Magnetic Properties 11 Two sources 1 The orbital

Magnetic Properties 11 • Two sources 1. The orbital angular momentum of electrons in atoms 2. The intrinsic electron angular momentum • Basic relationship

Review of Maxwell’s Equation: Gaussian Unit SI Unit

The Second Maxwell Equation:

Magnetic Property versus Electrical Property:

Coulombs law of the force between poles:

Again, the important concepts in magnetic properties. 1. Magnetic field strength: 2. Magnetic field can be enhanced within a material and is known by the names of magnetic induction B (or magnetic flux density) just like Displacement (D) 3. f is defined as magnetic flux while B is defined as magnetic flux density.

Again, the important concepts in magnetic properties. 4. Magnetic moment mm:

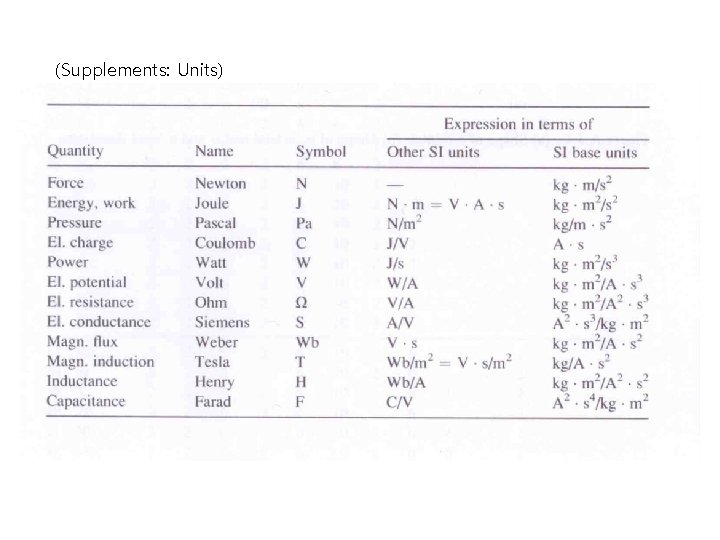
(Supplements: Units)

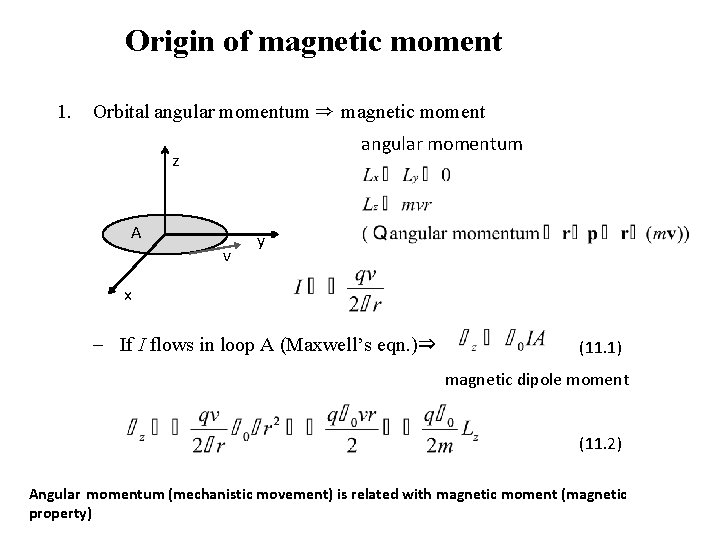
Origin of magnetic moment 1. Orbital angular momentum ⇒ magnetic moment angular momentum z A v y x - If I flows in loop A (Maxwell’s eqn. )⇒ (11. 1) magnetic dipole moment (11. 2) Angular momentum (mechanistic movement) is related with magnetic moment (magnetic property)
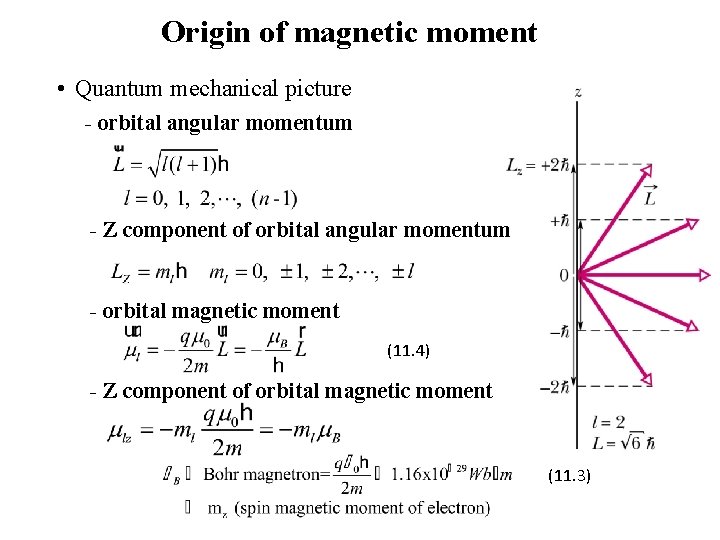
Origin of magnetic moment • Quantum mechanical picture - orbital angular momentum - Z component of orbital angular momentum - orbital magnetic moment (11. 4) - Z component of orbital magnetic moment (11. 3)
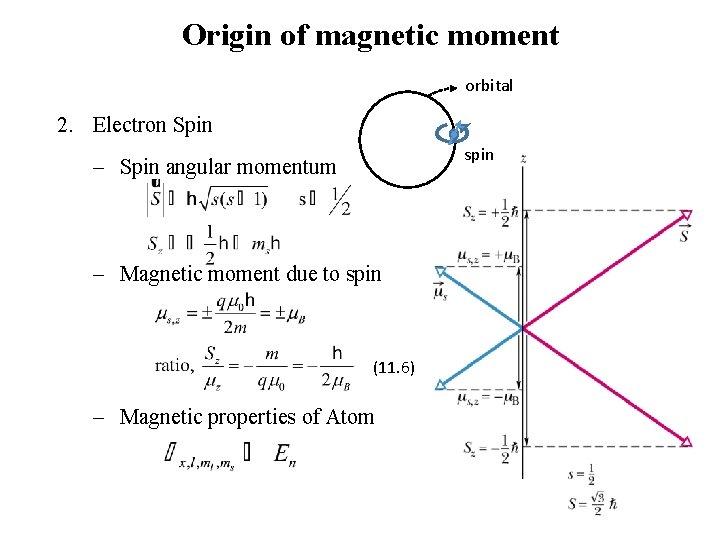
Origin of magnetic moment orbital 2. Electron Spin spin - Spin angular momentum - Magnetic moment due to spin (11. 6) - Magnetic properties of Atom

Origin of magnetic moment •

Magnetic moment in many electron atoms • In atom with many electrons - complex interact between orbital and spin moment • significant results 1. For a filled shell - only diamagnetism is possible. noble gas ionic solid

General description of magnetism • Diagnetism - No magnetic moment (not a function of temp) is present without field and a magnetic field is set up upon applied magnetic field, which opposes the applied field. (equivalent to Lenz’s law) - Slight repulsion - : small and negative (-1. 0× 10 -5 for copper ~ -3. 6× 10 -5 for gold) caused by orbital motion of electron • Paramagnetism - A magnetic moment is present in the absence of magnetic field, the magnetic moments line up with the magnetic field to decrease the total energy - Slight attraction of a material by a magnetic field - : small and positive (2 × 10 -5 for Al ~ +98× 10 -5 for Mn) - Thermal motion of the atoms tends to misalign the magnetic moments. paramagnetism decreases with increasing temperature

General description of magnetism • Ferromagnetism - The presence of a large magnetic moment that persists in the absence of a magnetic field due to spontaneous ordering of the moments by direct interaction at temperatures below curie temperature. - ~106 - Cooperative ordering → even when is removed • Antiferromagnetism - Results from internal interactions between magnetic moments. The magnetic moments of atoms on neighboring planes are oppositely oriented → Total magnetic moments over finite volume is zero - Antiferromagnetic ordering is destroyed above a critical temperature (Néel temp) • Ferrimagnetism - Ordered structure - The number of atoms with opposite spin are unequal • Net magnetic moment • Large field dependent susceptibility
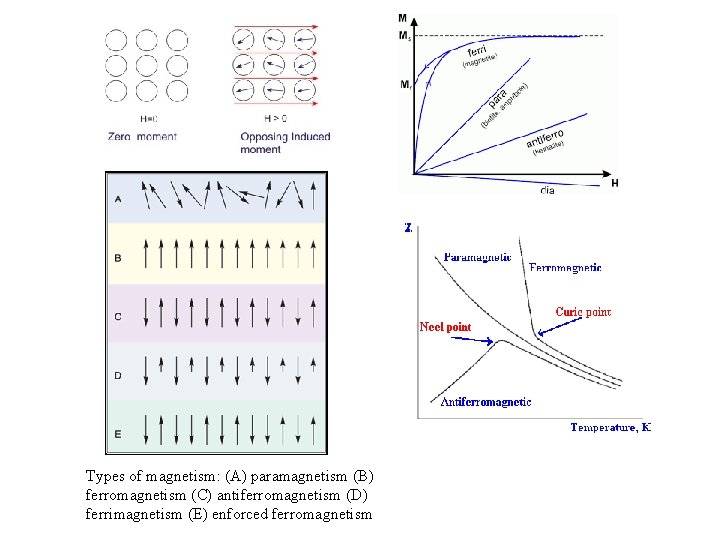
Types of magnetism: (A) paramagnetism (B) ferromagnetism (C) antiferromagnetism (D) ferrimagnetism (E) enforced ferromagnetism

Diamagnetism • without • Still there remains the interaction between applied and electrons “moving in orbits” increase the classical velocity of the orbital electron. • Every material has a diamagnetic contribution to H R - Effect of magnetic field is to increase angular momentum of the electron, resulting in the increase of the magnetic moment.

Diamagnetism (11. 10) - Larmor precession frequency (11. 11) - What is “I” due to Δω? (11. 12) atomic nucleus (11. 13) induced - diamagnetic atom has more contribution for large Z and r rr r

Diamagnetism (N: atoms per unit volume) (Large for heavier element) (11. 14) (11. 15) (classical Langevin equation)
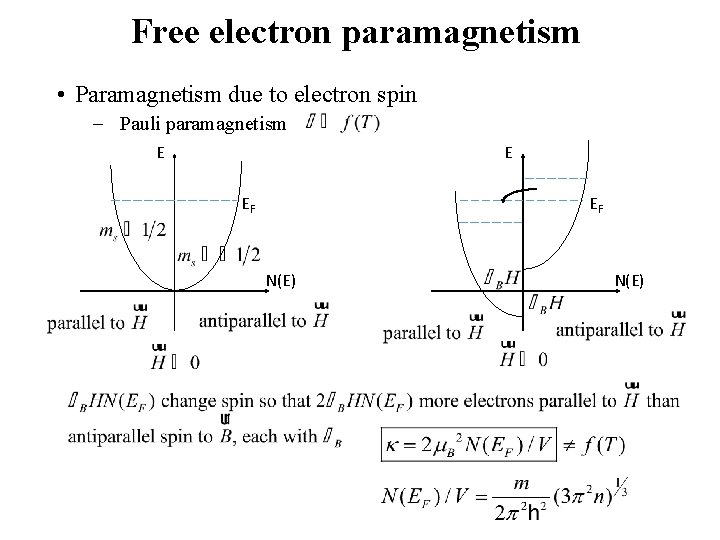
Free electron paramagnetism • Paramagnetism due to electron spin - Pauli paramagnetism E E EF EF N(E)
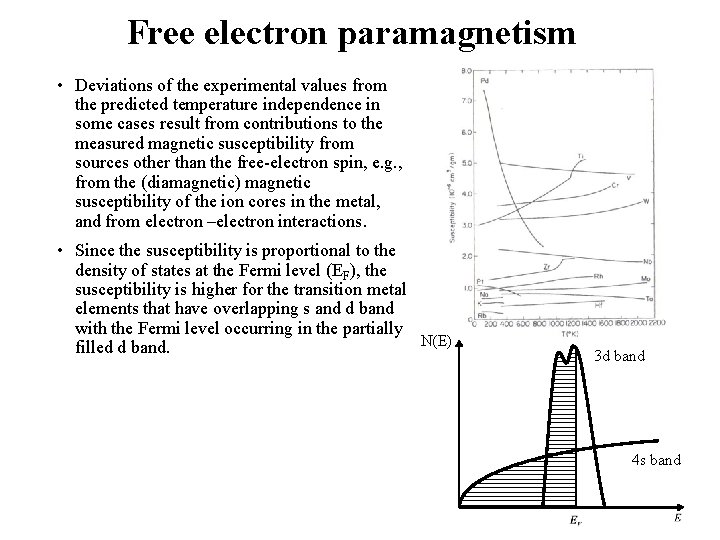
Free electron paramagnetism • Deviations of the experimental values from the predicted temperature independence in some cases result from contributions to the measured magnetic susceptibility from sources other than the free-electron spin, e. g. , from the (diamagnetic) magnetic susceptibility of the ion cores in the metal, and from electron –electron interactions. • Since the susceptibility is proportional to the density of states at the Fermi level (EF), the susceptibility is higher for the transition metal elements that have overlapping s and d band with the Fermi level occurring in the partially N(E) filled d band. 3 d band 4 s band

Bound electron paramagnetism • Paramagnetism may also arise from atoms or molecules with a net magnetic moment. (1) Atoms, molecules, or lattice defects with unpaired electrons, e. g. , an F-center in an alkali halide with an unpaired bound electron (2) Atoms or ions with partially filled shells, e. g. , transition elements, rare earth elements • Simple case of - electron antiparallel with has energy larger than electron

Bound electron paramagnetism The relative population of the two levels is given by a Boltzmann factor • (11. 20) (11. 23) • • The low-field limitation is usually satisfied for H = 104 G at 300 K. • For nonmetals the effect of the application of a magnetic field is to cause a flip in spin from ms = 1/2 to ms = -1/2 across energy difference . • In metals, however, unoccupied states with opposite spin are very close in energy near the Fermi level.

Ferro-magnetism • Fe, Co, Ni: incomplete d shell. • Alloys & rare earths: incomplete f shell. • Spins line up. • Interaction to order spin moments - Exchange energy between spins on neighboring atoms - Exchange energy between unpaired spins of neighboring atoms is lowest when these spins are parallel in a ferromagnet. - Overcome by T - Ferro → para when T > Tc (Curie temperature) - Dependence upon crystal structure • In isolated atoms, the exchange energy causes spins to be oriented in the same direction for less-than half filling of the shell. • Minimum energy condition requires spins to be paired.

Ferro-magnetism • What happens T > Tc (paramagnetic) • Represent “exchange” by an internal field (Weiss field) Curie-Weiss law • (from (11. 23)) Tc T

Ferro-magnetism • Calculate for Fe at 3 d 64 s 2 4 unpaired spin • • Expect per atom Actually per atom because of structure effect BCC structure

Anti-ferromagnetism • Cr, Mn (3 d 54 s) 5 unpaired spin in d shell • These are anti-ferromagnetic rather than ferromagnetic. • Cancellation of magnetic between planes results in antiferromagnetism. • The lowest energy is achieved by an antiparallel ordering of neighboring spins at low temperatures. • Interaction energy is negative rather than positive. - Mn, Cr, Mn. O, Mn. S, Ni. O • Spins in one atomic plane are oriented parallel to one another but that spins in neighboring planes are antiparallel. plane A B

Antiferromagnetism • Above the Néel temperature • At low temperature, the negative exchange forces dominate over thermal effects and the susceptibility is small. • As the temperature increases, the cancellation of magnetization due to antiferromagnetic arrangement is decreased, leading to an increase susceptibility. • At the Néel temperature, the antiferromagnetic coupling is totally overcome by thermal agitation, and the material becomes essentially paramagnetic at higher temperatures. paramagnetic T ferromagnetic TC T antiferromagnetic

Ferrimagnetism • MO·Fe 2 O 3 ; divalent cation (Mn, Fe, Co, Ni or Cu) • Low conductivity insulators • Induced currents from changing magnetic field are small. • Good for high frequency applications with low heating losses the magnetic properties are totally associated with metal ion. • Fe 3 O 4 ; we expect However we can get • Interpretation - Oxygen ions ~ FCC → no magnetic moment - Fe 3+ occupies both octahedral and tetrahedral equally distributed. - But the ions on different sites are coupled antiferromagnetically. v. Total magnetic moment is due to Fe 2+ ions that are located on octahedral sites → Total magnetic moment is 4. • 5 for Mn, 3 for Co

Ferromagnetic domains • All practical ferromagnetic materials consist of magnetic domain in the absence of an applied magnetic field. • The orientation of the magnetic moment in a domain corresponding to overall minimization of various forms of magnetic energy. • Domain exist in magnetic material in order to minimize the total magnetic energy which has several different contributions. 1. Magnetostatic energy ~ the energy in the external magnetic field: ~H 2 2. Magnetocrystalline anisotropy energy ~ the energy required to align magnetic moments in crystalline directions not Bloch domain walls favored by the particular crystalline structure (preferred direction in BCC ~ cubic edges, FCC ~ cube diagonals) 3. Magnetoelastic energy ~ A small change in physical dimensions in the direction of magnetization (20 ~ 40 ppm)
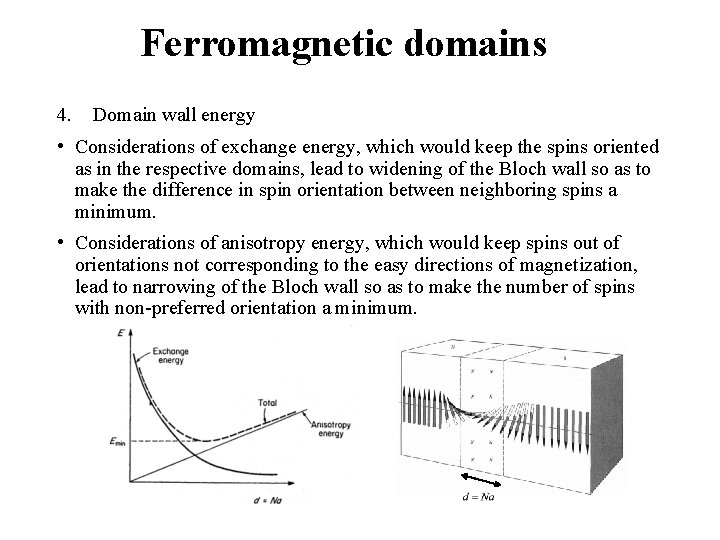
Ferromagnetic domains 4. Domain wall energy • Considerations of exchange energy, which would keep the spins oriented as in the respective domains, lead to widening of the Bloch wall so as to make the difference in spin orientation between neighboring spins a minimum. • Considerations of anisotropy energy, which would keep spins out of orientations not corresponding to the easy directions of magnetization, lead to narrowing of the Bloch wall so as to make the number of spins with non-preferred orientation a minimum.

Ferromagnetic domains 4. Domain wall energy • We assume that the wall has a width d corresponding to N atoms and a lattice constant a. Excess of interchange energy when • For N atoms in a line, therefore, and calculated per unit area, the increase in exchange energy becomes • The magnetocrystalline anisotropy energy is proportional to the width of the wall. (has a typical value of about 300 for Fe)

Ferromagnetic Hysteresis • Origin ~ defects and inclusions - Many defects in domains → large area hysteresis • “Hard” magnetic materials - High Mr, High Hc - Many defects - Useful as permanent magnet • “Soft” magnetic materials - Area is small - Few defects and inclusions - Useful for transformer cores or for electromangnets Remanent magnetization Coercive field

Ferromagnetic Hysteresis • Mechanisms ① For very small H, the curve is reversible since inclusions in the material prevent any motion of domains and only movable spins within domains are affected. ② As H↑, domains in the direction of H grow at the expense of those in other orientations by a series of discrete jumps known as Barkhausen jumps. ③ Finally, at high value of H, material has become a single domain and the saturation magnetization Ms is approached asymptotically as all spins are brought into exact alignment with H. ④ As the magnetic field is then reduced, magnetic walls reform and when the field is reduced to zero, a certain remanent magnetization Mr remains.

The end
- Slides: 37