MAGELLAN3 Study GLEPIB SOF RBV in patients who
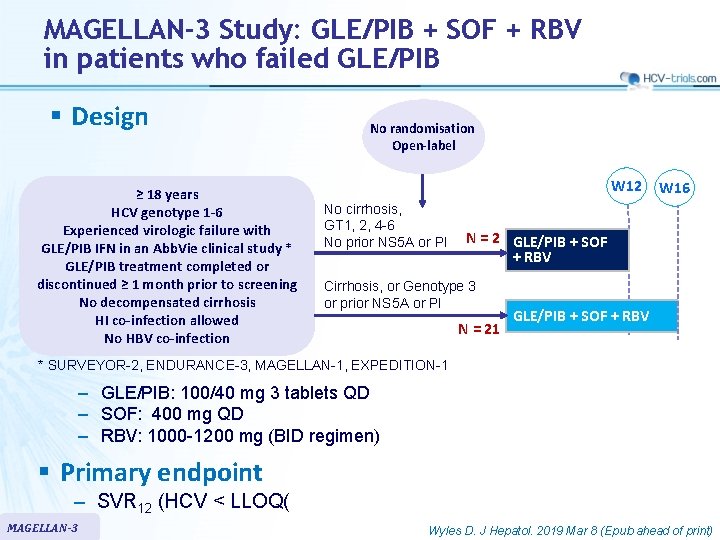
MAGELLAN-3 Study: GLE/PIB + SOF + RBV in patients who failed GLE/PIB § Design ≥ 18 years HCV genotype 1 -6 Experienced virologic failure with GLE/PIB IFN in an Abb. Vie clinical study * GLE/PIB treatment completed or discontinued ≥ 1 month prior to screening No decompensated cirrhosis HI co-infection allowed No HBV co-infection No randomisation Open-label W 12 No cirrhosis, GT 1, 2, 4 -6 No prior NS 5 A or PI W 16 N = 2 GLE/PIB + SOF + RBV Cirrhosis, or Genotype 3 or prior NS 5 A or PI N = 21 GLE/PIB + SOF + RBV * SURVEYOR-2, ENDURANCE-3, MAGELLAN-1, EXPEDITION-1 ‒ GLE/PIB: 100/40 mg 3 tablets QD ‒ SOF: 400 mg QD ‒ RBV: 1000 -1200 mg (BID regimen) § Primary endpoint – SVR 12 (HCV < LLOQ( MAGELLAN-3 Wyles D. J Hepatol. 2019 Mar 8 (Epub ahead of print)
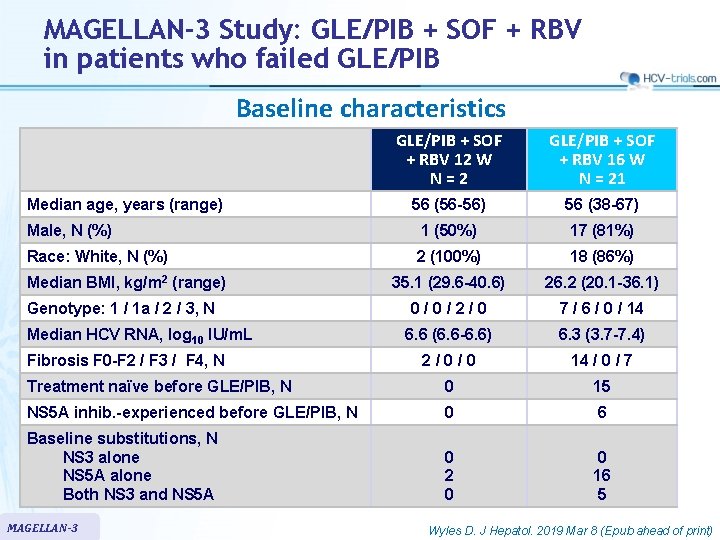
MAGELLAN-3 Study: GLE/PIB + SOF + RBV in patients who failed GLE/PIB Baseline characteristics GLE/PIB + SOF + RBV 12 W N = 2 GLE/PIB + SOF + RBV 16 W N = 21 56 (56 -56) 56 (38 -67) Male, N (%) 1 (50%) 17 (81%) Race: White, N (%) 2 (100%) 18 (86%) 35. 1 (29. 6 -40. 6) 26. 2 (20. 1 -36. 1) 0/0/2/0 7 / 6 / 0 / 14 6. 6 (6. 6 -6. 6) 6. 3 (3. 7 -7. 4) 2/0/0 14 / 0 / 7 Treatment naïve before GLE/PIB, N 0 15 NS 5 A inhib. -experienced before GLE/PIB, N 0 6 Baseline substitutions, N NS 3 alone NS 5 A alone Both NS 3 and NS 5 A 0 2 0 0 16 5 Median age, years (range) Median BMI, kg/m 2 (range) Genotype: 1 / 1 a / 2 / 3, N Median HCV RNA, log 10 IU/m. L Fibrosis F 0 -F 2 / F 3 / F 4, N MAGELLAN-3 Wyles D. J Hepatol. 2019 Mar 8 (Epub ahead of print)
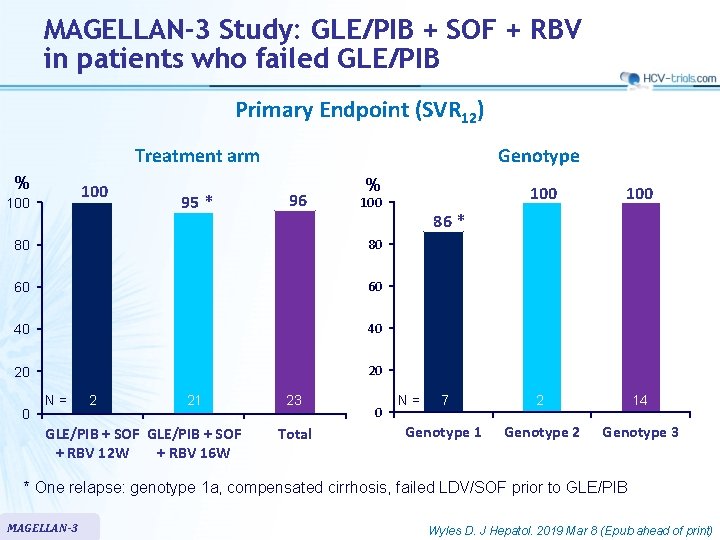
MAGELLAN-3 Study: GLE/PIB + SOF + RBV in patients who failed GLE/PIB Primary Endpoint (SVR 12) Treatment arm % 100 95 * Genotype 96 % 80 80 60 60 40 40 20 20 0 N= 2 21 GLE/PIB + SOF + RBV 12 W + RBV 16 W 23 Total 100 7 2 14 Genotype 1 Genotype 2 Genotype 3 100 0 86 * N= * One relapse: genotype 1 a, compensated cirrhosis, failed LDV/SOF prior to GLE/PIB MAGELLAN-3 Wyles D. J Hepatol. 2019 Mar 8 (Epub ahead of print)
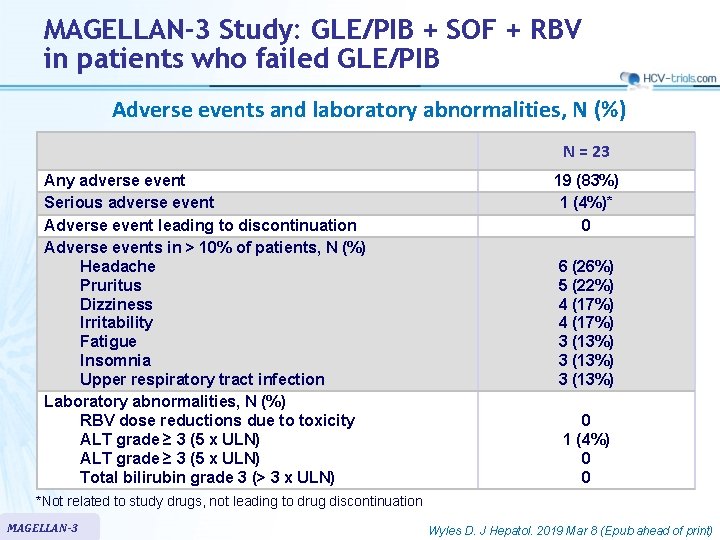
MAGELLAN-3 Study: GLE/PIB + SOF + RBV in patients who failed GLE/PIB Adverse events and laboratory abnormalities, N (%) N = 23 Any adverse event Serious adverse event Adverse event leading to discontinuation Adverse events in > 10% of patients, N (%) Headache Pruritus Dizziness Irritability Fatigue Insomnia Upper respiratory tract infection Laboratory abnormalities, N (%) RBV dose reductions due to toxicity ALT grade ≥ 3 (5 x ULN) Total bilirubin grade 3 (> 3 x ULN) 19 (83%) 1 (4%)* 0 6 (26%) 5 (22%) 4 (17%) 3 (13%) 0 1 (4%) 0 0 *Not related to study drugs, not leading to drug discontinuation MAGELLAN-3 Wyles D. J Hepatol. 2019 Mar 8 (Epub ahead of print)

MAGELLAN-3 Study: GLE/PIB + SOF + RBV in patients who failed GLE/PIB § Summary – GLE/PIB + SOF + RBV resulted in 96% SVR 12 in patients with previous GLE/PIB virologic failure • 100% SVR 12 in the 14 patients with genotype 3 – Treatment was well-tolerated MAGELLAN-3 Wyles D. J Hepatol. 2019 Mar 8 (Epub ahead of print)
- Slides: 5