MACROMOLECULES NOTES 2 3 Organic Compounds Organic Compounds

































































- Slides: 90

MACROMOLECULES NOTES – 2. 3

Organic Compounds �Organic Compounds: are CARBON compounds that come from living things �Produced by ORGANISMS �Contain CARBON atoms and bonds

Carbon �Carbon bonds easily with itself and other elements (this gives carbon the ability to create millions of three dimensional complex structures of compounds) 6 P 6 N

Carbon Cont. �Carbon can form 4 covalent bonds �Due to covalent bonds carbon atoms can combine to form: �Straight lines �Branched chains �Rings

Macromolecules �Many of the molecules in living cells are so large that they are known as MACROMOLECULES which means “giant molecules” �They are made from thousands or even hundreds of thousands of smaller molecules �MONOMER is one small unit �POLYMER form when many small units join together by COVALENT BONDS

Macromolecules �In living things there are 4 major classes of macromolecules: �Carbohydrates �Lipids �Proteins �Nucleic Acids

Carbohydrates �Carbohydrates: The main source of energy for living things �Also known as sugar SACCHARIDE �Made up of the elements C, H, and O in the ratio 2 : 1 for H : O

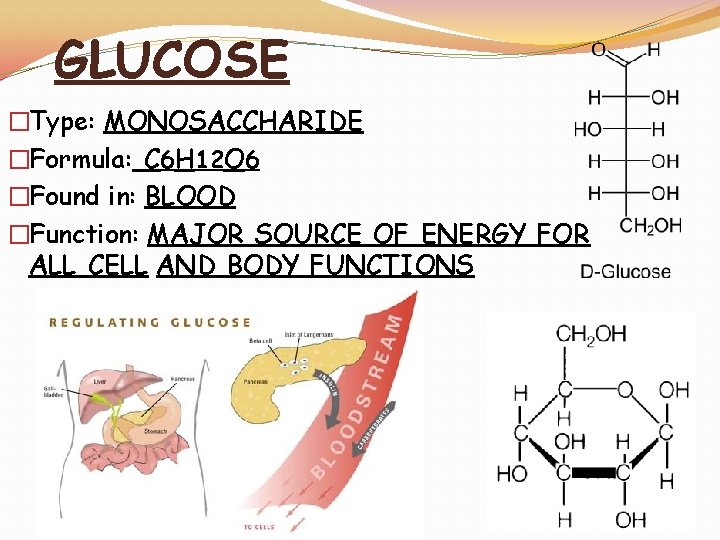
GLUCOSE �Type: MONOSACCHARIDE �Formula: C 6 H 12 O 6 �Found in: BLOOD �Function: MAJOR SOURCE OF ENERGY FOR ALL CELL AND BODY FUNCTIONS

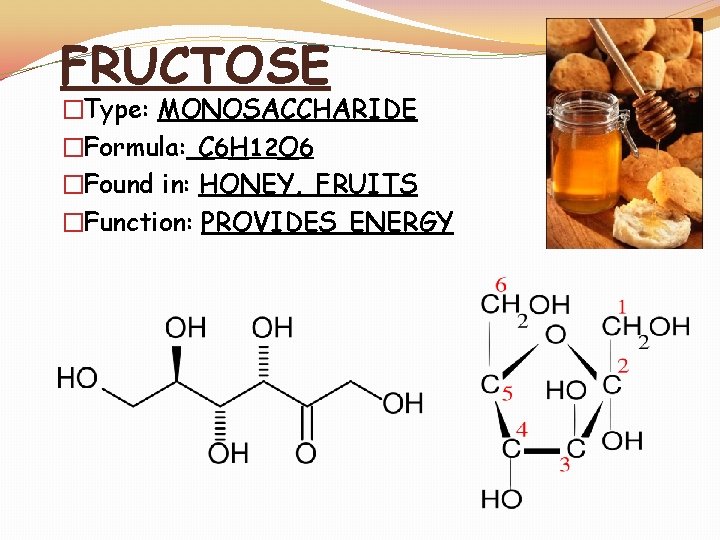
FRUCTOSE �Type: MONOSACCHARIDE �Formula: C 6 H 12 O 6 �Found in: HONEY, FRUITS �Function: PROVIDES ENERGY

SUCROSE �Type: DISACCHARIDE �Formula: C 12 H 22 O 11 �Found in: TABLE SUGAR �Function: PROVIDES ENERGY

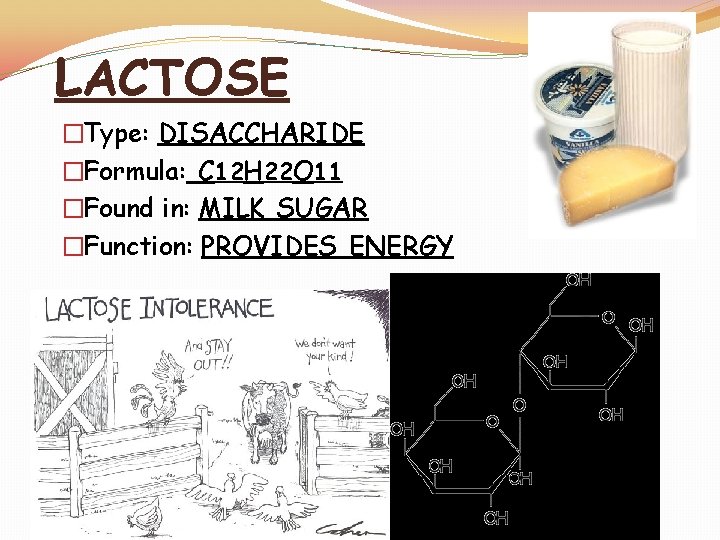
LACTOSE �Type: DISACCHARIDE �Formula: C 12 H 22 O 11 �Found in: MILK SUGAR �Function: PROVIDES ENERGY

MALTOSE �Type: DISACCHARIDE �Formula: C 12 H 22 O 11 �Found in: MALT SUGAR �Function: MAIN SUGAR IN BEER AND LIQUOR

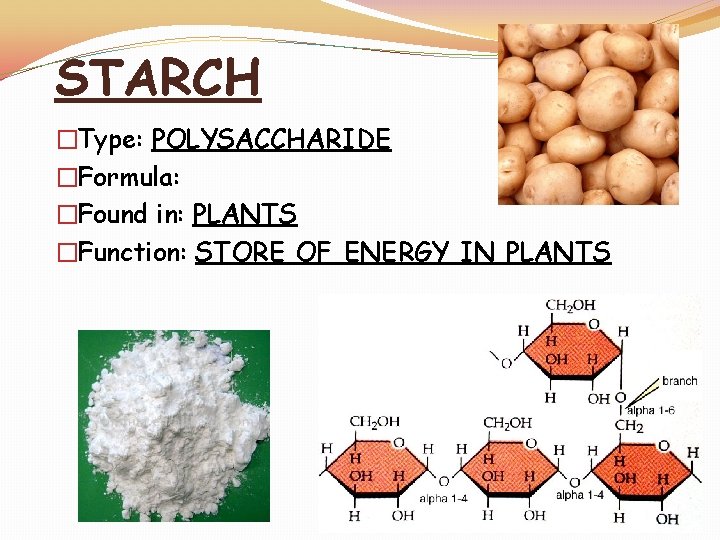
STARCH �Type: POLYSACCHARIDE �Formula: �Found in: PLANTS �Function: STORE OF ENERGY IN PLANTS

GLYCOGEN �Type: POLYSACCHARIDE �Formula: �Found in: ANIMALS �Function: CARB ENERGY STORED AS THIS IN ANIMALS

CELLULOSE �Type: POLYSACCHARIDE �Formula: �Found in: CELL WALL IN PLANTS �Function: TOUGH, PROTECTIVE, NOT DIGESTED (NO SOURCE OF ENERGY)

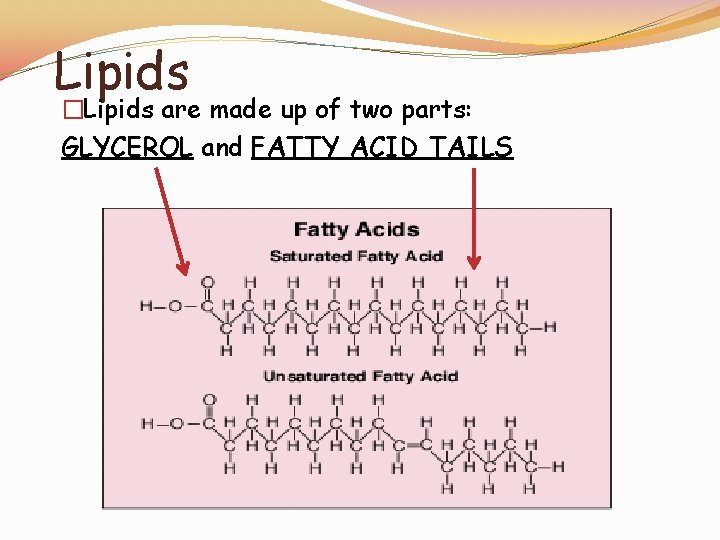
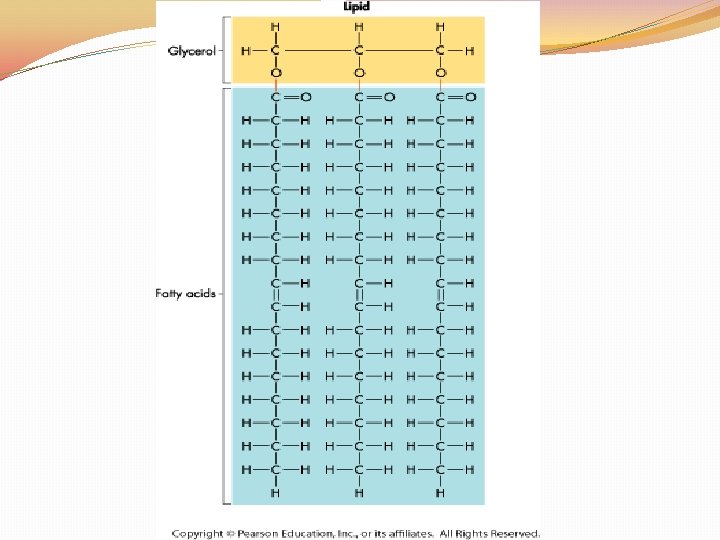
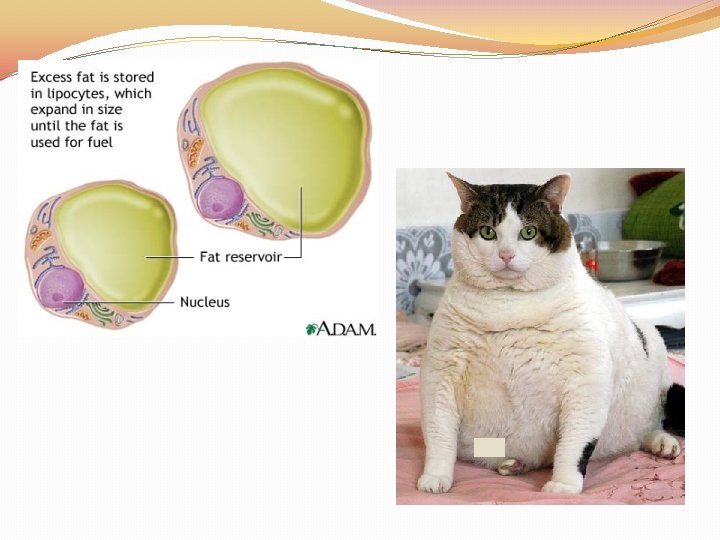
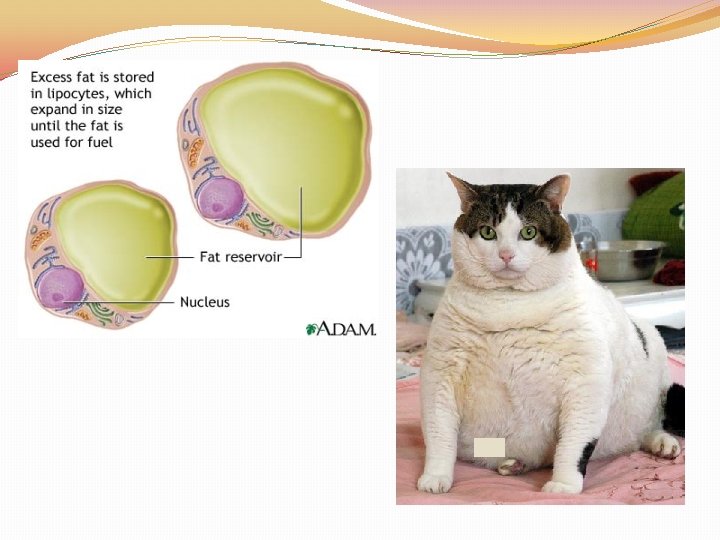
Lipids �Lipids: Fats, Oils, Waxes, Cholesterol, and Steroids �Made up of the elements C, H, and O in the ratio > 2 : 1 for H : O �Lipids are used to STORE and RELEASE energy, INSULATE and PROTECT �The body can release the MOST amount of energy from fat �Lipids DO NOT dissolve in WATER


Lipids �Lipids are made up of two parts: GLYCEROL and FATTY ACID TAILS

SATURATED FAT �Formula: H : O > 2: 1 (MAXIMUM AMOUNT OF H) �Structure: NO C TO C DOUBLE BONDS �At room Temp. : SOLID �Found in: ANIMAL FOOD STUFFS (MEAT/SKIN) AND DAIRY PRODUCTS �Function: INCREASES BLOOD CHOLESTEROL AND INCREASES RISK OF HEART DISEASE


UNSATURATED FAT �Formula: H : O > 2: 1 �Structure: AT LEAST ONE C TO C DOUBLE BOND �At room Temp. : LIQUID �Found in: VEGGIE OILS �Function: LOWERS BLOOD CHOLESTEROL


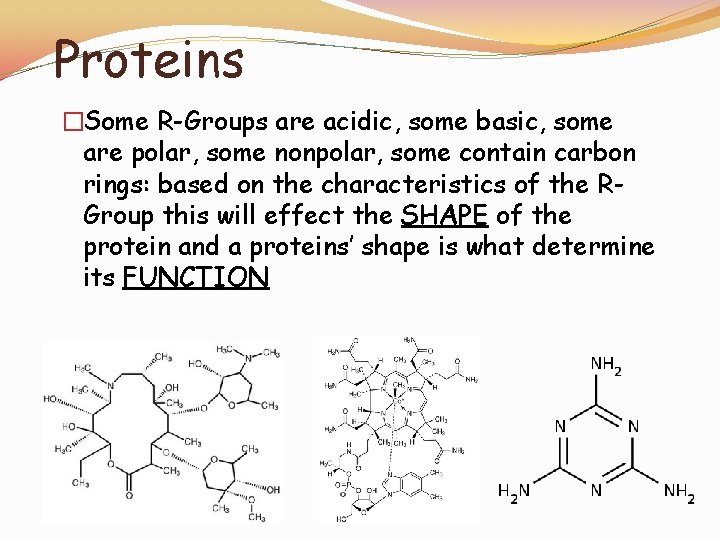
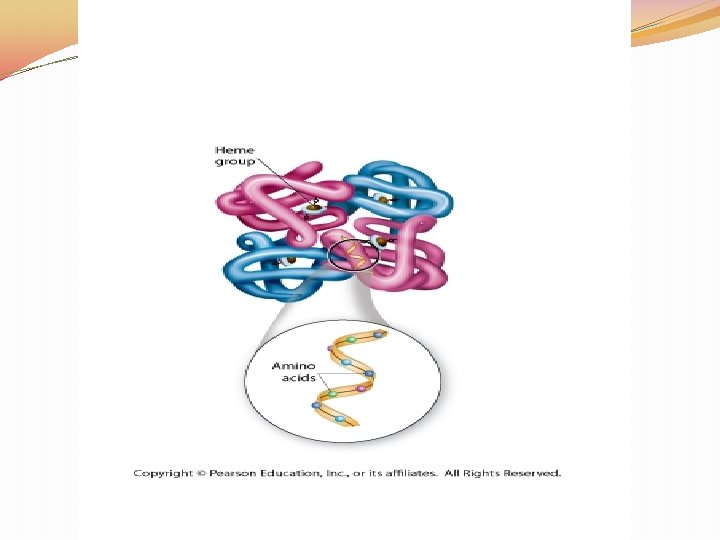
Proteins �Proteins: A large complex and diverse group of macromolecules �Made up of the elements C, H, O, and N �Unlike carbohydrates and lipids, proteins contain NITROGEN

Proteins �Proteins are made up of MONOmers called AMINO ACIDS �There are more than 20 different amino acids found in nature

Proteins �Some R-Groups are acidic, some basic, some are polar, some nonpolar, some contain carbon rings: based on the characteristics of the RGroup this will effect the SHAPE of the protein and a proteins’ shape is what determine its FUNCTION


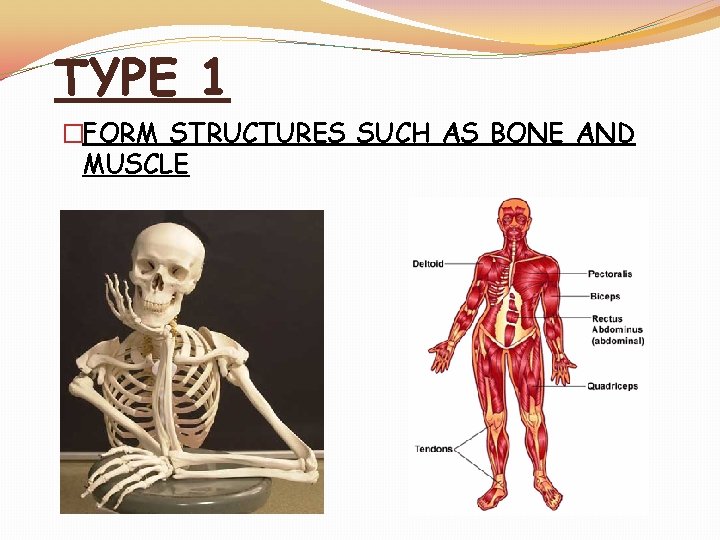
TYPE 1 �FORM STRUCTURES SUCH AS BONE AND MUSCLE

ANTIBODIES �HELP FIGHT DISEASE

TYPE 3 AND TYPE 4 �TRANSPORT SUBSTANCES INTO OR OUT OF CELLS �REGULATE CELL PROCESSES AND CONTROL THE RATE OF REACTIONS

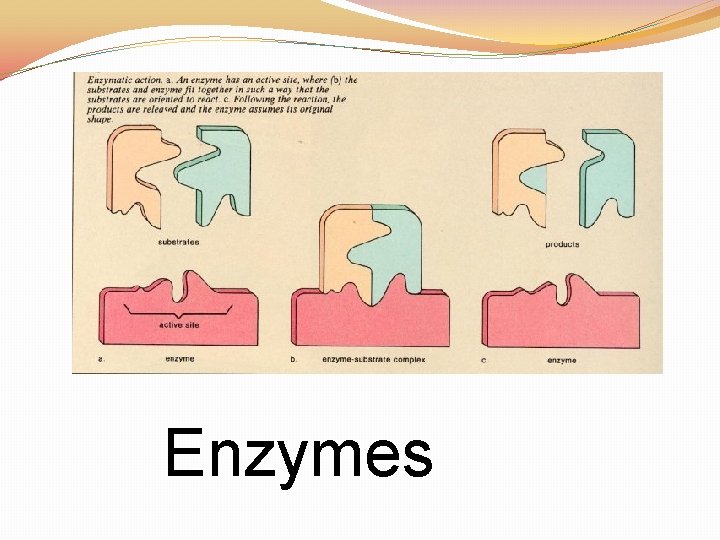
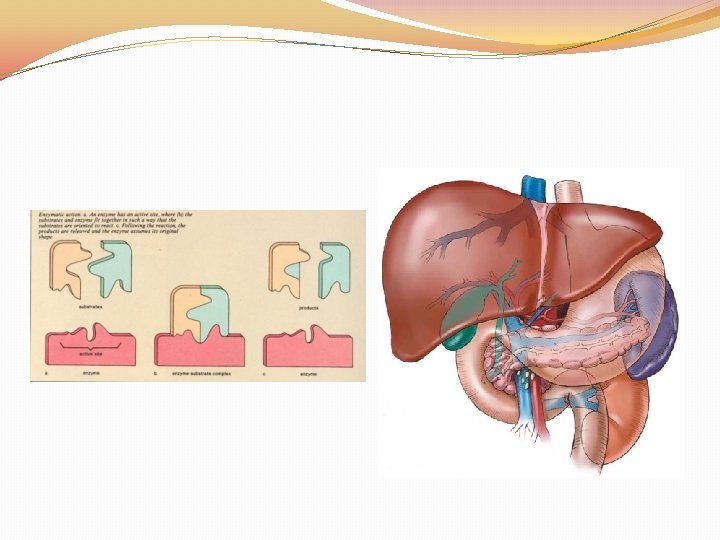
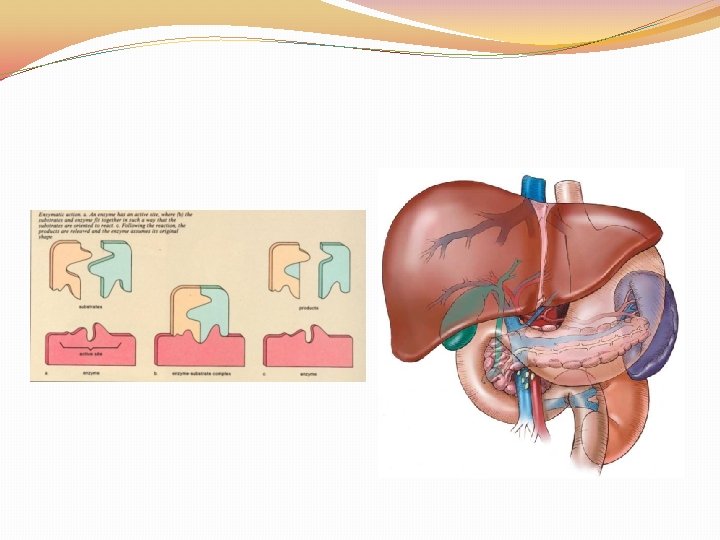
ENZYMES �CATALYST: speed up the rate of a chemical reaction �These are NOT used up during the chemical reaction (can be used over and over again) �Name ends in ASE �Enzymes are inactivated by HEAT and ACIDS (specific p. H)

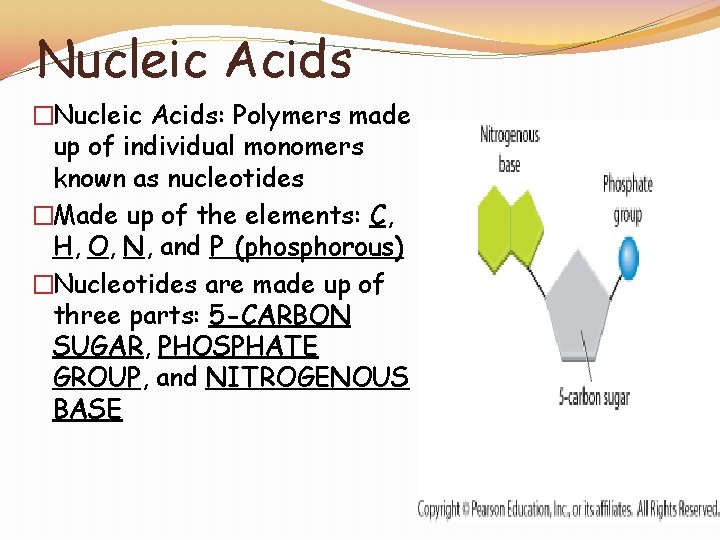
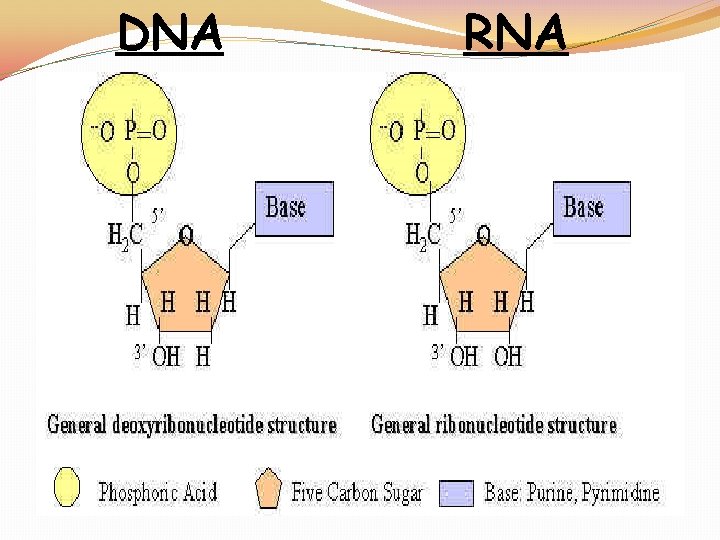
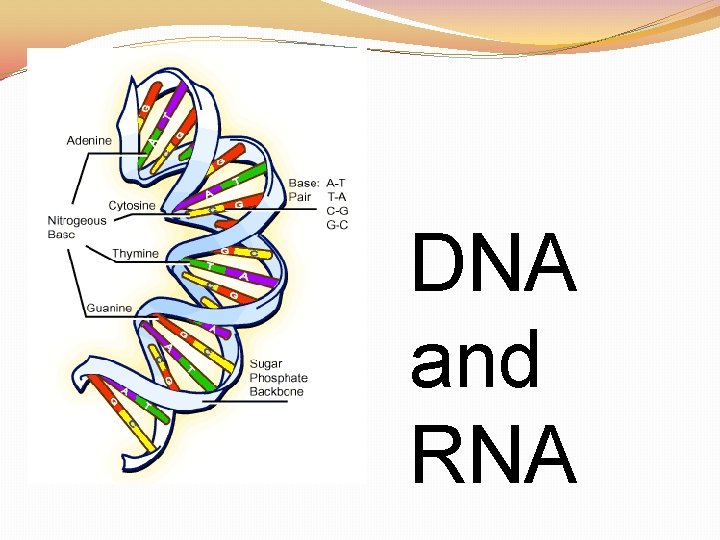
Nucleic Acids �Nucleic Acids: Polymers made up of individual monomers known as nucleotides �Made up of the elements: C, H, O, N, and P (phosphorous) �Nucleotides are made up of three parts: 5 -CARBON SUGAR, PHOSPHATE GROUP, and NITROGENOUS BASE

DNA RNA

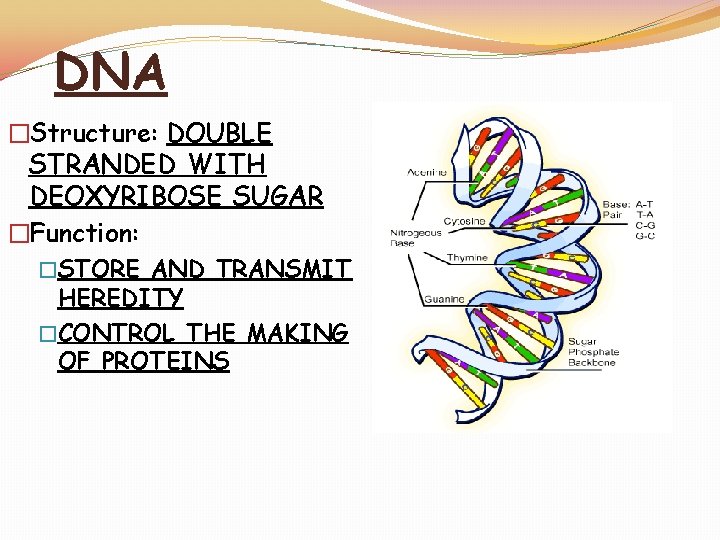
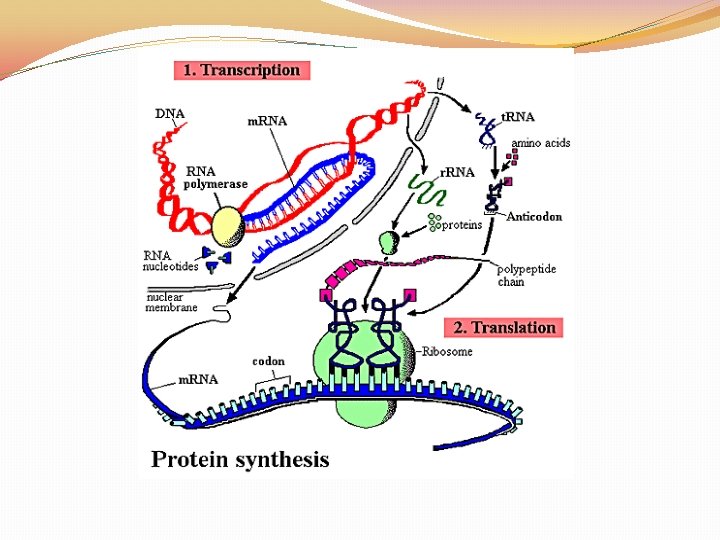
DNA �Structure: DOUBLE STRANDED WITH DEOXYRIBOSE SUGAR �Function: �STORE AND TRANSMIT HEREDITY �CONTROL THE MAKING OF PROTEINS

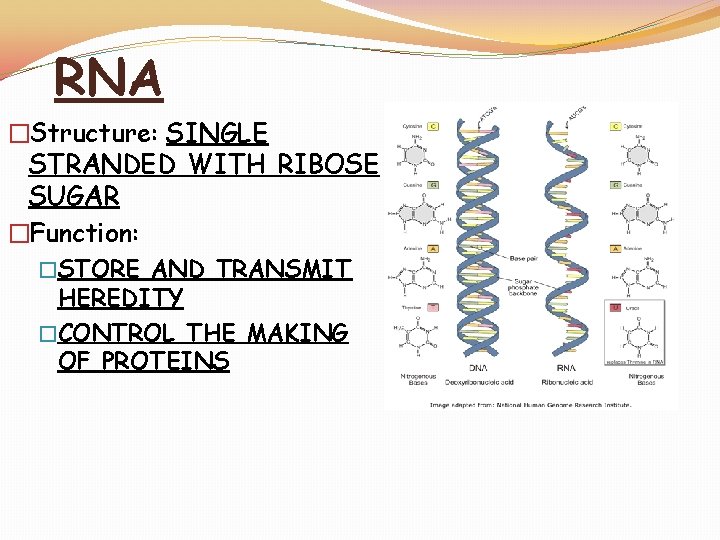
RNA �Structure: SINGLE STRANDED WITH RIBOSE SUGAR �Function: �STORE AND TRANSMIT HEREDITY �CONTROL THE MAKING OF PROTEINS

WHAT’S MY FUNCTION? .

Do not dissolve in water


Lipids

Form structures such as bone and muscle


Proteins

The main source of energy for living things.


Carbohydrates

Can store the greatest amount of energy in your body



Lipids

Helps fight disease

Antibodies

Proteins

Stores and transmits heredity

DNA and RNA

Nucleic Acids

Waxy organic compounds


Lipids

Controls the rate of reactions and regulates cell processes

Enzymes

Proteins

Controls the making of proteins


Nucleic Acids

Fats and oils


Lipids

Enzymes


Proteins

A glucose polymer that forms the cell wall of plants


Cellulose

Polysaccharide

Carbohydrate

A polysaccharide that can be used for energy in plants


Starch

Carbohydrate

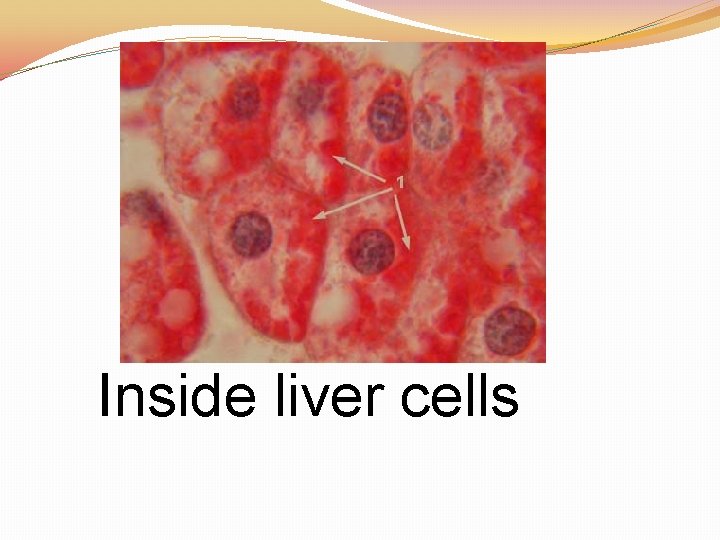
A polysaccharide that animals use to store excess energy

Inside liver cells

Glycogen

Carbohydrate

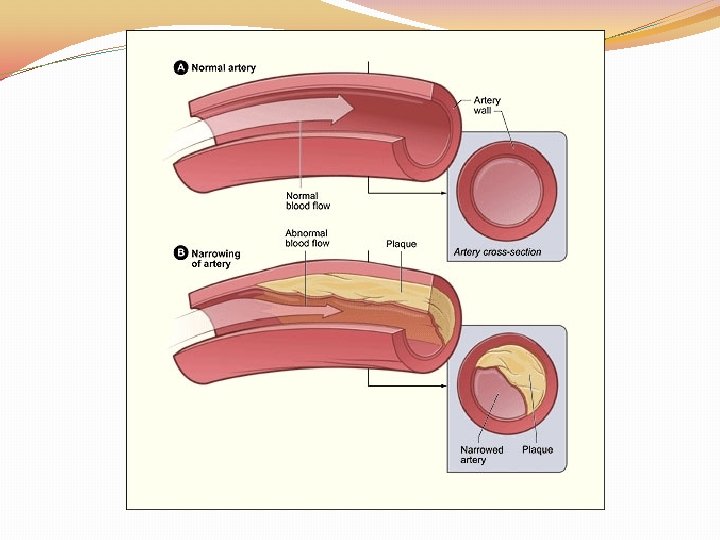
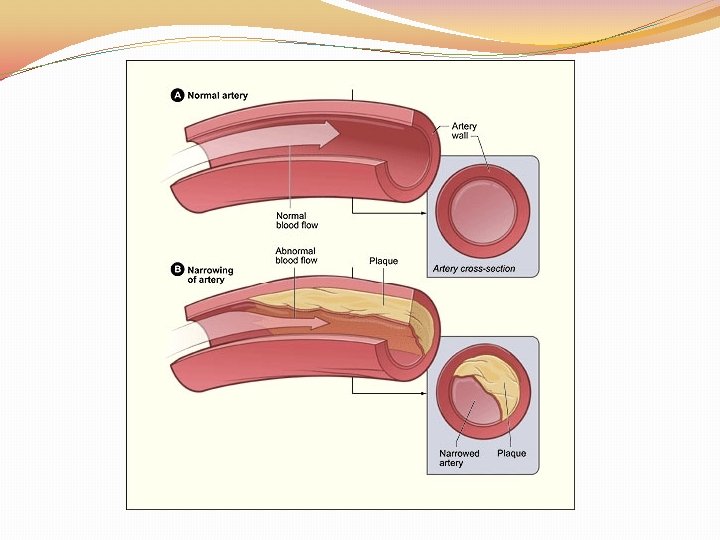
A lipid present in the blood and deposited in arteries


Cholesterol

Lipid

A protein molecule that serves as a catalyst in living things


Enzyme

Protein