Macromolecule analysis Analysis of amino acids and carbohydrates

Macro-molecule analysis Analysis of amino acids and carbohydrates

Ionization constant of water : equilibrium constant of water altered by temperature Kw = [H+][OH-] temp, ℃ Kw p. H of water 0 0. 114 x 10 -14 7. 47 10 0. 292 x 10 -14 7. 27 20 0. 681 x 10 -14 7. 08 25 1. 01 x 10 -14 7. 00 40 2. 92 x 10 -14 6. 77 At 25℃ and 1 atm, Kw = [H+][OH-] = 1. 0 x 10 -14 [H+] = [OH-] = 1. 0 x 10 -7 M p. H = - log[H+] = - log(10 -7) = - (-7) = 7 Molar = mole/liter mole
![Kw = [H+][OH-] = 1 x 10 -14 (at 25℃) (ex) when [H+] is Kw = [H+][OH-] = 1 x 10 -14 (at 25℃) (ex) when [H+] is](http://slidetodoc.com/presentation_image_h2/bc8931617f7b2dbc485d991aa24d2be5/image-3.jpg)
Kw = [H+][OH-] = 1 x 10 -14 (at 25℃) (ex) when [H+] is 10 -1 M, [OH-]는 10 -13 M Q) What are [H+] and [OH-] when HCl is added to make 0. 1 M HCl? Before After HCl is added [H+] 1 x 10 -7 0. 1 [OH-] 1 x 10 -7 1 x 10 -13
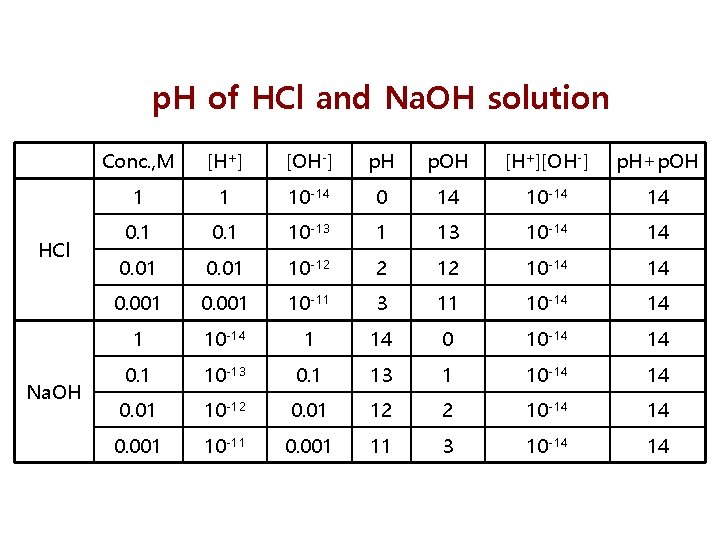
p. H of HCl and Na. OH solution HCl Na. OH Conc. , M [H+] [OH-] p. H p. OH [H+][OH-] p. H+p. OH 1 1 10 -14 0 14 10 -14 14 0. 1 10 -13 10 -14 14 0. 01 10 -12 2 12 10 -14 14 0. 001 10 -11 3 11 10 -14 14 1 10 -14 1 14 0 10 -14 14 0. 1 10 -13 0. 1 13 1 10 -14 14 0. 01 10 -12 0. 01 12 2 10 -14 14 0. 001 10 -11 0. 001 11 3 10 -14 14
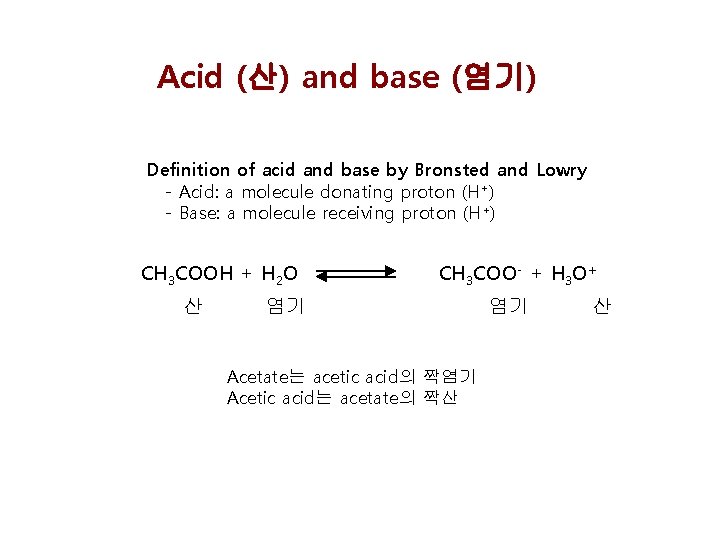
Acid (산) and base (염기) Definition of acid and base by Bronsted and Lowry - Acid: a molecule donating proton (H+) - Base: a molecule receiving proton (H+) CH 3 COOH + H 2 O 산 CH 3 COO- + H 3 O+ 염기 Acetate는 acetic acid의 짝염기 Acetic acid는 acetate의 짝산 염기 산
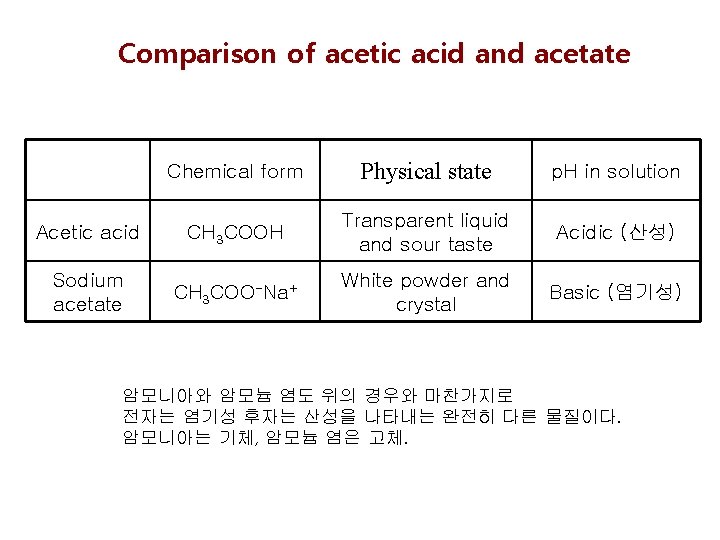
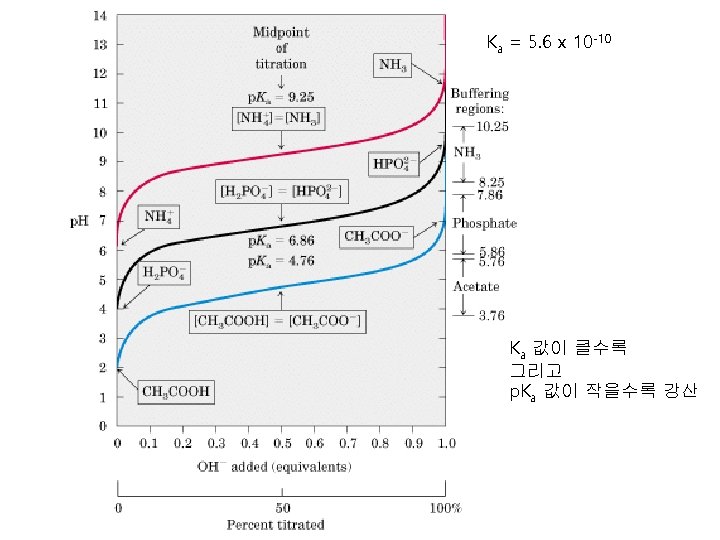
Comparison of acetic acid and acetate Chemical form Physical state p. H in solution Acetic acid CH 3 COOH Transparent liquid and sour taste Acidic (산성) Sodium acetate CH 3 COO-Na+ White powder and crystal Basic (염기성) 암모니아와 암모늄 염도 위의 경우와 마찬가지로 전자는 염기성 후자는 산성을 나타내는 완전히 다른 물질이다. 암모니아는 기체, 암모늄 염은 고체.
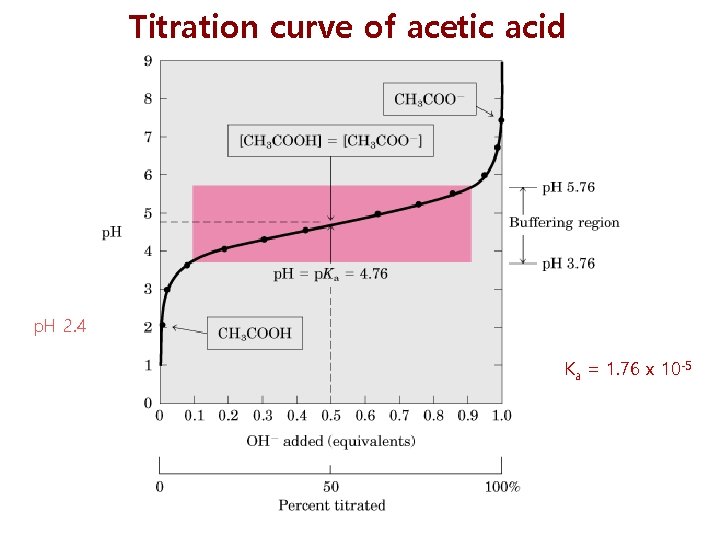
Titration curve of acetic acid p. H 2. 4 Ka = 1. 76 x 10 -5
![Henderson-Hasselbalch equation Ka = [H+][A-] = Ka [HA] [A-] -log[H+] = -log Ka - Henderson-Hasselbalch equation Ka = [H+][A-] = Ka [HA] [A-] -log[H+] = -log Ka -](http://slidetodoc.com/presentation_image_h2/bc8931617f7b2dbc485d991aa24d2be5/image-9.jpg)
Henderson-Hasselbalch equation Ka = [H+][A-] = Ka [HA] [A-] -log[H+] = -log Ka - log [HA] [A-]

Amino acids and Proteins - 20 amino acids and peptide bond Structure of proteins Sequencing and synthesis of polypetides
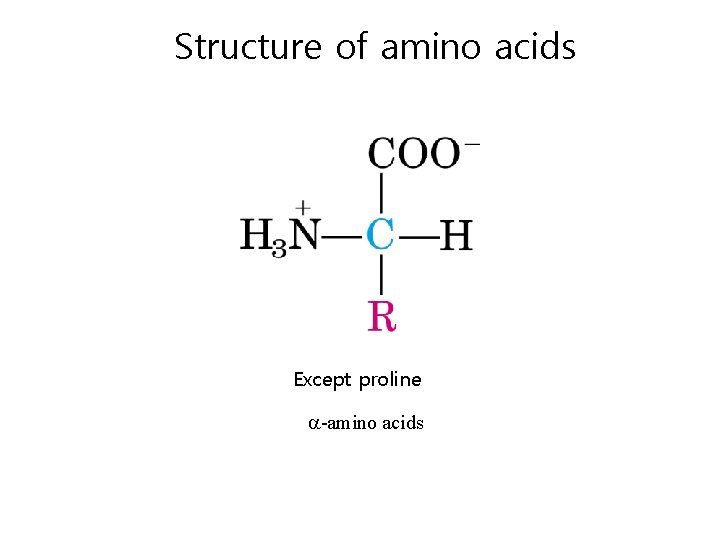
Structure of amino acids Except proline -amino acids
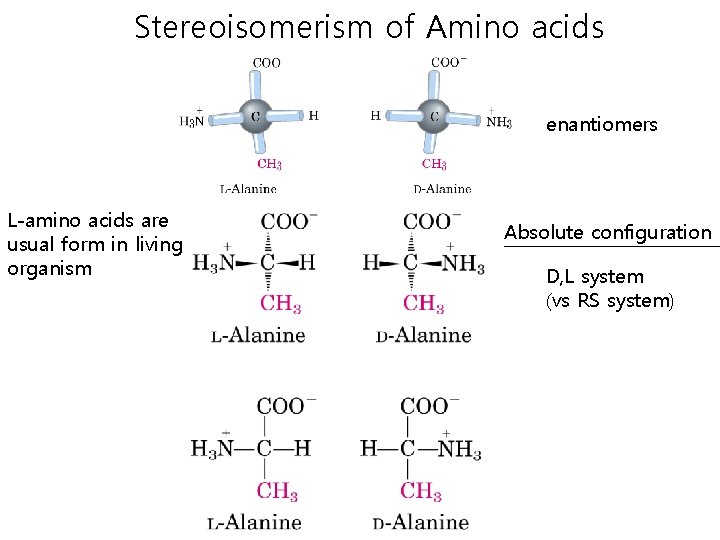
Stereoisomerism of Amino acids enantiomers L-amino acids are usual form in living organism Absolute configuration D, L system (vs RS system)
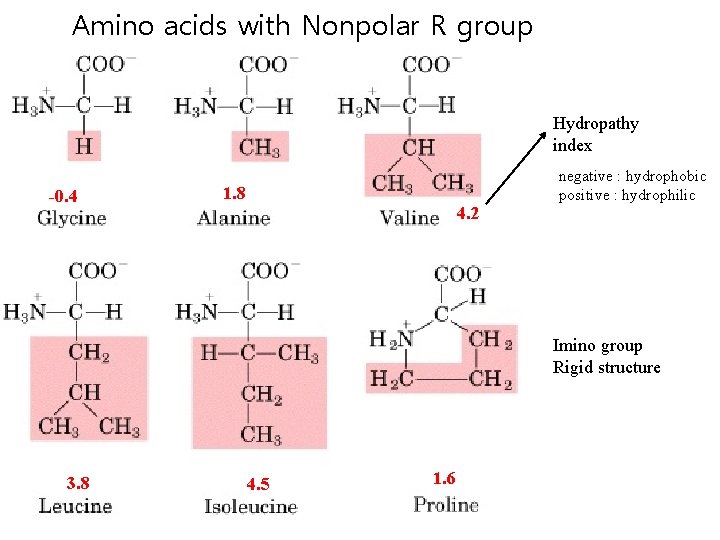
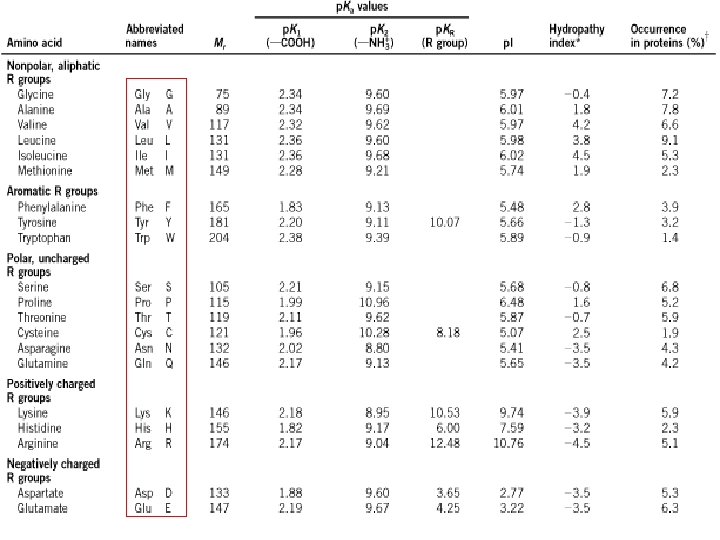
Amino acids with Nonpolar R group Hydropathy index -0. 4 1. 8 4. 2 negative : hydrophobic positive : hydrophilic Imino group Rigid structure 3. 8 4. 5 1. 6
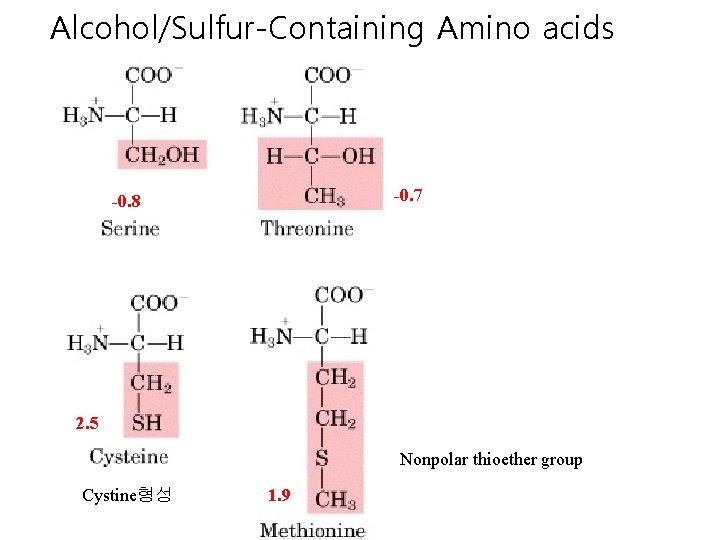
Alcohol/Sulfur-Containing Amino acids -0. 7 -0. 8 2. 5 Nonpolar thioether group Cystine형성 1. 9
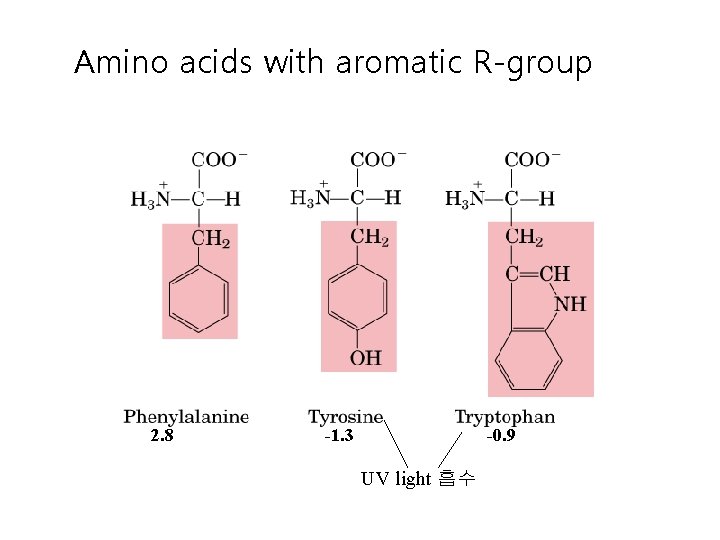
Amino acids with aromatic R-group 2. 8 -1. 3 -0. 9 UV light 흡수
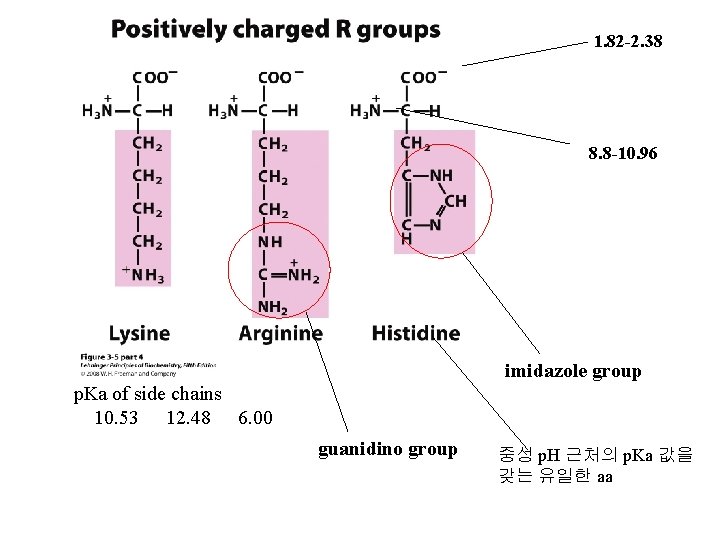
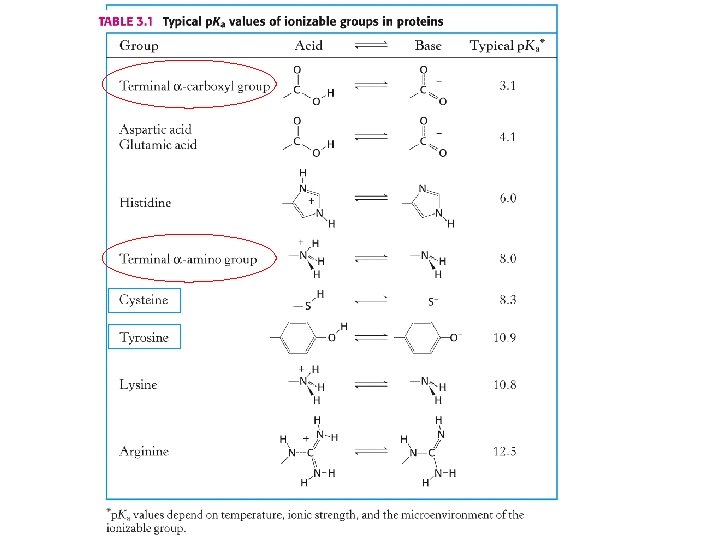
1. 82 -2. 38 8. 8 -10. 96 imidazole group p. Ka of side chains 10. 53 12. 48 6. 00 guanidino group 중성 p. H 근처의 p. Ka 값을 갖는 유일한 aa
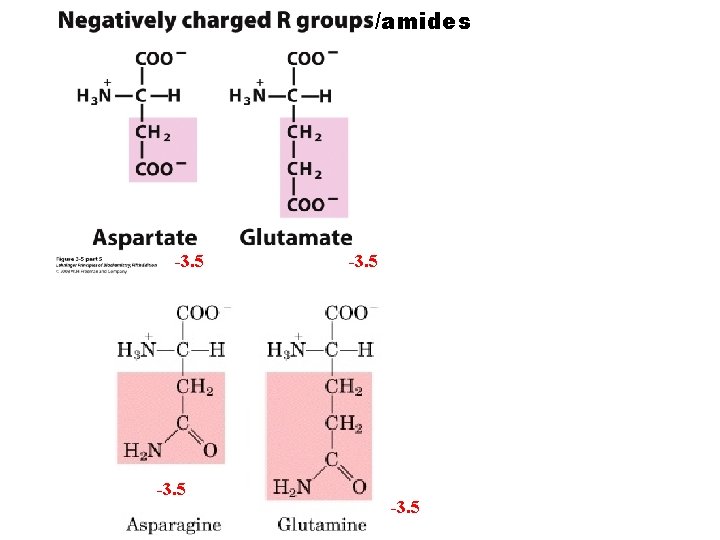
/amides -3. 5
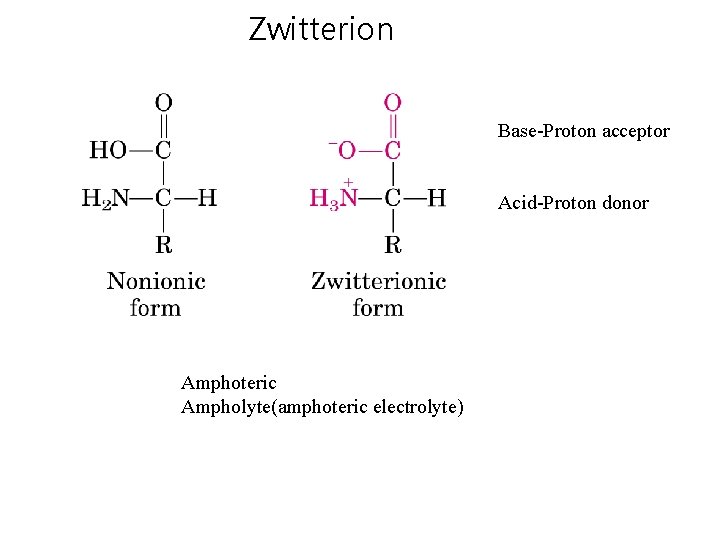
Zwitterion Base-Proton acceptor Acid-Proton donor Amphoteric Ampholyte(amphoteric electrolyte)
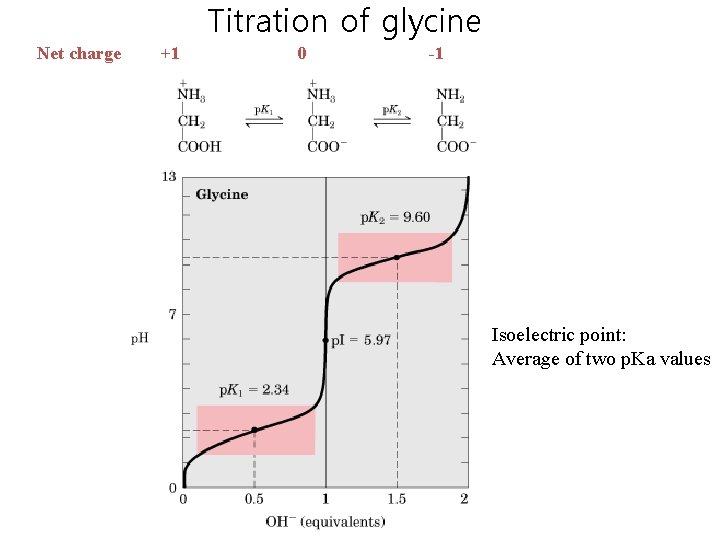
Titration of glycine Net charge +1 0 -1 Isoelectric point: Average of two p. Ka values
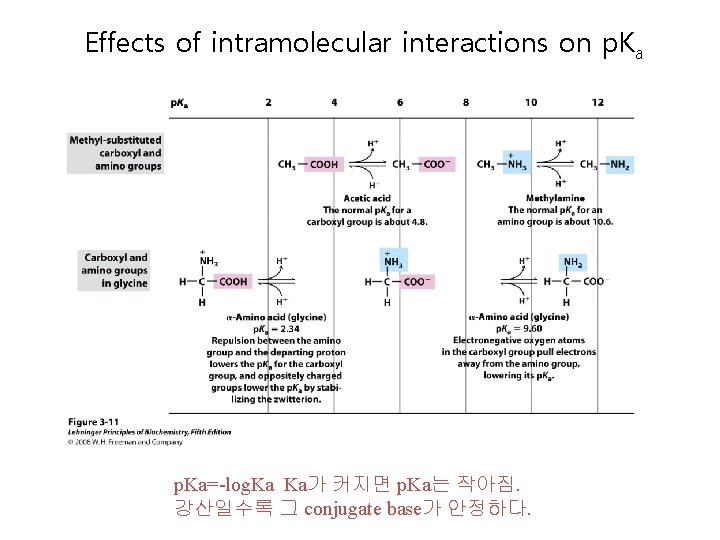
Effects of intramolecular interactions on p. Ka=-log. Ka Ka가 커지면 p. Ka는 작아짐. 강산일수록 그 conjugate base가 안정하다.
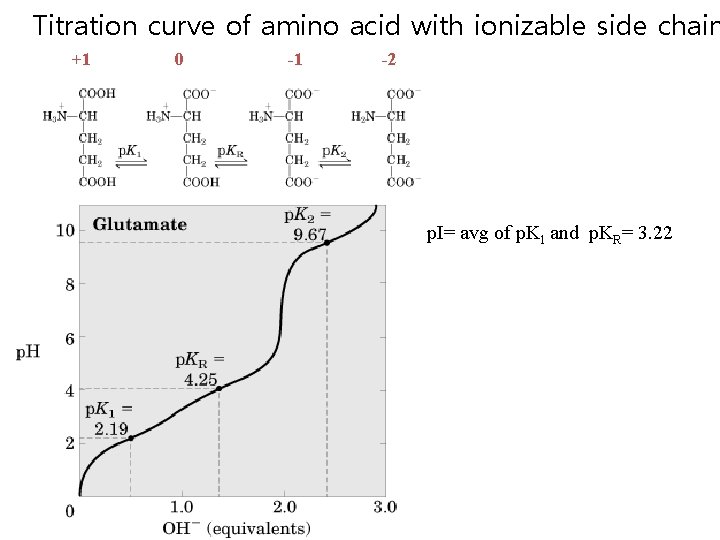
Titration curve of amino acid with ionizable side chain +1 0 -1 -2 p. I= avg of p. K 1 and p. KR= 3. 22
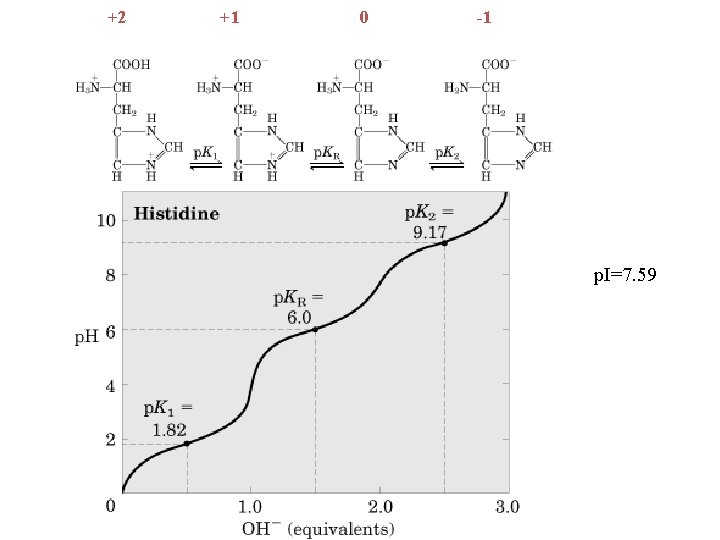
+2 +1 0 -1 p. I=7. 59
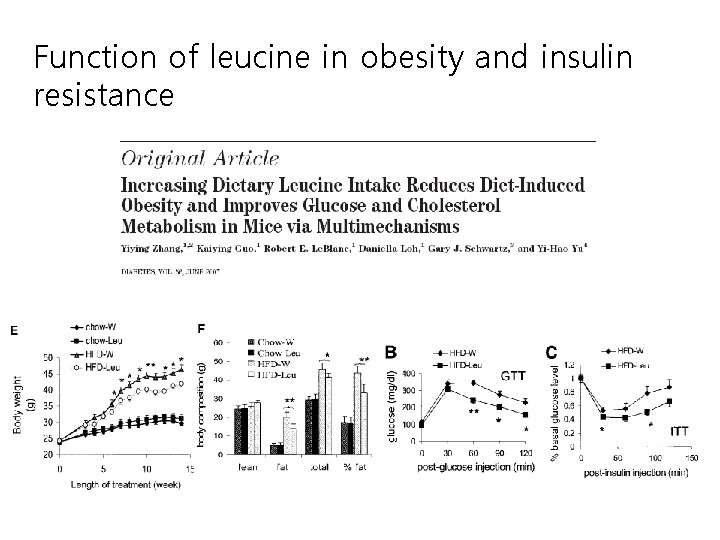
Function of leucine in obesity and insulin resistance

Monosodium glutamate and “chinese restaurant syndrome”- MSG allergy MSG, a flavor enhancer Dizziness, headaches, and chest pain after taking chinese meal Due to impure materials in fermented MSG, allergy is developed. Still inconclusive.

Asparagine reduces hangover by activating alcohol dehydrogenase European patent- Asparagine and hangover Soybean sprout soup.

Ionization behavior of peptide Only one a-amino group and a-carboxyl group p. Ka of peptide is different from individual amino acid p. I can be calculated.






Carbohydrates and Glycobiology Roles of Carbohydrates • • • Energy source ECM Lubricant Protection barrier Cell-cell communication Intracellular trafficking The most abundant biomolecules on Earth Many carbohydrates: (CH 2 O)n Polyhydroxy aldehydes or ketones Glycoconjugates • Carbohydrate polymers attached to proteins or lipids Carbohydrates
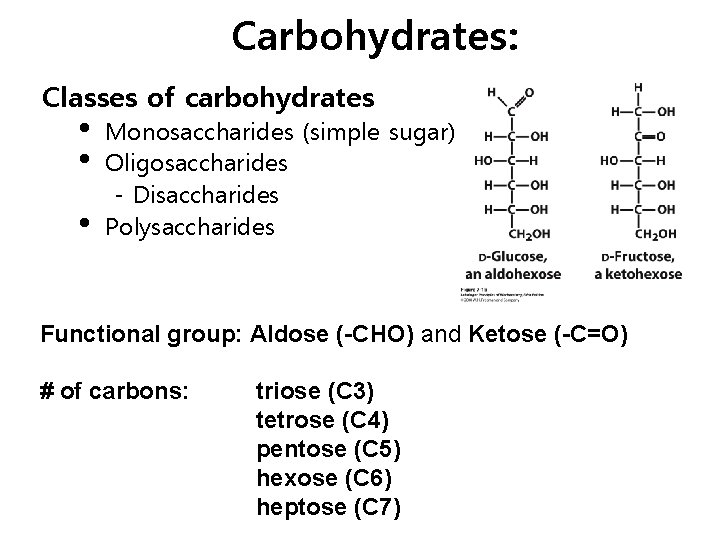
Carbohydrates: Classes of carbohydrates • • • Monosaccharides (simple sugar) Oligosaccharides - Disaccharides Polysaccharides Functional group: Aldose (-CHO) and Ketose (-C=O) # of carbons: triose (C 3) tetrose (C 4) pentose (C 5) hexose (C 6) heptose (C 7)
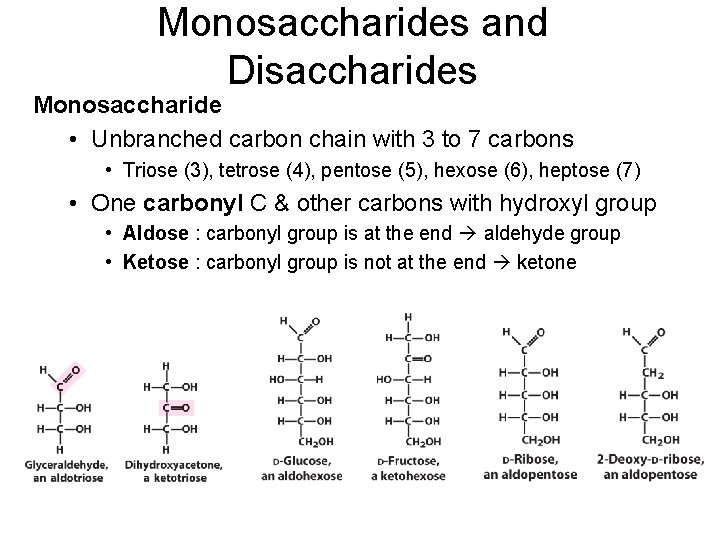
Monosaccharides and Disaccharides Monosaccharide • Unbranched carbon chain with 3 to 7 carbons • Triose (3), tetrose (4), pentose (5), hexose (6), heptose (7) • One carbonyl C & other carbons with hydroxyl group • Aldose : carbonyl group is at the end aldehyde group • Ketose : carbonyl group is not at the end ketone
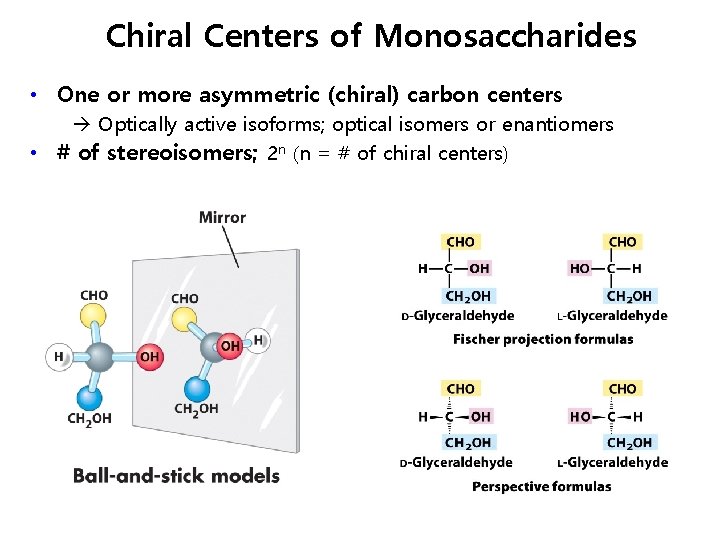
Chiral Centers of Monosaccharides • One or more asymmetric (chiral) carbon centers Optically active isoforms; optical isomers or enantiomers • # of stereoisomers; 2 n (n = # of chiral centers)
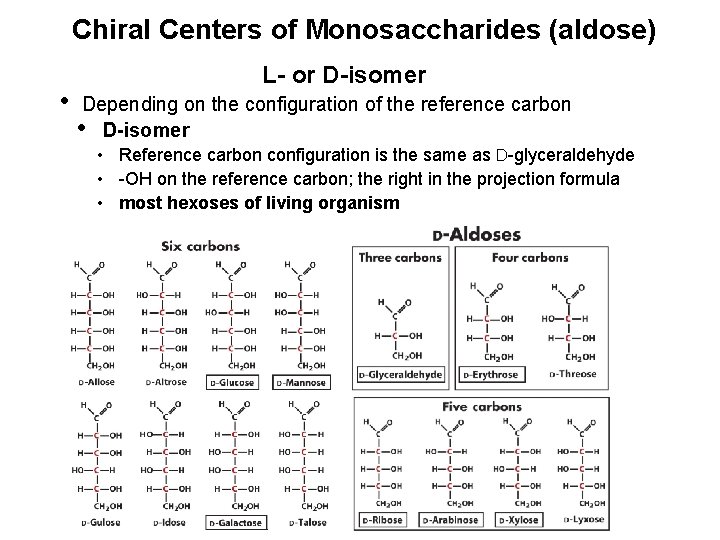
Chiral Centers of Monosaccharides (aldose) • L- or D-isomer Depending on the configuration of the reference carbon • D-isomer • Reference carbon configuration is the same as D-glyceraldehyde • -OH on the reference carbon; the right in the projection formula • most hexoses of living organism
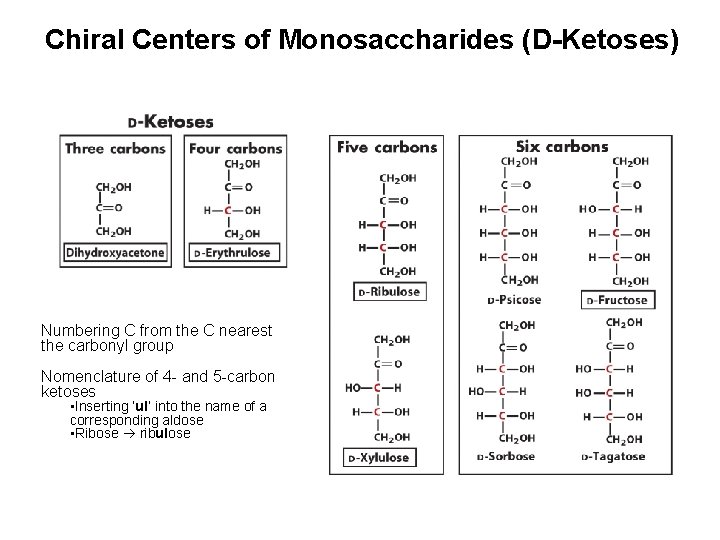
Chiral Centers of Monosaccharides (D-Ketoses) Numbering C from the C nearest the carbonyl group Nomenclature of 4 - and 5 -carbon ketoses • Inserting ‘ul’ into the name of a corresponding aldose • Ribose ribulose
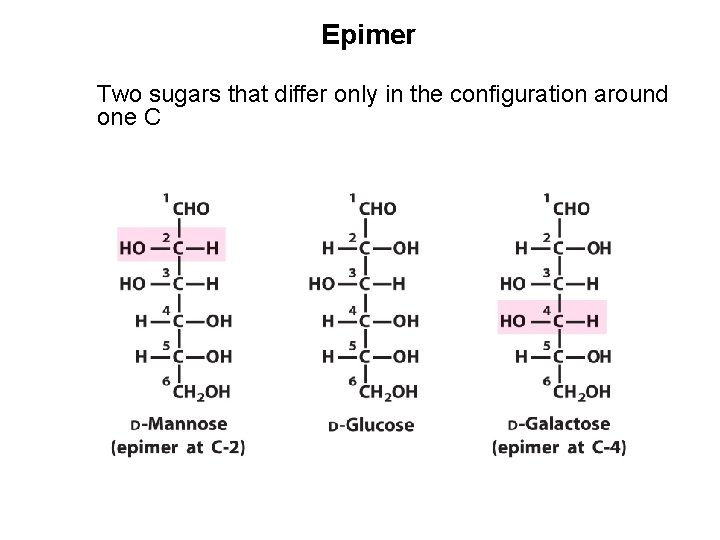
Epimer Two sugars that differ only in the configuration around one C
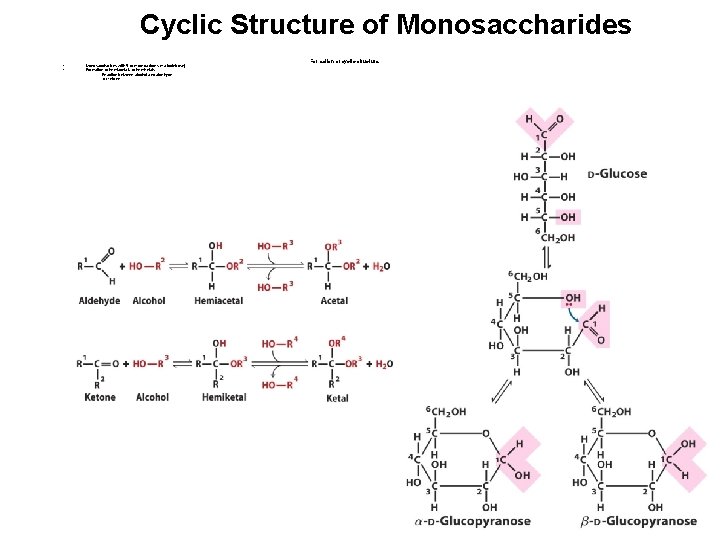
Cyclic Structure of Monosaccharides • • Monosaccharides with 5 or more carbons (+ aldotetrose) Formation of hemiacetals or hemiketals - Reaction between alcohol and aldehyde or ketone Formation of cyclic structure
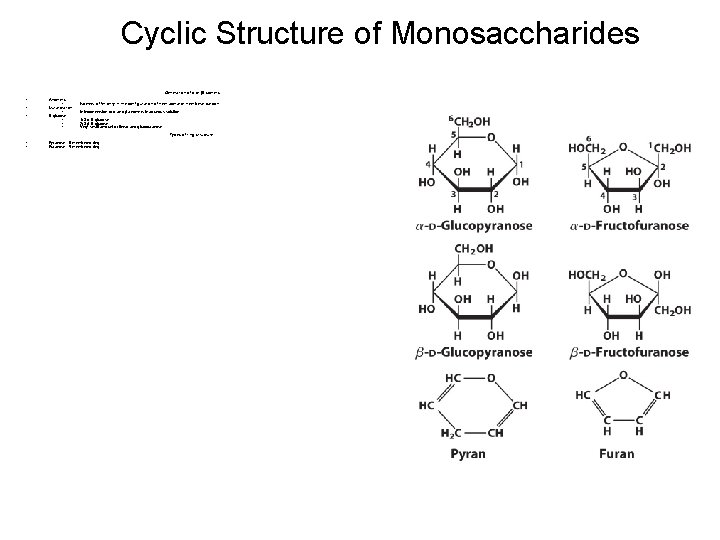
Cyclic Structure of Monosaccharides Generation of α or isomers • Anomers • Mutarotation • D-glucose • • Pyranose : 6 -membered ring Furanose : 5 -membered ring • Isomers differ only in the configuration of hemiacetal or hemiketal carbon • Interconversion of and anomers in aqueous solution • • • 1/3 -D-glucose 2/3 -D-glucose Very small amount of linear and glucofuranose Types of ring structure
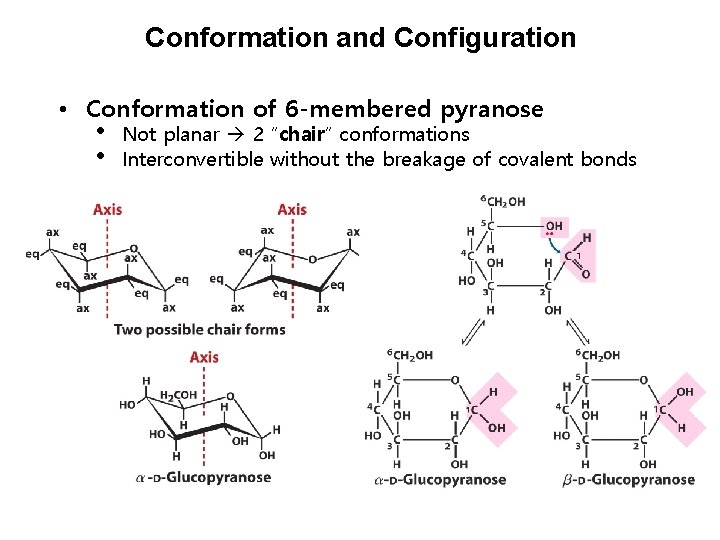
Conformation and Configuration • Conformation of 6 -membered pyranose • • Not planar 2 “chair” conformations Interconvertible without the breakage of covalent bonds
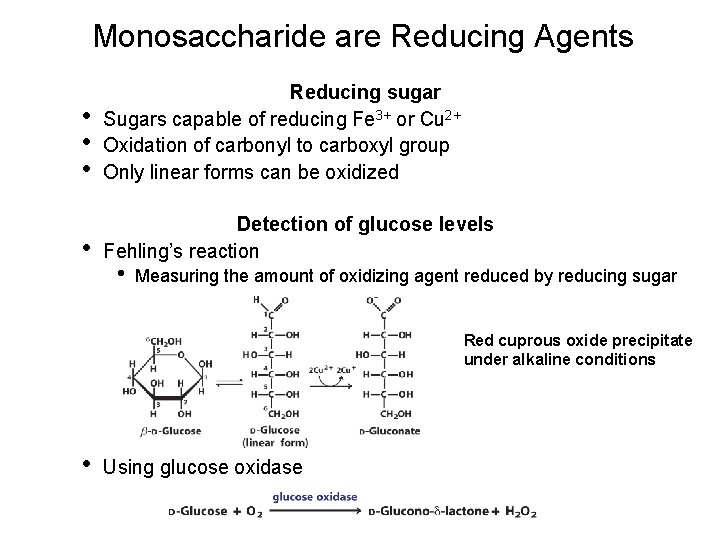
Monosaccharide are Reducing Agents • • • Reducing sugar Sugars capable of reducing Fe 3+ or Cu 2+ Oxidation of carbonyl to carboxyl group Only linear forms can be oxidized • Detection of glucose levels Fehling’s reaction • Measuring the amount of oxidizing agent reduced by reducing sugar Red cuprous oxide precipitate under alkaline conditions • Using glucose oxidase
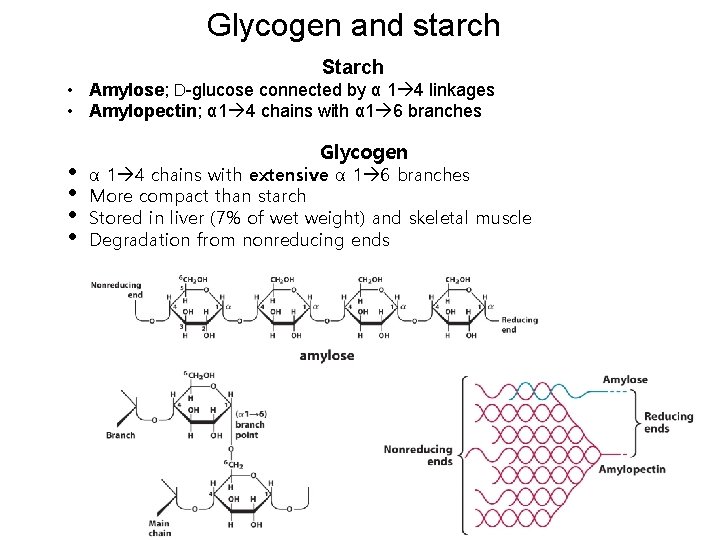
Glycogen and starch Starch • Amylose; D-glucose connected by α 1 4 linkages • Amylopectin; α 1 4 chains with α 1 6 branches • • Glycogen α 1 4 chains with extensive α 1 6 branches More compact than starch Stored in liver (7% of wet weight) and skeletal muscle Degradation from nonreducing ends



Results 당의 분석 Glucose, Fructose, Sucrose, 전분용액, 베네딕트 용액, 증류 수, 시험관, vortex mixer, hot plate, pipet, tip 변하는 순서 Glucose Fructose Sucrose 전분 용액 색깔 침전물 유무 환원당 여부
- Slides: 50