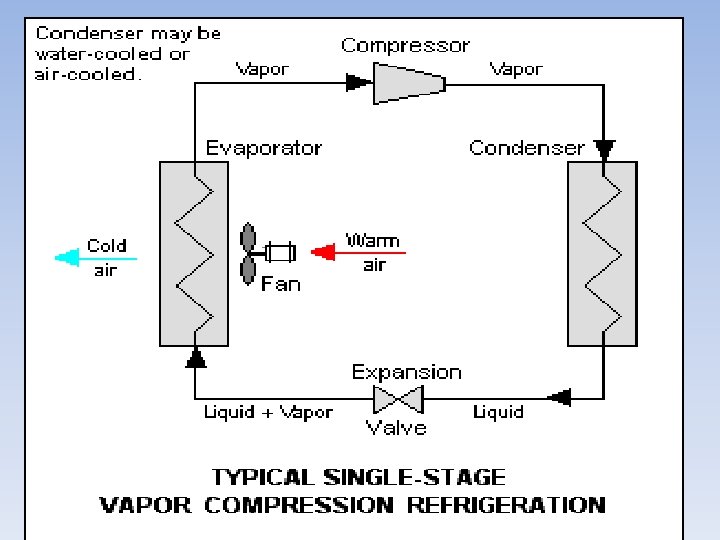
Vapor-compression cycle The vapor-compression cycle is used in most household refrigerators as well as in many large commercial and industrial refrigeration systems. provides a schematic diagram of the components of a typical vapor-compression refrigeration system. • • The thermodynamics of the cycle can be analyzed on a diagram[48] as shown in. In this cycle, a circulating refrigerant such as Freon enters the compressor as a vapor. From point 1 to point 2, the vapor is compressed at constant entropy and exits the compressor as a vapor at a higher temperature, but still below the vapor pressure at that temperature. From point 2 to point 3 and on to point 4, the vapor travels through the condenser which cools the vapor until it starts condensing, and then condenses the vapor into a liquid by removing additional heat at constant pressure and temperature. Between points 4 and 5, the liquid refrigerant goes through the expansion valve (also called a throttle valve) where its pressure abruptly decreases, causing flash evaporation and auto-refrigeration of, typically, less than half of the liquid.


• That results in a mixture of liquid and vapor at a lower temperature and pressure as shown at point 5. The cold liquid-vapor mixture then travels through the evaporator coil or tubes and is completely vaporized by cooling the warm air (from the space being refrigerated) being blown by a fan across the evaporator coil or tubes. The resulting refrigerant vapor returns to the compressor inlet at point 1 to complete thermodynamic cycle. • The above discussion is based on the ideal vapor-compression refrigeration cycle, and does not take into account real-world effects like frictional pressure drop in the system, slight thermodynamic irreversibility during the compression of the refrigerant vapor, or non-ideal gas behavior, if any.

Absorption cycle • In the early years of the twentieth century, the vapor absorption cycle using water-ammonia systems water was popular and widely used. After the development of the vapor compression cycle, the vapor absorption cycle lost much of its importance because of its low coefficient of performance (about one fifth of that of the vapor compression cycle). Today, the vapor absorption cycle is used mainly where fuel for heating is available but electricity is not, such as in recreational vehicles that carry LP gas. It is also used in industrial environments where plentiful waste heat overcomes its inefficiency.



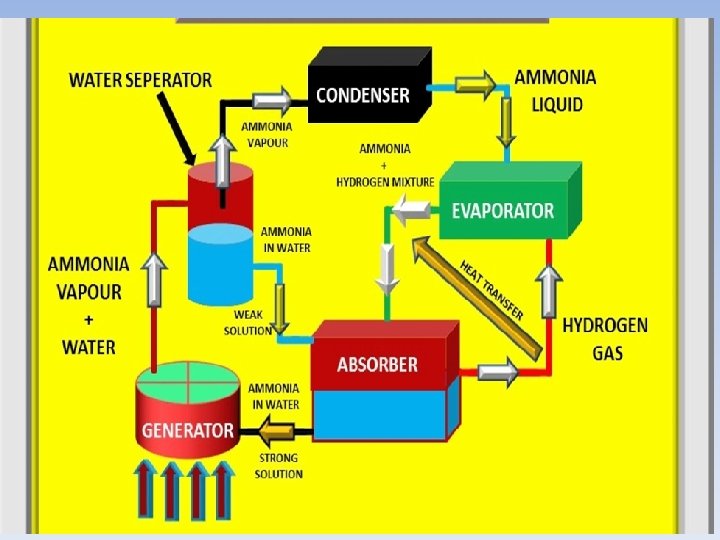
• The absorption cycle is similar to the compression cycle, except for the method of raising the pressure of the refrigerant vapor. In the absorption system, the compressor is replaced by an absorber which dissolves the refrigerant in a suitable liquid, a liquid pump which raises the pressure and a generator which, on heat addition, drives off the refrigerant vapor from the high-pressure liquid. Some work is needed by the liquid pump but, for a given quantity of refrigerant, it is much smaller than needed by the compressor in the vapor compression cycle. In an absorption refrigerator, a suitable combination of refrigerant and absorbent is used. The most common combinations are ammonia (refrigerant) with water (absorbent), and water (refrigerant) with lithium bromide (absorbent).

• Absorption refrigerators are often used for food storage in recreational vehicles. The principle can also be used to air-condition buildings using the waste heat from a gas turbine or water heater. Using waste heat from a gas turbine makes the turbine very efficient because it first produces electricity, then hot water, and finally, air-conditioning

Absorption cooling process • Both absorption and compressor refrigerators use a refrigerant with a very low boiling point (less than − 18 °C (0 °F)). In both types, when this refrigerant evaporates (boils), it takes some heat away with it, providing the cooling effect. The main difference between the two systems is the way the refrigerant is changed from a gas back into a liquid so that the cycle can repeat. An absorption refrigerator changes the gas back into a liquid using a method that needs only heat, and has no moving parts other than the refrigerant itself.

• Evaporation: A liquid refrigerant evaporates in a low partial pressure environment, thus extracting heat from its surroundings (e. g. the refrigerator's compartment). Because of the low partial pressure, the temperature needed for evaporation is also low. • Absorption: The now gaseous refrigerant is absorbed by another liquid (e. g. a salt solution). • Regeneration: The refrigerant-saturated liquid is heated, causing the refrigerant to evaporate out. The hot gaseous refrigerant passes through a heat exchanger, transferring its heat outside the system (such as to surrounding ambient -temperature air), and condenses. The condensed (liquid) refrigerant supplies the evaporation phase.

• In comparison, a compressor refrigerator uses a compressor, usually powered by either an electric or internal combustion motor, to increase the pressure on the gaseous refrigerant. The resulting hot, high-pressure gas is condensed to a liquid form by cooling in a heat exchanger ("condenser") that is exposed to the external environment (usually air in the room). The condensed refrigerant, now at a temperature near to that of the external environment, then passes through an orifice or a throttle valve into the evaporator section. The orifice or throttle valve creates a pressure drop between the high pressure condenser section and the low pressure evaporator section. The lower pressure in the evaporator section allows the liquid refrigerant to evaporate, which absorbs heat from the refrigerator food compartment. The now-vaporized refrigerant then goes back into the compressor to repeat the cycle

Refrigerator Maintenance • • • • How to care for your refrigerator Clean off the condenser coils twice per year. Condenser coils are responsible for removing heat from refrigerators and freezers. . Clean and replace the gasket (rubber door seal) as needed. . Defrost. . Clean the interior. . Deodorize. . Replace the ice maker or water dispenser water filter. Set the right temperature. Keep the fridge between 37 and 40 degrees Fahrenheit and the freezer at 0 degrees. Fill it up (even if you never cook and only have takeout). Refrigerators need “thermal mass” Do the swipe and protect daily. A good rule of thumb is to be diligent every day to prevent cross contamination and just ickiness in general. . Purge weekly. Once a week, grab leftovers and have a toss day. . Deep clean seasonally. . Change the water filter every six months.

Refrigerant • A refrigerant is a substance or mixture, usually a fluid, used in a heat pump and refrigeration cycle. In most cycles it undergoes phase transitions from a liquid to a gas and back again. Many working fluids have been used for such purposes. Fluorocarbons, especially chlorofluorocarbons, became commonplace in the 20 th century, but they are being phased out because of their ozone depletion effects. Other common refrigerants used in various applications are ammonia, sulfur dioxide, and non-halogenated hydrocarbons such as propane

Refrigerant • The ideal working fluid or often called refrigerant would have favorable thermodynamic properties, be noncorrosive to mechanical components, and be safe, including freedom from toxicity and flammability. It would not cause ozone depletion or climate change. Since different fluids have the desired traits in different degree, choice is a matter of trade-offs. • The desired thermodynamic properties are a boiling point somewhat below the target temperature, a high heat of vaporization, a moderate density in liquid form, a relatively high density in gaseous form, and a high critical temperature. Since boiling point and gas density are affected by pressure, refrigerants may be made more suitable for a particular application by appropriate choice of operating pressures.

Ammonia • • • Properties. Ammonia is a colourless gas with a characteristic pungent smell. It is lighter than air, its density being 0. 589 times that of air. It is easily liquefied due to the strong hydrogen bonding between molecules; the liquid boils at − 33. 3 °C (− 27. 94 °F), and freezes at − 77. 7 °C (− 107. 86 °F) to white crystals. Anhydrous ammonia is a clear liquid that boils at a temperature of -28°F. In refrigeration systems, the liquid is stored in closed containers under pressure. When the pressure is released, the liquid evaporates rapidly, generally forming an invisible vapor or gas. Liquid anhydrous ammonia weighs less than water. About eight gallons of ammonia weighs the same as five gallons of water. Liquid and gas ammonia expand contract with changes in pressure and temperature. Liquid ammonia will expand by 850 times when evaporating: Anhydrous ammonia gas is considerably lighter than air and will rise in dry air. Anhydrous ammonia is easily absorbed by water Ammonia, especially in the presence of moisture, reacts with and corrodes copper, zinc, and many alloys. Anhydrous ammonia is classified as nonflammable. However, ammonia vapor in high concentrations Anhydrous ammonia is an alkali. Ammonia is not, strictly speaking, a poison and repeated exposure to it produces no additive (chronic) effects on the human body. However, even in small concentrations in the air it can be extremely irritating to the eyes, throat, and breathing passages.
