Magical Chiral Spirobiindane Skeletons Supervisor Yong Huang Reporter

Magical Chiral Spirobiindane Skeletons Supervisor: Yong Huang Reporter: Qian Wang Date: 2015 -10 -26

Contents 1. The origin of spirobiindane skeletons 2. The development and application 3. Comparison of biaryls and spiranes 4. Summary and Outlook 5. Acknowledgement 2
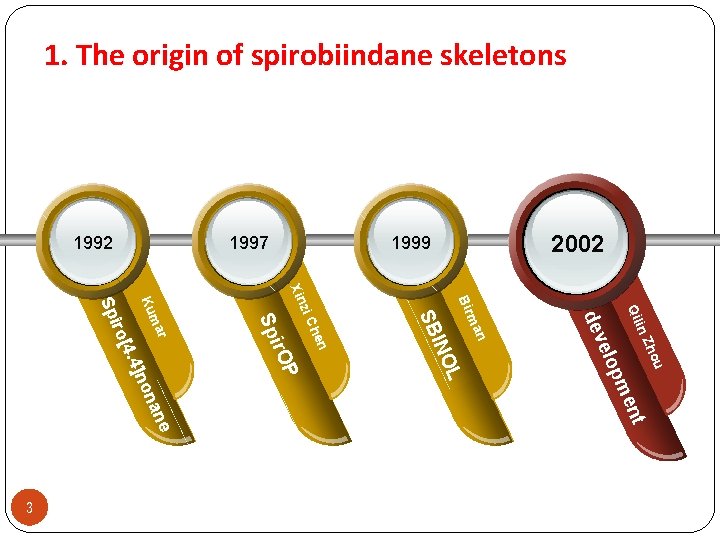
1. The origin of spirobiindane skeletons ho in Z Qil u L on ane ent pm elo dev n ma Bir P INO SB hen zi C Xin ir. O Sp r ma Ku . 4]n o[4 ir Sp 3 2002 1999 1997 1992
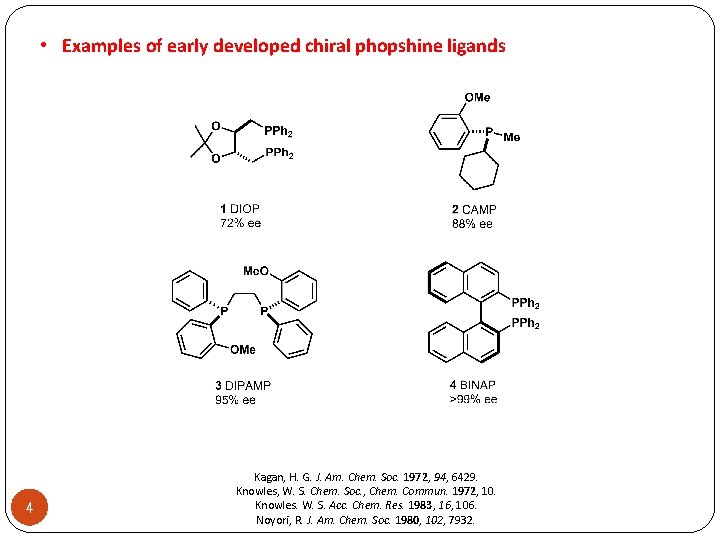
• Examples of early developed chiral phopshine ligands 4 Kagan, H. G. J. Am. Chem. Soc. 1972, 94, 6429. Knowles, W. S. Chem. Soc. , Chem. Commun. 1972, 10. Knowles. W. S. Acc. Chem. Res. 1983, 16, 106. Noyori, R. J. Am. Chem. Soc. 1980, 102, 7932.

The Nobel Prize in Chemistry 2001 W. S. Knowles R. Noyori “chirally catalysed Hydrogenation reactions” 5 K. B. Sharpless “chirally catalysed oxidation reactions”
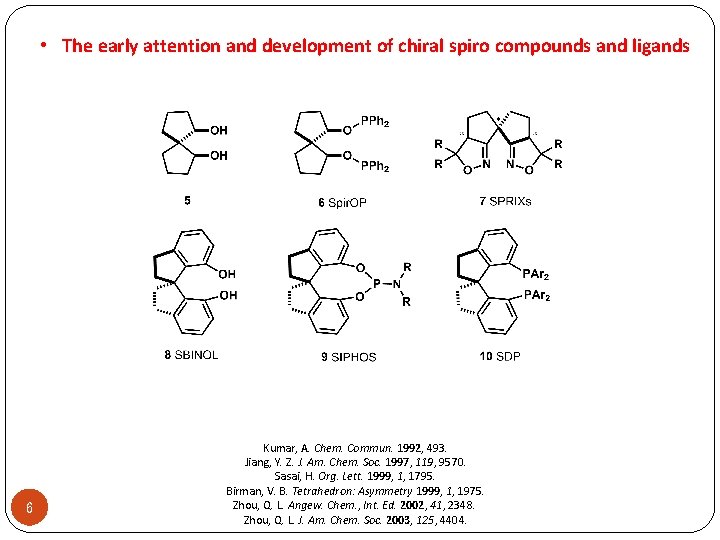
• The early attention and development of chiral spiro compounds and ligands 6 Kumar, A. Chem. Commun. 1992, 493. Jiang, Y. Z. J. Am. Chem. Soc. 1997, 119, 9570. Sasai, H. Org. Lett. 1999, 1, 1795. Birman, V. B. Tetrahedron: Asymmetry 1999, 1, 1975. Zhou, Q. L. Angew. Chem. , Int. Ed. 2002, 41, 2348. Zhou, Q. L. J. Am. Chem. Soc. 2003, 125, 4404.
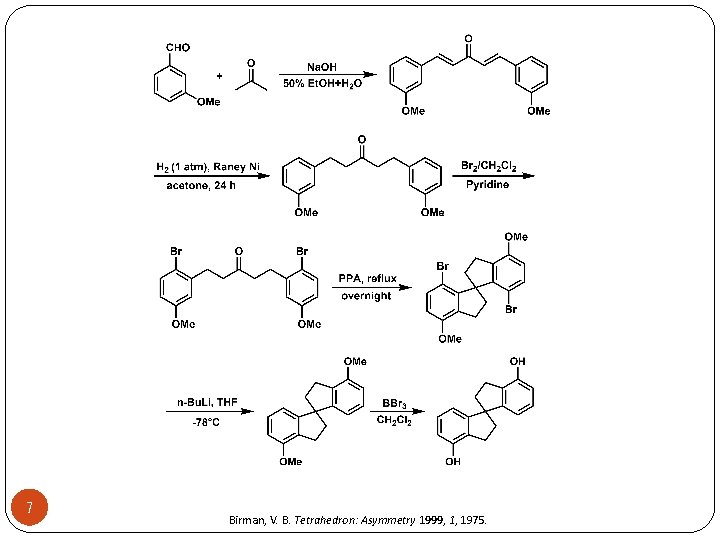
7 Birman, V. B. Tetrahedron: Asymmetry 1999, 1, 1975.
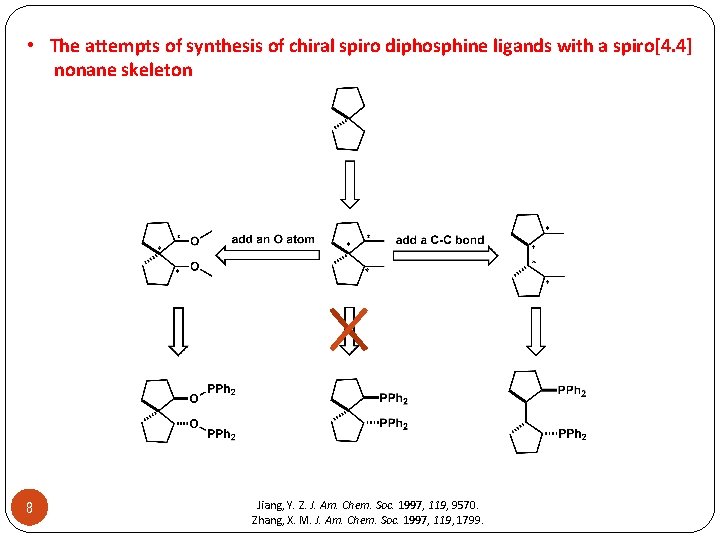
• The attempts of synthesis of chiral spiro diphosphine ligands with a spiro[4. 4] nonane skeleton 8 Jiang, Y. Z. J. Am. Chem. Soc. 1997, 119, 9570. Zhang, X. M. J. Am. Chem. Soc. 1997, 119, 1799.
![• The spirobiindane and spiro[4. 4]nonadiene skeletons and their chiral spiro ligands 9 • The spirobiindane and spiro[4. 4]nonadiene skeletons and their chiral spiro ligands 9](http://slidetodoc.com/presentation_image_h/1510b364fcbc3ecaa93a084c275bd95c/image-9.jpg)
• The spirobiindane and spiro[4. 4]nonadiene skeletons and their chiral spiro ligands 9 Zhou, Q. L. Chem. Commun. 2002, 480. Ding, K. L. Angew. Chem. , Int. Ed. 2009, 48, 5345.

2. The development and application 10

Chiral Spirobiindane 1. As metal ligands Ø Monophosphorous ligands Ø Diphosphine ligands Ø Phosphine-nitrogen ligands Ø Aminophosphine ligands Ø Bidentate nitrogen ligands Ø Phosphine-ene ligands 2. As organic catalysts 11
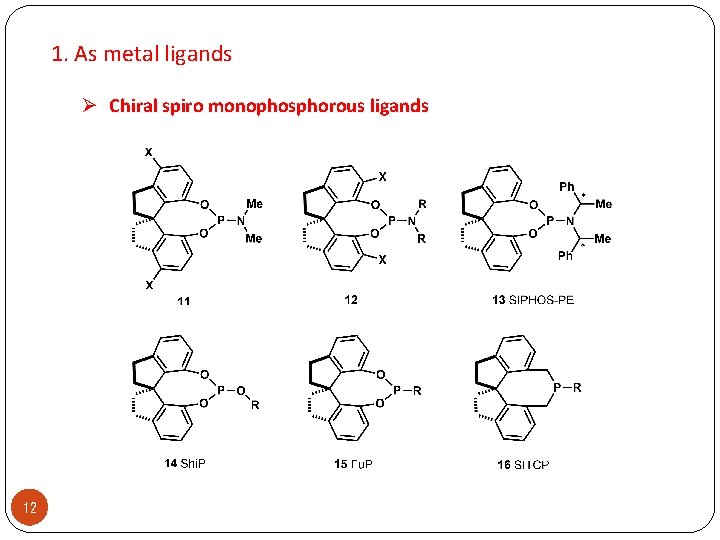
1. As metal ligands Ø Chiral spiro monophosphorous ligands 12
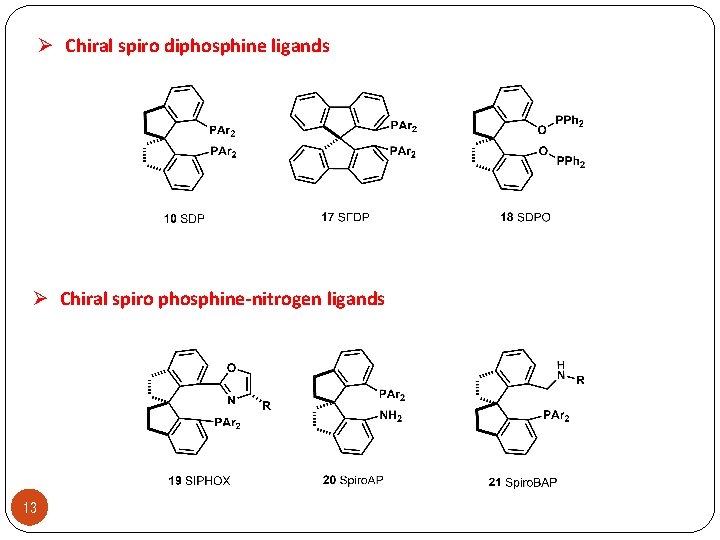
Ø Chiral spiro diphosphine ligands Ø Chiral spiro phosphine-nitrogen ligands 13
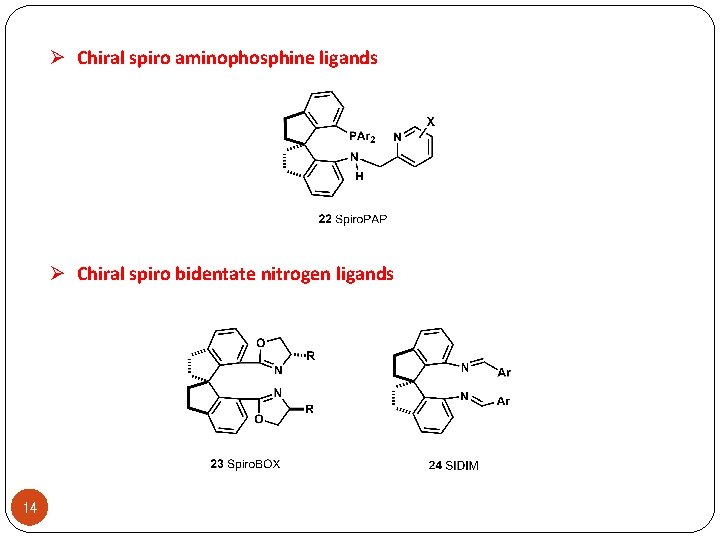
Ø Chiral spiro aminophosphine ligands Ø Chiral spiro bidentate nitrogen ligands 14
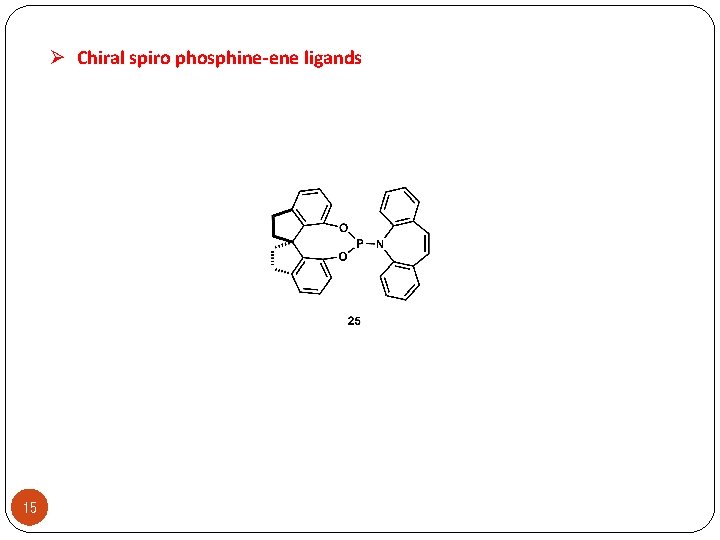
Ø Chiral spiro phosphine-ene ligands 15
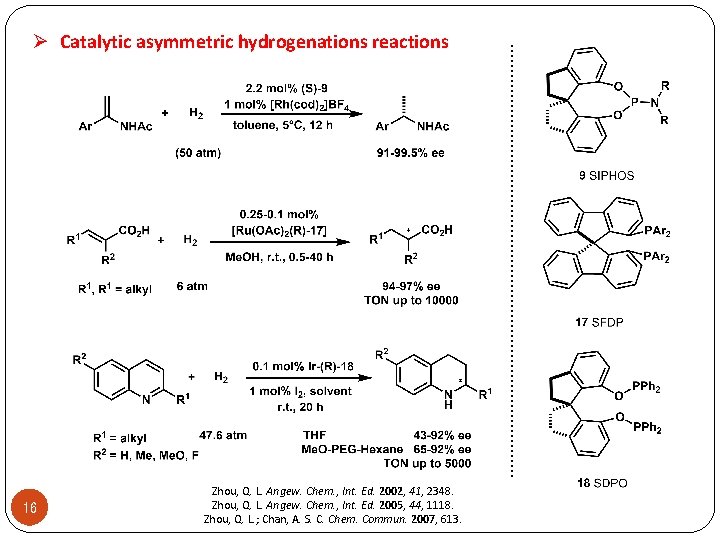
16 Zhou, Q. L. Angew. Chem. , Int. Ed. 2002, 41, 2348. Zhou, Q. L. Angew. Chem. , Int. Ed. 2005, 44, 1118. Zhou, Q. L. ; Chan, A. S. C. Chem. Commun. 2007, 613. . . . . . Ø Catalytic asymmetric hydrogenations reactions
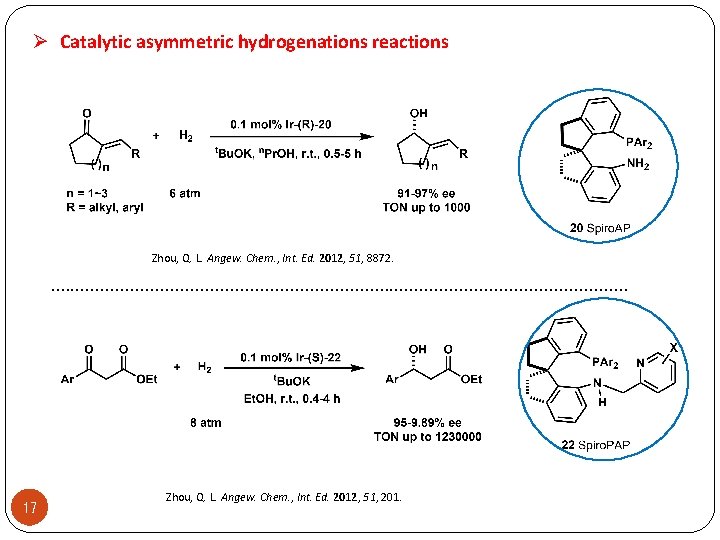
Ø Catalytic asymmetric hydrogenations reactions Zhou, Q. L. Angew. Chem. , Int. Ed. 2012, 51, 8872. . . . . . . . 17 Zhou, Q. L. Angew. Chem. , Int. Ed. 2012, 51, 201.
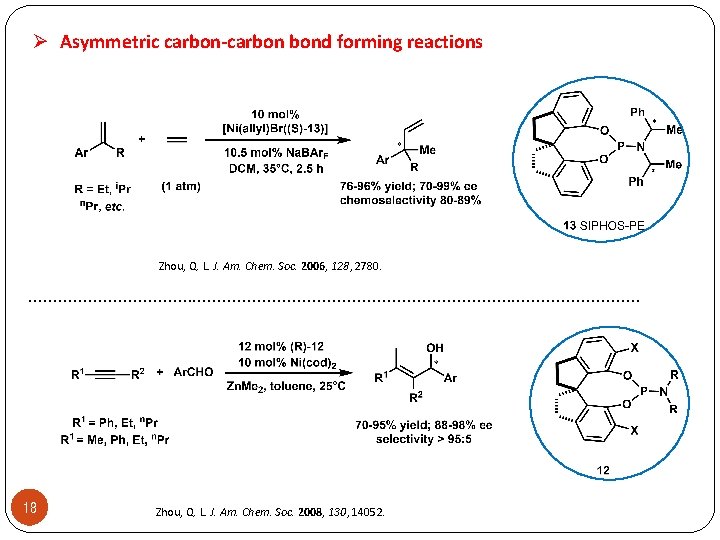
Ø Asymmetric carbon-carbon bond forming reactions Zhou, Q. L. J. Am. Chem. Soc. 2006, 128, 2780. . . . . . . . 18 Zhou, Q. L. J. Am. Chem. Soc. 2008, 130, 14052.
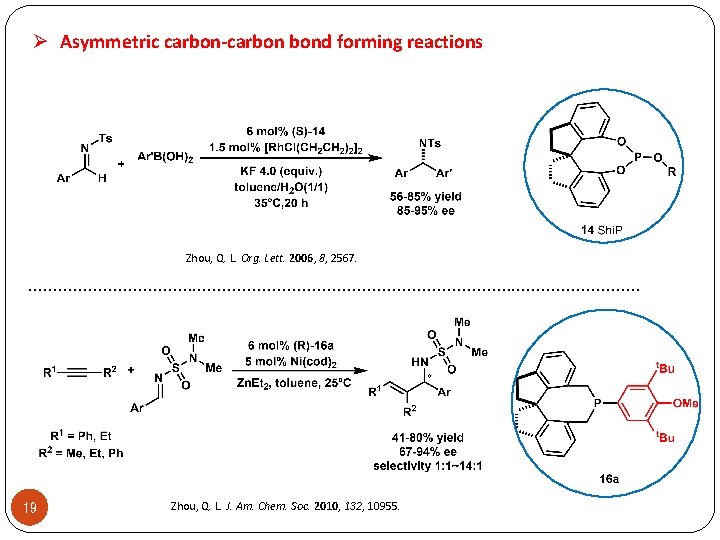
Ø Asymmetric carbon-carbon bond forming reactions Zhou, Q. L. Org. Lett. 2006, 8, 2567. . . . . . . . 19 Zhou, Q. L. J. Am. Chem. Soc. 2010, 132, 10955.
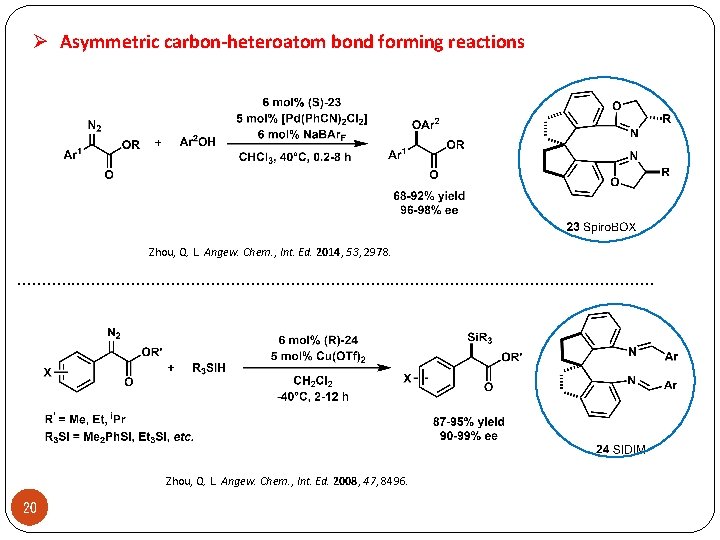
Ø Asymmetric carbon-heteroatom bond forming reactions Zhou, Q. L. Angew. Chem. , Int. Ed. 2014, 53, 2978. . . . . . . . Zhou, Q. L. Angew. Chem. , Int. Ed. 2008, 47, 8496. 20
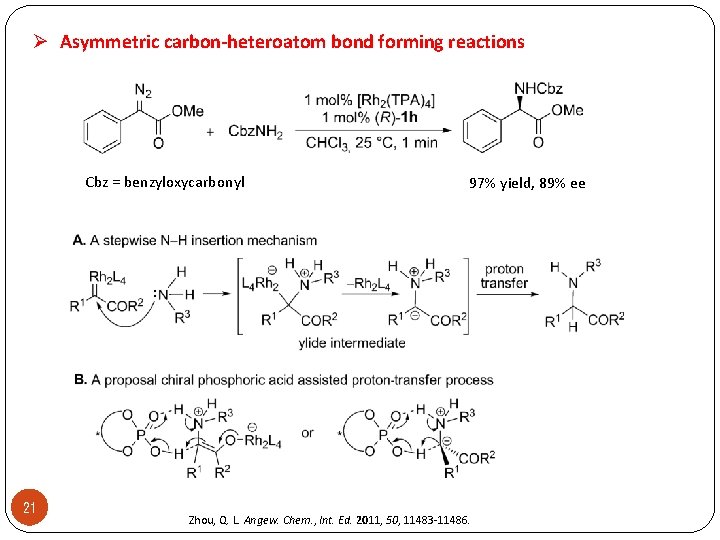
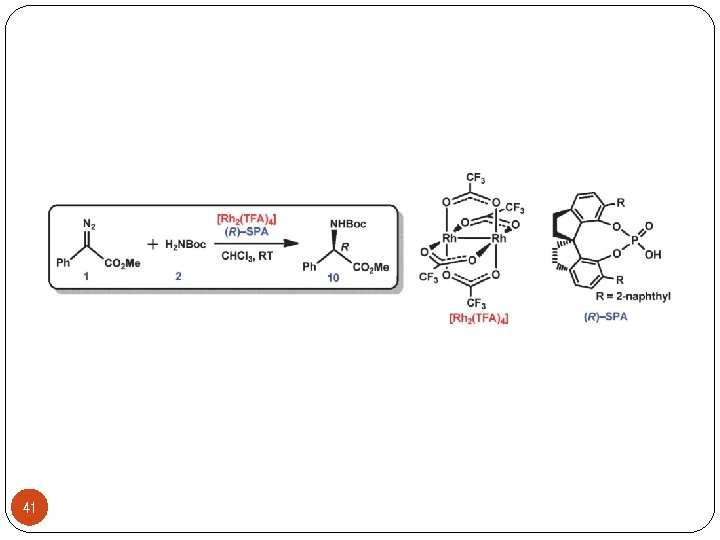
Ø Asymmetric carbon-heteroatom bond forming reactions Cbz = benzyloxycarbonyl 21 97% yield, 89% ee Zhou, Q. L. Angew. Chem. , Int. Ed. 2011, 50, 11483 -11486.
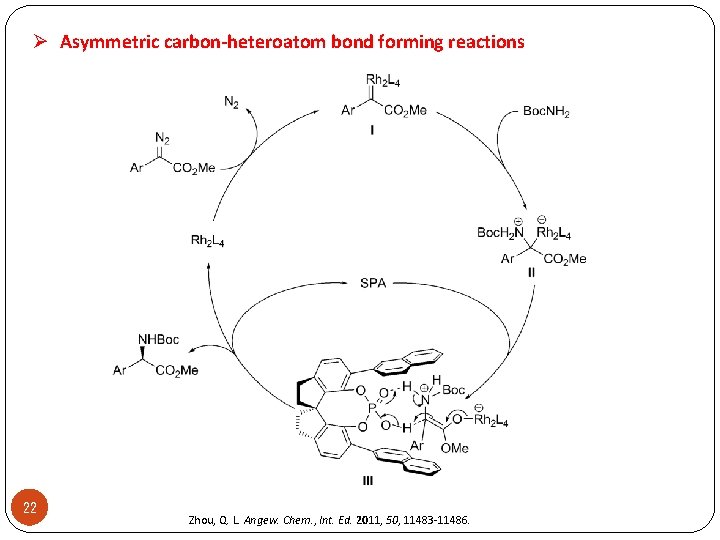
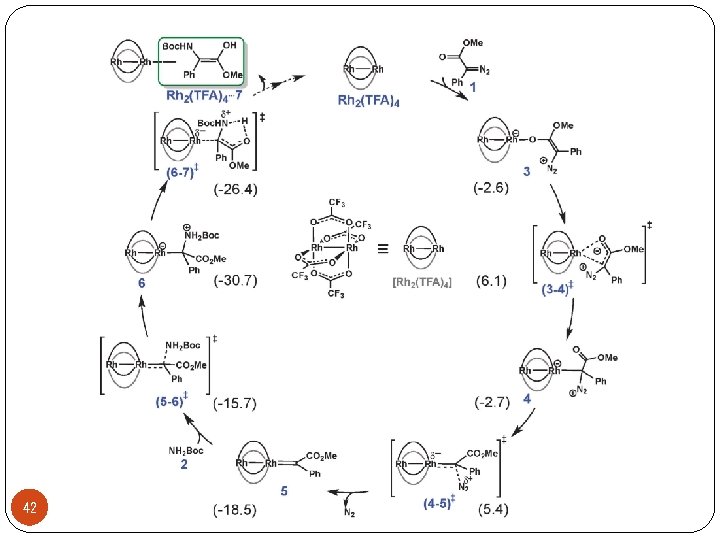
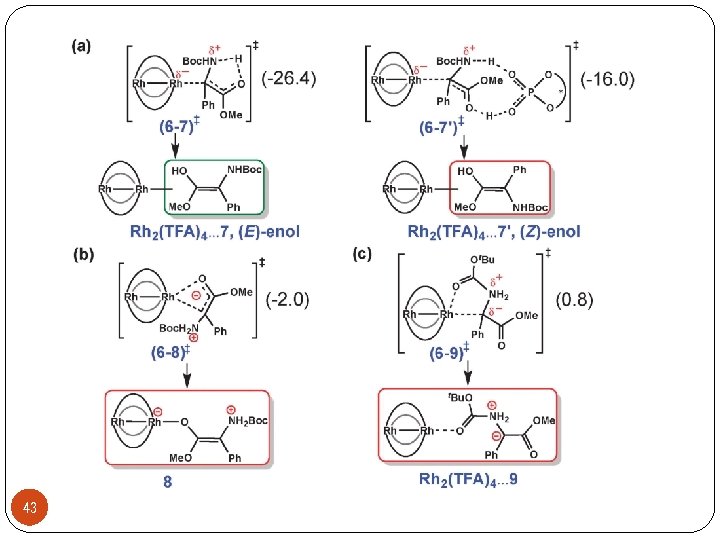
Ø Asymmetric carbon-heteroatom bond forming reactions 22 Zhou, Q. L. Angew. Chem. , Int. Ed. 2011, 50, 11483 -11486.
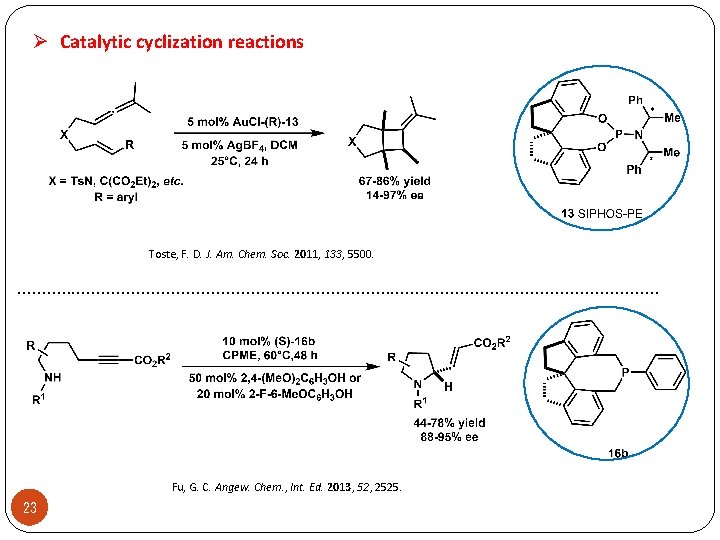
Ø Catalytic cyclization reactions Toste, F. D. J. Am. Chem. Soc. 2011, 133, 5500. . . . . . . . Fu, G. C. Angew. Chem. , Int. Ed. 2013, 52, 2525. 23
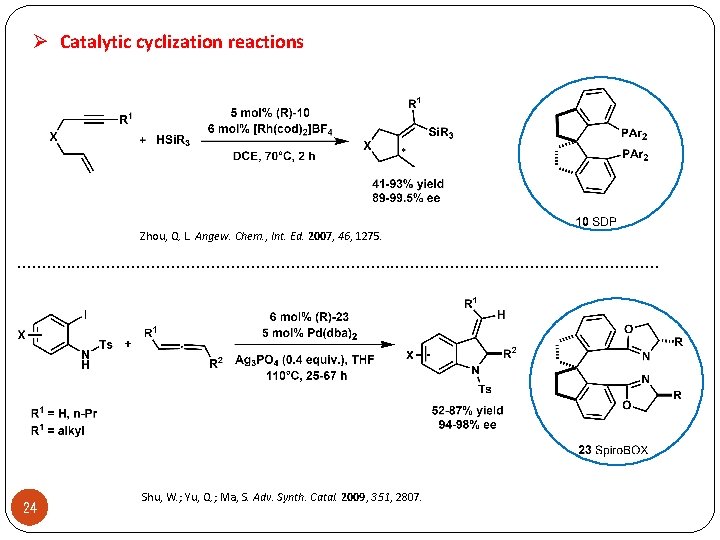
Ø Catalytic cyclization reactions Zhou, Q. L. Angew. Chem. , Int. Ed. 2007, 46, 1275. . . . . . . . 24 Shu, W. ; Yu, Q. ; Ma, S. Adv. Synth. Catal. 2009, 351, 2807.
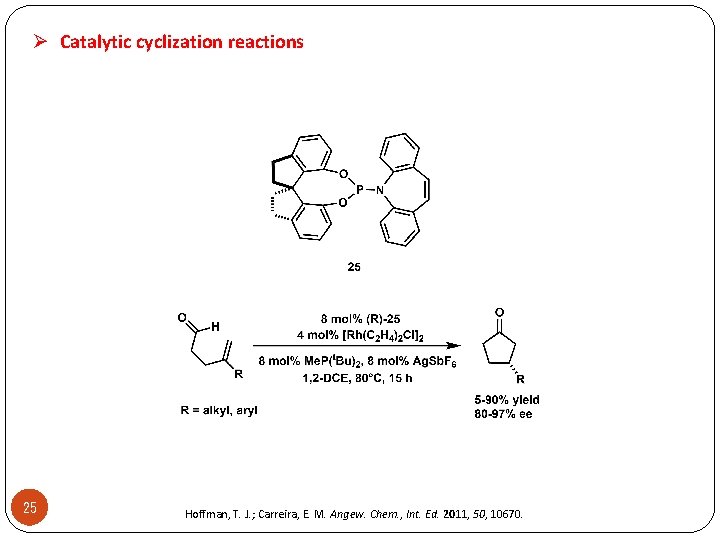
Ø Catalytic cyclization reactions 25 Hoffman, T. J. ; Carreira, E. M. Angew. Chem. , Int. Ed. 2011, 50, 10670.
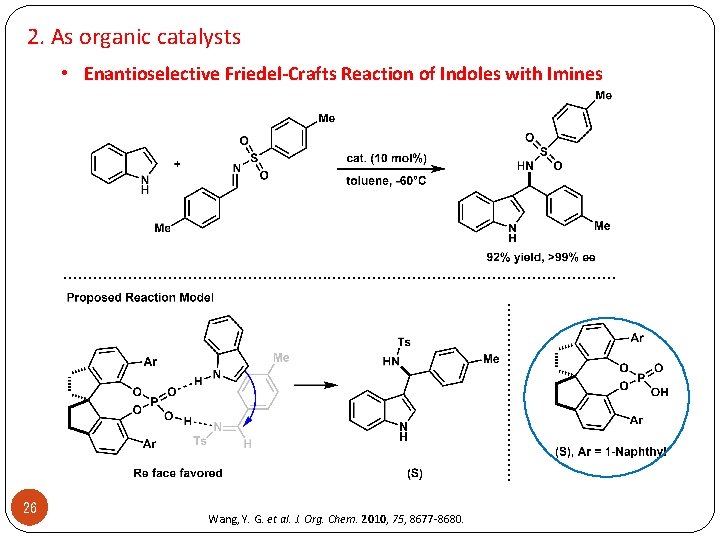
2. As organic catalysts • Enantioselective Friedel-Crafts Reaction of Indoles with Imines . . . . . . . . . 26 Wang, Y. G. et al. J. Org. Chem. 2010, 75, 8677 -8680.
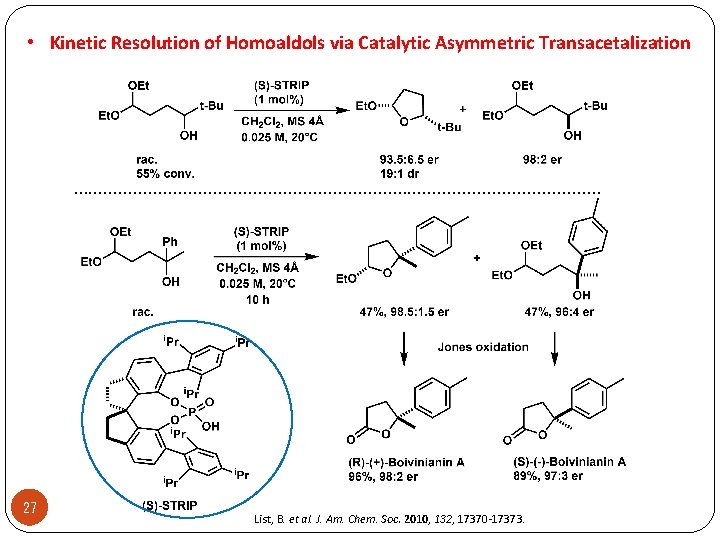
• Kinetic Resolution of Homoaldols via Catalytic Asymmetric Transacetalization . . . . . . 27 List, B. et al. J. Am. Chem. Soc. 2010, 132, 17370 -17373.
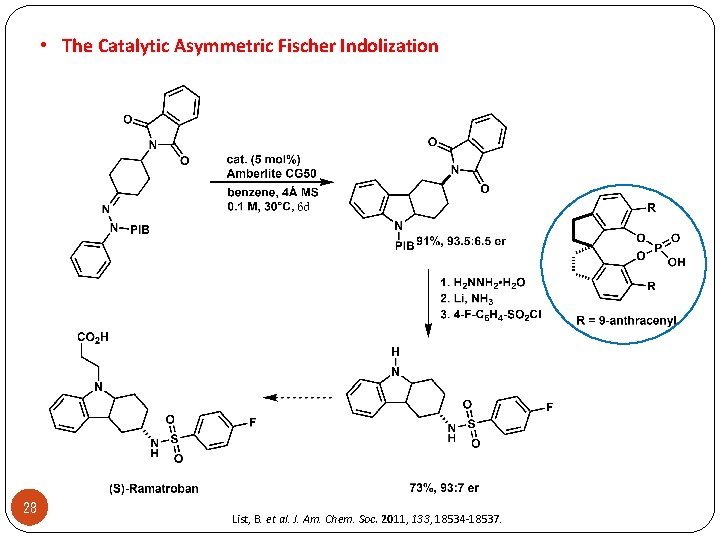
• The Catalytic Asymmetric Fischer Indolization 28 List, B. et al. J. Am. Chem. Soc. 2011, 133, 18534 -18537.
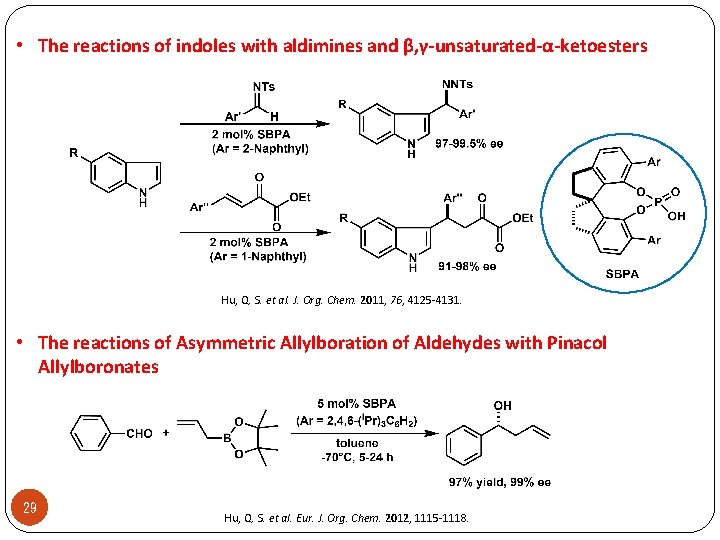
• The reactions of indoles with aldimines and β, γ-unsaturated-α-ketoesters Hu, Q. S. et al. J. Org. Chem. 2011, 76, 4125 -4131. • The reactions of Asymmetric Allylboration of Aldehydes with Pinacol Allylboronates 29 Hu, Q. S. et al. Eur. J. Org. Chem. 2012, 1115 -1118.
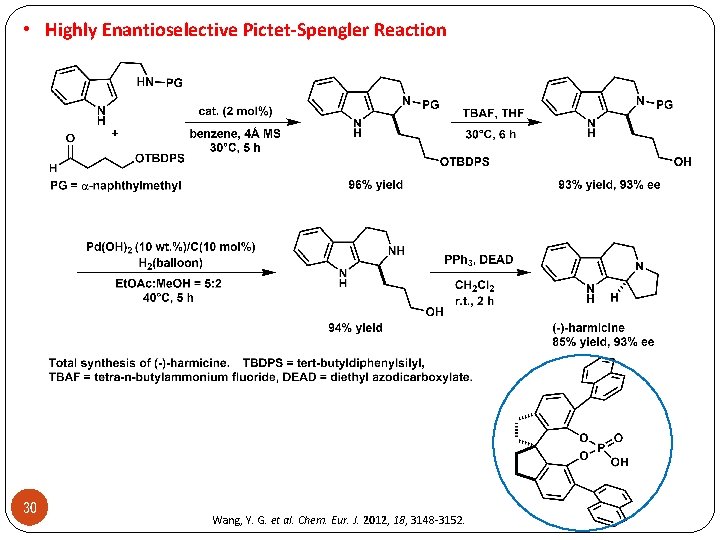
• Highly Enantioselective Pictet-Spengler Reaction 30 Wang, Y. G. et al. Chem. Eur. J. 2012, 18, 3148 -3152.
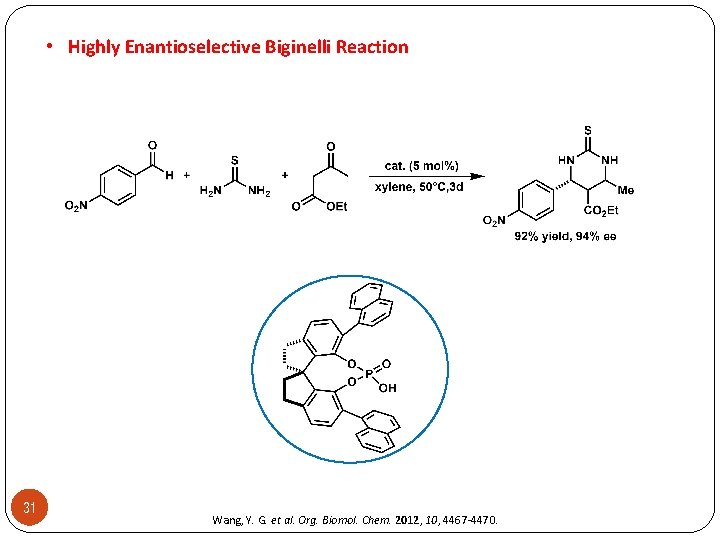
• Highly Enantioselective Biginelli Reaction 31 Wang, Y. G. et al. Org. Biomol. Chem. 2012, 10, 4467 -4470.
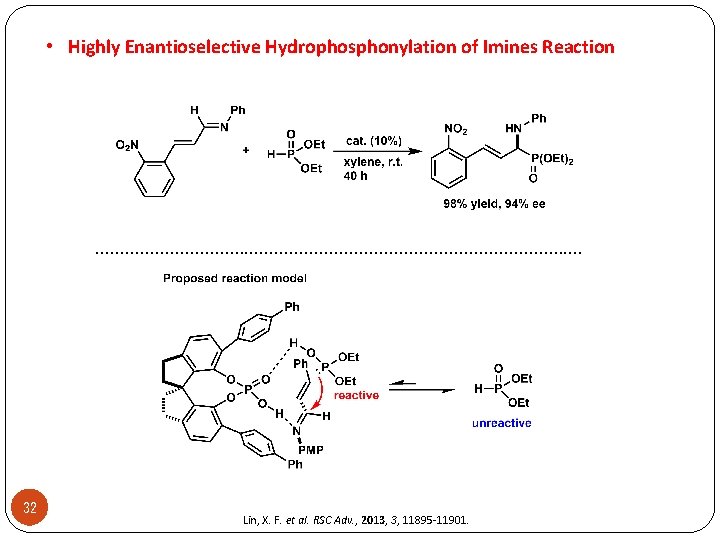
• Highly Enantioselective Hydrophosphonylation of Imines Reaction . . . . . . 32 Lin, X. F. et al. RSC Adv. , 2013, 3, 11895 -11901.
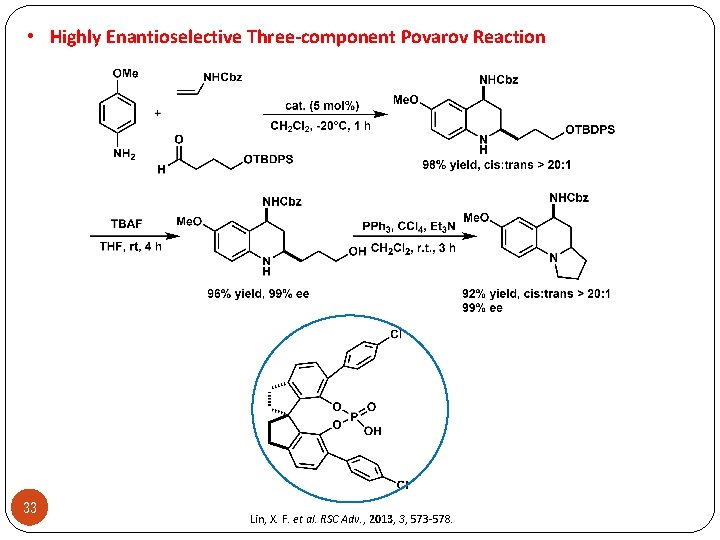
• Highly Enantioselective Three-component Povarov Reaction 33 Lin, X. F. et al. RSC Adv. , 2013, 3, 573 -578.
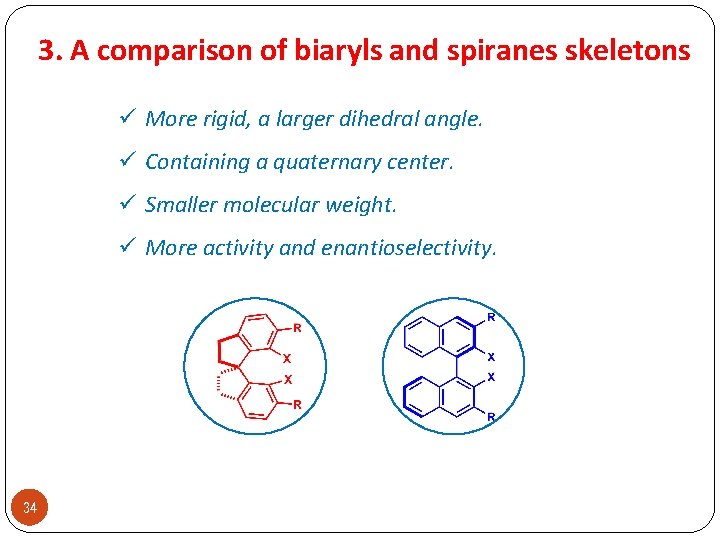
3. A comparison of biaryls and spiranes skeletons ü More rigid, a larger dihedral angle. ü Containing a quaternary center. ü Smaller molecular weight. ü More activity and enantioselectivity. 34
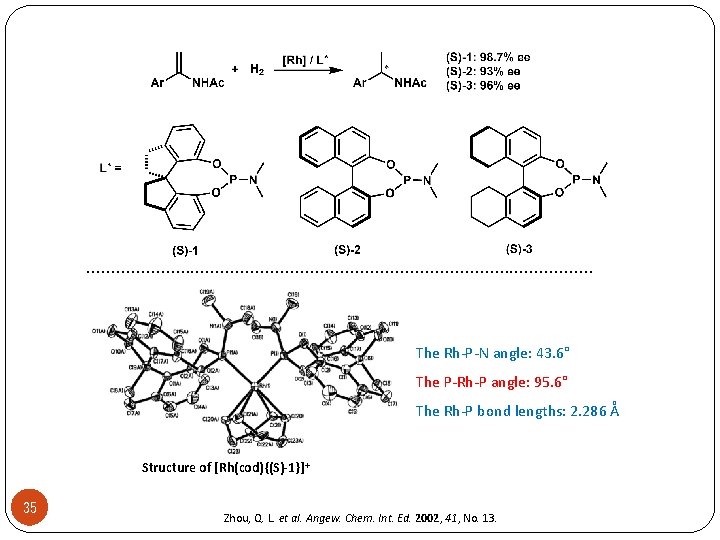
. . . . . . . The Rh-P-N angle: 43. 6° The P-Rh-P angle: 95. 6° The Rh-P bond lengths: 2. 286 Å Structure of [Rh(cod){(S)-1}]+ 35 Zhou, Q. L. et al. Angew. Chem. Int. Ed. 2002, 41, No. 13.
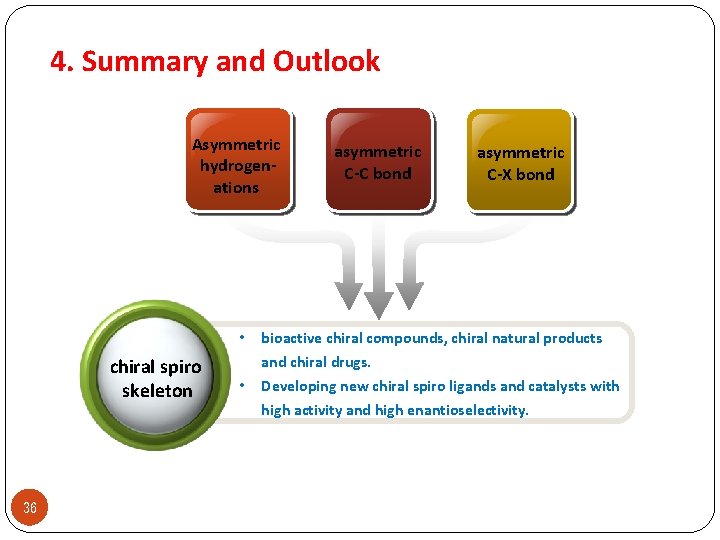
4. Summary and Outlook Asymmetric hydrogenations chiral spiro skeleton 36 asymmetric C-C bond asymmetric C-X bond • bioactive chiral compounds, chiral natural products • and chiral drugs. Developing new chiral spiro ligands and catalysts with high activity and high enantioselectivity.

105 135 2006 -2010 21/44 2002 -2005 3/19 Prof. Zhou’s Research Group 37 2011 -2015 21/42
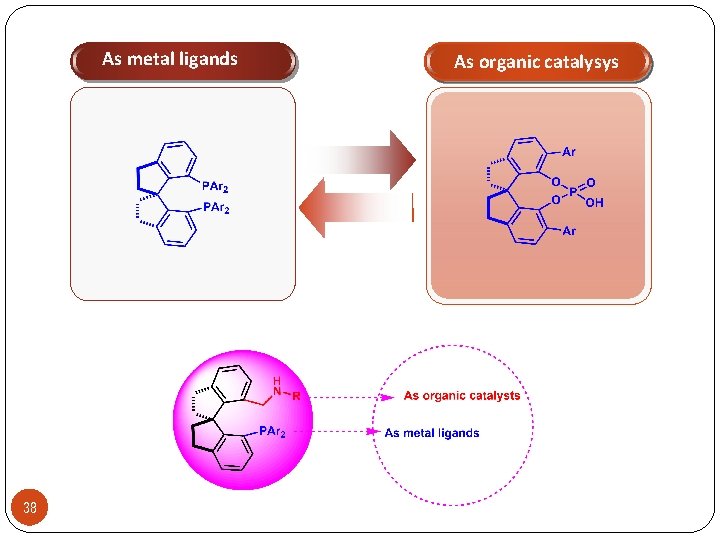
As metal ligands 38 As organic catalysys

5. Acknowledgement ØProf. Huang ØMr. Chen ØAll members here Thanks for your attention! 39
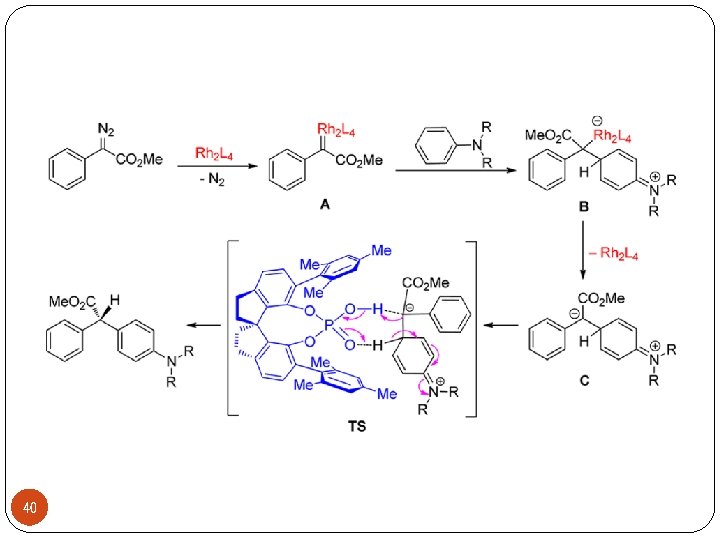
40
41
42
43
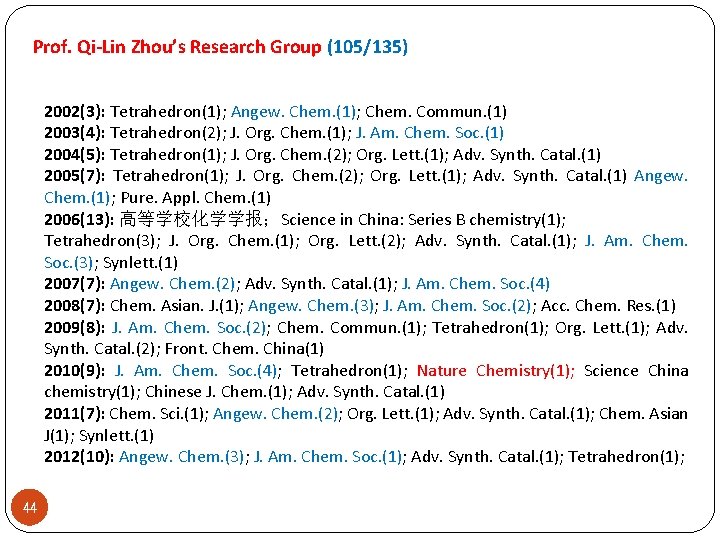
Prof. Qi-Lin Zhou’s Research Group (105/135) 2002(3): Tetrahedron(1); Angew. Chem. (1); Chem. Commun. (1) 2003(4): Tetrahedron(2); J. Org. Chem. (1); J. Am. Chem. Soc. (1) 2004(5): Tetrahedron(1); J. Org. Chem. (2); Org. Lett. (1); Adv. Synth. Catal. (1) 2005(7): Tetrahedron(1); J. Org. Chem. (2); Org. Lett. (1); Adv. Synth. Catal. (1) Angew. Chem. (1); Pure. Appl. Chem. (1) 2006(13): 高等学校化学学报;Science in China: Series B chemistry(1); Tetrahedron(3); J. Org. Chem. (1); Org. Lett. (2); Adv. Synth. Catal. (1); J. Am. Chem. Soc. (3); Synlett. (1) 2007(7): Angew. Chem. (2); Adv. Synth. Catal. (1); J. Am. Chem. Soc. (4) 2008(7): Chem. Asian. J. (1); Angew. Chem. (3); J. Am. Chem. Soc. (2); Acc. Chem. Res. (1) 2009(8): J. Am. Chem. Soc. (2); Chem. Commun. (1); Tetrahedron(1); Org. Lett. (1); Adv. Synth. Catal. (2); Front. Chem. China(1) 2010(9): J. Am. Chem. Soc. (4); Tetrahedron(1); Nature Chemistry(1); Science China chemistry(1); Chinese J. Chem. (1); Adv. Synth. Catal. (1) 2011(7): Chem. Sci. (1); Angew. Chem. (2); Org. Lett. (1); Adv. Synth. Catal. (1); Chem. Asian J(1); Synlett. (1) 2012(10): Angew. Chem. (3); J. Am. Chem. Soc. (1); Adv. Synth. Catal. (1); Tetrahedron(1); 44
- Slides: 44