MACROMOLECULES Carbohydrates and Lipids Mr Winch Centre Wellington

MACROMOLECULES Carbohydrates and Lipids Mr. Winch Centre Wellington DHS

Types of Carbohydrates 1. 2. 3. Monosaccharides Disaccharides Polysaccharides

Carbs… Monosaccharides “ONE” “SUGAR” v simple sugar containing 3 to 7 carbons v examples: glucose, fructose, galactose v provides instant energy “STRAIGHT CHAIN” FORM “RING” FORM (in water)

Carbs… Characteristics of Sugars 1) An –OH group is attached to each carbon except one; this carbon is double bonded to an oxygen (carbonyl group Start numbering the carbons of your sugars at the end closest to the carbonyl group!!! 1 1 2 2 3 3 4 4 5 5 6 6

Carbs… Characteristics of Sugars 2) Size of carbon skeleton varies from 3 to 7 carbons. Classification Number of Carbons Example Triose 3 Glyceraldehyde Pentose 5 Ribose Hexose 6 Glucose

Carbs… Characteristics of Sugars 3) Spatial arrangements around asymmetric carbon may vary. For example, glucose and galactose are stereoisomers. Can you spot the only difference? ? ? Notice how the –OH’s are on different sides. This affects the properties of these molecules & makes them chemically different 1 1 2 2 3 3 4 4 5 5 6 6

Carbs… Characteristics of Sugars 4) In aqueous solutions, many monosaccharides form rings. Equilibrium favours the ring structure. The #5 C –OH reacts within the molecule at the terminal aldehyde group, breaking the double bond and creates a closed ring 6 5 1 2 4 2 3 4 5 6 3 1
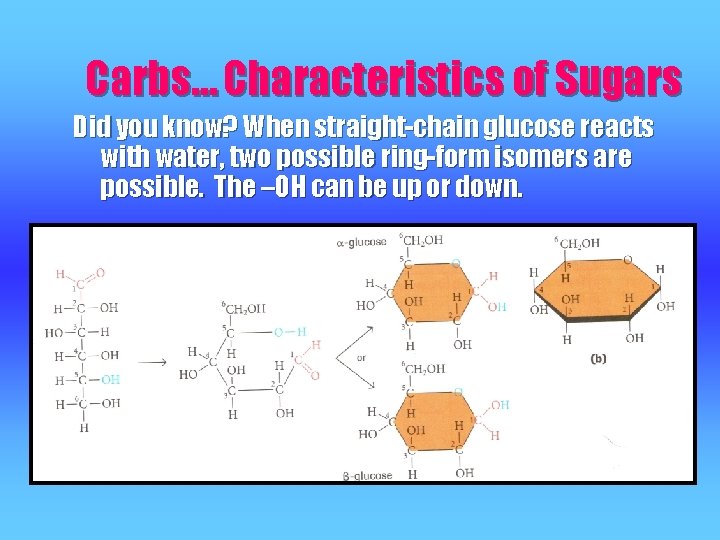
Carbs… Characteristics of Sugars Did you know? When straight-chain glucose reacts with water, two possible ring-form isomers are possible. The –OH can be up or down.

Carbs… Dissaccharides “TWO” “SUGARS” made up of two monosaccharides v common examples: glucose + glucose = maltose (malt sugar) glucose + fructose = sucrose (table sugar) glucose + galactose = lactose (milk sugar) v MOO!!!
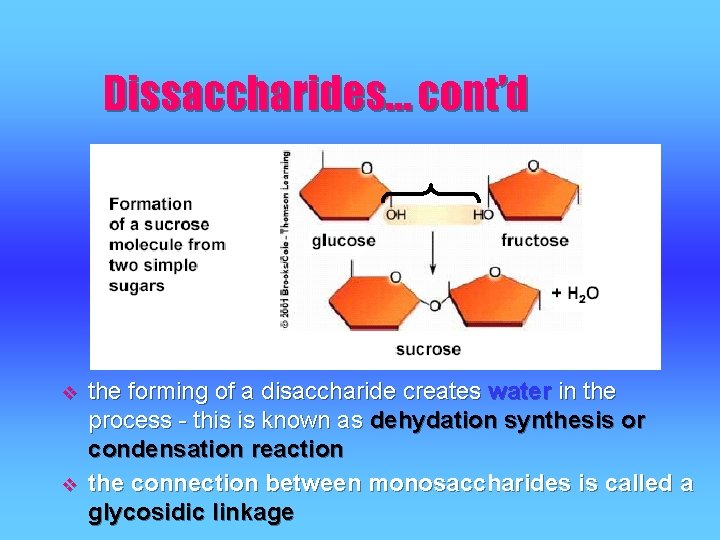
Dissaccharides… cont’d v v the forming of a disaccharide creates water in the process - this is known as dehydation synthesis or condensation reaction the connection between monosaccharides is called a glycosidic linkage

Polysaccharides “MANY” “SUGARS” v a complex carbohydrate consisting of many simple sugars linked together Functions: Energy Storage Starch - in plants Glycogen - in animals Structural Support Cellulose – component of plant cell wall Chitin – exoskeleton of arthropods
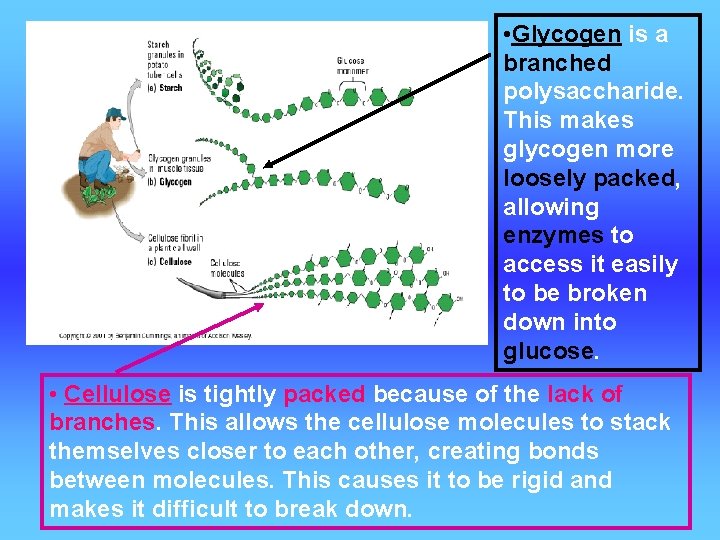
• Glycogen is a branched polysaccharide. This makes glycogen more loosely packed, allowing enzymes to access it easily to be broken down into glucose. • Cellulose is tightly packed because of the lack of branches. This allows the cellulose molecules to stack themselves closer to each other, creating bonds between molecules. This causes it to be rigid and makes it difficult to break down.

Macromolecule #2 LIPIDS Functions: v Long-term energy storage (triglycerides) v Form cell membrane (phospholipids) v Messaging (steroids act as hormones) v Insulation v Cushioning of Internal Organs

Lipids Why are lipids well suited for long term energy storage? v Contain many high energy bonds between carbon and hydrogen v Contain twice as much energy per gram than carbohydrates (very concentrated)

Types of Lipids 1. 2. 3. Fats (triglycerides) Phospholipids Steroids

Fats v Made up of glycerol and 3 fatty acids. v There are many kinds of fatty acids. They differ in two ways: 1. In length 2. In the # of hydrogen atoms attached to it
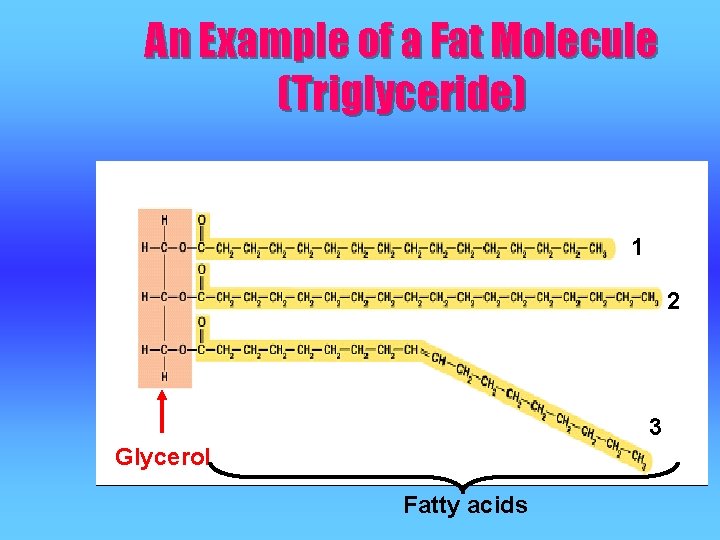
An Example of a Fat Molecule (Triglyceride) 1 2 3 Glycerol Fatty acids

Digestion of a Fat Molecule + 3 H 2 O triglyceride Fig 1. 21 p. 18 + glycerol The above is a hydrolysis reaction. What is the reverse of this reaction called? 3 fatty acids

Where have you seen these fats? Glycerol + 1 Fatty Acid Monoglyceride + H 2 O Glycerol + 2 Fatty Acids Diglyceride + 2 H 2 O Glycerol + 3 Fatty Acids Triglyceride + 3 H 2 O

Types of Fatty Acids SATURATED (palmitic acid) v No double bonds between carbons MONOUNSATURATED (oleic acid) v 1 double bond POLYUNSATURATED (linoleic acid) v More than 2 double bonds
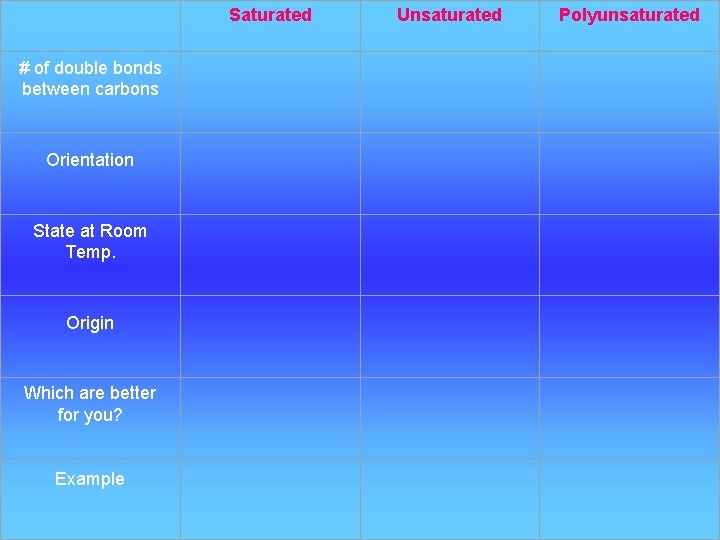
Saturated Unsaturated # of double bonds between carbons Orientation Polyunsaturated State at Room Temp. Origin Which are better for you? Example
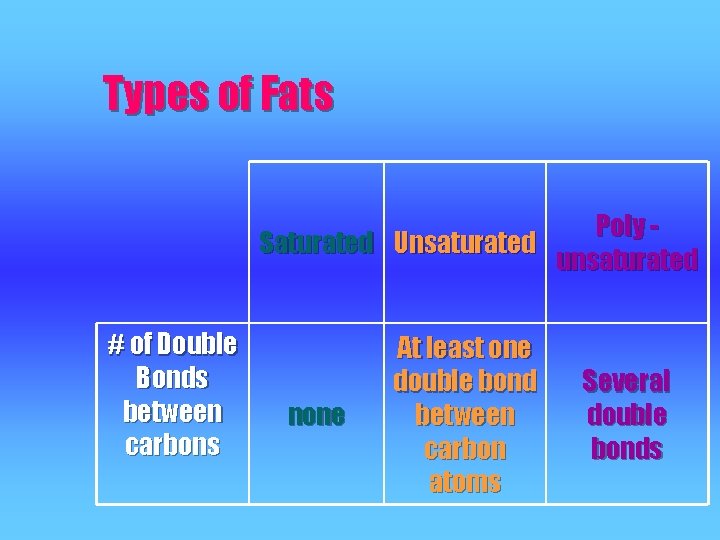
Types of Fats Poly Saturated Unsaturated unsaturated # of Double Bonds between carbons none At least one double bond between carbon atoms Several double bonds

Types of Fats… cont’d Poly Saturated Unsaturated unsaturated Orientation of Fatty Acids Straight chains Kinks / bends at the double bonds

Types of Fats… cont’d Poly Saturated Unsaturated unsaturated State at Room Temp. Solid (tightly packed) Liquid (less tightly packed) Liquid (even less tightly packed)

Types of Fats… cont’d Poly Saturated Unsaturated unsaturated Origin Animals (meat, dairy) Plants

Types of Fats… cont’d Poly Saturated Unsaturated unsaturated Bad for Which are healthier? Stored in adipose tissue Healthier

Types of Fats… cont’d Poly Saturated Unsaturated unsaturated Examples Butter, lard Olive oil, vegetable oil, peanut oil, canola oil (genetically modified)

What up with TRANS-FATS? NOT GOOD FOR YOU!!! (FRIES, PEANUT BUTTER) v Created from oils (unsaturated) that are hydrogenated (hydrogen added to double bonds) v Done to increase shelf life, flavour, & workability (eg. semi-solid for baking) v Only partial hydrogenation occurs & get a change in the orientation of hydrogens around some double bonds (cis- turns into trans) v This fat is packaged by your body as LDL (aka BAD cholesterol) leaving you @ risk for heart disease, artheriosclerosis, diabetes & obesity v

Type of Lipids #2 PHOSPHOLIPIDS v are fat derivatives in which one fatty acid has been replaced by a phosphate group and one of several nitrogencontaining molecules. v an important part of the cell membrane (phospholipid bilayer)
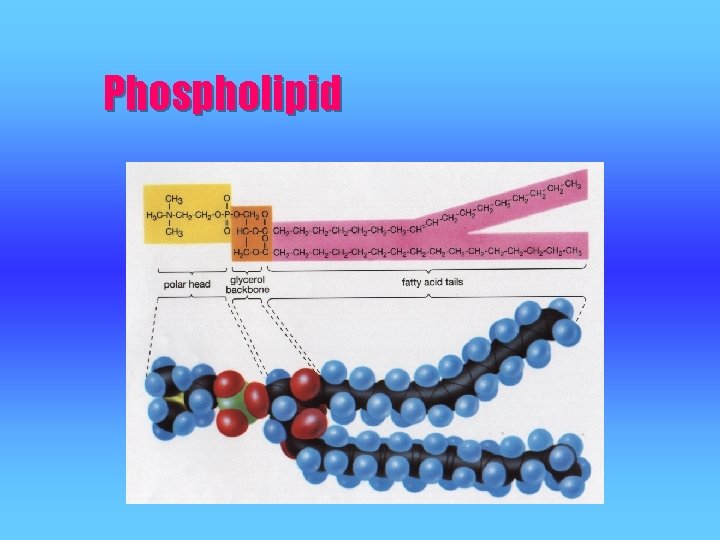
Phospholipid

Phospholipid • The phospholipid can also be represented as: Polar Head – hydrophilic (water-loving) Non-Polar Tails (fatty acids) - hydrophobic (water-hating)
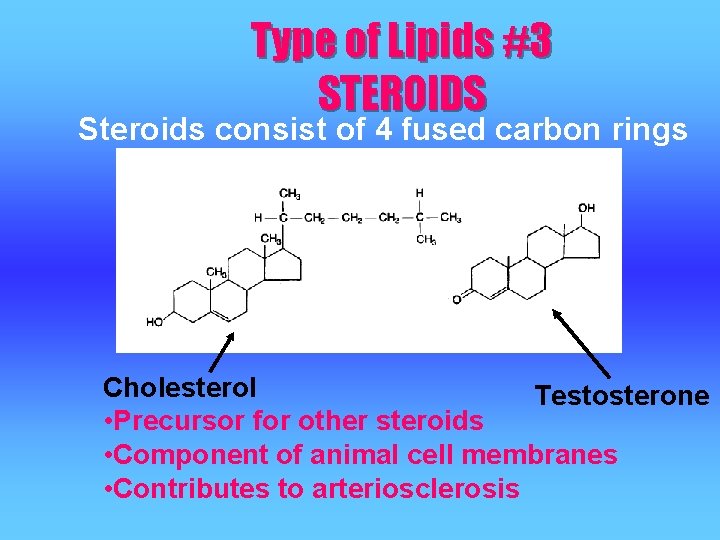
Type of Lipids #3 STEROIDS Steroids consist of 4 fused carbon rings Cholesterol Testosterone • Precursor for other steroids • Component of animal cell membranes • Contributes to arteriosclerosis

MACROMOLECULES Proteins Mr. Blair Winch Centre Wellington DHS

Proteins FUNCTIONS v Act as enzymes (to control chemical reactions) v Provide support and help shape cells v Act as transporters (hemoglobin) v Act as hormones v Make up structures (hair, cartilage) v Act as antibodies (immunoglobulins)
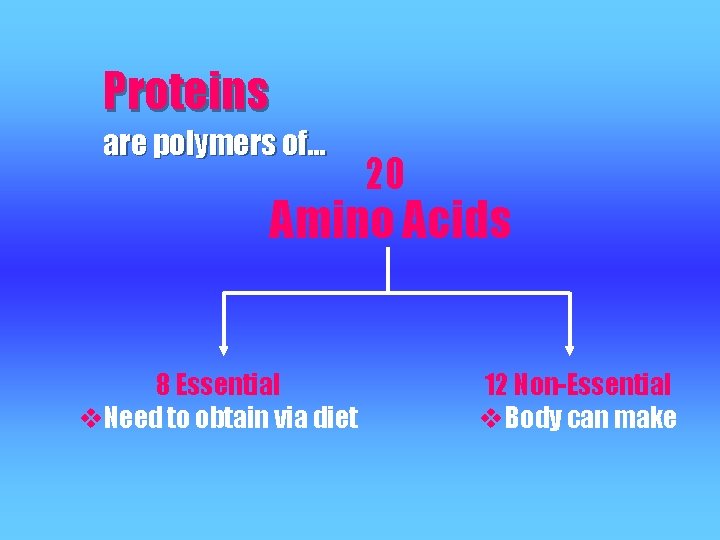
Proteins are polymers of… 20 Amino Acids 8 Essential v. Need to obtain via diet 12 Non-Essential v. Body can make
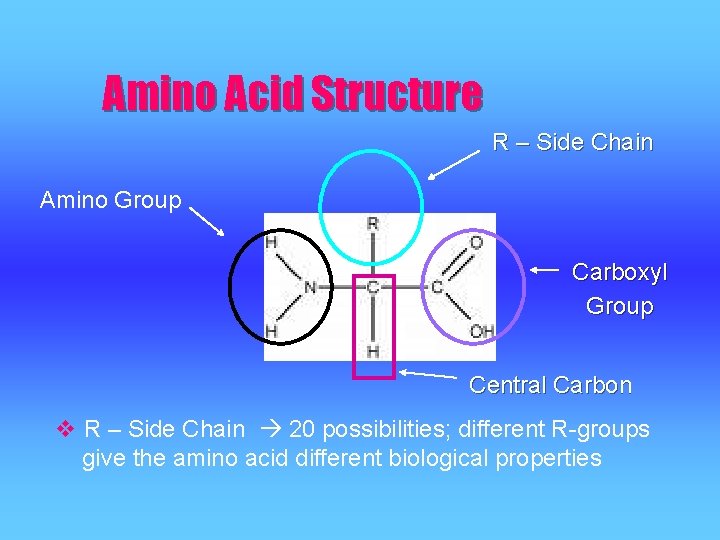
Amino Acid Structure R – Side Chain Amino Group Carboxyl Group Central Carbon v R – Side Chain 20 possibilities; different R-groups give the amino acid different biological properties

Proteins Are often very large polymers of many amino acids (monomers) linked together to form POLYPEPTIDES v Proteins are built by condensation reactions forming peptide bonds. v aa 1 – aa 2 -- aa 3 – aan v dipeptide tripeptide polypeptide Proteins are broken down through hydrolysis reactions

Polypeptides Polypeptide Chain Amino Acid Peptide Bonds (Amide Linkage) v. This polypeptide will begin to fold over on itself until it has reached its 3 -dimensional shape v. The folding will be determined by the R-group interactions of the specific amino acid sequence v. It is only the final shape that will determine the protein’s specific function
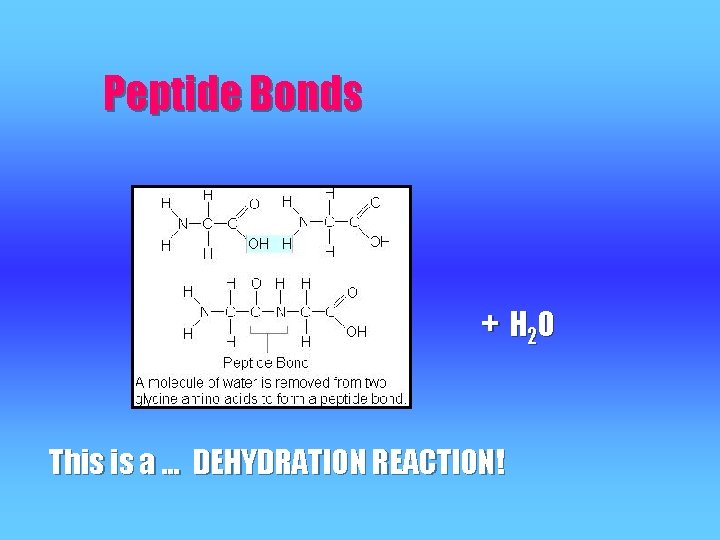
Peptide Bonds + H 2 O This is a … DEHYDRATION REACTION!
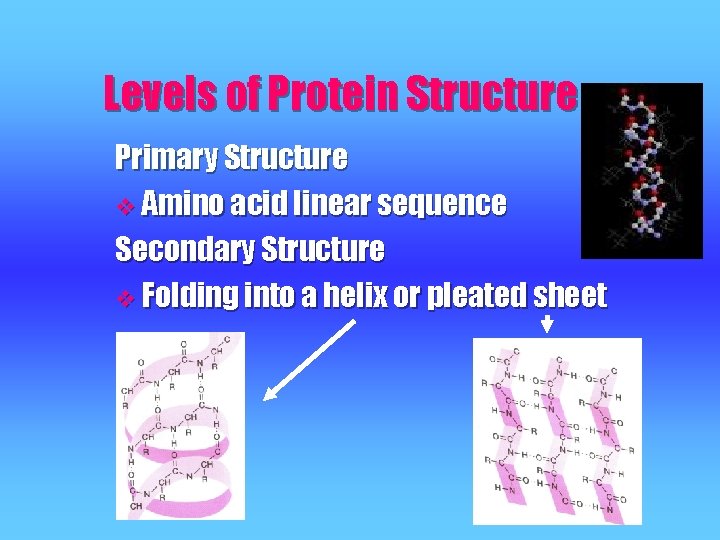
Levels of Protein Structure Primary Structure v Amino acid linear sequence Secondary Structure v Folding into a helix or pleated sheet
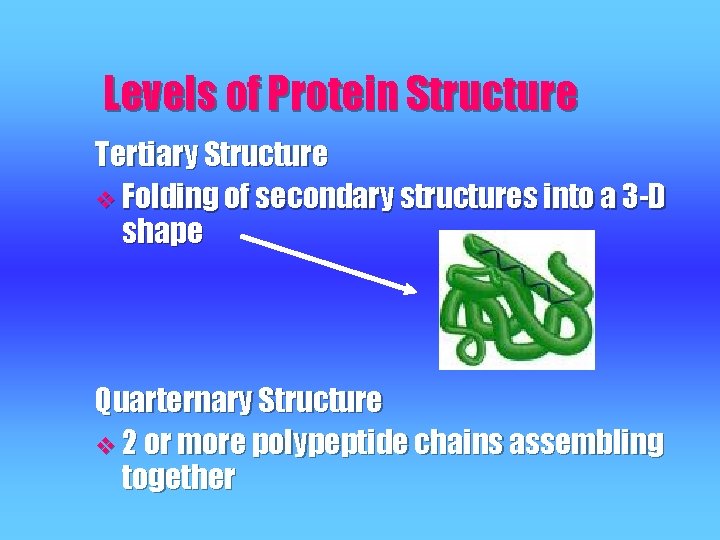
Levels of Protein Structure Tertiary Structure v Folding of secondary structures into a 3 -D shape Quarternary Structure v 2 or more polypeptide chains assembling together

Hemoglobin – A Complex Protein v Found in RBCs, is responsible for oxygen transport to your cells for respiration v Scientists believe the protein dates back 4 billion years to the start of life v A quarternary protein consisting of 4 polypeptides v 2 of the polypeptides (α subunits) contain 141 amino acids while the other 2 (β subunits) contain 146 amino acids
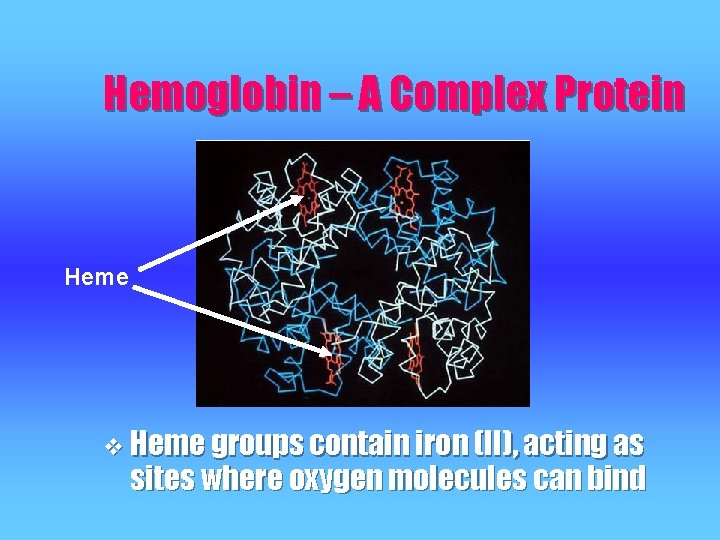
Hemoglobin – A Complex Protein Heme v Heme groups contain iron (II), acting as sites where oxygen molecules can bind
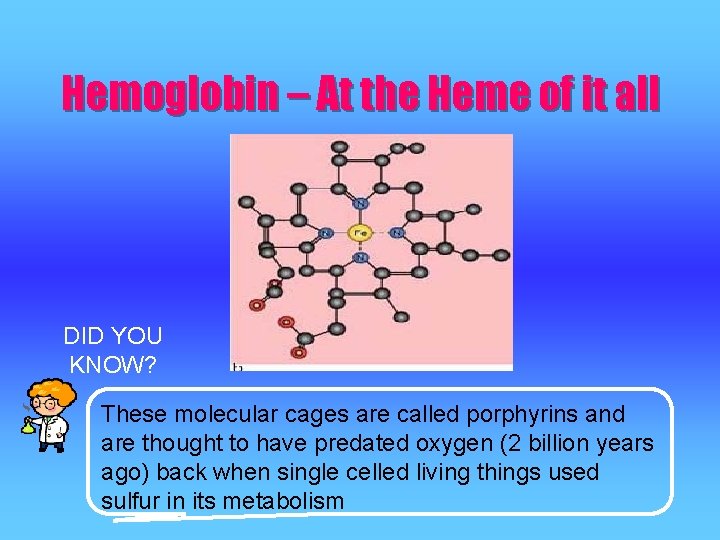
Hemoglobin – At the Heme of it all DID YOU KNOW? These molecular cages are called porphyrins and are thought to have predated oxygen (2 billion years ago) back when single celled living things used sulfur in its metabolism
- Slides: 44