MACROMOLECULES The Big Idea Living organisms are made

MACROMOLECULES

The Big Idea… Living organisms are made up of carbon-bases molecules

Objectives � � � Describe the role of carbon in living organisms Summarize the four major families of biological macromolecules Compare the functions of each group of macromolecules

4 essential elements to life {Q#1} 95% of your body weight

The element CARBON

Carbon � Component of almost all biologic molecules � Life on Earth is thought to be “carbon-based. ” � Organic Chemistry – a branch of chemistry devoted completely to the study of carbonbased compounds
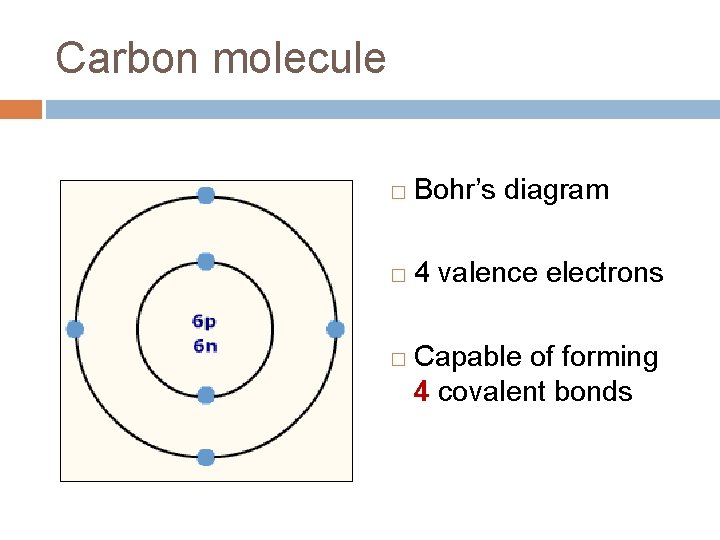
Carbon molecule � Bohr’s diagram � 4 valence electrons � Capable of forming 4 covalent bonds
Carbon molecules

Macromolecules � Prefix: Macro- means: large � aka: Biomolecules � aka: Polymers � Prefix: Poly- means: many � Repeating units of identical monomers ■ building blocks � 4 categories (pg. 167) ■ ■ Carbohydrates Lipids Proteins Nucleic acids
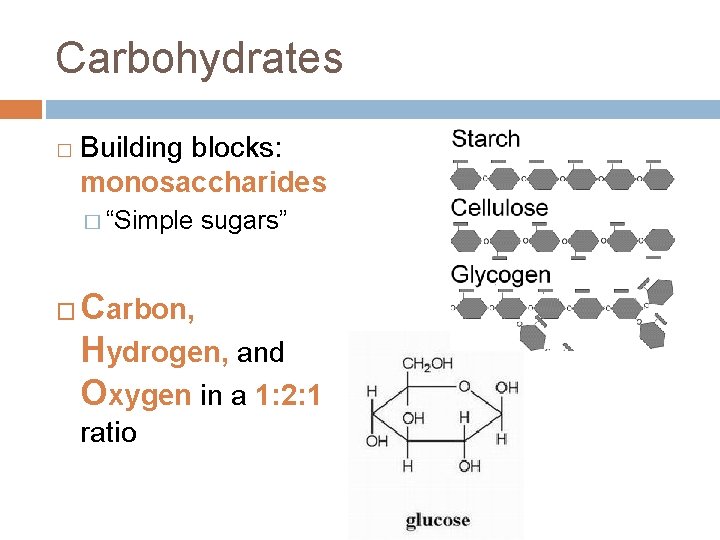
Carbohydrates � Building blocks: monosaccharides � “Simple � sugars” Carbon, Hydrogen, and Oxygen in a 1: 2: 1 ratio

How we use carbohydrates � Mono- and disaccharides are an energy source � Glucose (chief energy source) � Sucrose � Lactose � Polysaccharides are energy storage molecules � Glycogen (liver and muscle cells)

Polysaccharides in nature � Cellulose � Plant � cell walls Chitin � Exoskeleton of insects and crustaceans � Fungi cell wall

Lipids � Fats, oils, and waxes � Carbon, Hydrogen, Oxygen (CHO) � Building blocks of lipids are fatty acids � Large, non-polar molecules � Hydrophobic - will not dissolve in water � Serve as barriers
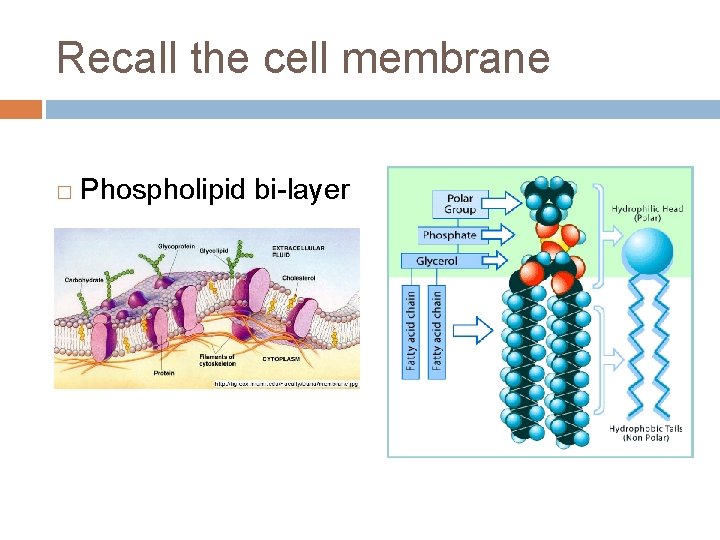
Recall the cell membrane � Phospholipid bi-layer

Phospholipid review � Heads are polar and attract water � hydrophilic � Tails are non-polar and repel water � hydrophobic
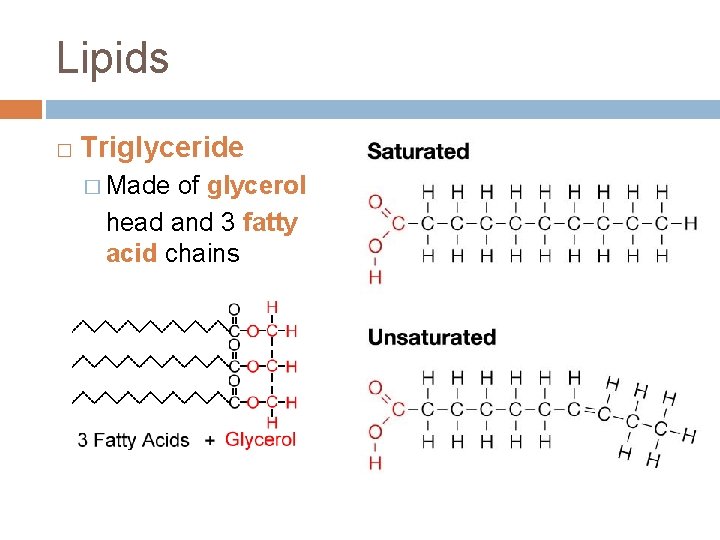
Lipids � Triglyceride � Made of glycerol head and 3 fatty acid chains
Lipids � Steroids � Cholesterol � Hormones ■ ■ Testosterone Estrogen

Lipids in nature � Cutin – a waxy coating covering leaves � Chlorophyll – green pigment molecule � Beeswax – honeycomb

Proteins � Building blocks – amino acids �Carbon, Hydrogen, Oxygen, Nitrogen, and sometimes Sulfur (CHON S) � 2 ■ ■ functional groups Amino Group … -NH 2 Carboxyl Group … -COOH

Amino Acids � Same general structure � 20 different amino acids � � Combine in various ways to form polypeptides which join together to make a protein Peptide bonds join amino acids together to form proteins in a condensation reaction (removal of water molecule)
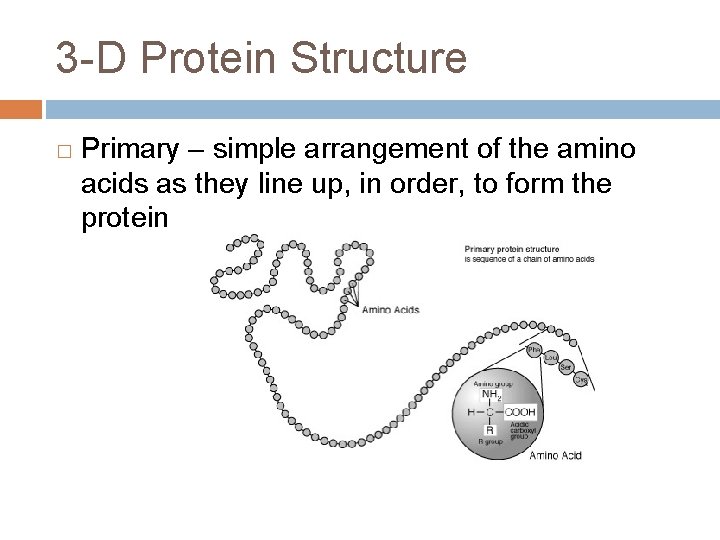
3 -D Protein Structure � Primary – simple arrangement of the amino acids as they line up, in order, to form the protein
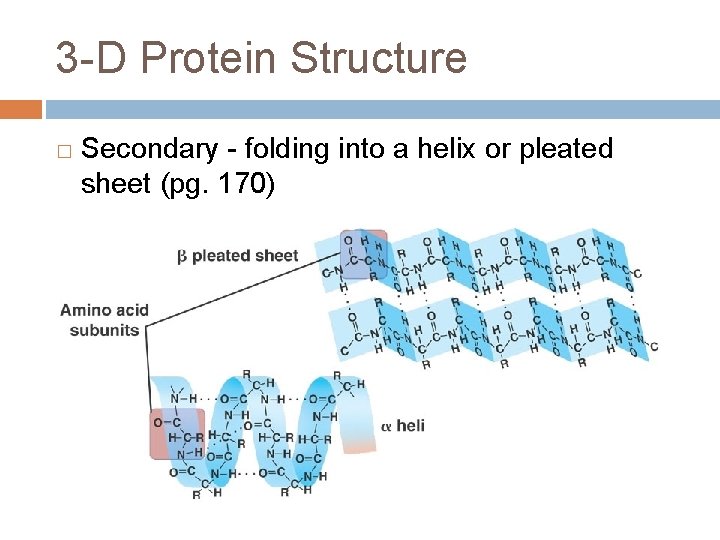
3 -D Protein Structure � Secondary - folding into a helix or pleated sheet (pg. 170)
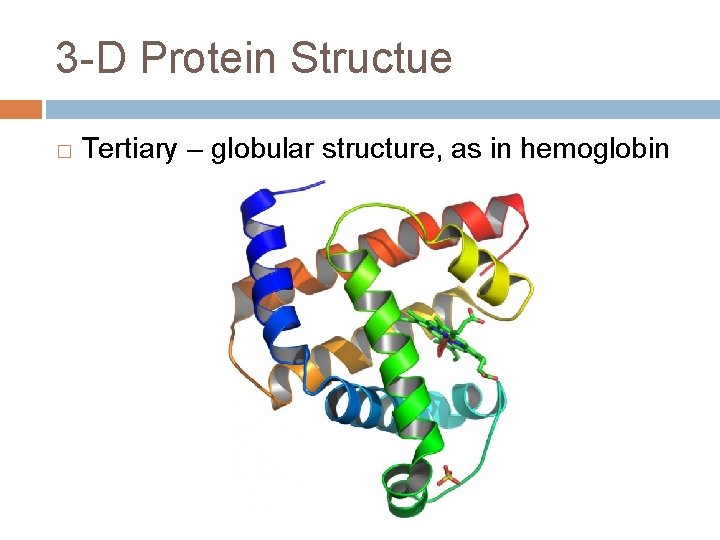
3 -D Protein Structue � Tertiary – globular structure, as in hemoglobin
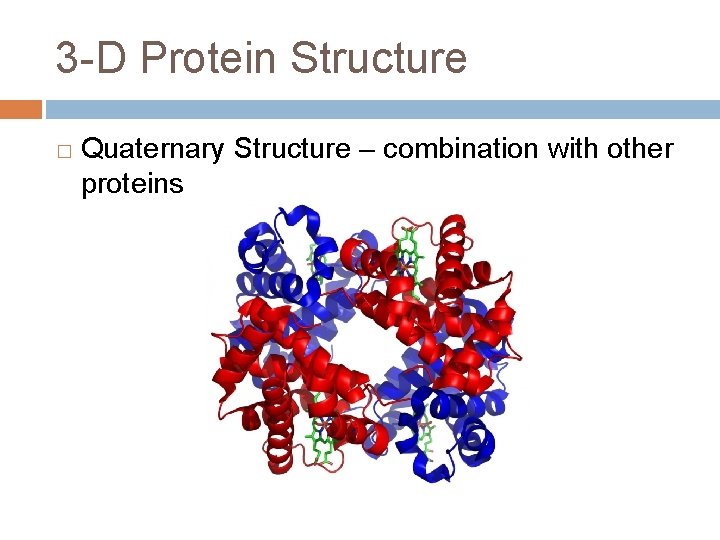
3 -D Protein Structure � Quaternary Structure – combination with other proteins

Protein Functions � 15 percent of total human body mass � Hair, skin, muscles � Cells have about 10, 000 different proteins � Transport proteins � Enzymes – thousands to control chemical reactions (aka biologic catalysts)

Nucleic Acids � Building blocks – nucleotides Carbon, Hydrogen, Oxygen, Nitrogen, and Phosphorus atoms �Composed of � Nucleotides are made of 3 parts: � Phosphate � Nitrogenous base � Ribose (5 carbon) sugar
Functions of Nucleic Acids � Carry the genetic information in cells � DNA ■ Contains all the instructions for making every protein needed in a living organism � RNA ■ – deoxyribose nucleic acid – ribose nucleic acid Copies and transfers the genetic information in DNA to ribosomes so that proteins can be made

Nucleotide ATP � � ATP is a special nucleotide with 3 phosphate groups Storehouse of chemical energy � You will learn more about ATP in chapter 8

8 review questions…
REVIEW � � Carbohydrates, lipids, proteins, and nucleic acids are M__________. Proteins are made from A________ that are joined by P____ B_____. L______ make up fats, oils, and waxes. DNA and RNA are examples of N______ A____.

REVIEW � Which two elements are always found in amino acids? A. Nitrogen and Sulfur � Carbon and Oxygen � Hydrogen and Phosphorus � Sulfur and Oxygen �

REVIEW � Which joins amino acids together? A. Peptide bonds � B. Hydrogen Bonds � C. van der Waals forces � D. ionic bonds �

REVIEW � Which substance is not part of a nucleotide? A. a phosphate � B. a base � C. a sugar � D. water �
- Slides: 33