Macromolecules Lipids IBBiology Topic 2 3 Meat Alternatives

Macromolecules Lipids IB-Biology Topic 2. 3

Meat & Alternatives What is inside these? Protein. . . Fat. . . What does Protein & Fat look like molecularly?
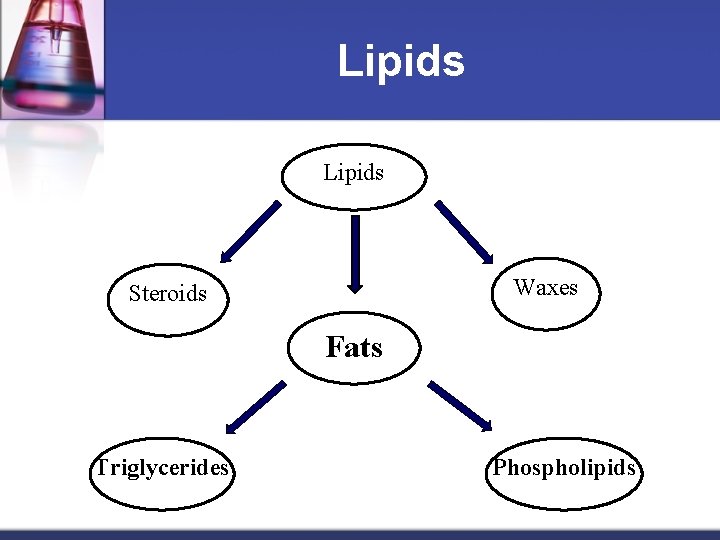
Lipids Waxes Steroids Fats Triglycerides Phospholipids

Lipids’ Importance 1) Insulation: look how round this seal is!

2) Energy Storage: the fat on this bacon was a piggy’s bank of energy
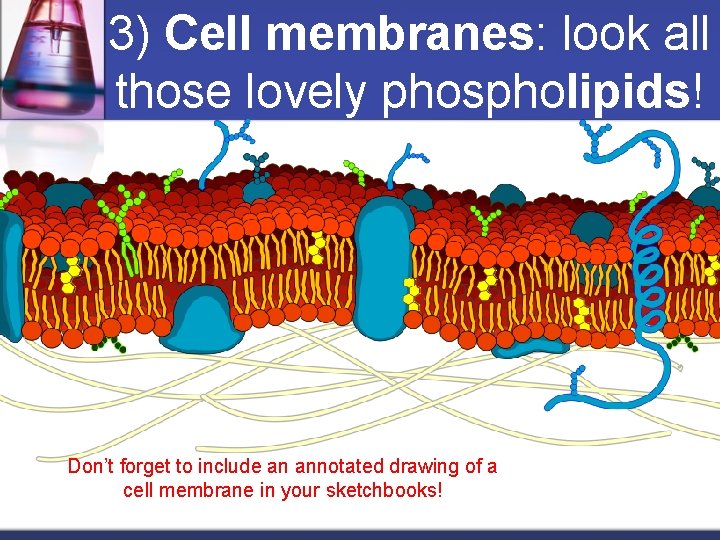
3) Cell membranes: look all those lovely phospholipids! Don’t forget to include an annotated drawing of a cell membrane in your sketchbooks!

Energy Ratio in Lipids Energy Ratio by mass Fat : Carbs : Protein 2: 1: 1 The energy stored in lipids is primarily in the fatty acid side chains, so triglycerides are used for energy storage rather than diglycerides or monoglycerides So it makes the most sense for animals to store long term energy reserves as fat. The same amount of energy stored as carbohydrates would mass twice as much.

Glycogen is the medium-term energy storage molecule in animals. It is stored in the liver and muscles. The energy stored in glycogen is more readily available than the energy stored in fat. Glucose in the bloodstream is for immediate use and will either be used in respiration to yield ATP or converted to glycogen or fat An analogy: in t it y r ou Wallet (Glycogen) Pu You are paid in cash(Glucose) easy to get to, would be too big if you put in all your money Deposit in t he Bank (Fat) Spend it! (Respiration) Can put lots of money here, more of a hassle to get it back out

Unlike most animals, most plants do not need to move, so they store their energy as carbohydrates. Especially plants with storage roots, shoots or tubers

Seeds are the exception, they need to disperse to spread the species. So they use lipids for energy storage. Thus sunflower oil, canola oil, etc.

Lipids’ Importance Stores energy n Insulate and protects vital organs n Fuel n Cell membrane component n Hormones n “I am lipid, hear me roar!” HYDROPHOBIC n 1 gram of fat = 2 x energy of gram of carbs n Excess glucose not converted into glycogen becomes fat! n
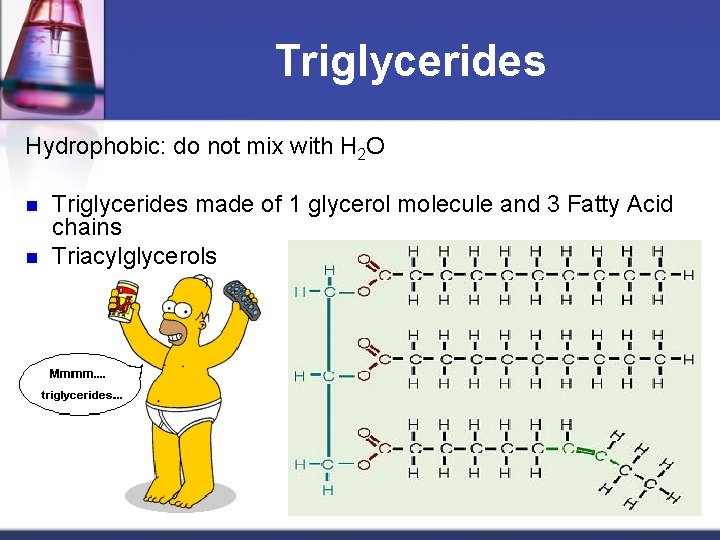
Triglycerides Hydrophobic: do not mix with H 2 O n n Triglycerides made of 1 glycerol molecule and 3 Fatty Acid chains Triacylglycerols
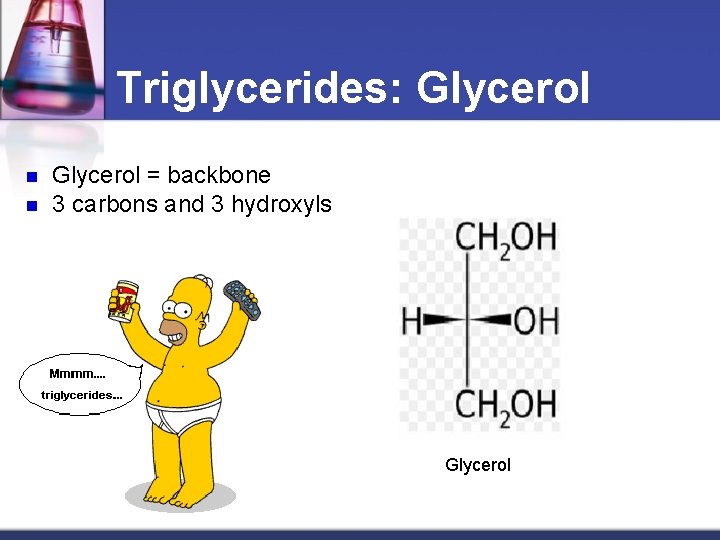
Triglycerides: Glycerol n n Glycerol = backbone 3 carbons and 3 hydroxyls Glycerol
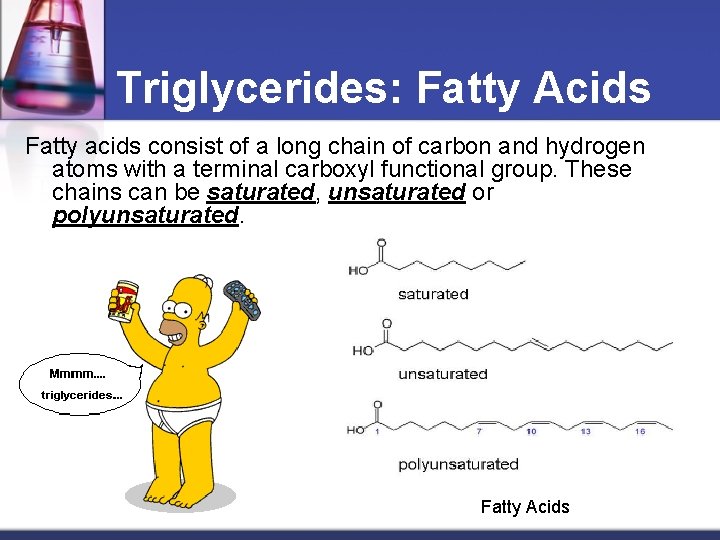
Triglycerides: Fatty Acids Fatty acids consist of a long chain of carbon and hydrogen atoms with a terminal carboxyl functional group. These chains can be saturated, unsaturated or polyunsaturated. Fatty Acids

Unsaturated FA (Fats) Healthy (Athlete) - Unsaturated - Multiple bond carbons - Missing hydrogens - Nuts, fish, avocado, olive oil, etc.
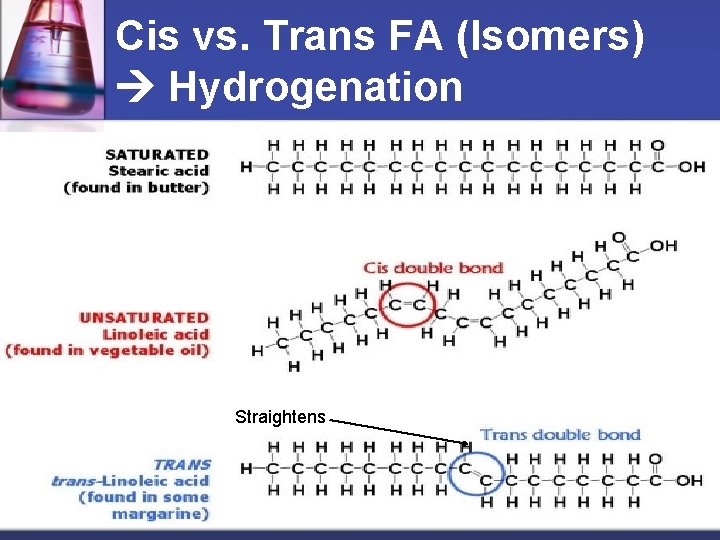
Cis vs. Trans FA (Isomers) Hydrogenation Straightens

Omega-3 and Omega-6 Omega-end

Saturated FA (Fats) Unhealthy (Homer) - Saturated - Single bond carbons - Maximum hydrogens - Animal fat, dairy fat, fried food, etc.
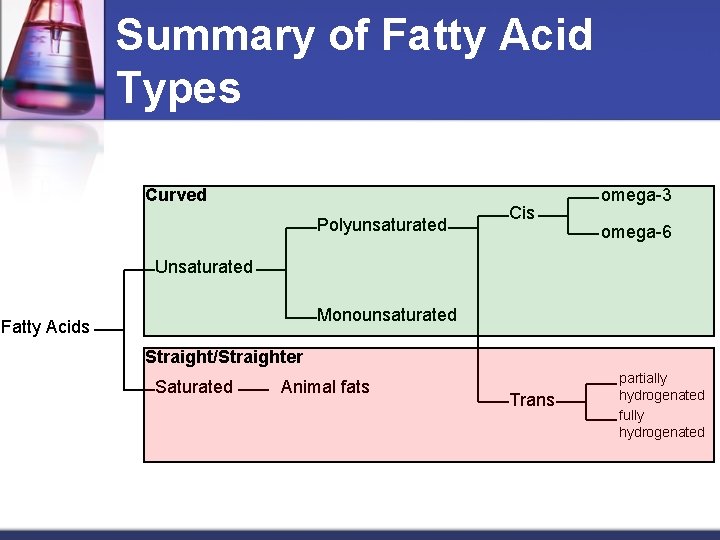
Summary of Fatty Acid Types Curved Polyunsaturated Cis omega-3 omega-6 Unsaturated Monounsaturated Fatty Acids Straight/Straighter Saturated Animal fats Trans partially hydrogenated fully hydrogenated

Triglyceride Synthesis Dehydration Synthesis; (removal of H 2 O) Ester Linkage; (between Carbon & Oxygen)

Triglyceride Synthesis
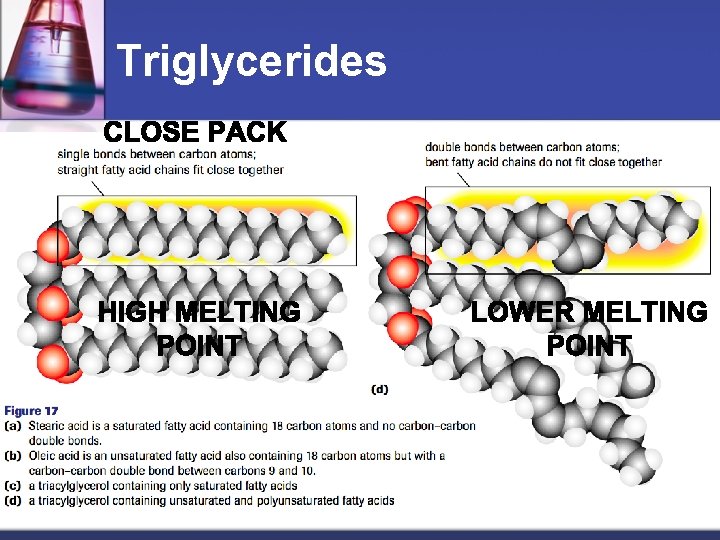
Triglycerides

SPECIAL CASE Special Case. . .

Phospholipids Glycerol + FFA + PO 4 - (phosphate)

Steroids n Steroids are an important group of lipids that consist of four linked carbon rings. Steroids are hydrophobic and include cholesterol and cortisol as well as hormones such as estrogen and testosterone. Steroids are naturally produced in the body and are necessary for proper growth and development

Waxes n Waxes are composed of long chains of fatty acids that are attached to an alcohol or a carbon ring. Waxes are hydrophobic and are firm yet pliable.
- Slides: 26