Macromolecules BIIIIIG MOLECULES Macromolecules The Legos of the

Macromolecules BIIIIIG MOLECULES

Macromolecules: The Legos of the Cellular world. What they are. - Large molecules that are used to make all living things. - There are 4 types - Proteins - Carbohydrates - Lipids - Nucleic Acids

Macromolecules: The Legos of the Cellular world. Made of Monomers: - Single, small molecules. - Similar to Atoms in Chemistry or a single lego block

Polymers Monomers combine to make Polymers - Long chains of monomers - Like making a lego Sculpture. The four types of macromolecules are all Polymers Other Polymers include: Nylon, Plastic, Adhesives
Carbohydrates YEAH TOAST!!!

Structures • Always end in -ose • Carbohydrates exist as: – Monosaccharides • Single structure which can contain up to 8 carbon atoms • Not degraded by hydrolysis – Disaccharides • Two monosaccharides bonded together – Polysaccharides • More than two monosaccharides bonded together
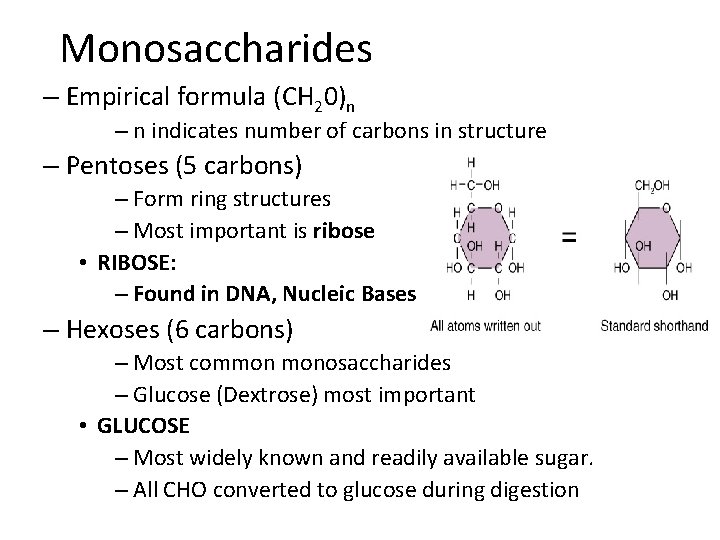
Monosaccharides – Empirical formula (CH 20)n – n indicates number of carbons in structure – Pentoses (5 carbons) – Form ring structures – Most important is ribose • RIBOSE: – Found in DNA, Nucleic Bases – Hexoses (6 carbons) – Most common monosaccharides – Glucose (Dextrose) most important • GLUCOSE – Most widely known and readily available sugar. – All CHO converted to glucose during digestion
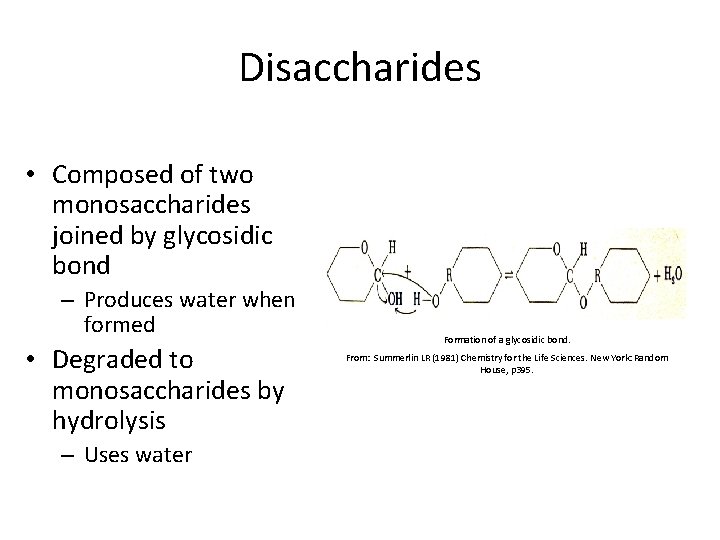
Disaccharides • Composed of two monosaccharides joined by glycosidic bond – Produces water when formed • Degraded to monosaccharides by hydrolysis – Uses water Formation of a glycosidic bond. From: Summerlin LR (1981) Chemistry for the Life Sciences. New York: Random House, p 395.
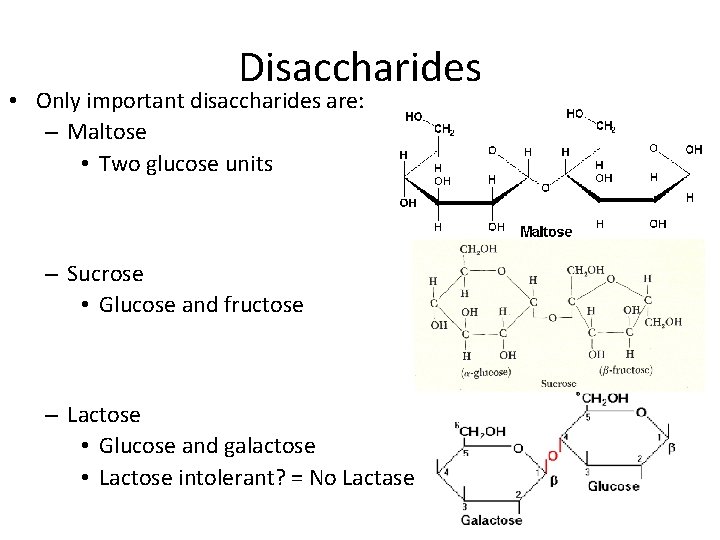
Disaccharides • Only important disaccharides are: – Maltose • Two glucose units – Sucrose • Glucose and fructose – Lactose • Glucose and galactose • Lactose intolerant? = No Lactase
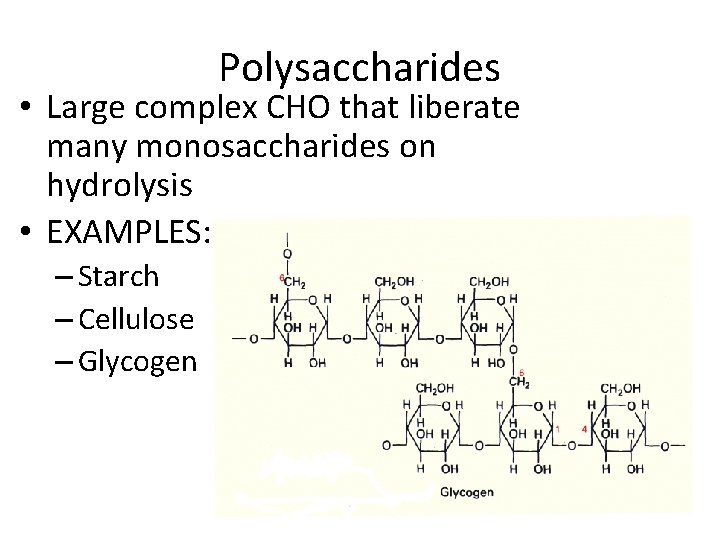
Polysaccharides • Large complex CHO that liberate many monosaccharides on hydrolysis • EXAMPLES: – Starch – Cellulose – Glycogen

GLYCOGEN • Glycogen one of most important – Short Term Storage form of CHO in animals • Liver and muscle • Used as quick energy – Formed by strings of glucose (1 -4 bonds) – Strings joined approx. every 10 glucose units (1 -6 bonds) • Glycogenesis - formation of glycogen • Glycogenolysis - hydrolysis of glycogen

Functions • Carbohydrates serve as: – Stores (usually converted to lipids) – Fuels • Catabolised in glycolysis to produce ATP – Structural molecules • Molecules on surface of cell membranes involved in: – Cell recognition – Cell adherence – Hydrophilic properties attract water layer as lubricant

Where you Get it! • Sugary sweet things – Fruit, Candy, Pastries • Breads and Grains • Starchy stuff like Potatoes, Taro, Plantains, Roots, Tubers • UNUSABLE but a source is Cellulose. Found in many plants including Pickled Okra. • Pretty much everything you eat gets converted by the body to sugar in some way

PROTEIN (polypeptides)

Function • Proteins do the work of the body. You name it, a protein is involved. – Examples: • Muscles • Digestion • Nerve impulses • Body Structure • ETC.

Protein Structure

Levels Of Protein Organization Primary Structure � Primary Structure - The sequence of amino acids in the protein Met-Gly-Ala-Pro-His-Ile-Asp-Glu-Met-Ser-Thr-. . . This sequence comes from the translated strand of m. RNA that is created during the transcription step of protein synthesis. It uses the chart on the next slide.
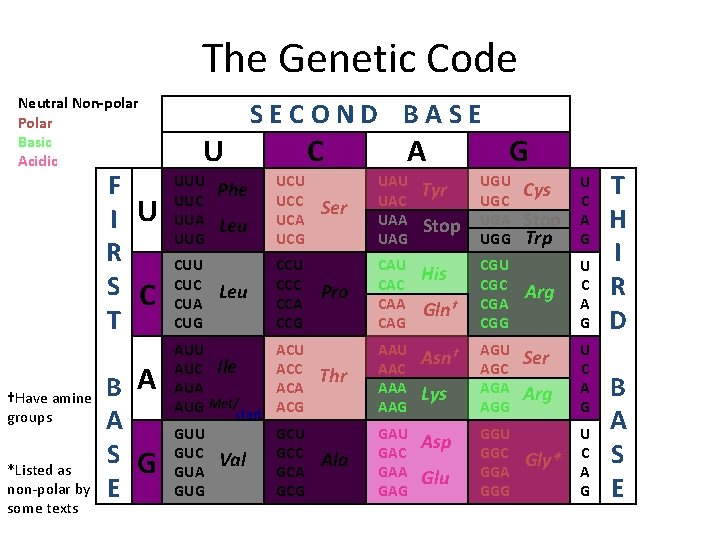
The Genetic Code Neutral Non-polar Polar Basic Acidic F I U R S C T †Have amine groups *Listed as non-polar by some texts B A A S G E U UUC UUA UUG CUU CUC CUA CUG SECOND BASE Phe Leu C UCU UCC UCA UCG CCU CCC CCA CCG AUU AUC Ile AUA AUG Met/start ACU ACC ACA ACG GUU GUC GUA GUG GCU GCC GCA GCG Val A Ser UAU UAC UAA UAG Tyr Pro CAU CAC CAA CAG His Thr AAU AAC AAA AAG Asn† Ala GAU GAC GAA GAG Asp Stop Gln† Lys Glu G UGU UGC UGA UGG CGU CGC CGA CGG AGU AGC AGA AGG GGU GGC GGA GGG Cys Stop Trp U C A G Arg U C A G Ser Arg Gly* U C A G T H I R D B A S E

Levels of Organization Secondary Structure • Secondary Structure - The formation of a helices and b pleated sheets due to hydrogen bonding between the peptide backbone

Protein Secondary Structure • The peptide backbone of DNA has areas of positive charge and negative charge • These areas can interact with one another to form hydrogen bonds • The result of these hydrogen bonds are two types of structures: - a helices - b pleated sheets
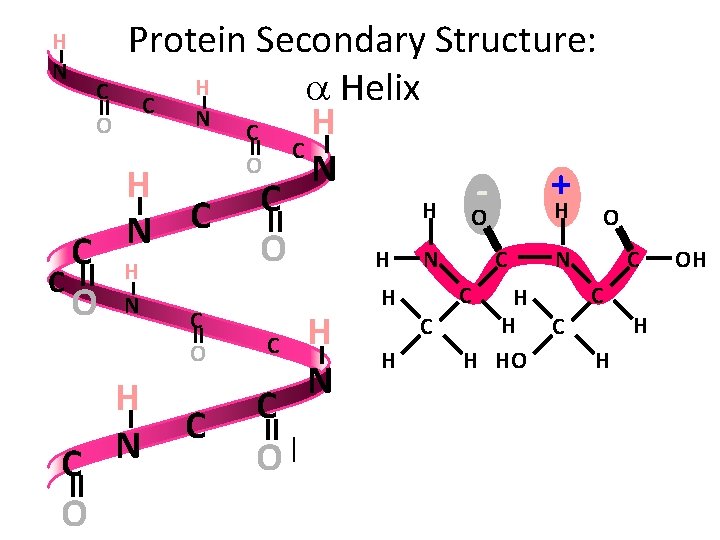
Protein Secondary Structure: H C a Helix C N O H C C O N + H C H O C N O C H H N C C C C H H O N C H C H C O H H HO H N H C C N O C O H N OH
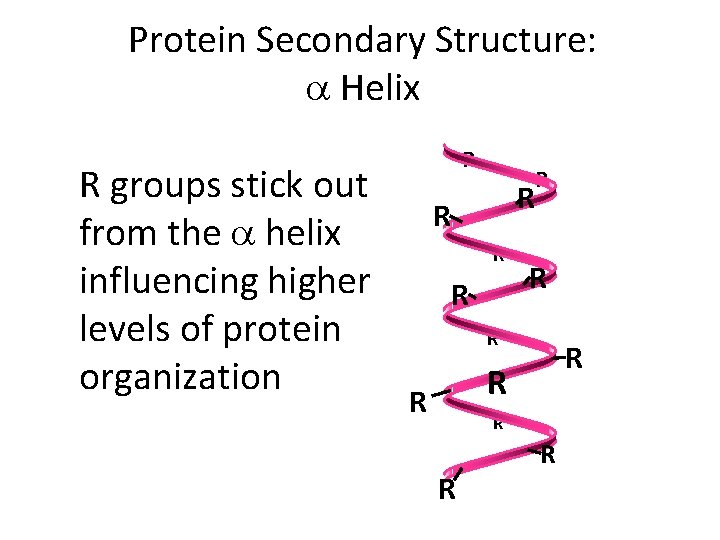
Protein Secondary Structure: a Helix R groups stick out from the a helix influencing higher levels of protein organization R R R R
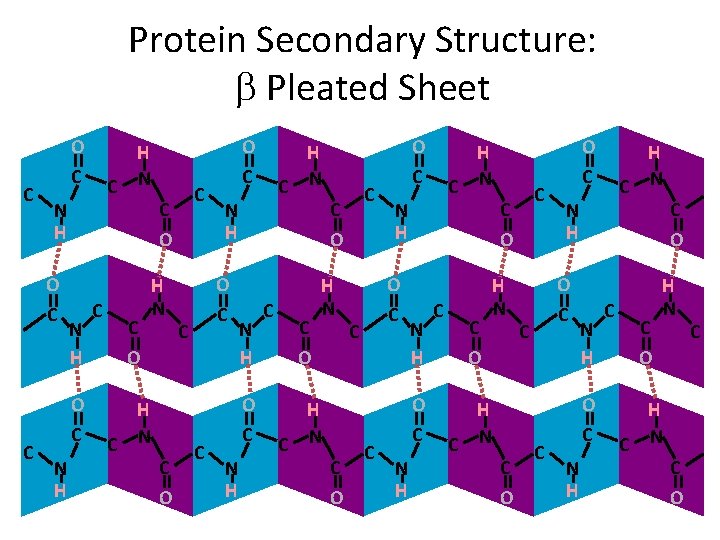
Protein Secondary Structure: b Pleated Sheet C O C N H O C C H C N N H O C N H C C O C O H N H C N C C O O C N H O C C C H C N N H O C N H C C O H N H C N C O C

Levels Of Protein Organization Tertiary Structure • Tertiary structure results from the folding of a helices and b pleated sheets • Factors influencing tertiary structure include: – Hydrophobic/hydrophilic interactions – Hydrogen bonding – Disulfide linkages – Folding by chaperone proteins
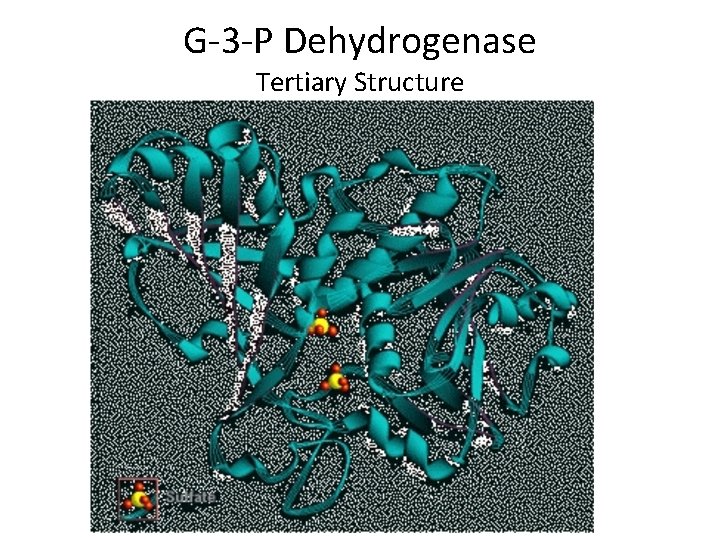
G-3 -P Dehydrogenase Tertiary Structure

Levels Of Protein Organization Quaternary Structure • Quaternary structure results from the interaction of independent polypeptide chains • Factors influencing quaternary structure include: – Hydrophobic/hydrophilic interactions – Hydrogen bonding – The shape and charge distribution on associating polypeptides
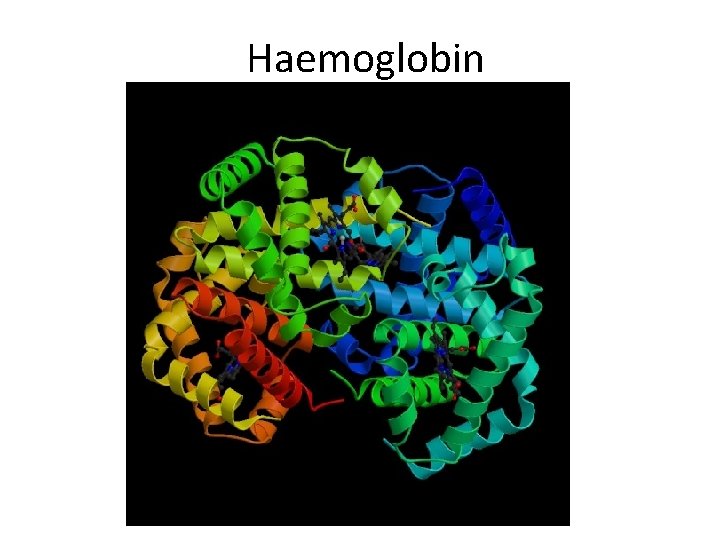
Haemoglobin

3 Main types • Enzymes • Structural • Cell Signaling

ENZYMES • Proteins that help to speed up the chemical rxns (reactions) of the body.

Structural • Collagen: – The rebar of the body – Major component in bones, cartilage, and other structural substances of the body. – Extremely strong stuff
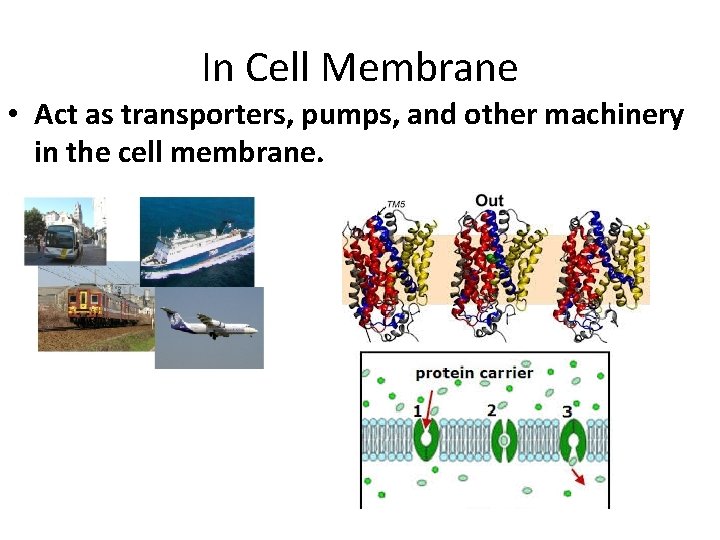
In Cell Membrane • Act as transporters, pumps, and other machinery in the cell membrane.

Lipids: The things that make you soft, squishy, and smelly.

What is it? Lipids: Compounds that consist mainly of carbon and hydrogen linked together, that are found in substances such as a fat, oil or wax that dissolve in alcohol but not in water.

Types There are 4 main types of Lipids: – Triglycerides (Fats + oils) – Phospholipids – Waxes – Steroids

Anatomy of a Triglyceride • Glycerol Backbone - made of a 3 carbon chain with hydroxyl groups. • Fatty acids - made with a carboxyl group and a hydrocarbon chain
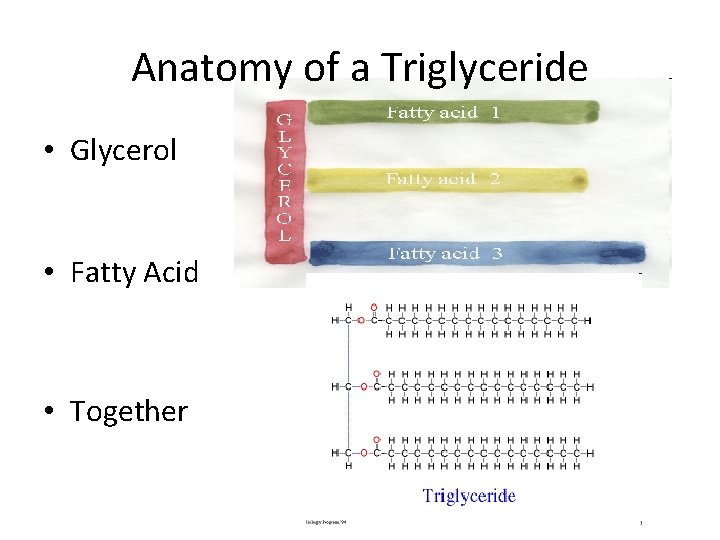
Anatomy of a Triglyceride • Glycerol • Fatty Acid • Together

Triglyceride Importance The Main functions of Triglycerides are: – Energy Storage • 1 g of fat contains nearly 2 x as much energy as 1 g of a polysaccharide. – Insulation (mainly sea critters) • Blubber

Saturated vs. Unsaturated Fat • These terms refer to the bonds between the carbon molecules of the lipid. A fat is saturated when all the carbon to carbon bonds are single bonds (-c-c-) • An unsaturated fat has at least one double bond between carbons ( -c=c-)
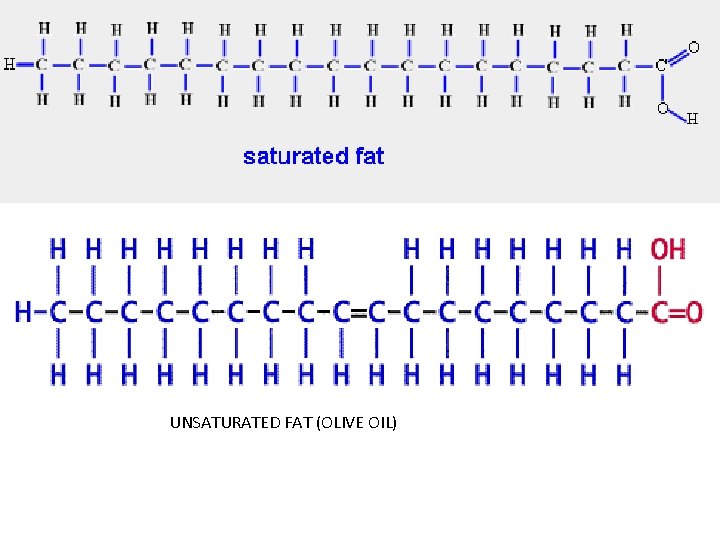
UNSATURATED FAT (OLIVE OIL)
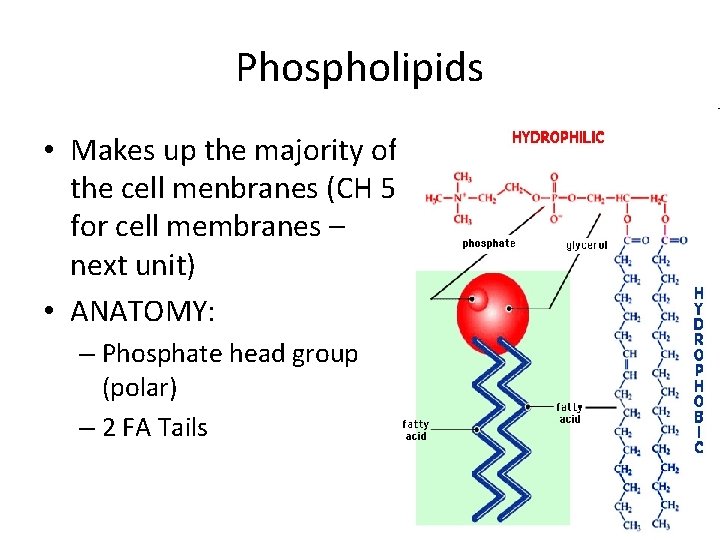
Phospholipids • Makes up the majority of the cell menbranes (CH 5 for cell membranes – next unit) • ANATOMY: – Phosphate head group (polar) – 2 FA Tails

Waxes • STRUCTURE: – 1 FA tail attached to an Alcohol • Extremely hydrophobic(water bad mmkay) – Means it is a natural protective coat found on apples and pears, but it really shines as a dehydration preventive coating for many animals like insects.
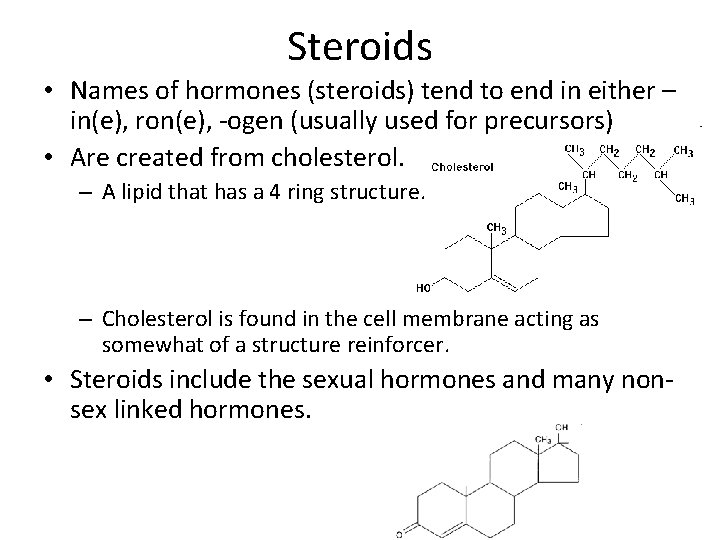
Steroids • Names of hormones (steroids) tend to end in either – in(e), ron(e), -ogen (usually used for precursors) • Are created from cholesterol. – A lipid that has a 4 ring structure. – Cholesterol is found in the cell membrane acting as somewhat of a structure reinforcer. • Steroids include the sexual hormones and many nonsex linked hormones.

Steroids Use - Major part of the Endocrine system usually referred to as hormones. - They are produced in glands - Roles of hormones: - Growth Gender characteristics Messengers Total Body responses (Adrenaline) Generally gradual changes to the body - NOT QUICK LIKE NERVES

Fun with Roids! ANABOLIC STEROIDS - These are synthetic types of steroids derived from male testosterone. - BENEFITS: - Helps to treat anemia (muscle attacking disease) - Quickly builds muscle - COSTS: - Roid Rage due to chemicals in brain being thrown off Liver damage Decreased libido or sterility Bloated face

Steroids anyone? Tammy in 2007 Tammy Thomas -US Cycling Gold Medalist Tammy in 2001
- Slides: 45