Magma Rocks Classification Textures MAGMA MAGMA Larutan silikat






















- Slides: 54

Magma, Rocks Classification & Textures

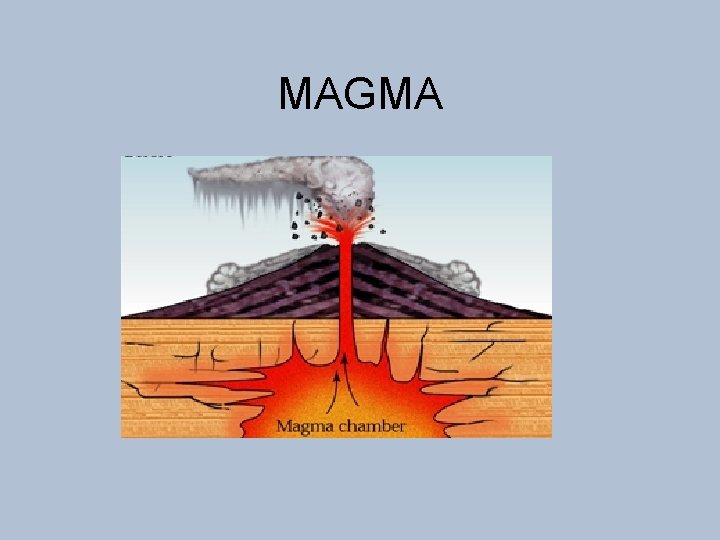
MAGMA

MAGMA • Larutan silikat yang sangat panas • Mengandung oksida, sulfida serta volatiles (CO 2, sulfur, chlorine, fluorin, boron dll) • Temperatur antara 600°C (magma asam) sampai 1250°C (magma basa)

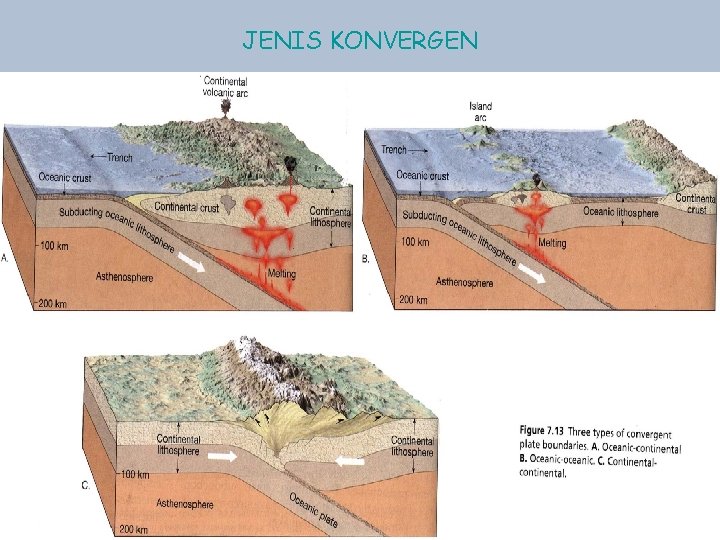
JENIS KONVERGEN


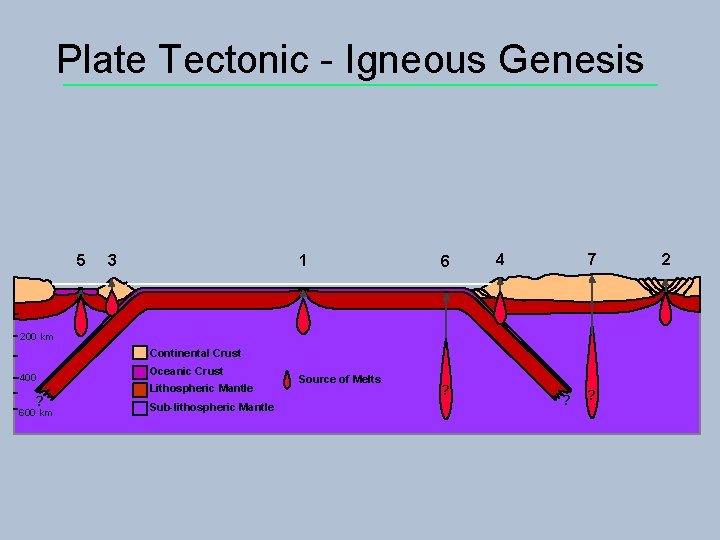
Plate Tectonic - Igneous Genesis 5 3 1 6 7 4 200 km Continental Crust 400 ? 600 km Oceanic Crust Lithospheric Mantle Sub-lithospheric Mantle Source of Melts ? ? ? 2

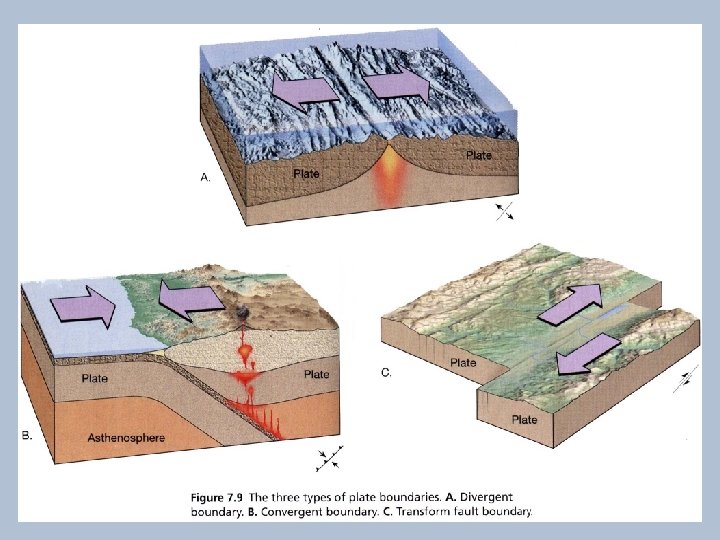
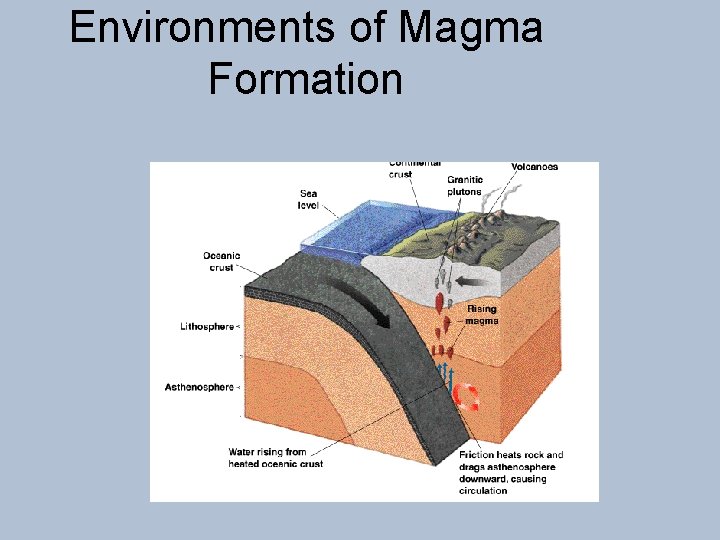
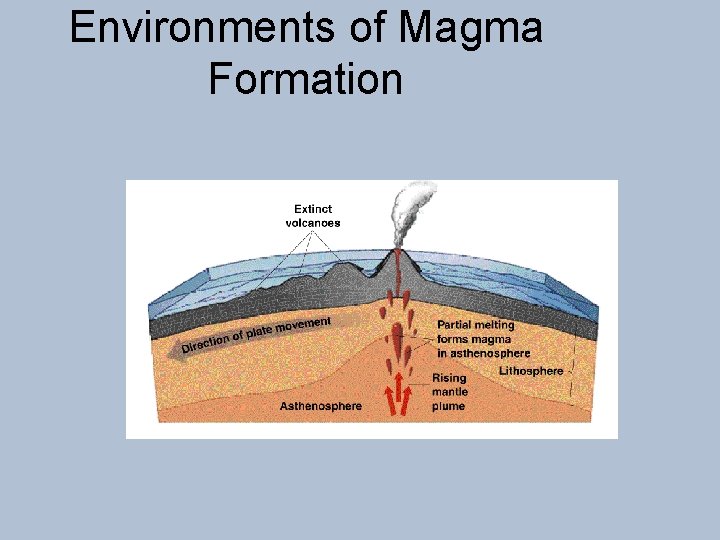
Environments of Magma Formation

Environments of Magma Formation

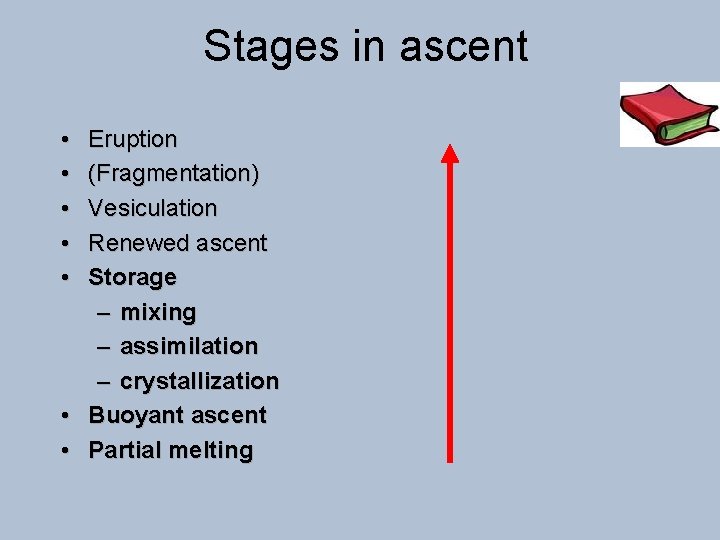
Stages in ascent • • • Eruption (Fragmentation) Vesiculation Renewed ascent Storage – mixing – assimilation – crystallization • Buoyant ascent • Partial melting

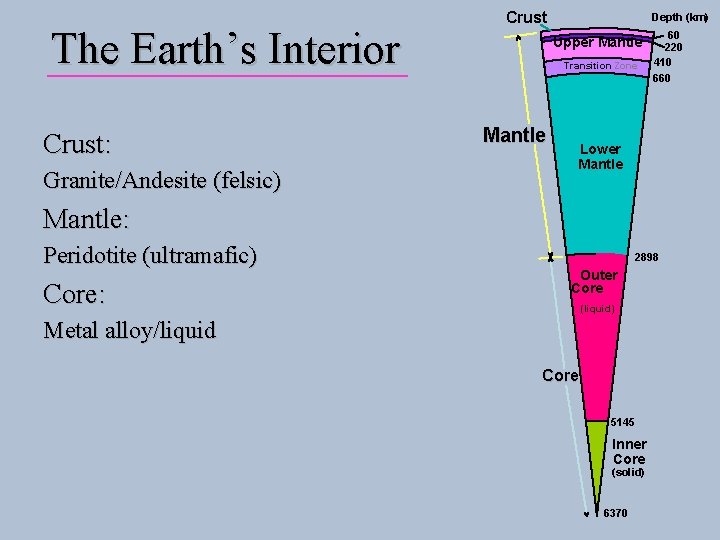
The Earth’s Interior Crust: Granite/Andesite (felsic) Crust Depth (km) Upper Mantle Transition Zone Mantle 60 220 410 660 Lower Mantle: Peridotite (ultramafic) Core: 2898 Outer Core (liquid) Metal alloy/liquid Core 5145 Inner Core (solid) 6370

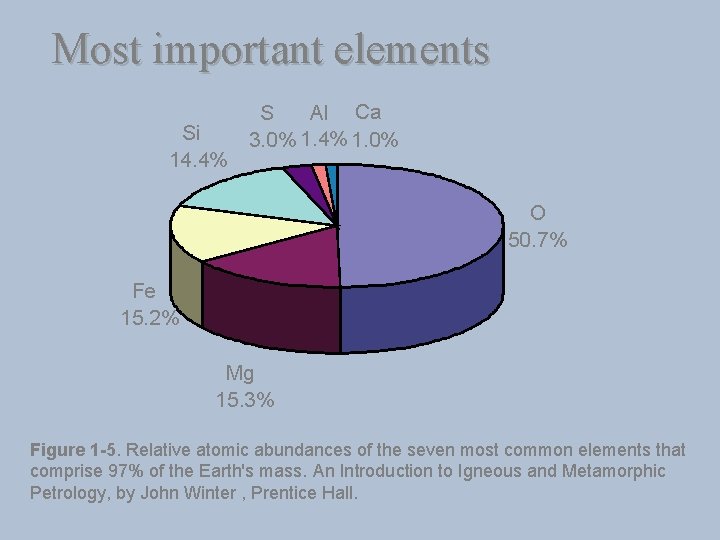
Most important elements Si 14. 4% Al Ca S 3. 0% 1. 4% 1. 0% O 50. 7% Fe 15. 2% Mg 15. 3% Figure 1 -5. Relative atomic abundances of the seven most common elements that comprise 97% of the Earth's mass. An Introduction to Igneous and Metamorphic Petrology, by John Winter , Prentice Hall.

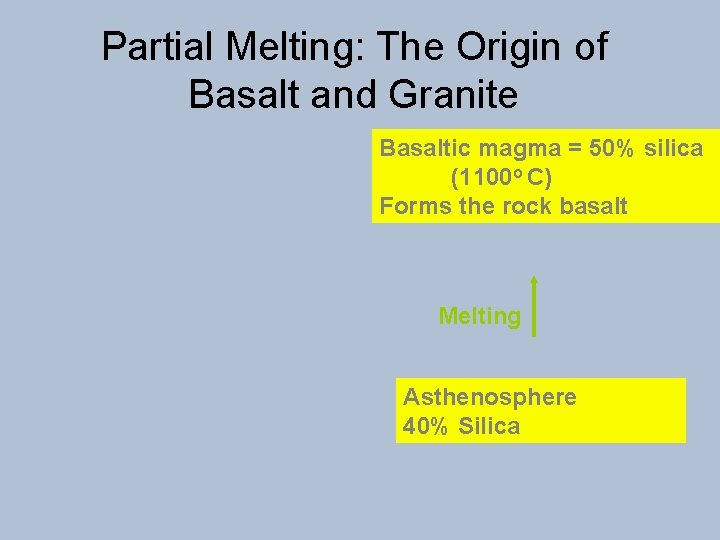
Partial Melting: The Origin of Basalt and Granite Basaltic magma = 50% silica (1100 o C) Forms the rock basalt Melting Asthenosphere 40% Silica

Partial Melting: The Origin of Basalt and Granite Granitic magma ~ 70% silica (700 -900 o C) Forms granite (a mixture of quartz and feldspar) Melting Continental Crust (Mainly low melting point minerals such as quartz, feldspar, mica)

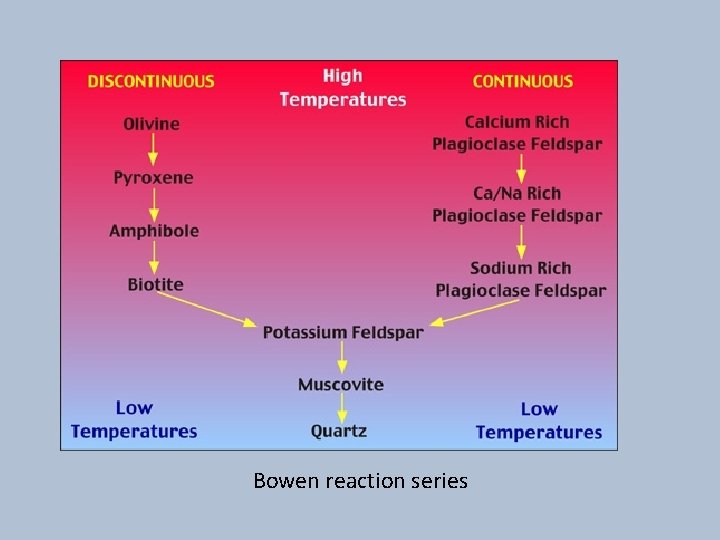
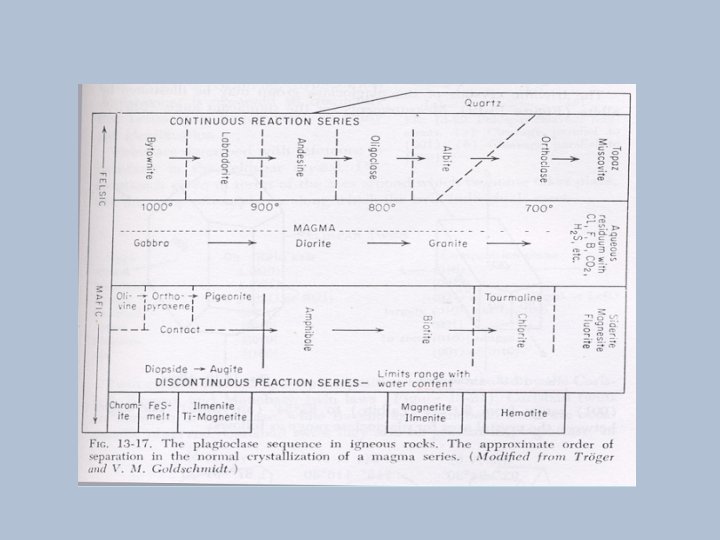
Urutan pembekuan magma • Pada pembekuan magma, pada awalnya mineral yang terbentuk adalah yang anhydrous (tidak mengandung air) tidak mengandung gugus OH, disebut mineral pyrogenetik. • Cairan selanjutnya akan lebih banyak mengandung komponen gas dan terbentuk mineral-mineral yang mengandung gugusan hydroksil (OH), disebut mineral hydratogenetik.

Diferensiasi Magma • Proses diferensiasi meliputi semua kegiatan yang mengakibatkan suatu jenis magma induk yang semula relatif homogen terpecah-pecah menjadi beberapa bagian atau fraksi dengan komposisi yang berbeda-beda. Hal ini disebabkan karena migrasi ion atau molekul dalam larutan magma karena adanya perubahan temperatur dan tekanan. Yang pada akhirnya akan membentuk berbagai jenis batuan beku dengan komposisi yang berbeda-beda pula.

Bowen reaction series


DIAGRAM FASE • Fase : padat, cair, gas • Diagram fase : menggambarkan kondisi magma pada kondisi P & T tertentu • Parameter penting dalam sistem magma : fase, komponen, variabel intensif

DIAGRAM f. ASE • fase : padat, cair • komponen : komponen terkecil yang diperlukan utk pembentukan fase-fase • dalam sistem (OH, H 2 O, Mg. O, Na. Al. Si 3 O 8, dll) • • variabel intensif : temperatur dan tekanan, jumlah komponen

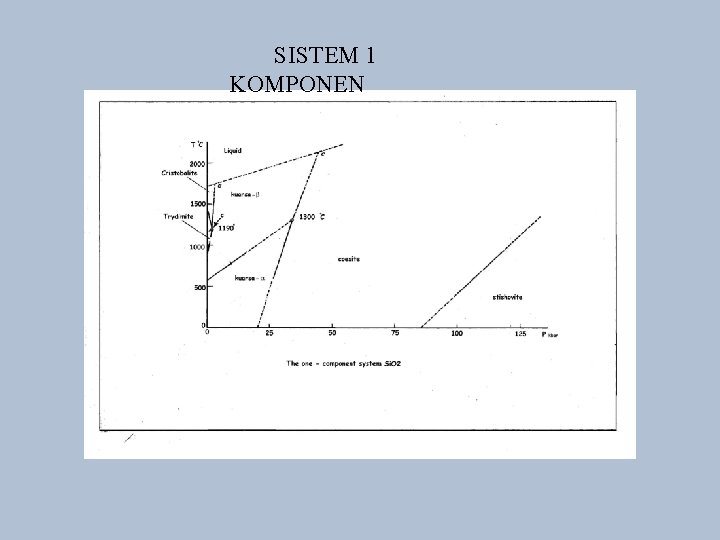
DIAGRAM FASE • • • Rumus fase : F = C – P + 2 F : degree of freedom : jumlah kondisi minimum C : jumlah komponen; P : jumlah fase contoh utk air – es ------ C = 1 (H 2 O) ; P = 2 (es dan air) F = C – P + 2 ---- F = 1 – 2 + 2 = 1 (unary system)

SISTEM 1 KOMPONEN

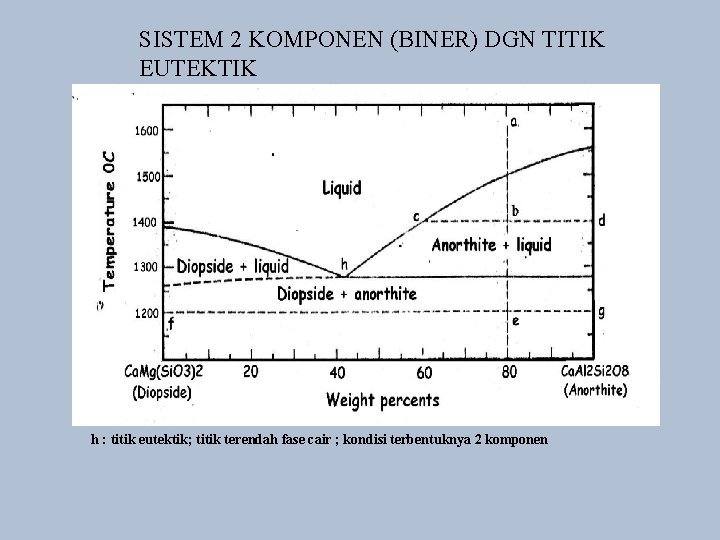
SISTEM 2 KOMPONEN (BINER) DGN TITIK EUTEKTIK h : titik eutektik; titik terendah fase cair ; kondisi terbentuknya 2 komponen

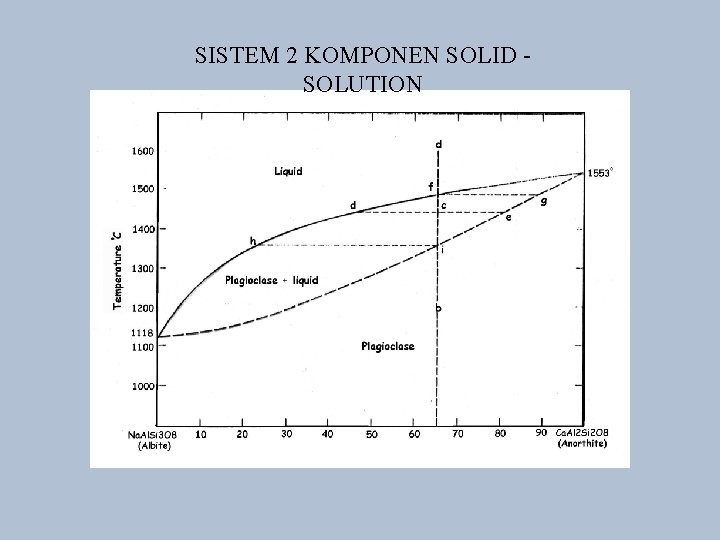
SISTEM 2 KOMPONEN SOLID - SOLUTION

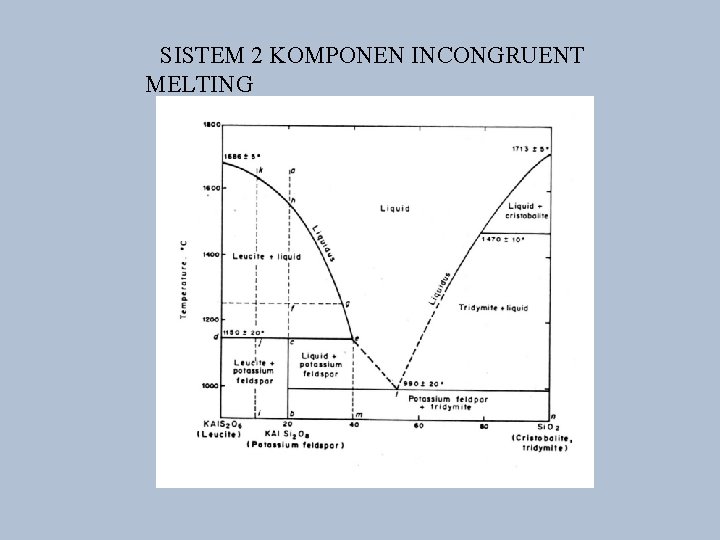
SISTEM 2 KOMPONEN INCONGRUENT MELTING

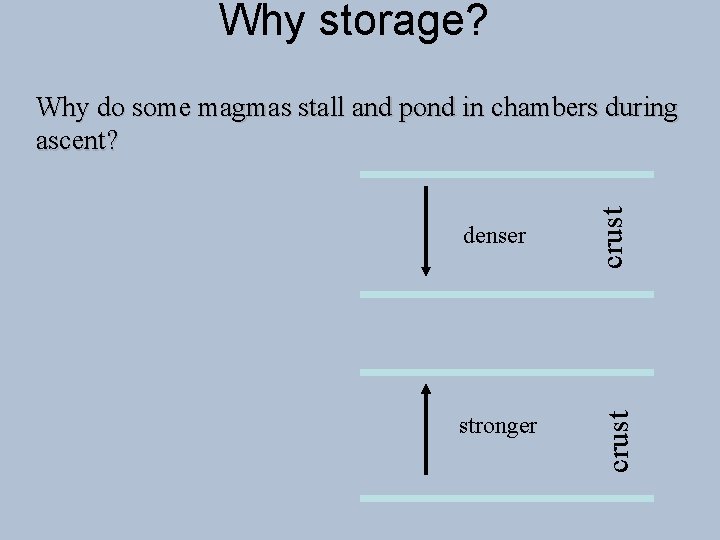
Why storage? stronger crust denser crust Why do some magmas stall and pond in chambers during ascent?

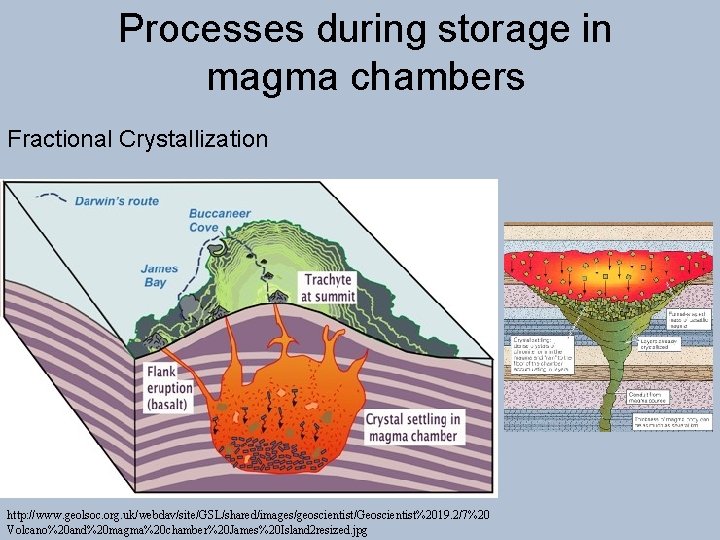
Processes during storage in magma chambers Fractional Crystallization http: //www. geolsoc. org. uk/webdav/site/GSL/shared/images/geoscientist/Geoscientist%2019. 2/7%20 Volcano%20 and%20 magma%20 chamber%20 James%20 Island 2 resized. jpg

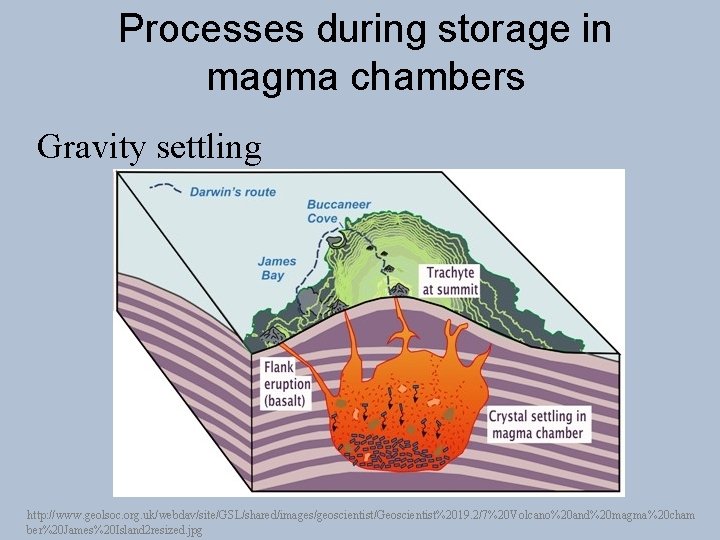
Processes during storage in magma chambers Gravity settling http: //www. geolsoc. org. uk/webdav/site/GSL/shared/images/geoscientist/Geoscientist%2019. 2/7%20 Volcano%20 and%20 magma%20 cham ber%20 James%20 Island 2 resized. jpg

Gravity settling and cumulates http: //www. geol. lsu. edu/henry/Geology 3041/lectures/12 Layered. Mafic/Fig 12 -15. jpg

Buoyancy, sinking: Stoke’s Law 2 gr (r s - r l ) V= 9 h 2 V = the settling velocity (cm/sec) g = the acceleration due to gravity (980 cm/sec 2) r = the radius of a spherical particle (cm) rs = the density of the solid spherical particle (g/cm 3) rl = the density of the liquid (g/cm 3) h = the viscosity of the liquid (1 c/cm sec = 1 poise)

Sinking olivine in basalt Olivine in basalt F Olivine (rs = 3. 3 g/cm 3, r = 0. 1 cm) F Basaltic liquid (rl = 2. 65 g/cm 3, h = 1000 poise) F V = 2· 980· 0. 12 (3. 3 -2. 65)/9· 1000 = 0. 0013 cm/sec that’s ~1 m per day

Sinking x’tal in rhyolite Rhyolitic melt F F F h = 107 poise and rl = 2. 3 g/cm 3 hornblende crystal (rs = 3. 2 g/cm 3, r = 0. 1 cm) -7 s V = 2 x 10 cm/sec, or 6 cm/year feldspars (rl = 2. 7 g/cm 3) s V = 2 cm/year 4 s = 200 m in the 10 years that a stock might cool s If 0. 5 cm in radius (1 cm diameter) settle at 0. 65 meters/year, or 6. 5 km in 104 year cooling of stock

IGNEOUS ROCKS CLASSIFICATION

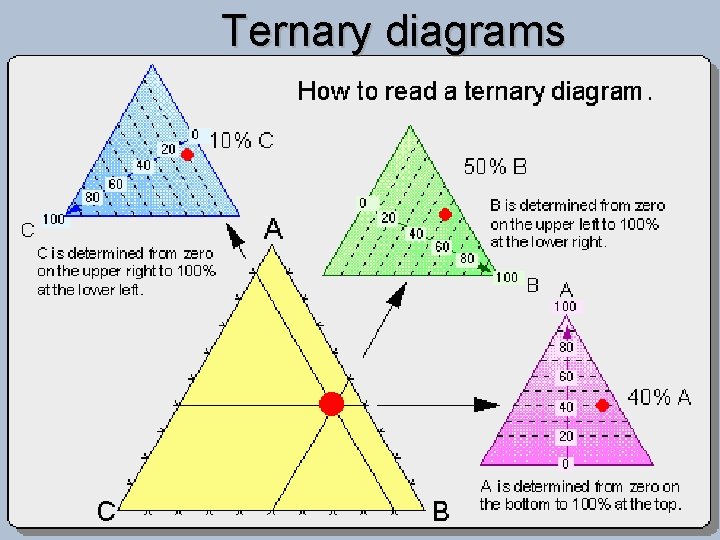
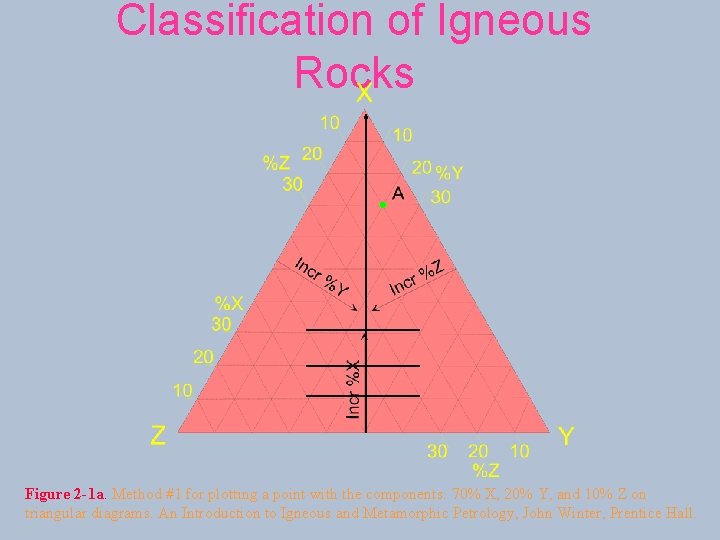
Ternary diagrams

Classification of Igneous Rocks Figure 2 -1 a. Method #1 for plotting a point with the components: 70% X, 20% Y, and 10% Z on triangular diagrams. An Introduction to Igneous and Metamorphic Petrology, John Winter, Prentice Hall.

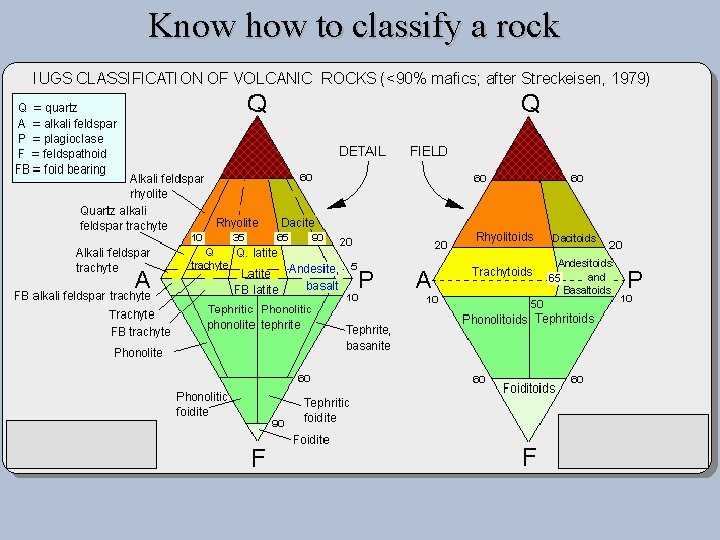
Know how to classify a rock



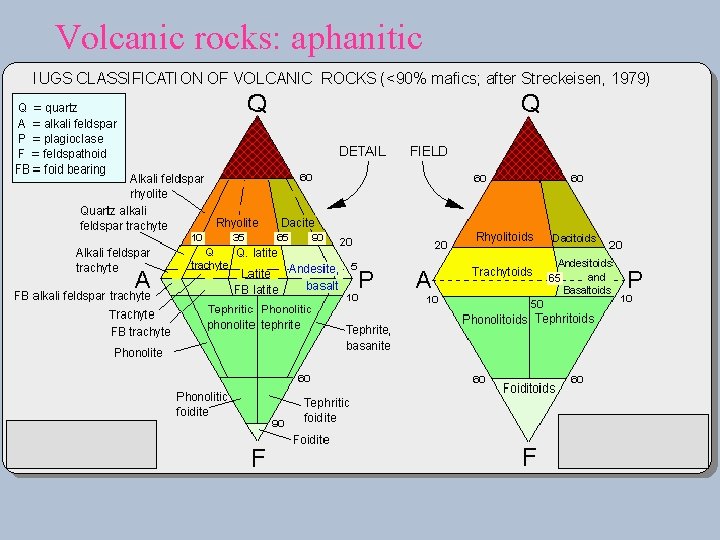
Volcanic rocks: aphanitic

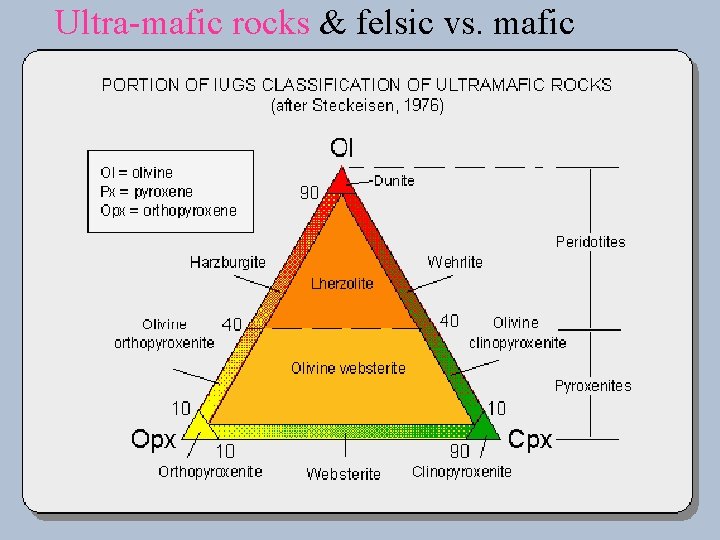
Ultra-mafic rocks & felsic vs. mafic

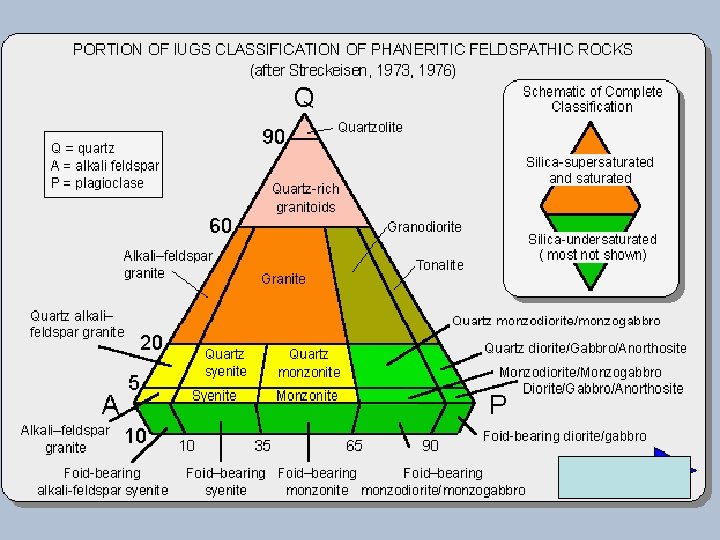
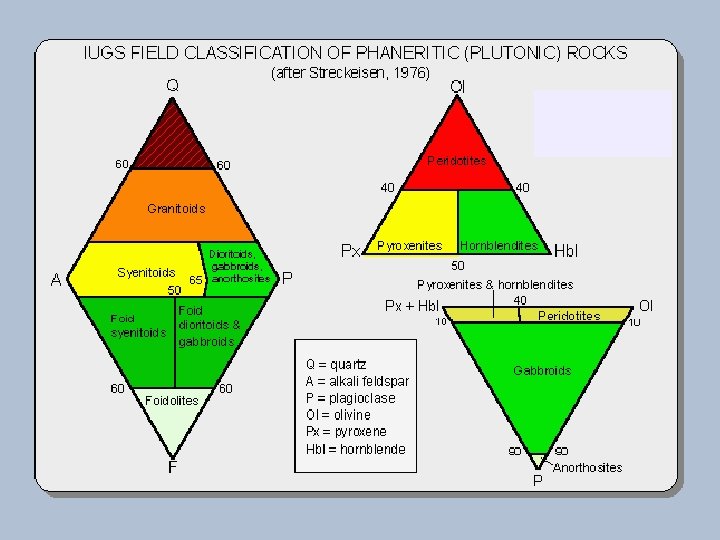
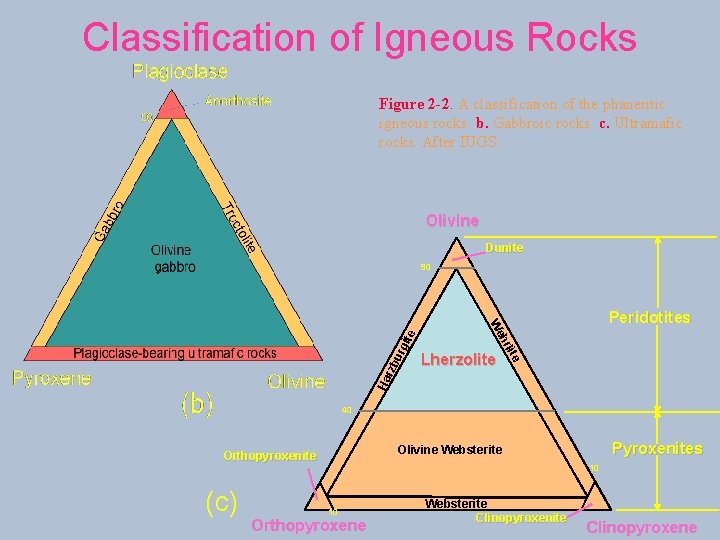
Classification of Igneous Rocks Figure 2 -2. A classification of the phaneritic igneous rocks. b. Gabbroic rocks. c. Ultramafic rocks. After IUGS. Olivine Dunite urg Ha Lherzolite hr rzb Peridotites We ite 90 40 (c) Pyroxenites Olivine Websterite Orthopyroxenite 10 10 Orthopyroxene Websterite Clinopyroxene

Classification of Igneous Rocks Figure 2 -4. A chemical classification of volcanics based on total alkalis vs. silica. After Le Bas et al. (1986) J. Petrol. , 27, 745 -750. Oxford University Press.

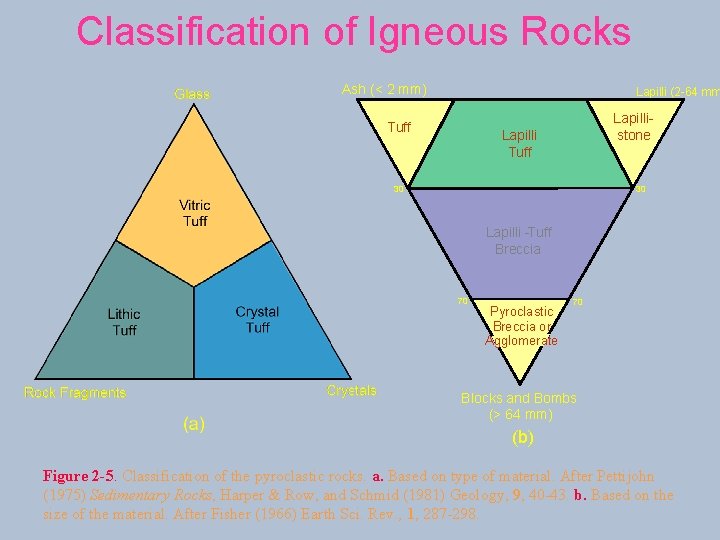
Classification of Igneous Rocks Ash (< 2 mm) Lapilli (2 -64 mm Tuff Lapillistone Lapilli Tuff 30 30 Lapilli -Tuff Breccia 70 Pyroclastic Breccia or Agglomerate 70 Blocks and Bombs (> 64 mm) (b) Figure 2 -5. Classification of the pyroclastic rocks. a. Based on type of material. After Pettijohn (1975) Sedimentary Rocks, Harper & Row, and Schmid (1981) Geology, 9, 40 -43. b. Based on the size of the material. After Fisher (1966) Earth Sci. Rev. , 1, 287 -298.

TEXTURES IN IGNEOUS ROCKS

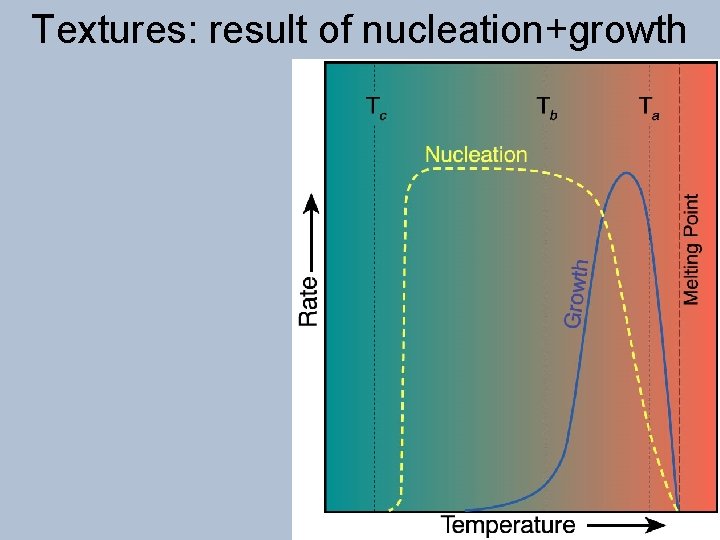
Textures: result of nucleation+growth

Grain size

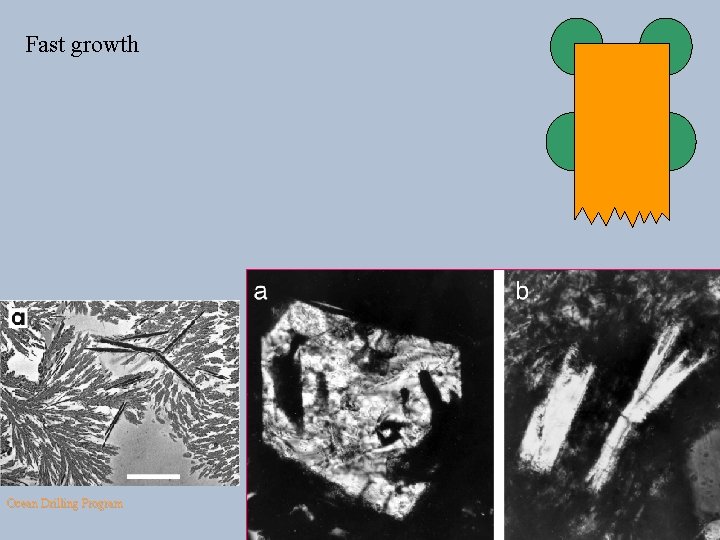
Fast growth a Ocean Drilling Program

Crystal zoning

Crystal shape

Growth order

Quartz - feldspar intergrowth

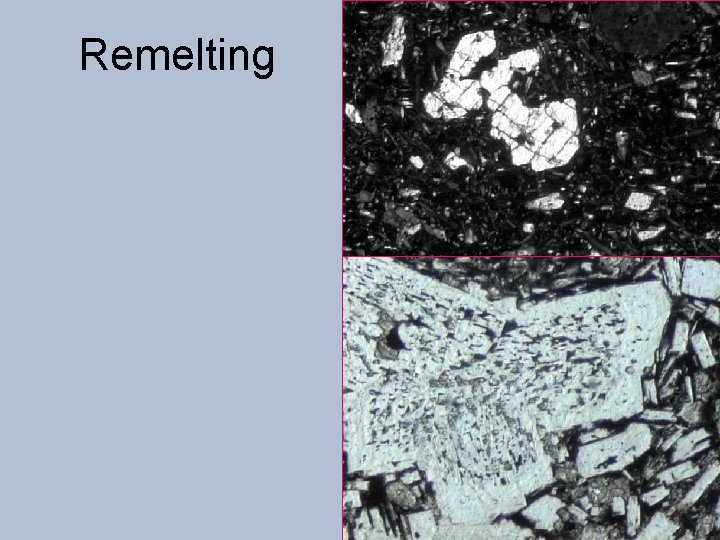
Remelting

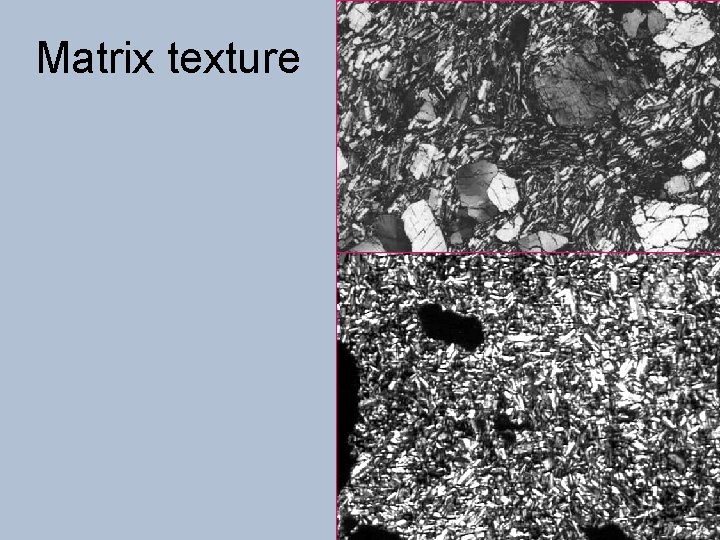
Matrix texture

Twinning

Replacements North Carolina State University Smith College