Macromolecules Biomolecules Building Blocks of Life Monomers Build

Macromolecules/ Biomolecules Building Blocks of Life

Monomers Build Polymers � Monomers: Small units that join together. Also referred to as building blocks � Polymers: Large compounds built from monomers › Ex: carbohydrates, lipids, proteins, nucleic acids Monomers Polymers

Carbohydrates � Made up of C, H, O � Function: source of energy (short term) � Examples: sugar, rice, bread, potatoes � Monomer: monosaccharide (simple sugar)
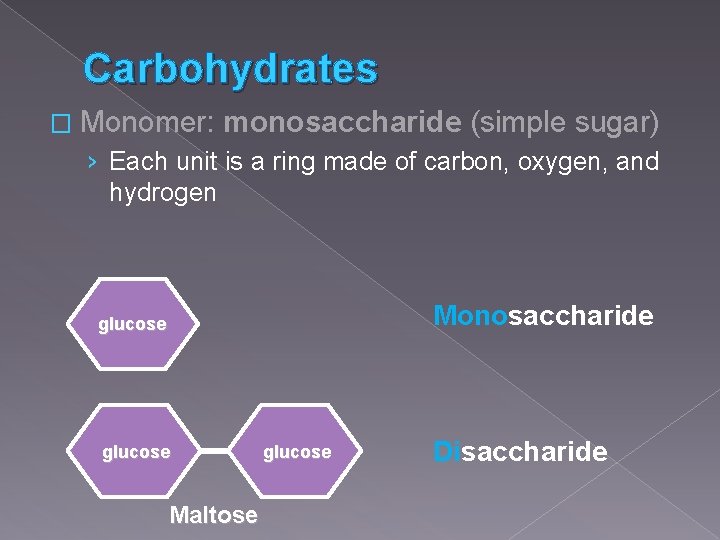
Carbohydrates � Monomer: monosaccharide (simple sugar) › Each unit is a ring made of carbon, oxygen, and hydrogen Monosaccharide glucose Maltose glucose Disaccharide
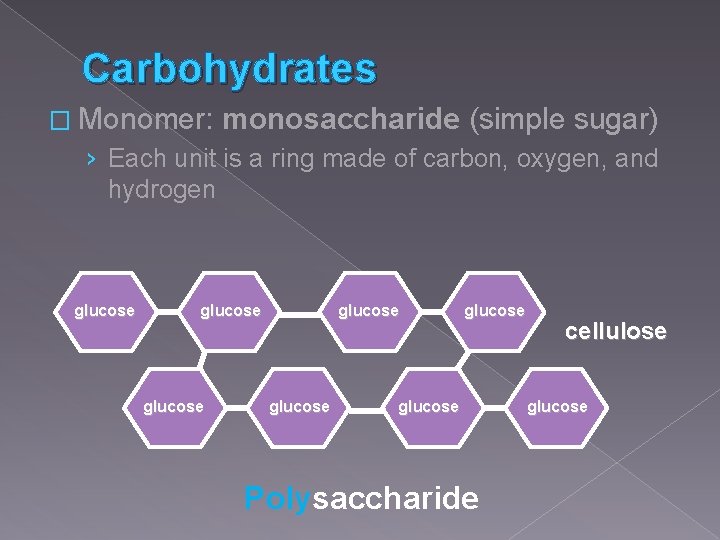
Carbohydrates � Monomer: monosaccharide (simple sugar) › Each unit is a ring made of carbon, oxygen, and hydrogen glucose glucose Polysaccharide cellulose glucose
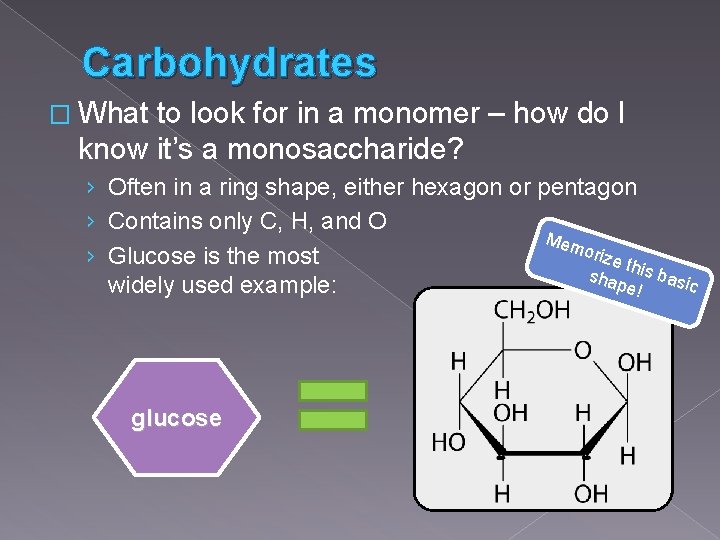
Carbohydrates � What to look for in a monomer – how do I know it’s a monosaccharide? › Often in a ring shape, either hexagon or pentagon › Contains only C, H, and O Mem oriz › Glucose is the most e th sha is basi pe! c widely used example: glucose

Lipids � Made of C, H, O � Functions: Store energy (long term), building blocks of membranes � Examples: wax, oil, butter, steroids, makeup � Monomers: glycerol and fatty acids
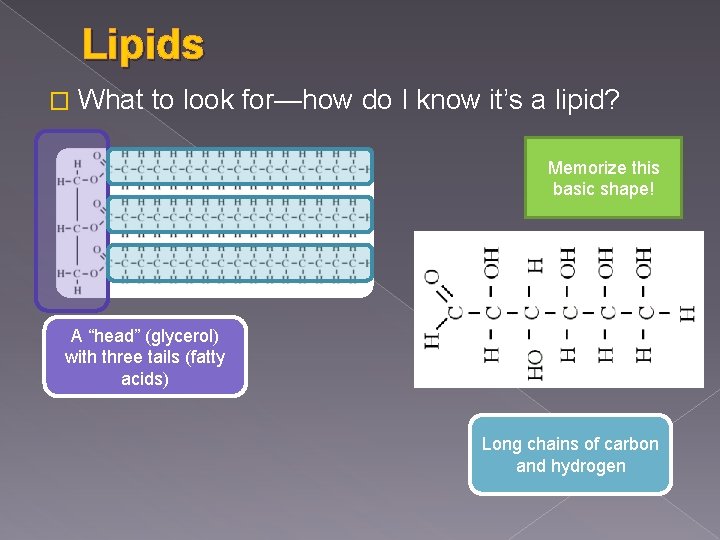
Lipids � What to look for—how do I know it’s a lipid? Memorize this basic shape! A “head” (glycerol) with three tails (fatty acids) Long chains of carbon and hydrogen

Proteins � Made of C, H, O, N � Functions: Build body structures, control chemical reactions, do cellular work � Example: meat/muscle, hair, nails, enzymes, peanut butter, milk � Monomer: amino acid
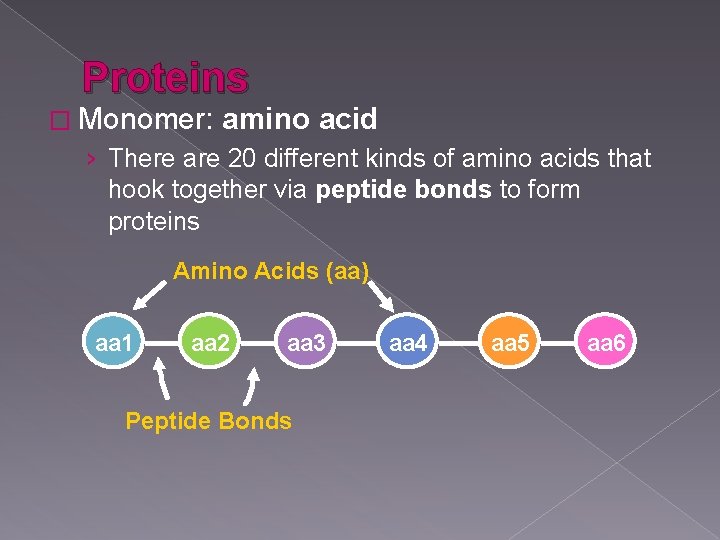
Proteins � Monomer: amino acid › There are 20 different kinds of amino acids that hook together via peptide bonds to form proteins Amino Acids (aa) aa 1 aa 2 aa 3 Peptide Bonds aa 4 aa 5 aa 6
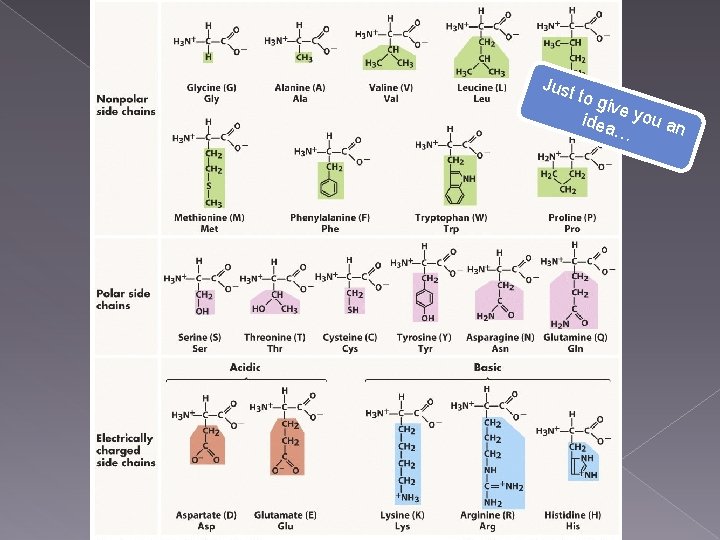
Just to g ive y ou a idea n …
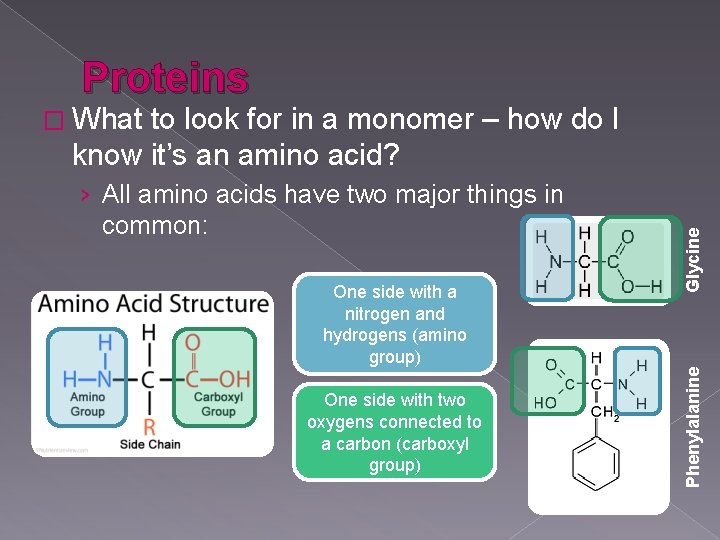
Proteins to look for in a monomer – how do I know it’s an amino acid? One side with a nitrogen and hydrogens (amino group) One side with two oxygens connected to a carbon (carboxyl group) Phenylalanine › All amino acids have two major things in common: Glycine � What

Building/Breaking down Polymers � Building Polymers= Dehydration Synthesis “Taking away water” � Breaking the Polymers= Hydrolysis “Adding Water”

Activity! � If you received a paper on the way in, come to the front of the classroom, bring your paper!
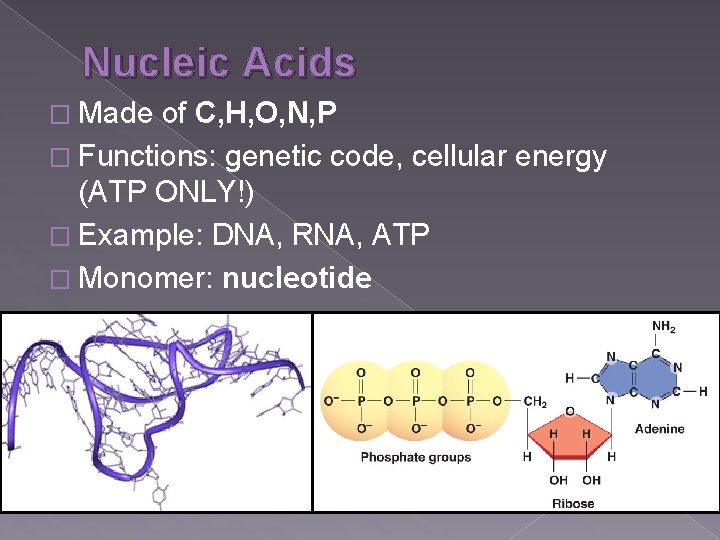
Nucleic Acids � Made of C, H, O, N, P � Functions: genetic code, cellular energy (ATP ONLY!) � Example: DNA, RNA, ATP � Monomer: nucleotide
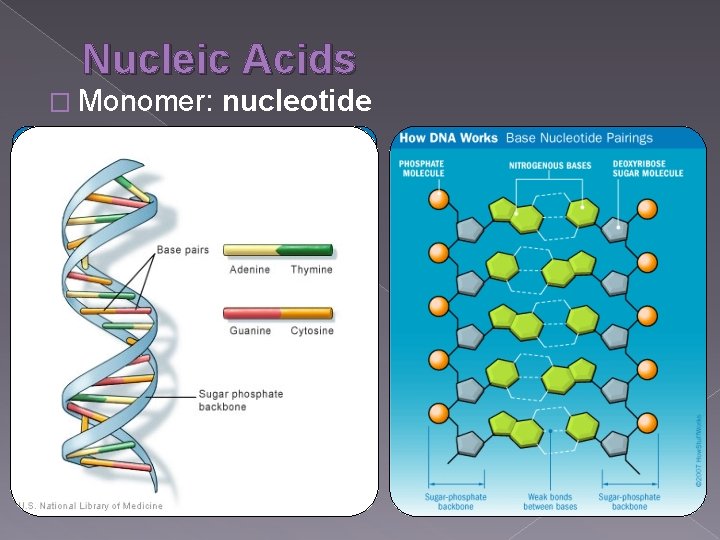
Nucleic Acids � Monomer: nucleotide
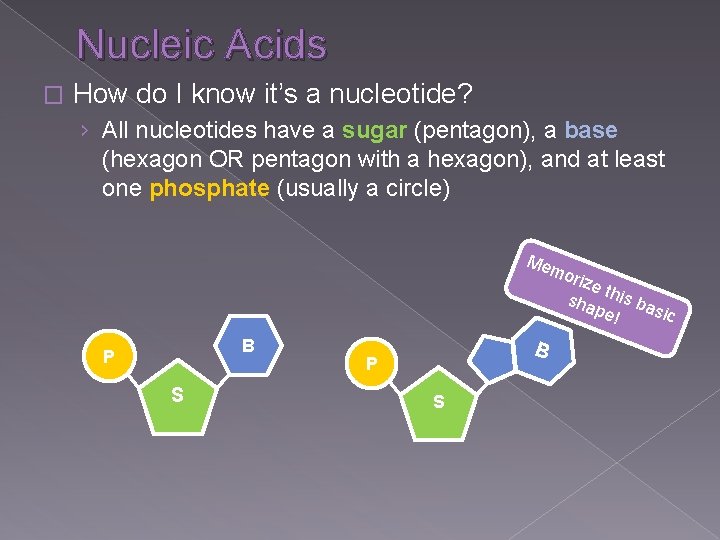
Nucleic Acids � How do I know it’s a nucleotide? › All nucleotides have a sugar (pentagon), a base (hexagon OR pentagon with a hexagon), and at least one phosphate (usually a circle) Me mo B P S rize sha this ba pe! sic

Nucleic Acids � How do I know it’s a nucleotide? › All nucleotides have a sugar (pentagon), a base (hexagon OR pentagon with a hexagon), and at least one phosphate (usually a circle) › A special nucleotide called ATP has three phosphates instead of one P P B P S
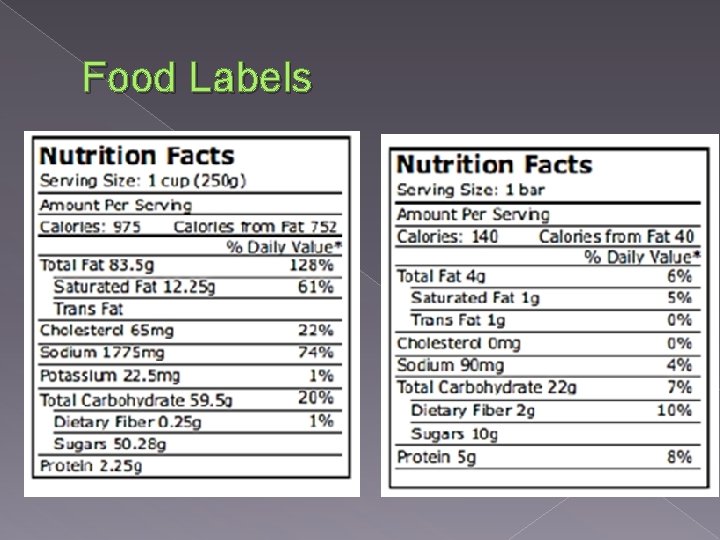
Food Labels

� 1. Glue in Macromolecules Sheet � 2. Put away Notebook � 3. Get out Biomolecules packet and a pencil and highlighter

� Glue in Biomolecules Grid Sheet � Pack up � Keep out the Food Label Investigation sheet

Stomach Contents Carbs Positive Lipids Positive Sugar Positive Proteins Positive
- Slides: 22