Magnetism Overview of magnetic materials Magnetic objects Magnetic

Magnetism

Overview of magnetic materials

Magnetic objects

Magnetic field lines

Molecular magnetism, example 1 Paramagnets are attracted to magnetic fields
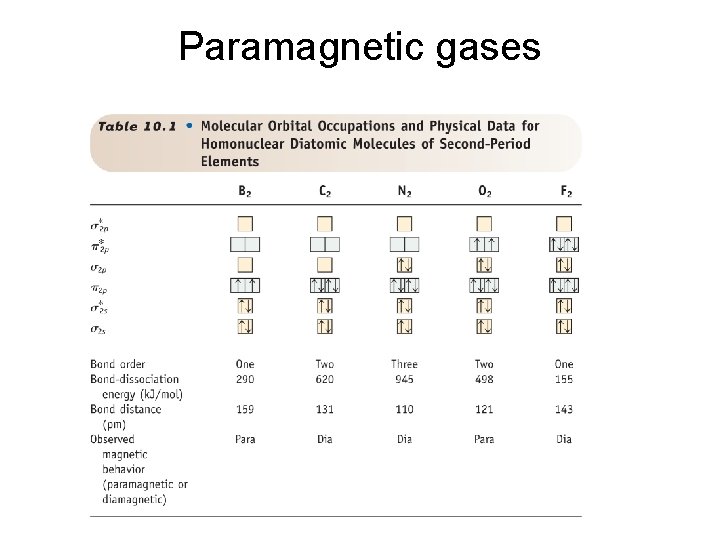
Paramagnetic gases
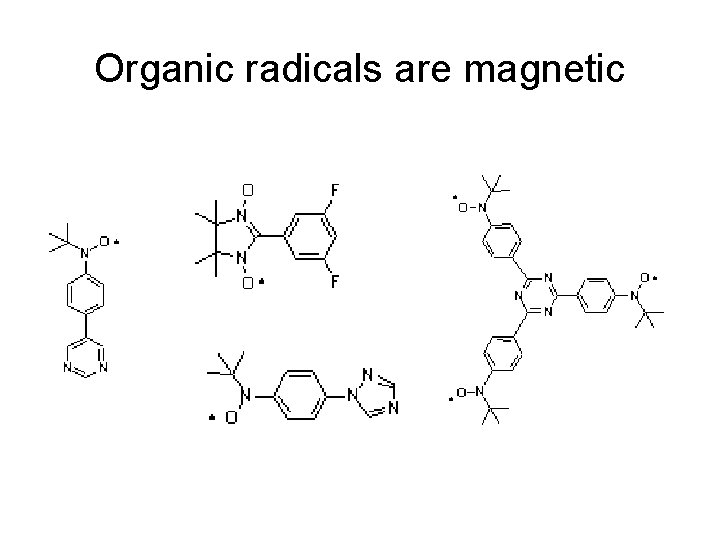
Organic radicals are magnetic
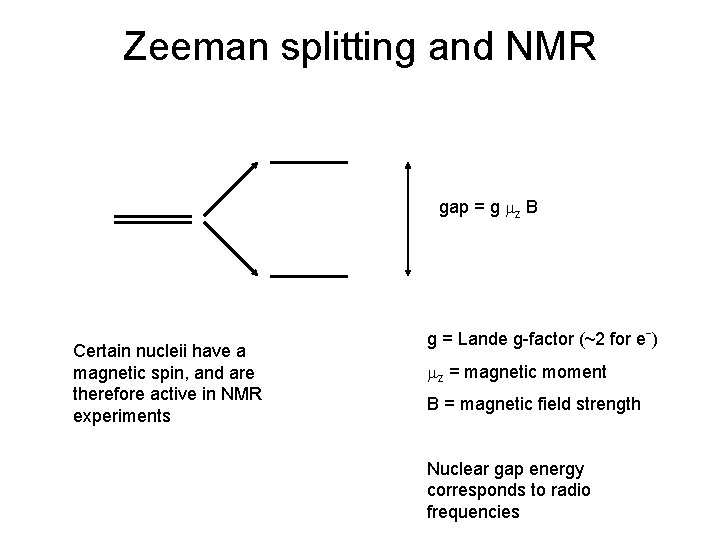
Zeeman splitting and NMR gap = g mz B Certain nucleii have a magnetic spin, and are therefore active in NMR experiments g = Lande g-factor (~2 for eˉ) mz = magnetic moment B = magnetic field strength Nuclear gap energy corresponds to radio frequencies
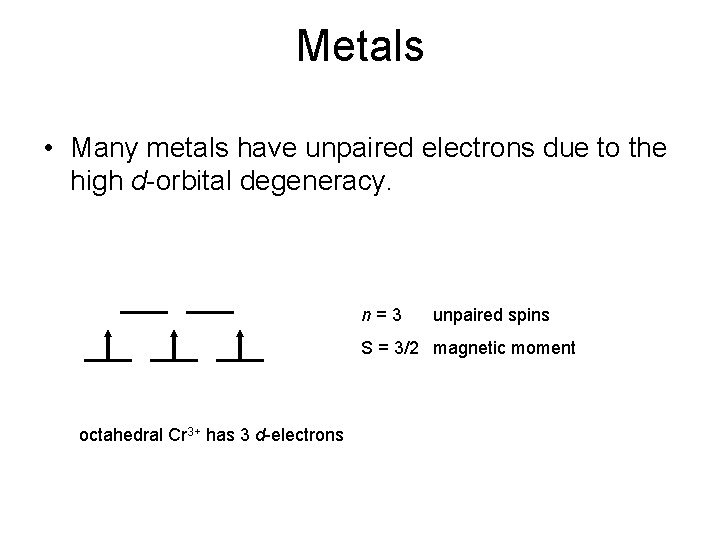
Metals • Many metals have unpaired electrons due to the high d-orbital degeneracy. n=3 unpaired spins S = 3/2 magnetic moment octahedral Cr 3+ has 3 d-electrons
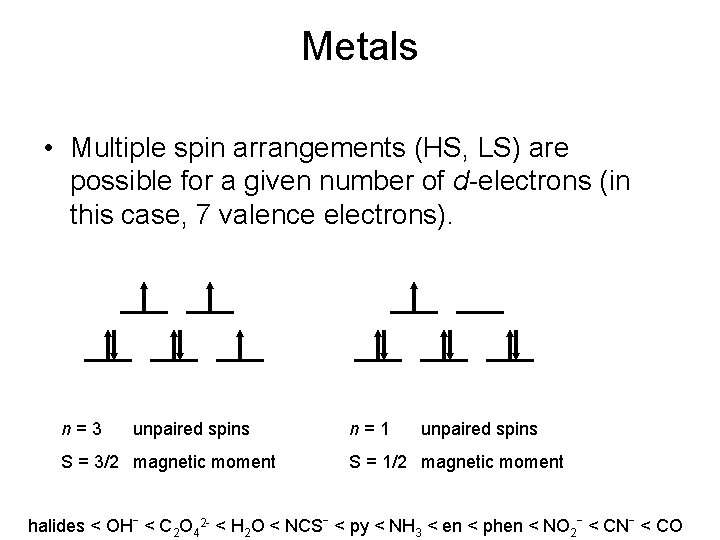
Metals • Multiple spin arrangements (HS, LS) are possible for a given number of d-electrons (in this case, 7 valence electrons). n=3 unpaired spins S = 3/2 magnetic moment n=1 unpaired spins S = 1/2 magnetic moment halides < OHˉ < C 2 O 42 - < H 2 O < NCSˉ < py < NH 3 < en < phen < NO 2ˉ < CNˉ < CO
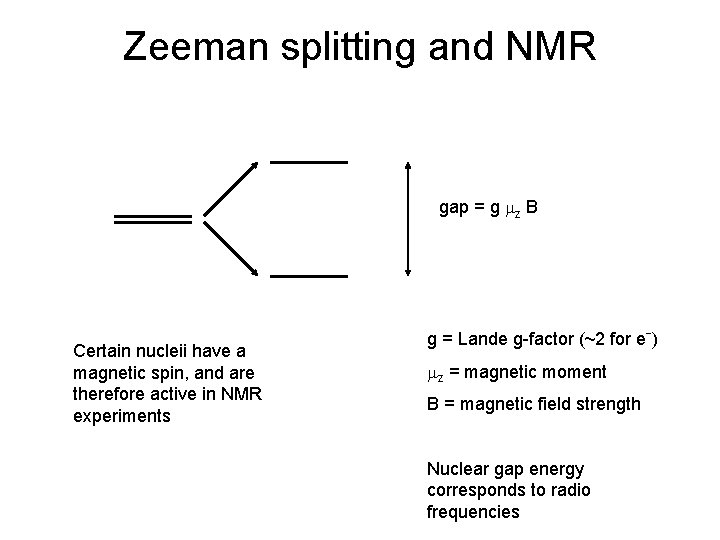
Zeeman splitting and NMR gap = g mz B Certain nucleii have a magnetic spin, and are therefore active in NMR experiments g = Lande g-factor (~2 for eˉ) mz = magnetic moment B = magnetic field strength Nuclear gap energy corresponds to radio frequencies
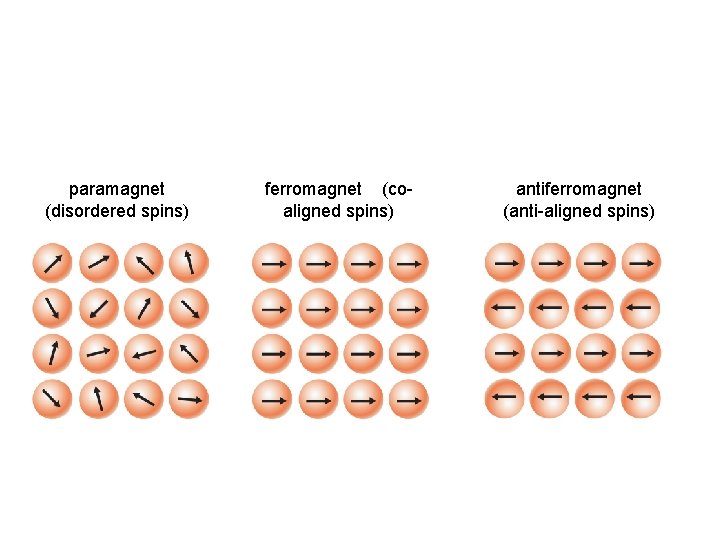
paramagnet (disordered spins) ferromagnet (coaligned spins) antiferromagnet (anti-aligned spins)

Ferromagnets have a persistent moment, even without a magnetic field

c. M vs T for ferromagnets ferromagnetic paramagnetic

The math of magnetism c. M – Molar susceptibility meff – Effective moment m. B – Bohr magneton

Useful experimental quantity • Molar magnetic susceptibility, c. M • Magnetic susceptibility per mole – Why use “molar” and not the “gram” susceptibility?

Moment per metal atom S = total spin (1/2 per e ˉ) n = # unpaired eˉ meff = moment in Bohr magnetons (m. B) g = magnetogyric ratio (Landé g-facor, ~2) m. B = 9. 27 x 10 -24 J / T

Magnetic susceptibility, c. M (Curie Law – assumes no spin-spin interactions) (units are slightly fudged) c. M = measurable quantity (M / H) Can calculate meff from c. M Can calculate # of unpaired electrons from meff

Magnetic susceptibility, c. M (Curie Law) (Curie-Weiss law) Simple approximations break down when spins are not isolated from each other
- Slides: 19