Mad Cow Disease Zoonotic Disease FKHUB MAD COW

Mad Cow Disease Zoonotic Disease FKH-UB

MAD COW DISEASE v Mad Cow Disease (Bovine Spongiform Encepalopathy) in Cows Scrapie (Scrapie Spongiform Encepalopathy) in Sheep Creutzfeldt-Jakob Disease in human Transmissible Spongiform Encephalopathy (TSE) v Initially thought to be due to “slow viruses”, due to the long incubation period between time of infection and appearance of disease

TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHY (TSE) v Transmissible Spongiform Encephalopathy (TSE) are rare forms of progressive neurodegenerative disorders that affect both humans & animals They are caused by similar agents caused by agents called prions v They are called so because they produce spongiform changes in the brain v The causative agent is found at the highest titer in the brain
PRION • 1960 2 London-based researchers, radiation biologist Tikvah Alper & biophysicist John Stanlley Griffith develop hypothesis some TSE are caused by an infectious agent consisting solely of proteins • E. J. Field scrapie & kuru identified transfer of pathological inert polysaccharides that only become infectious in the host • Alper & Griffith infectious agent of Creutzfeldt-Jakop disease & scrapie resistant from ionizing radiation • 1982, Stanley B. Prusiner (California University) Prion Protein (Pr. P) Nobel Prize in Physiology or Medicine
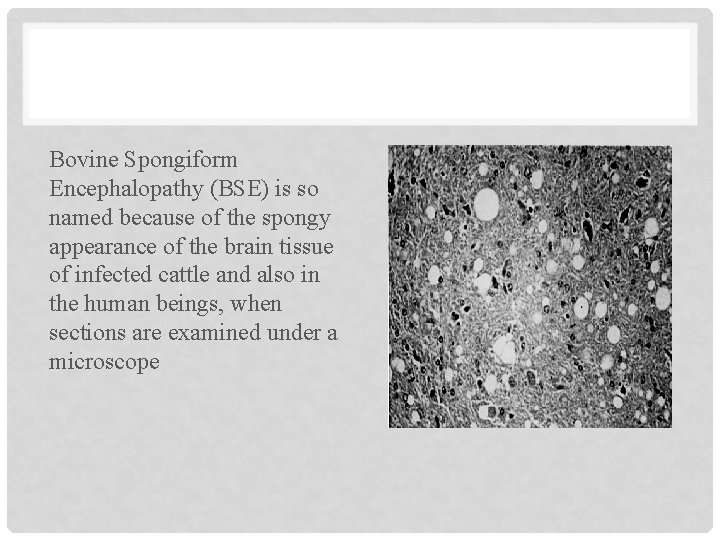
Bovine Spongiform Encephalopathy (BSE) is so named because of the spongy appearance of the brain tissue of infected cattle and also in the human beings, when sections are examined under a microscope

HOW DOES IT PROGRESS? v The incubation period varies from 2 to 8 years. v Following the onset of clinical signs, the animals condition deteriorates until it either dies. v This process usually takes from 2 weeks to 6 months. v Most cases in Great Britain (where it was first detected) have occurred in dairy cows between 3 to 6 years of age.

THREE PHASES OF BSE IN CATTLE The first phase: v Low infectivity rate, and at this level the cow does not pose a large threat to humans & animals The second phase: v Symptoms are not evident, but the infectivity level is very high v The prion agent is abundant in both the spinal chord and the brain – the cow is a risk to public health The third phase: v Clinical symptoms, & then death follows shortly

WHAT CAUSES MAD COW DISEASE?

v Initially thought to have been caused by a “slow” viruses, these were classified as “slow Viral Diseases” v Now there is evidence to point out prions as the causative factor

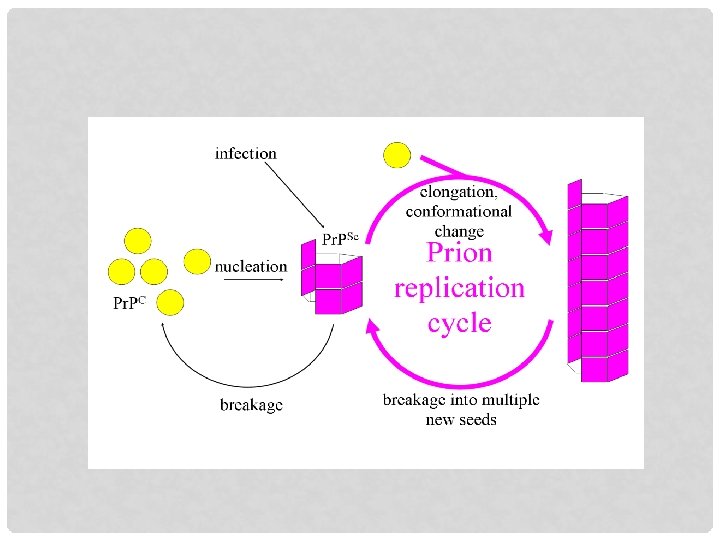
PRION HYPOTHESIS v Pr. P is a normal cellular protein referred to as Pr. Pc, found throughout the body, even in healthy human and animal Pr. Pc is found on the membrane of cells v Diseased brain contains aberrant Pr. P which is referred to as Pr. PSc v Pr. PSc has the ability to convert Pr. Pc to itself v There are multiple strain of classical scrapie proin ---- atypical (Nor 98) scrapie prion Prion CH 1614 ---- have similarities to BSE prion v A chain reaction follows, resulting in a cluster of tangled, nonfunctional proteins called plaques, which are aggregates of Pr. PSc in the brain

PRION • Shortened form /Smaller than smallest known virus • Not yet completely characterized • Most widely accepted theory • Prion = Proteinaceous infectious particle • Normal Protein • Pr. PC protein (C for cellular) • Glycoprotein normally found at cell surface inserted in plasma membrane

• The physiological function of the prion protein remains a controversial matter • Research in knockout mice cleavage Pr. P in peripheral nerves activation of myelin repair in Schwan cells • The lack mice of Pr. P protein demyelination
PRIONS Prions are single molecules containing about 250 amino acids They are abnormal variants of proteins which normally occur in cells Prions have the ability to convert the normal forms that they come into contact with into abnormal forms
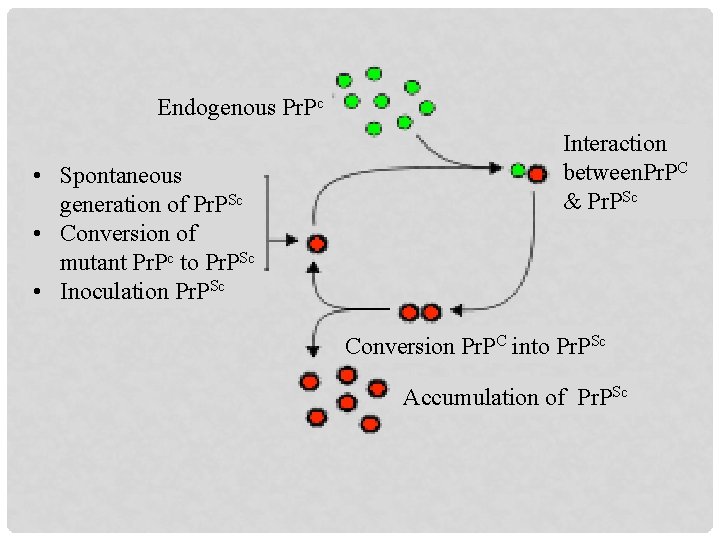
Endogenous Pr. Pc • Spontaneous generation of Pr. PSc • Conversion of mutant Pr. Pc to Pr. PSc • Inoculation Pr. PSc Interaction between. Pr. PC & Pr. PSc Conversion Pr. PC into Pr. PSc Accumulation of Pr. PSc

NORMAL PROTEIN • Secondary structure dominated by alpha helices • Easily soluble • Easily digested by proteases • Encoded by PRNP gene Located on human chromosome 20 Center for Food Security and Public Health, Iowa State University, 2011

ABNORMAL PROTEIN • Pr. PSc (Sc for scrapie) /Pr. PTSE/ Pr. Pd (disease-associated) • Same amino acid sequence and primary structure as normal protein • Secondary structure dominated by beta conformation • When Pr. PSc contacts Pr. PC • Converts it to the abnormal form Center for Food Security and Public Health, Iowa State University, 2011
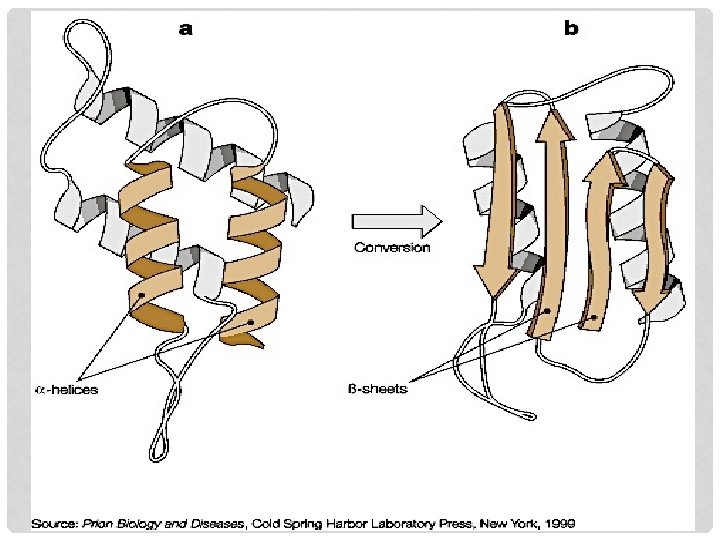

ABNORMAL PROTEIN • Insoluble in all but strongest solvents • Highly resistant to digestion by proteases ---- Pr. Pres (proteinase K-resistant nature of prion) • Survives in tissues post-mortem • Extremely resistant • Heat, normal sterilization processes, sunlight Center for Food Security and Public Health, Iowa State University, 2011

v The body defenses remove these Pr. PSc aggregates leaving behind holes v This causes degeneration of the brain cells leading to mental changes and ultimately, death
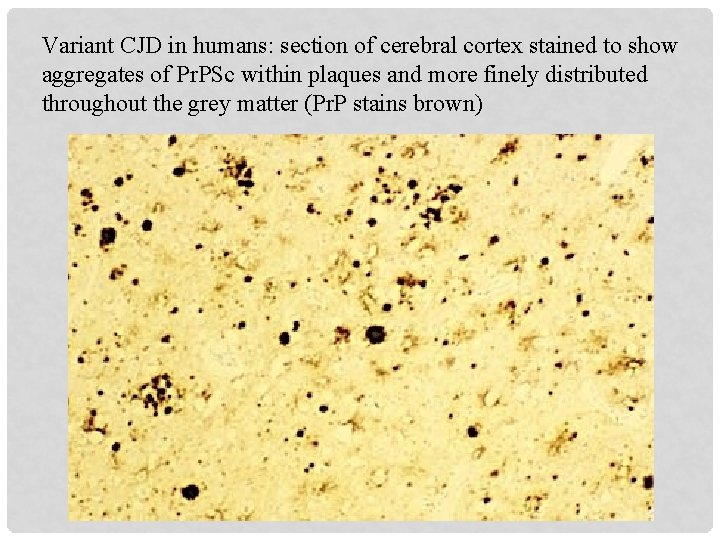
Variant CJD in humans: section of cerebral cortex stained to show aggregates of Pr. PSc within plaques and more finely distributed throughout the grey matter (Pr. P stains brown)

Prion Characteristics No antibiotics can cure disease caused by prions Not typical of a prokaryotic or a eukaryotic organism Prions are non immunogens and do not induce an immune response The prion does not evoke a detectable immune or inflammatory response --- the body does not react to it as an invader. PSc is resistant to any form of digestion Prions are not easy to decompose biologically Resistant to high temperatures & disinfectants

HOW IS IT DIAGNOSED IN CATTLE?

CLINICAL SIGNS - 1 § Cattle affected by BSE experience progressive degeneration of the nervous system. § Affected animals may display changes in temperament, such as nervousness or aggression, abnormal posture, incoordination and difficulty in rising, decreased milk production, decreased rumination or loss of body weight despite continued appetite § 2 -3 % morbidity § 100% mortality

CLINICAL SIGNS - 2 The clinical signs are: § Apprehension § Progressive paresis & ataxia
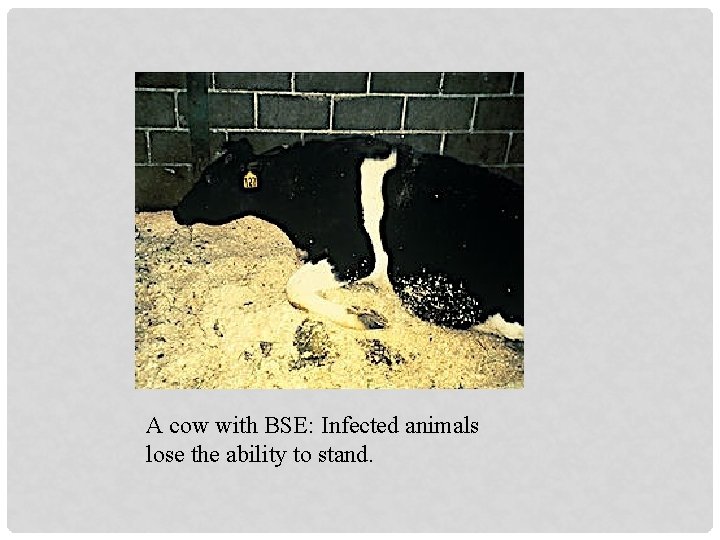
A cow with BSE: Infected animals lose the ability to stand.
DIAGNOSIS § Currently, there is no test to detect the disease in a live animal or in humans (no ante mortem testing available § Veterinary pathologists confirm BSE by postmortem microscopic examination of brain/medulla, spinal cord tissue or by the detection of the abnormal form of the prion protein

Histological Findings on Post Mortem Histological findings include § Vacuolation of the neurones and neuronal ground substance in cerebella/cortex (gray matter) § Perivascular fibrils of amyloid in which Pr. Psc can be demonstrated by immunostaining and congo red bifringence § Astrocyte infiltration.

POST MORTEM TESTS FOR BSE • All are based on antibodies to detect prion protein in tissue • Immunohistochemistry (IHC) is considered the gold standard • Internationally recognized • Expensive, labor intensive • Rapid diagnostic tests • Western blotting, ELISA
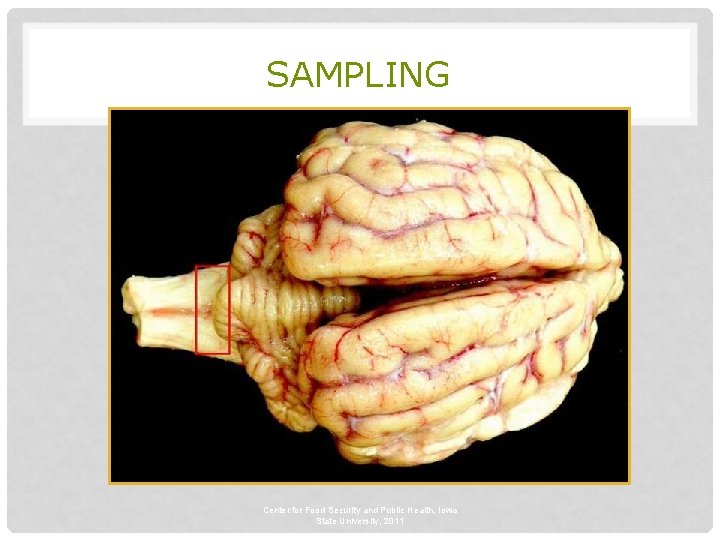
SAMPLING Obex Center for Food Security and Public Health, Iowa State University, 2011

SAMPLING • Before collecting or sending any samples, the proper authorities should be contacted • Samples should only be sent under secure conditions and to authorized laboratories to prevent the spread of the disease Center for Food Security and Public Health, Iowa State University, 2011

Differentials • Nervous ketosis, hypomagnesemia, listeriosis, polioencephalomalacia, rabies, brain tumor, lead poisoning spinal cord trauma

How Does it Spread?

THE SOURCE OF PRION v Prion in cattle is mainly are from the carcasses of scrapie-infected sheep. v After these infected sheep having died , their brains and other sheep byproducts infected with scrapie is used to feed cattle with the meat and bone mill (MBM)

HOW DOES IT SPREAD FROM ANIMAL TO ANIMAL? Feeding cattle animal bi-products such as meat-n-bone mill that has an infected prion causes the infection in the cattle The prions are concentrated in the brain, and spinal cord of these animals There is no evidence that it is concentrated in the muscle mass of cattle, and they are considered safe as long as they are not in contact with the brain and spinal cord during the slaughter process

HOW DID BSE TRANSFER TO HUMANS v Sheep with Scrapie used in Meat and Bone Meal (MBM) – known as “Offal” v Infected Beef eaten by humans § Not affected by cooking § Brain & spinal tissue v Maternal transmission § Low risk v Genetic susceptibility § Human have been homozygous for methionine at codon 129 of Pr. PC
HUMAN TRANSMISSION Possible modes • Transmission from surgical instruments used on tonsils, appendix, or brain tissue • Growth hormone injections • Vaccines Center for Food Security and Public Health, Iowa State University, 2011

Should Humans be Worried?

MAD COW DISEASE IN HUMANS When cattle brains and other cattle byproducts infected with BSE are ingested by humans, there is a risk of developing the Creutzfeldt-Jakob Disease

Is there a Cure?

At Present, there is no Cure for the Mad Cow Disease (Bovine Spongiform Encephalitis) and for the Creutzfeldt-Jakob Disease Prevention is the only available option

PREVENTION v Don’t feed cattle animal bi-products v Watch to make sure you are feeding your animals safe feeds v Always vaccinate cattle properly

PREVENTION v USDA requires all imported meat to be inspected v US will not import cattle from Britian v Submit brain, medulla: incinerate the carcass v Animals suspected of the disease are quarantined

DISINFECTION • • • Porous load autoclaving Sodium hypochlorite 2 -N sodium hydroxide Rendering at high temperature and pressure Resistant in tissues, dried organic material, high titer Center for Food Security and Public Health, Iowa State University, 2011
- Slides: 44