Magnesiumbased Alloys Magnesium is HCP at all temperatures


Magnesium-based Alloys Magnesium is HCP at all temperatures up to its melting point of 649 o. C It has relatively high strength – but limited ductility at room temperature It can be easily worked at high temperatures – i. e. , at 400 o. C Mg is a highly reactive metal It reacts with air and moisture – so must be covered with a flux during melting For covered crucibles the flux is 20% KCl, 50% Mg 2 Cl, and 15% Ca. F 2. For open pots the flux is 55% KCl, 34% Mg 2 Cl, 9% Ba. Cl 2 and 2% Ca. F 2. - strong reducing agents.

Magnesium-based Alloys Magnesium reacts with the Si. O 2 in clays to form Mg 2 Si – but it can be safely melted in iron or graphite crucibles. To obtain a bright, clean casting – the mold is covered with sulphur boric acid or KBF 4. To dissolve magnesium alloy precipitates, it is solution treated at 390 – 410 o. C If the solution temperature is too high – 1) It will “burn” where low melting grain boundary phases are exuded at the surface. 2) A grey-black powder appears on the surface 3) Internal voids form due to evolution of gaseous phases.

General Properties of Mg-Alloys • The corrosion resistance of Mg alloys is improved by using high purity starting materials and modifying practices with respect to caustic fluxes. • Mg alloys are still susceptible to corrosion in salt atmospheres – a problem for “mag” wheels in snow belt regions – and for marine applications. • Aircraft are not so critical – but low flying over the ocean – or the use of reactive de-icing fluids – can create problems

General Properties of Mg-Alloys • Mg is also “notch sensitive” – so care has to be taken in design to remove sharp corners – and abrupt changes in section • Mg has excellent machining properties - but poor machining practice can introduce severe notch brittle effects • Mg has a modulus of elasticity of 45 GPa – compared to 71 GPa for Al and 200 GPa for steel

General Properties of Mg-Alloys • Mg density is 1. 8 g/cm – compared to 2. 8 g/cm for Al and 7. 9 g/cm for steel – on a mass basis, Mg has the greatest stiffness/weight – and steel the least • Mg is relatively difficult to weld – as it must be protected from the atmosphere by an inert gas – using a tungsten arc or consumable Mg • It can be welded - like Al – using a gas torch with suitable flux – for temporary repairs in the field.

Mg-Al Alloy System Al is soluble up in Mg up to ~12. 6 wt% Alloys containing up to 3 wt% Al are solution strengthened Alloys with 6 -9 wt% Al can be precipitation hardened

Mg-Al Alloy System At 437 o. C a. Mg forms a eutectic with an intermediate phase d – which has a mean composition of 32 wt%Al – or 33 at%Al – Its chemical formula is actually Mg 17 Al 12 As d is brittle - the eutectic is also brittle – as d is the major constituent – the eutectic contains 71. 4% d and 28. 6% a. Mg

Mg-Zn Alloy System Zn is soluble in Mg up to 8. 4 wt% At 341 o. C a. Mg forms a eutectic with an intermediate phase Mg. Zn – which has a mean composition of 54 wt%Zn As Mg. Zn is brittle – the eutectic is also brittle – as Mg. Zn is the major constituent
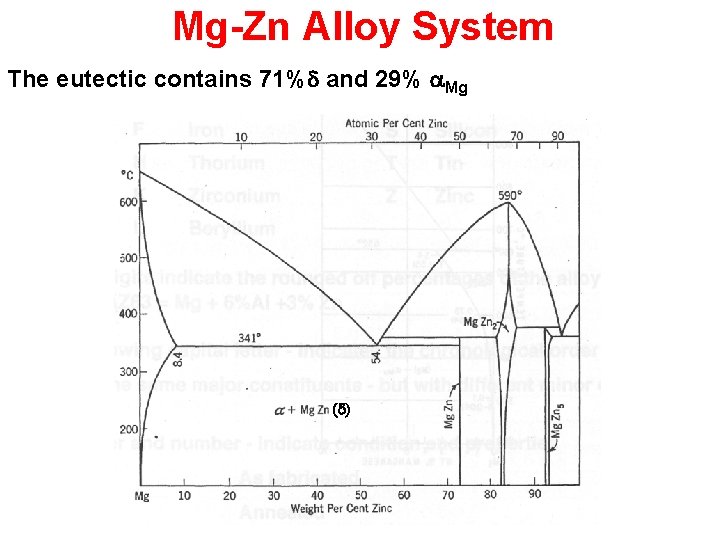
Mg-Zn Alloy System The eutectic contains 71%d and 29% a. Mg (d)

Mg-Mn Alloy System Mn is soluble in Mg up to 3. 4 wt% The a. Mg phase forms by a peritectic reaction at 652 o. C. As there are no intermediate phases for precipitation hardening – Mg-Mn alloys are strengthened by solid solution hardening alone.

ASTM Designation for Mg Alloys 1. Two capital letters indicate the two principal alloying elements. A Aluminium M Manganese B Bismuth N Nickel C Copper P Lead D Cadium Q Silver E Rare Earth R Chromium F Iron S Silicon H Thorium T Tin K Zirconium Z Zinc L Beryllium

ASTM Designation for Mg Alloys 2. 3. 4. Two digits indicate the rounded off percentages of the alloying elements, e. g. , AZ 63 = Mg + 6%Al + 3%Zn A following capital letter – indicates the chronological order of an alloy – with the same major constituents – but with different minor elements. A letter and number – indicate condition and properties F As fabricated O Annealed H 10, H 11 Slightly strain hardened H 23, F 24, H 26 Strain harden and partially annealed T 4 Solution treated T 5 Artificially aged T 6 Heat treated and artificially aged Similar to Al alloys

Compositions of Mg-Alloys Mg – Mn (1. 2 – 1. 5%) – solution hardening Mg – Al (3 -6%) + Zn (0. 4 – 1. 5%) – solution hardening Mg – Al (6 – 10%) + Zn (2 -3%) – precipitation hardening Mg – Zn (3. 5 – 6. 5%) + Zr (0. 55 – 1. 0%) – precipitation hardening Mg – Rare Earths* (0. 75 – 1. 75%) + Zn (3. 5 – 5. 0%) + Zr (0. 4 – 1. 0%) – precipitation hardening * - Mo, Nb, Ta, W Mg – Ce (6%) – precipitation hardening These alloys are solution treated at 390 – 410 o. C and then air cooled. Due to the low melting temperature – this allows ageing at room temperature, i. e. , natural ageing – after solution treatment – they do not have to be tempered.
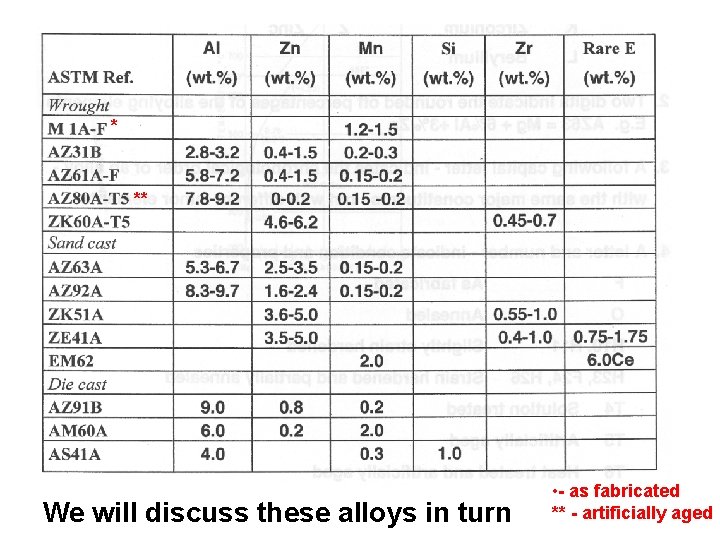
* ** We will discuss these alloys in turn • - as fabricated ** - artificially aged

Wrought Mg-Alloys • All solid solution Mg alloys can be hot forged at 300 – 400 o. C in hydraulic presses – rather like hammers. • Extrusions can also be made from alloys – to obtain a fine grain size extrusions are made from very fine pellets • M 1 A, AZ 31 B and AZ 61 A – can be rolled into sheet at temperatures ~200 o. C • These alloys are not heat treatable.

Wrought Mg-Alloys • AZ 80 A and ZK 60 A are effectively solution treated after forging – because of the hot working temperature is close to 400 o. C – so precipitation hardening during subsequent aging at room temperature occurs. • AZ 80 A and ZK 60 A are used for “high” temperature ~150 o. C – applications • ZK 60 A – T 5 –contains no Al – so is more expensive – but has greater strength and ductility than AZ 80 A.

Microstructures of Mg-Alloys - 1 AZ 31 Alloy – Annealed after hot working AZ 31 Alloy – cold rolled into sheet – work hardened

Microstructures of Mg-Alloys - 2 M 1 A Alloy – Annealed Particles in grain boundaries are impurities

Microstructures of Mg-Alloys - 2 ZK 60 Alloy – Extruded from pellets To obtain fine grain size (0. 001 mm)

Sand Cast Mg-Alloys • Mg reacts with Si. O 2 – causing the skin of the casting to be blackened (oxidized) to an appreciable depth below the surface. • To obtain a bright surface – “inhibitors” – such as sulphur, boric acid or KBF 4 – are mixed with the molding sand. • The reactive nature of Mg also means that sand cast alloys are subject to microporosity – caused by evolution of hydrogen* – with a consequent deterioration of its mechanical properties • Insoluble gases – such as He and Cl – are bubbled through the melt before casting to remove reactive gases such as H. • * - similar to Al alloys

Sand Cast Mg-Alloys • It is also evident from the phase diagrams that sand cast alloys will contain brittle networks of eutectic constituents • To improve the ductility of these castings they can be solution treated to dissolve the eutectic constituents – and this treatment also increases the tensile strength • Aging a solution-treated alloy strongly increases the yield point – and slightly lowers the ductility – but has relatively little effect on the ultimate strength • Increasing the amount of Al increases the strength – compared AZ 63 with AZ 92 – but lowers the casting quality and increases the amount of microporosity • The stronger Mg-Zn-Zr alloys are also more difficult to cast.

Microstructures of Sand Cast Mg-Alloys - 1 Grain boundary constituent is Mg 17 Al 12 Grain boundary constituent is local Mg 17 Al 12

Microstructures of Sand Cast Mg-Alloys - 2 EM 62 Alloy – As Cast The eutectic constituent is Mg 9 Ce AZ 91 B Die Casting Alloy – As Cast The Mg 17 Al 12 eutectic is very fine because of chill casting

Die Cast Mg-Alloys • Die cast alloys have excellent dimensional tolerances – and can be formed in complicated shapes as the liquid is forced into a steel mold under pressure. • Alloy AM 60 A is used for auto wheels. • Alloy AS 41 A is used for crankcases for air cooled engines like VWs • AZ 91 B is a general purpose alloy – recently used for dash boards in GM trucks Die cast alloys are significantly stronger than sand cast alloys – as they are not susceptible to microporosity.

Mechanical Properties of Mg-Alloys (intermediate step)
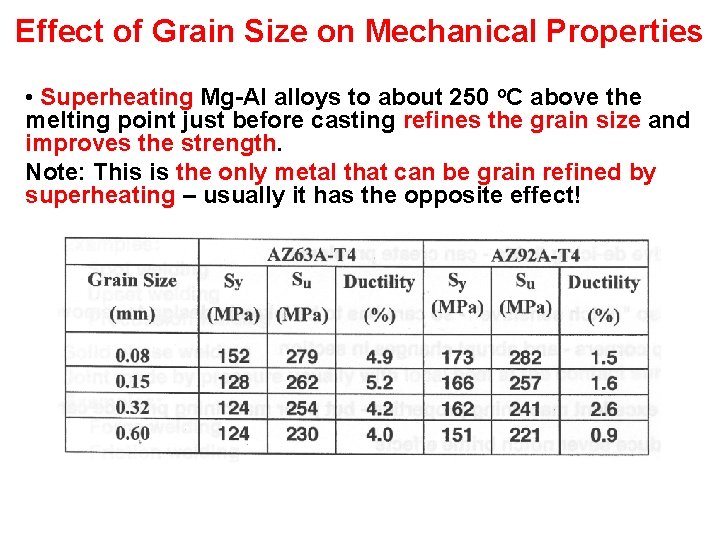
Effect of Grain Size on Mechanical Properties • Superheating Mg-Al alloys to about 250 o. C above the melting point just before casting refines the grain size and improves the strength. Note: This is the only metal that can be grain refined by superheating – usually it has the opposite effect!

Effect of Grain Size on Mechanical Properties The grain size can also be refined by applying one of the following treatments at 760 o. C – i. e. , just before casting: 1) Vigorous stirring 2) Bubbling acetylene, methane, propane or carbon tetrachloride 3) Stirring in 0. 003% carbon – as graphite or lamp black – or Al 4 C 3.

The End Any questions or comments?
- Slides: 29