Magnesium Hydrochloric Acid LAB 19 Mg s H
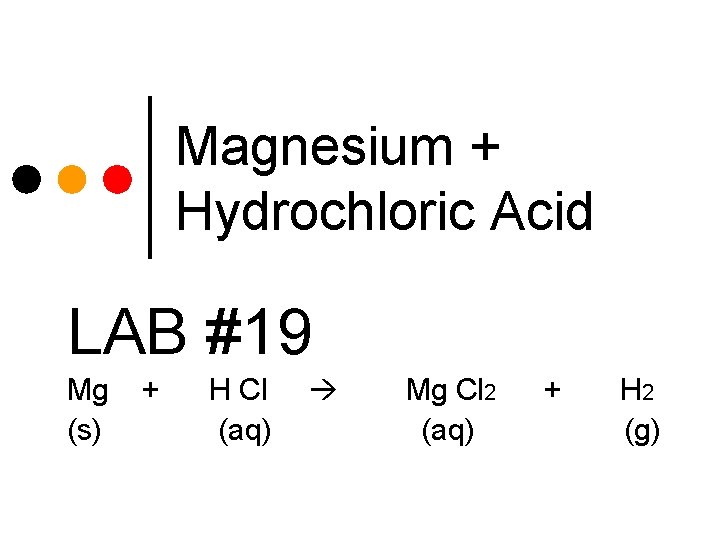
Magnesium + Hydrochloric Acid LAB #19 Mg (s) + H Cl (aq) Mg Cl 2 (aq) + H 2 (g)

MATERIALS q Ring stand q Eudiometer tube q Tray q String
MATERIALS q Magnesium ribbon q 5 ml of HCl q Goggles q Thermometer q Ruler q Beaker

PROCEDURE 1) With your goggles on, measure out 5 ml of the concentrated hydrochloric acid HCl) in the fume hood.

PROCEDURE 2) Pour the acid into the eudiometer tube. Fill the tube with water. Clamp it into the ring stand.

PROCEDURE 3) Measure the length of your magnesium ribbon to the nearest centimeter. 4) Fill the gray tray with 2 inches of tap water.
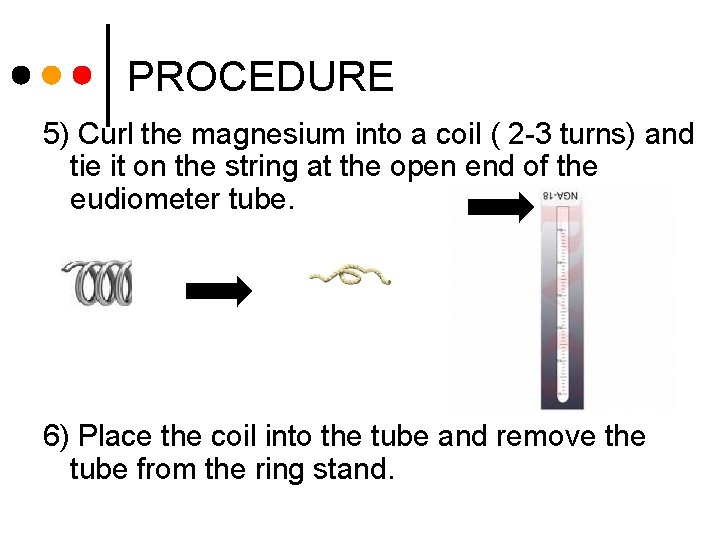
PROCEDURE 5) Curl the magnesium into a coil ( 2 -3 turns) and tie it on the string at the open end of the eudiometer tube. 6) Place the coil into the tube and remove the tube from the ring stand.
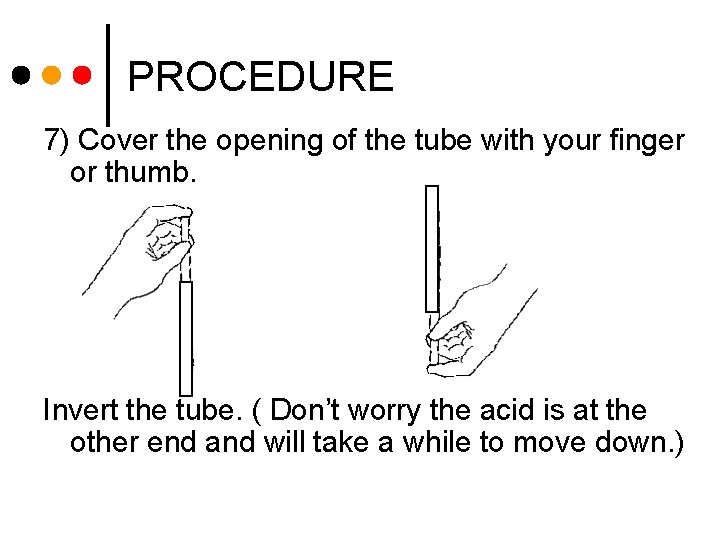
PROCEDURE 7) Cover the opening of the tube with your finger or thumb. Invert the tube. ( Don’t worry the acid is at the other end and will take a while to move down. )
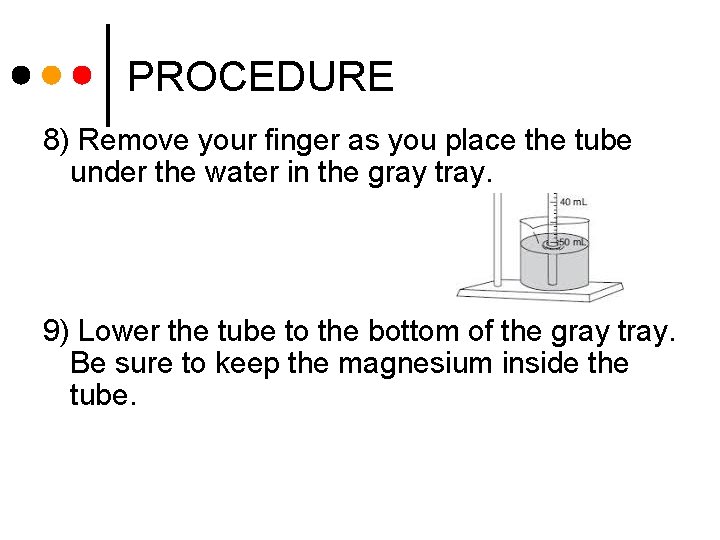
PROCEDURE 8) Remove your finger as you place the tube under the water in the gray tray. 9) Lower the tube to the bottom of the gray tray. Be sure to keep the magnesium inside the tube.

10) Observe the acid as it flows down the tube toward the magnesium. As it reacts hydrogen gas bubbles will be produced.

12) After the hydrogen bubbles have stopped wait 2 minutes before moving the tube. 13) Read the amount of hydrogen gas produced on the side of the eudiometer tube.

14) Record the following data = centimeters of magnesium = temperature of water degrees C = volume of hydrogen gas collected in milliliters =pressure of hydrogen gas = air pressure = inches of mercury

CALCULATIONS IDEAL GAS LAW P X V = n X R X T P = PRESSURE ATMOSPHERES V = VOLUME LITERS n = NUMBER OF MOLES R= GAS CONSTANT T = TEMPERATURE DEGREES KELVIN

CALCULATIONS PRESSURE = is measured in ATMOSPHERES an atmosphere = 760 mm of mercury 1 inch = 2. 54 centimeters ____ in x 2. 54 cm =_______ cm 1 inch Convert the centimeters into mm _______ cm X 10 mm =_____ mm of mercury 1 cm

CALCULATIONS Convert the mm of mercury into atmospheres _____ mm x 1 ATM = _______ ATM 760 mm PRESSURE =

CALCULATIONS VOLUME = is measured in liters Convert the ml of hydrogen gas collected into liters 1 liter = 1000 ml ____ ml x 1 liter = _______ liters 1000 ml VOLUME =

CALCULATIONS TEMPERATURE = is measured in degrees Kelvin = degrees Celsius + 273 _____ C + 273 = degrees Kelvin TEMPERATURE =

CALCULATIONS R = gas constant R= 0. 0821 ATM x Liter / mole x K n = NUMBER OF MOLES OF HYDROGEN MADE

CALCULATIONS SUBSTITUTE ALL VALUES AND SOLVE FOR n Px. V Rx. T = n (number of moles)
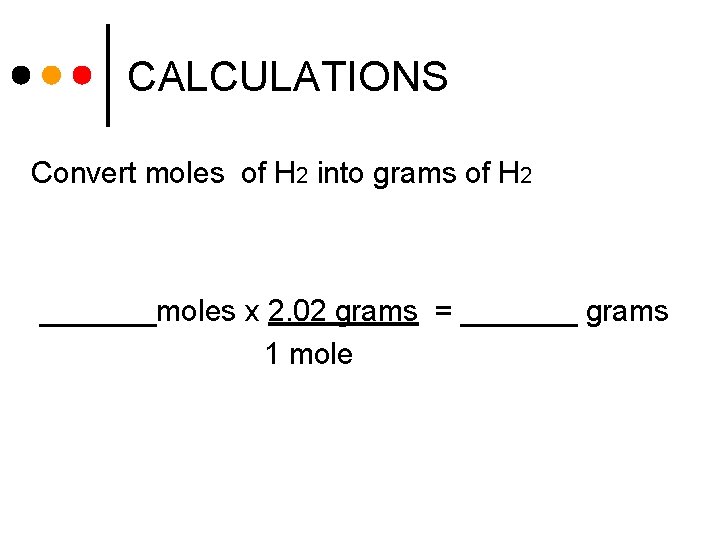
CALCULATIONS Convert moles of H 2 into grams of H 2 _______moles x 2. 02 grams = _______ grams 1 mole

LAB FOLLOW UP In the follow up you will compare the grams of hydrogen you made to the grams of hydrogen you expected to make by using 2. 5 cm of the Mg. 1)Use FLM to convert cm of Mg into grams of Mg 2) Use the 5 step method to predict how much H 2 should be made 3) Determine the % Yield

Convert centimeters of magnesium into grams of magnesium. 100 cm = 1. 57 grams 2. 5 cm X 1. 57 grams = _____grams used 100 cm

Convert centimeters of magnesium into grams of magnesium. 100 cm = 1. 57 grams 2. 5 cm X 1. 57 grams = 0. 0393 grams used 100 cm

CALCULATIONS 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) _____grams X 1. 00 mole =____moles 24. 3 g 3) ratio G: ? = 1 : 1 4) ____: ____ 5)_____ moles X 2. 02 grams =______grams 1. 00 mole expected

CALCULATIONS 0. 0393 g ? ? ? 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) _____grams X 1. 00 mole =____moles 24. 3 g 3) ratio G: ? = 1 : 1 4) ____: ____ 5)_____ moles X 2. 02 grams =______grams 1. 00 mole expected

CALCULATIONS 0. 0393 g ? ? ? 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) 0. 0393 grams X 1. 00 mole =0. 00162 moles 24. 3 g 3) ratio G: ? = 1 : 1 4) ____: ____ 5)_____ moles X 2. 02 grams =______grams 1. 00 mole expected

CALCULATIONS 0. 0393 g ? ? ? 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) 0. 0393 grams X 1. 00 mole =0. 00162 moles 24. 3 g 3) ratio G: ? = 1 : 1 4) ____: ____ 5)_____ moles X 2. 02 grams =______grams 1. 00 mole expected

CALCULATIONS 0. 0393 g ? ? ? 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) 0. 0393 grams X 1. 00 mole =0. 00162 moles 24. 3 g 3) ratio G: ? = 1 : 1 4) 0. 00162 : 0. 00162 5)_____ moles X 2. 02 grams =______grams 1. 00 mole expected

CALCULATIONS 0. 0393 g ? ? ? 1) 2 HCl + 1 Mg --> Mg. Cl 2 + 1 H 2 2) 0. 0393 grams X 1. 00 mole =0. 00162 moles 24. 3 g 3) ratio G: ? = 1 : 1 4) 0. 00162 : 0. 00162 5) 0. 00162 moles X 2. 02 grams =. 00327 grams 1. 00 mole expected

CALCULATIONS DETERMINE THE PERCENT YIELD grams of H 2 formed in the lab = X 100 = % YIELD grams of H 2 expected How could your percentage be over 100%?
- Slides: 30