M Capitelli Dipartimento di Chimica Universit di Bari

M. Capitelli Dipartimento di Chimica, Università di Bari, Italy IMIP-CNR, Bari, Italy

COLLABORATORS
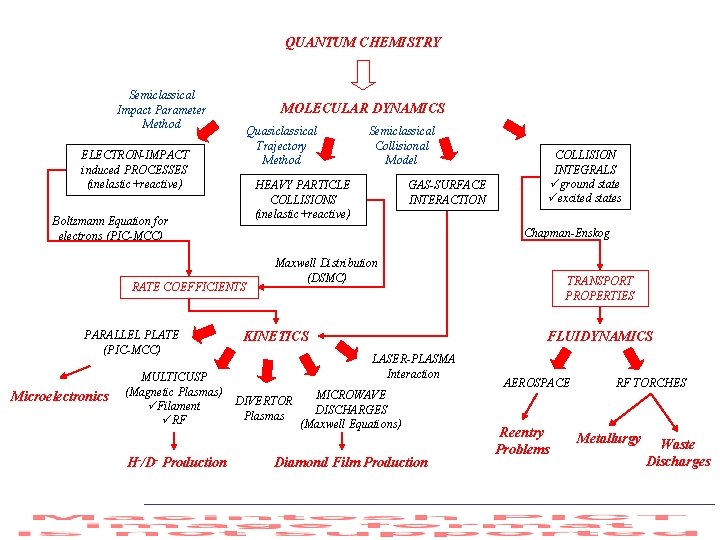
QUANTUM CHEMISTRY Semiclassical Impact Parameter Method MOLECULAR DYNAMICS Quasiclassical Trajectory Method ELECTRON-IMPACT induced PROCESSES (inelastic+reactive) HEAVY PARTICLE COLLISIONS (inelastic+reactive) Boltzmann Equation for electrons (PIC-MCC) Microelectronics MULTICUSP (Magnetic Plasmas) üFilament üRF H-/D- Production GAS-SURFACE INTERACTION COLLISION INTEGRALS üground state üexcited states Chapman-Enskog RATE COEFFICIENTS PARALLEL PLATE (PIC-MCC) Semiclassical Collisional Model Maxwell Distribution (DSMC) KINETICS TRANSPORT PROPERTIES FLUIDYNAMICS LASER-PLASMA Interaction MICROWAVE DIVERTOR DISCHARGES Plasmas (Maxwell Equations) Diamond Film Production AEROSPACE Reentry Problems RF TORCHES Metallurgy Waste Discharges

OUTLINE - Open problems (1985 -1995) in Multipole Magnetic Plasmas a) Validation of Bari code with FOM experiments b) Extension to D 2 plasmas c) Pulsed discharges d) Rydberg states e) Wall effects - Cross Section Improvements (1995 -2005) a) Electron-molecule cross sections b) Heavy particle collision cross sections c) Gas surface interaction - Kinetic models Improvements(1995 -2005) a) New multipole zerodimensional code b) Parallel-plate 1 D code c) RF and MW quasi 1 D code - Air Plasmas a) N 2 and O 2 State-to-State Cross Sections

OUTLINE - Open problems (1985 -1995) in Multipole Magnetic Plasmas a) Validation of Bari code with FOM experiments b) Extension to D 2 plasmas c) Pulsed discharges d) Rydberg states e) Wall effects - Cross Section Improvements (1995 -2005) a) Electron-molecule cross sections b) Heavy particle collision cross sections c) Gas surface interaction - Kinetic models Improvements(1995 -2005) a) New multipole zerodimensional code b) Parallel-plate 1 D code c) RF and MW quasi 1 D code - Air Plasmas a) N 2 and O 2 State-to-State Cross Sections

VIBRATIONAL EXCITATION and NEGATIVE ION KINETICS MULTIPOLE MAGNETIC PLASMAS: !! Self-consistent non equilibrium vibrational kinetics coupled to the Boltzmann equation for the electron energy distribution function!! Time Evolution (heavy species) Electron source term: Flux of electrons along energy axis due to elastic collisions Flux of electrons along energy axis due to electron-electron collisions Term due to inelastic collisions Term due to ionization collisions Term due to superelastic collisions Electron loss term

VALIDATION of BARI CODE with the FOM EXPERIMENTS (GORSE ET AL. 1992) comparison between experimental (a) and theoretical (b) vdfs at several pressures comparison between experimental (a) and theoretical (b) vdfs at several discharge currents Id

!!PROBLEMS!! - calculations overestimate by a factor 10 the high lying vibrational levels giving satisfactory agreement with negative ion concentrations - FOM experimental vibrational distributions limited to v=5 !!New: experimental determination by Mosbach and Dobele up to v=13!! vibrational distribution Nv measured in a H 2 multicusp source (p = 11. 25 mtorr, Id = 0. 5 A, Vd = 100 V)

EXTENSION to D 2 PLASMAS theoretical EEDF (a) and Nv (b) in H 2 and D 2 sources (p = 4. 5 mtorr, Id = 10 A, Vd = 115 V, plasma potential Vp = 2. 9 V) dissociative attachment rates versus vibrational quantum number for H 2 and D 2 molecules
![(a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D] (a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D]](http://slidetodoc.com/presentation_image_h/ac869d64df8dc36450a9b5f68f88b6fa/image-10.jpg)
(a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D] (d) negative ion concentration [H-]/[D-] for H 2 and D 2 systems versus pressure p (Id = 10 A, Vd = 115 V)
![(a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D] (a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D]](http://slidetodoc.com/presentation_image_h/ac869d64df8dc36450a9b5f68f88b6fa/image-11.jpg)
(a) behavior of electron density ne (b) electron temperature Te (c) atomic concentration [H]/[D] (d) negative ion concentration [H-]/[D-] for H 2 and D 2 systems versus current Id (p = 7. 5 mtorr, Vd = 115 V)

DISSOCIATIVE ATTACHMENT: H 2 (X 1 g, ) + e H 2 - H- + H E = 4. 5 e. V

RESONANT VIBRATIONAL EXCITATION: H 2 (X 1 g, i) + e H 2 - H 2 (X 1 g, f) + e E = 5 e. V

PULSED DISCHARGES: MODEL Relaxation of several quantities in the D 2 post-discharge regime (a) EEDF, (b) e-da rate coefficient, (c) Nv, (d) D- density

PULSED DISCHARGES: EXPERIMENT extracted H- current in a pulsed hydrogen discharge with a 2. 7 ms pulse length and a 87 Hz repetition rate (p = 2. 4 mtorr, Id = 15 A)
![RYDBERG STATES: HYSTORICAL SCENARIO/1 Pinnaduwage et al. [Phys. Rev. Lett. 70, 754 (1993)] e RYDBERG STATES: HYSTORICAL SCENARIO/1 Pinnaduwage et al. [Phys. Rev. Lett. 70, 754 (1993)] e](http://slidetodoc.com/presentation_image_h/ac869d64df8dc36450a9b5f68f88b6fa/image-16.jpg)
RYDBERG STATES: HYSTORICAL SCENARIO/1 Pinnaduwage et al. [Phys. Rev. Lett. 70, 754 (1993)] e + H 2* H + H- Kda(Ryd)=10 -6 cm 3/sec Garscadden and Nagpal [Plasma Sources Sci. Technol. 4, 268 (1995)] Simplified model: (lumped excitation cross section on Rydberg states + lifetime of Rydberg states of 10 -6 sec + Kda(Ryd)=10 -6 cm 3/sec) Result: Contribution from Rydberg states 10 times the one from vibrationally excited states Gorse et al. [AIP Conf. Proc. 380, 109 (1995)] Model: Insertion of Garscadden model in the self-consistent kinetics in multipole magnetic plasmas Result: enhancement by a factor 2 Hiskes [Appl. Phys. Lett. 69, 755 (1996)] Model: collisional radiative model for H 2* Rydberg states + Kda(Ryd)=10 -6 cm 3/sec Result: lifetime of Rydberg states of the order of 10 -8 sec Consequence: contribution of Rydberg states 1%
![RYDBERG STATES: HYSTORICAL SCENARIO/2 Pinnaduwage et al. [Phys. Rev. A 55, 4131 (1997)] e RYDBERG STATES: HYSTORICAL SCENARIO/2 Pinnaduwage et al. [Phys. Rev. A 55, 4131 (1997)] e](http://slidetodoc.com/presentation_image_h/ac869d64df8dc36450a9b5f68f88b6fa/image-17.jpg)
RYDBERG STATES: HYSTORICAL SCENARIO/2 Pinnaduwage et al. [Phys. Rev. A 55, 4131 (1997)] e + H 2* = H + H- Kda(Ryd) = 5 10 -5 cm 3/sec Hassouni et al. [Chem. Phys. Lett. 290, 502 (1998)] Model: collisional radiative model for H 2* Rydberg states + Kda(Ryd)=5 10 -5 cm 3/sec Result: enhancement by factor 2. 7 Problem: Rydberg state from n>3 Pinnaduwage et al. [J. Appl. Phys. 85, 7064 (1999)] scaling law for Rydberg states which corresponds to n=12 An estimation For a plateau between 1010 -1012 cm-3 Rydberg concentrations of the order of 1/6 107 to 1/6 109 cm-3 can be of the same importance as the dissociative attachment from vibrationally excited molecules

FUTURE IMPROVEMENTS • COLLISIONAL RADIATIVE MODEL FOR RYDBERG STATES • SCALING LAW FOR THE EXCITATION OF RYDBERG STATES • LIFETIMES OF RYDBERG STATES • SCALING LAW FOR DISSOCIATIVE ATTACHMENT FROM RYDBERG STATES

WALL EFFECTS: HYSTORICAL SCENARIO Hiskes & Karo Model: trajectory calculations Results: strong deactivation of vibrationally excited molecules on iron surfaces - widely used in multicusp modelling Billing & Cacciatore Model: semiclassical/classical for describing atoms and molecules reaching the surface; quantum description of the interaction of the molecule/atom with the phononic and electronic structure of the metal Results : small deactivation of vibrationally excited molecules on copper surfaces
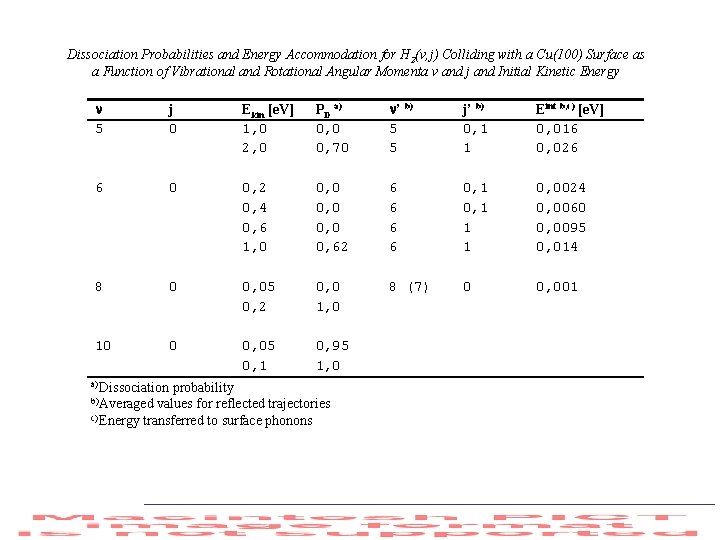
Dissociation Probabilities and Energy Accommodation for H 2(v, j) Colliding with a Cu(100) Surface as a Function of Vibrational and Rotational Angular Momenta v and j and Initial Kinetic Energy 5 j 0 Ekin [e. V] 1, 0 2, 0 PD a) 0, 0 0, 70 ’ b) 5 5 j’ b) 0, 1 1 Eint b, c) [e. V] 0, 016 0, 026 6 0 0, 2 0, 4 0, 6 1, 0 0, 62 6 6 0, 1 1 1 0, 0024 0, 0060 0, 0095 0, 014 8 0 0, 05 0, 2 0, 0 1, 0 8 (7) 0 0, 001 10 0 0, 05 0, 1 0, 95 1, 0 a) Dissociation probability b) Averaged values for reflected trajectories c) Energy transferred to surface phonons

FORMATION OF VIBRATIONALLY EXCITED STATES from HETEROGENEOUS ATOM RECOMBINATION H(gas) + Hads H 2( ) DIRECT ELEY RIDEAL (E-R) MECHANISM H(gas) H (trapped) H(trapped) + Hads H 2( ) HOT ATOM (HA) MECHANISM Hads + Hads H 2( ) HINSHELWOOD-LANGMUIR (H-L) MECHANISM !!Different energetics depending on the nature of the adsorbed atom e. g. physi-adsorbed; chemi-adsorbed!! PHYSI-ADSORBED: practically all the recombination energy can go into vibrational excitation of desorbed molecules in both E-R and H-L mechanisms CHEMI-ADSORBED: only the difference between the dissociation energy of the diatom and the adsorption energy of atom(s) can go into vibrational energy of the desorbed molecules
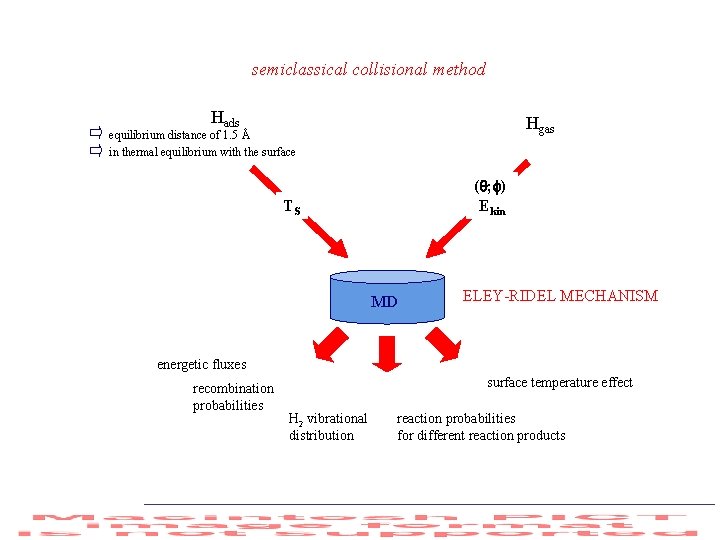
semiclassical collisional method Hads Hgas equilibrium distance of 1. 5 Å in thermal equilibrium with the surface ( ; ) Ekin TS MD ELEY-RIDEL MECHANISM energetic fluxes recombination probabilities surface temperature effect H 2 vibrational distribution reaction probabilities for different reaction products

VIBRATIONAL DISTRIBUTIONS from PHYSIADSORBED H and D ATOMS on COPPER (E-R mechanism, BILLING&CACCIATORE)
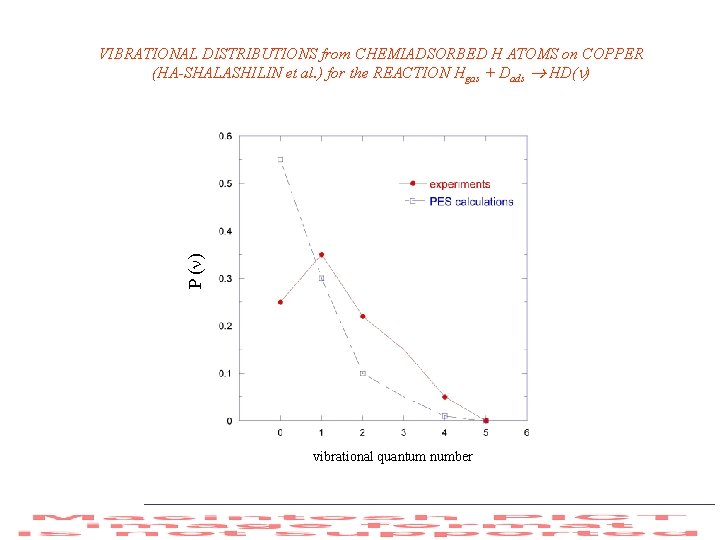
P ( ) VIBRATIONAL DISTRIBUTIONS from CHEMIADSORBED H ATOMS on COPPER (HA-SHALASHILIN et al. ) for the REACTION Hgas + Dads HD( ) vibrational quantum number

VIBRATIONAL DISTRIBUTIONS from PHYSIADSORBED H ATOMS on GRAPHITE (E-R mechanism, H-L mechanism, SIDIS&MORISSET) SCHEME OF THE REACTION PATH (v, j) distribution of the H 2 product
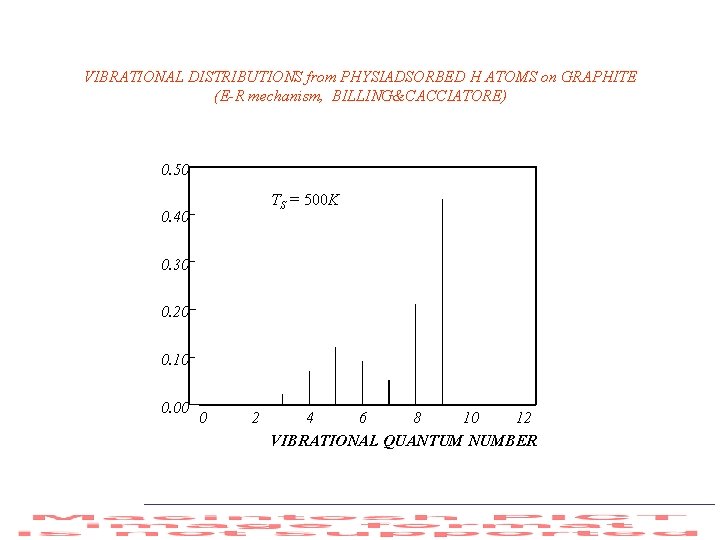
VIBRATIONAL DISTRIBUTIONS from PHYSIADSORBED H ATOMS on GRAPHITE (E-R mechanism, BILLING&CACCIATORE) 0. 50 TS = 500 K 0. 40 0. 30 0. 20 0. 10 0. 00 0 2 4 6 8 10 12 VIBRATIONAL QUANTUM NUMBER

OUTLINE - Open problems (1985 -1995) in Multipole Magnetic Plasmas a) Validation of Bari code with FOM experiments b) Extension to D 2 plasmas c) Pulsed discharges d) Rydberg states e) Wall effects - Cross Section Improvements (1995 -2005) a) Electron-molecule cross sections b) Heavy particle collision cross sections c) Gas surface interaction - Kinetic models Improvements(1995 -2005) a) New multipole zerodimensional code b) Parallel-plate 1 D code c) RF and MW quasi 1 D code - Air Plasmas a) N 2 and O 2 State-to-State Cross Sections

CROSS SECTIONS IMPROVEMENTS ELECTRONIC EXCITATION to the lowest SINGLETS X 1 g ( i) B 1 u X 1 g ( i) C 1 u
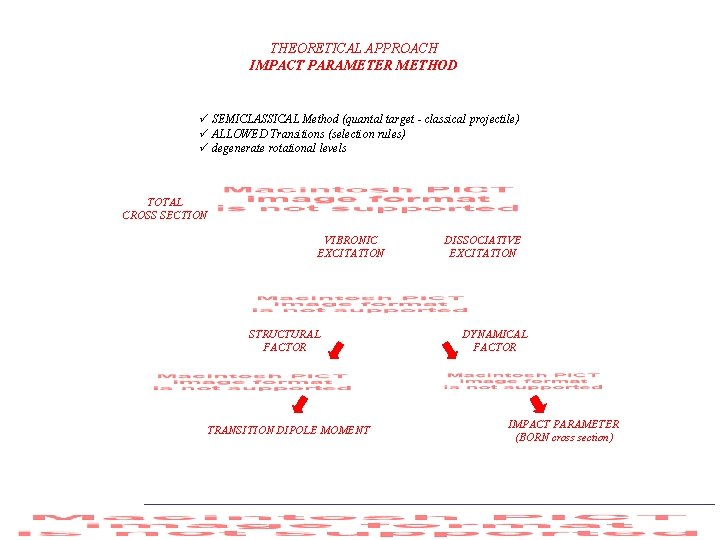
THEORETICAL APPROACH IMPACT PARAMETER METHOD ü SEMICLASSICAL Method (quantal target - classical projectile) ü ALLOWED Transitions (selection rules) ü degenerate rotational levels TOTAL CROSS SECTION VIBRONIC EXCITATION STRUCTURAL FACTOR TRANSITION DIPOLE MOMENT DISSOCIATIVE EXCITATION DYNAMICAL FACTOR IMPACT PARAMETER (BORN cross section)

CROSS SECTIONS for H 2 ISOTOPIC VARIANTS X 1 g ( i) B 1 u E = 40 e. V

RESONANT VIBRATIONAL EXCITATION H 2 (X 1 g, ni) + e H 2 - H 2 (X 1 g, nf) + e E = 5 e. V

EXCITATION of low-lying RYDBERG STATES X 1 g ( i) B’ 1 u X 1 g ( i) D 1 u X 1 g ( i) B” 1 u X 1 g ( i) D’ 1 u

s n-4

DISSOCIATION DIRECT DISSOCIATION through EXCITED STATES excited bound state ’ B 1 u and C 1 u + low-lying RYDBERG STATES B’, B” 1 u , D, D’ 1 u DISSOCIATION pure repulsive state i

theoretical approach in atom-molecule collisions: QCT Method atomic motion is considered classical on the potential energy surface (PES) initial and final states are approximated with pseudoquantization rules COMPUTATIONAL LOAD months on fast processor RELIABILITY of METHOD good
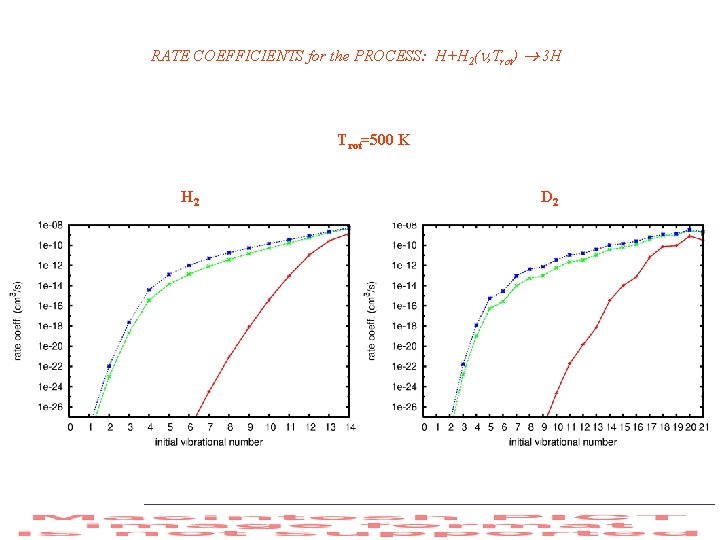
RATE COEFFICIENTS for the PROCESS: H+H 2( , Trot) 3 H Trot=500 K H 2 D 2
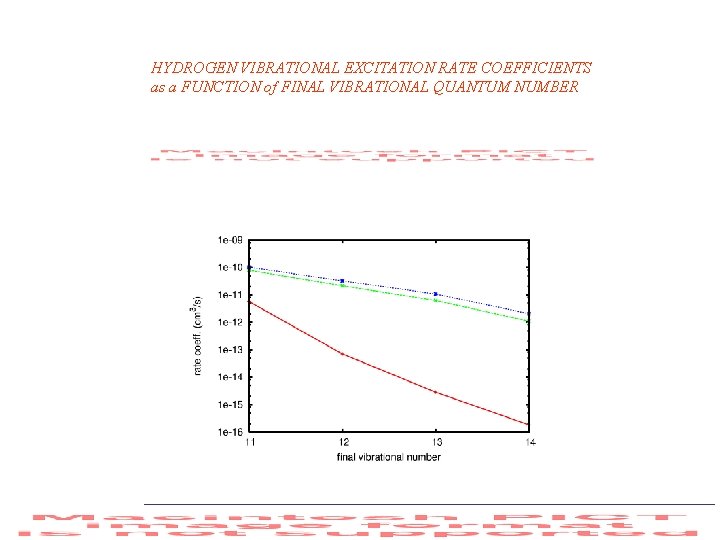
HYDROGEN VIBRATIONAL EXCITATION RATE COEFFICIENTS as a FUNCTION of FINAL VIBRATIONAL QUANTUM NUMBER
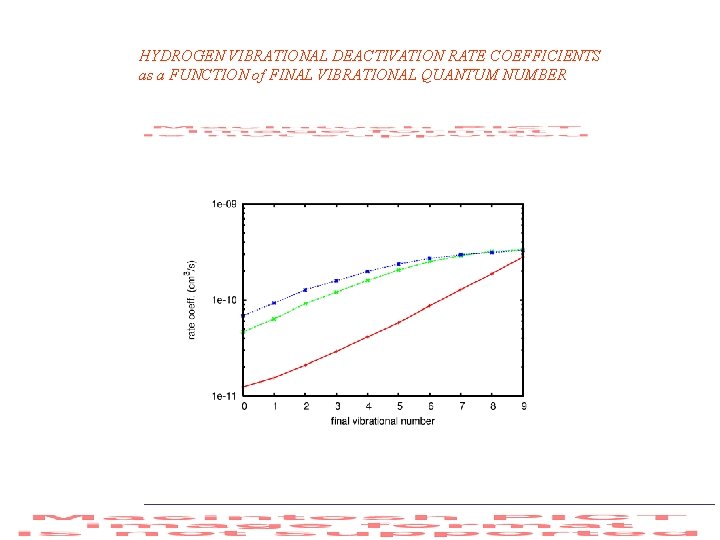
HYDROGEN VIBRATIONAL DEACTIVATION RATE COEFFICIENTS as a FUNCTION of FINAL VIBRATIONAL QUANTUM NUMBER
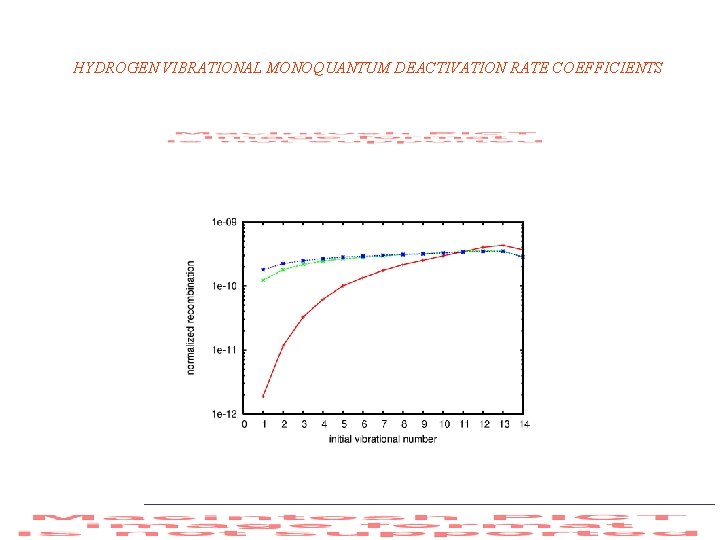
HYDROGEN VIBRATIONAL MONOQUANTUM DEACTIVATION RATE COEFFICIENTS
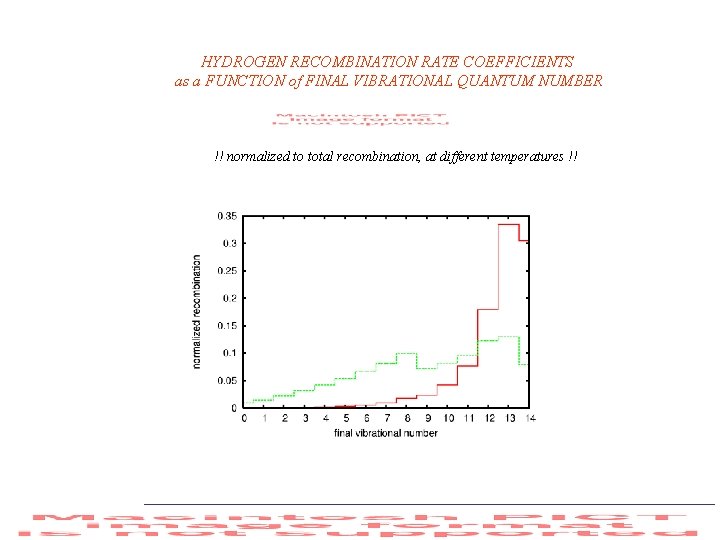
HYDROGEN RECOMBINATION RATE COEFFICIENTS as a FUNCTION of FINAL VIBRATIONAL QUANTUM NUMBER !! normalized to total recombination, at different temperatures !!

OUTLINE - Open problems (1985 -1995) in Multipole Magnetic Plasmas a) Validation of Bari code with FOM experiments b) Extension to D 2 plasmas c) Pulsed discharges d) Rydberg states e) Wall effects - Cross Section Improvements (1995 -2005) a) Electron-molecule cross sections b) Heavy particle collision cross sections c) Gas surface interaction - Kinetic models Improvements(1995 -2005) a) New multipole zerodimensional code b) Parallel-plate 1 D code c) RF and MW quasi 1 D code - Air Plasmas a) N 2 and O 2 State-to-State Cross Sections
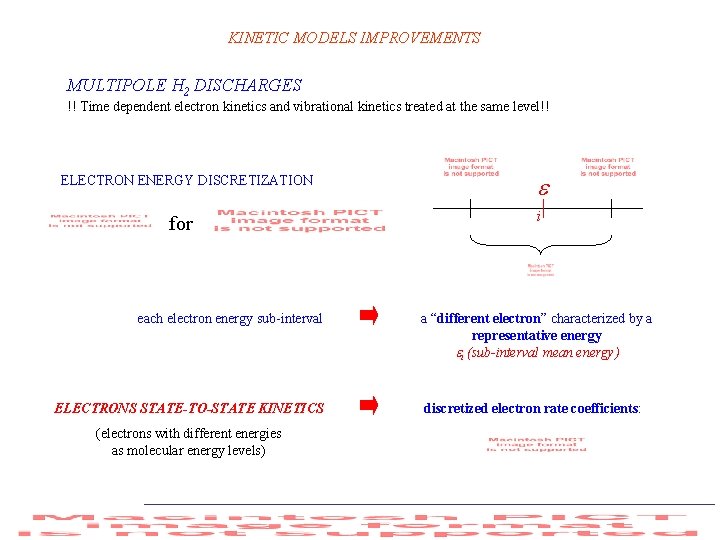
KINETIC MODELS IMPROVEMENTS MULTIPOLE H 2 DISCHARGES !! Time dependent electron kinetics and vibrational kinetics treated at the same level!! ELECTRON ENERGY DISCRETIZATION for each electron energy sub-interval ELECTRONS STATE-TO-STATE KINETICS (electrons with different energies as molecular energy levels) i a “different electron” characterized by a representative energy i (sub-interval mean energy) discretized electron rate coefficients:

VDF and EEDF as a function of PRESSURE TG =500 K DISCHARGE CURRENT=10 A DISCHARGE VOLTAGE=100 V

VDF and EEDF as a function of CURRENT TG =500 K PRESSURE=7. 5 mtorr DISCHARGE VOLTAGE=100 V
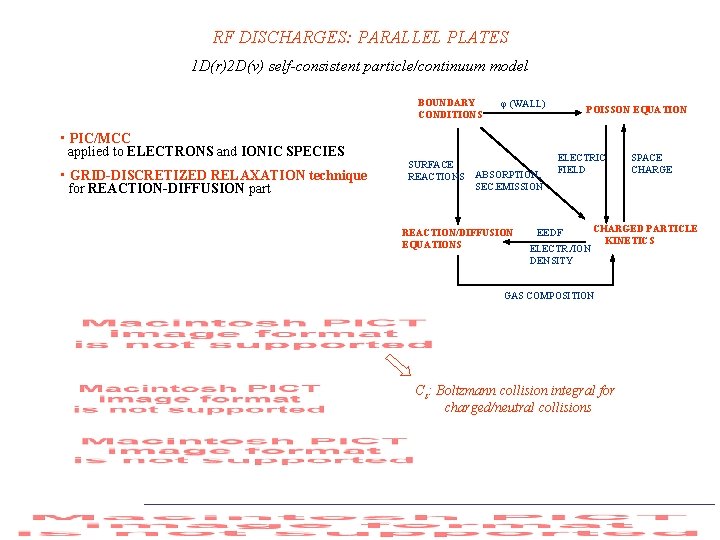
RF DISCHARGES: PARALLEL PLATES 1 D(r)2 D(v) self-consistent particle/continuum model BOUNDARY CONDITIONS (WALL) • PIC/MCC applied to ELECTRONS and IONIC SPECIES • GRID-DISCRETIZED RELAXATION technique for REACTION-DIFFUSION part SURFACE REACTIONS ABSORPTION, SEC. EMISSION REACTION/DIFFUSION EQUATIONS POISSON EQUATION ELECTRIC FIELD EEDF ELECTR. /ION DENSITY SPACE CHARGED PARTICLE KINETICS GAS COMPOSITION Cs: Boltzmann collision integral for charged/neutral collisions
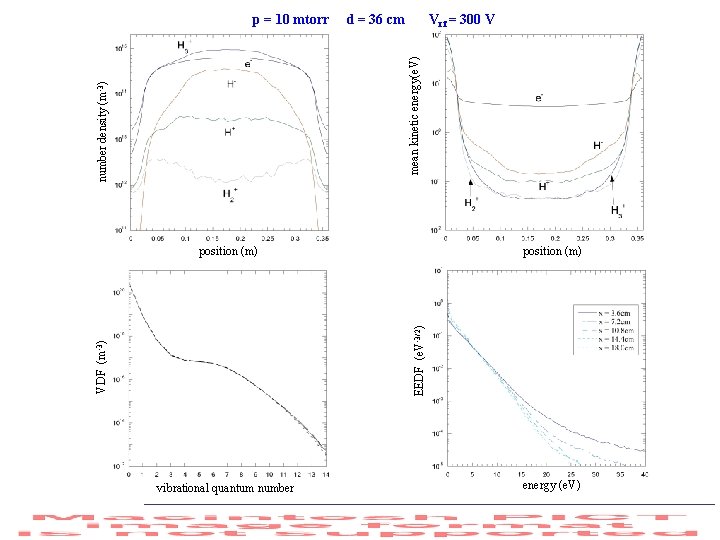
d = 36 cm Vrf = 300 V number density (m-3) mean kinetic energy(e. V) p = 10 mtorr position (m) VDF (m-3) EEDF (e. V-3/2) position (m) vibrational quantum number energy (e. V)

FUTURE STEPS • CONSTRUCTION OF A DATA BASE OF CROSS SECTIONS FOR H 2 AND ISOTOPES • RYDBERG KINETICS AND GAS-SURFACE INTERACTIONS • INSERTION OF THE COMPLETE DATA BASE IN 1 D-2 D CODES • EXTENSION TO SURFACE SOURCES • VALIDATION OF THE PREDICTIVE CODE WITH DEDICATED EXPERIMENTS • AGREEMENT PROTOCOL WITH ITER PROGRAMME

HARPOON REACTION INVOLVING CESIUM ATOM AND HYDROGEN MOLECULE Asymptotic Approach

OUTLINE - Open problems (1985 -1995) in Multipole Magnetic Plasmas a) Validation of Bari code with FOM experiments b) Extension to D 2 plasmas c) Pulsed discharges d) Rydberg states e) Wall effects - Cross Section Improvements (1995 -2005) a) Electron-molecule cross sections b) Heavy particle collision cross sections c) Gas surface interaction - Kinetic models Improvements(1995 -2005) a) New multipole zerodimensional code b) Parallel-plate 1 D code c) RF and MW quasi 1 D code - Air Plasmas a) N 2 and O 2 State-to-State Cross Sections

AIR PLASMAS ELECTRON IMPACT induced PROCESSES in HOMONUCLEAR DIATOMIC MOLECULES NON-DISSOCIATIVE IONIZATION of N 2 VIBRONIC EXCITATION and (PRE)DISSOCIATION of O 2 and N 2 ATOM-DIATOM COLLISION PROCESSES DISSOCIATION/RECOMBINATION of O 2 and N 2 ENERGY EXCHANGE (VT Processes) of O 2 and N 2

IONIZATION N 2 -N 2+ system POTENTIAL ENERGY CURVES F. R. Gilmore, J. Q. R. S. T. 5, 369 (1965)
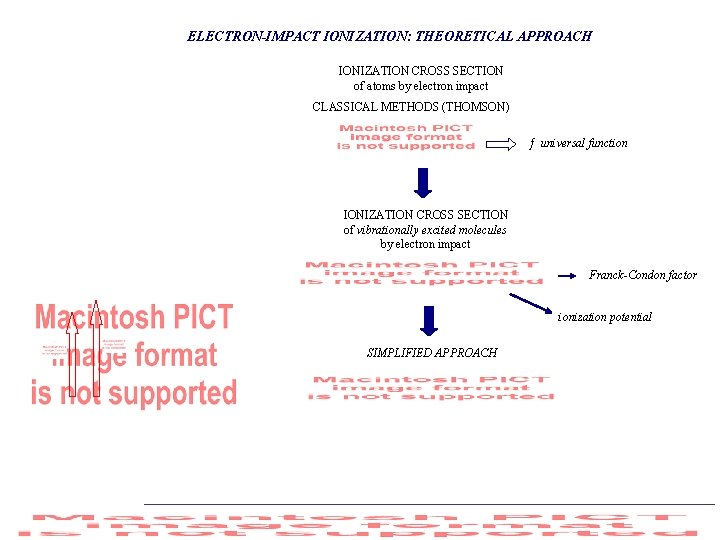
ELECTRON-IMPACT IONIZATION: THEORETICAL APPROACH IONIZATION CROSS SECTION of atoms by electron impact CLASSICAL METHODS (THOMSON) ƒ universal function IONIZATION CROSS SECTION of vibrationally excited molecules by electron impact Franck-Condon factor ionization potential SIMPLIFIED APPROACH
![ELECTRON-IMPACT IONIZATION from GROUND STATE cross section [10 -17 cm 2] ionic state Van ELECTRON-IMPACT IONIZATION from GROUND STATE cross section [10 -17 cm 2] ionic state Van](http://slidetodoc.com/presentation_image_h/ac869d64df8dc36450a9b5f68f88b6fa/image-53.jpg)
ELECTRON-IMPACT IONIZATION from GROUND STATE cross section [10 -17 cm 2] ionic state Van Zyl 0. 320 0. 535 0. 145 this work 0. 30 0. 50 0. 20 E=100 e. V [J. Geophys. Res. 100, 23755 (1995)]

ELECTRON-IMPACT IONIZATION from EXCITED STATE

O 2 system POTENTIAL ENERGY CURVES: Schumann-Runge transition

DISSOCIATIVE O 2 CHANNELS

The N 2 Birge-Hopfield system Direct Dissociation through the excited state Vibronic Excitation Dissociation through Predissociative Channels D. Spelsberg, W. Meyer, Journal of Chemical Physics 115 (2001) 6438

X 1 g ( i) b 1 u E=40 e. V X 1 g ( i) b 1 u (continuum) E=40 e. V

CROSS SECTION DEPENDENCE on the INITIAL VIBRATIONAL QUANTUM NUMBER
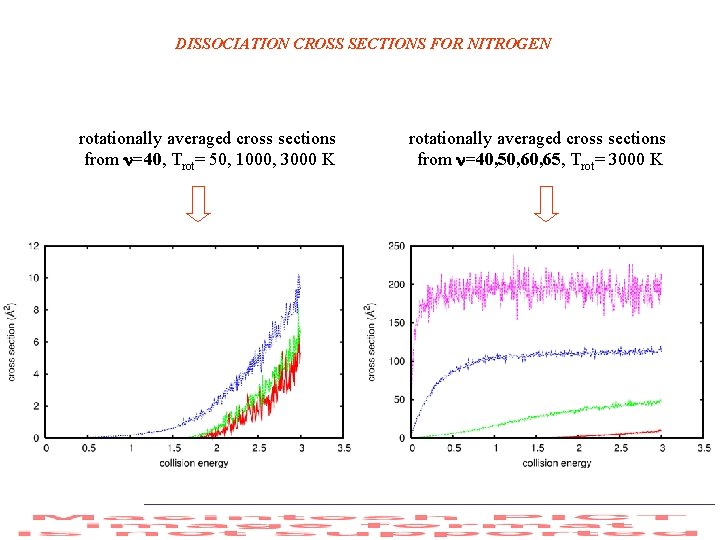
DISSOCIATION CROSS SECTIONS FOR NITROGEN rotationally averaged cross sections from =40, Trot= 50, 1000, 3000 K rotationally averaged cross sections from =40, 50, 65, Trot= 3000 K
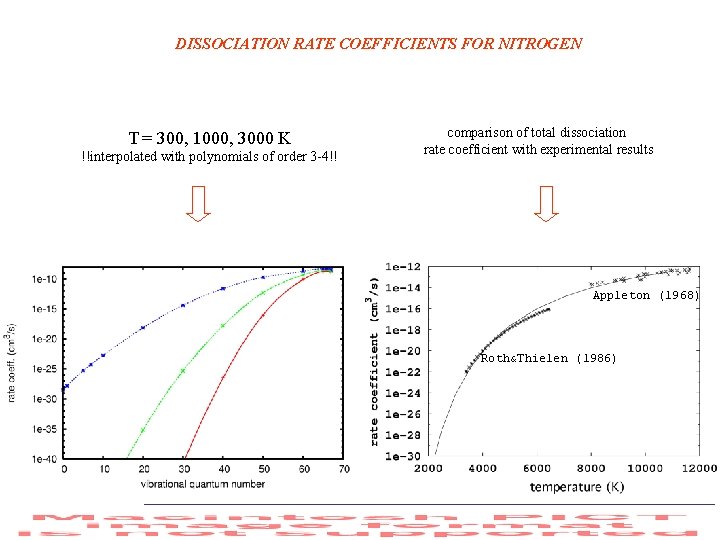
DISSOCIATION RATE COEFFICIENTS FOR NITROGEN T = 300, 1000, 3000 K !!interpolated with polynomials of order 3 -4!! comparison of total dissociation rate coefficient with experimental results Appleton (1968) Roth&Thielen (1986)
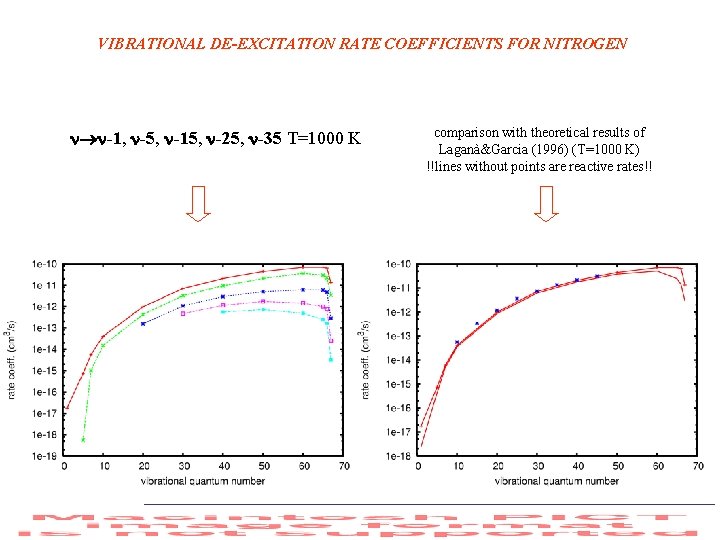
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR NITROGEN -1, -5, -15, -25, -35 T=1000 K comparison with theoretical results of Laganà&Garcia (1996) (T=1000 K) !!lines without points are reactive rates!!
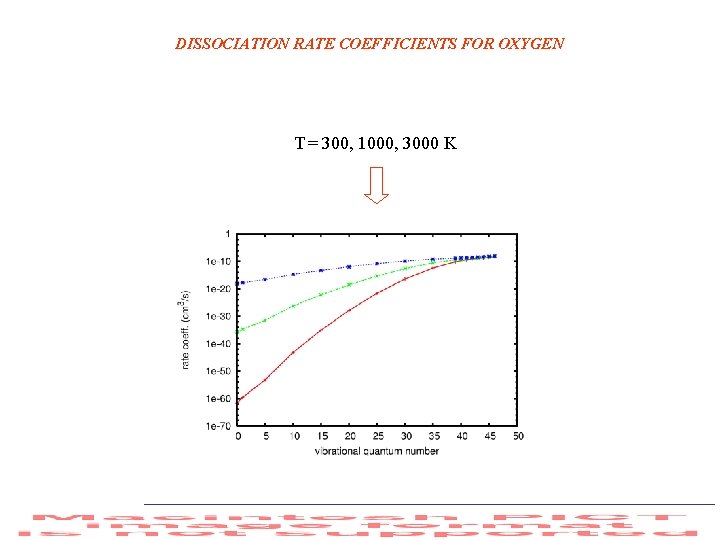
DISSOCIATION RATE COEFFICIENTS FOR OXYGEN T = 300, 1000, 3000 K
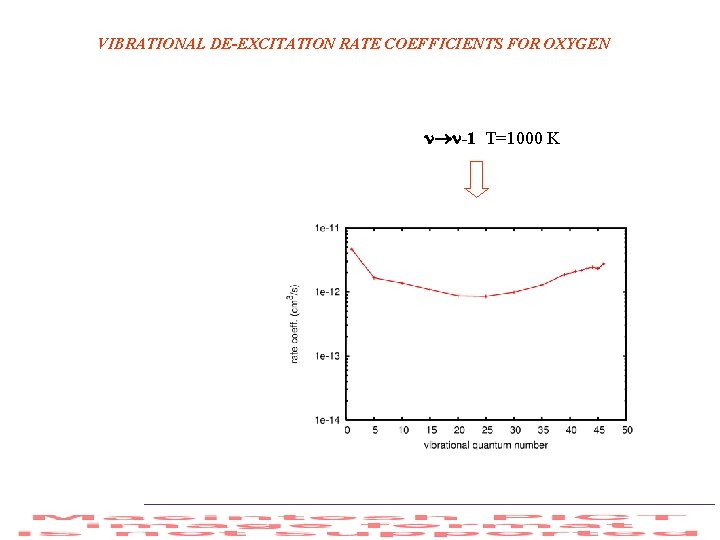
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN -1 T=1000 K
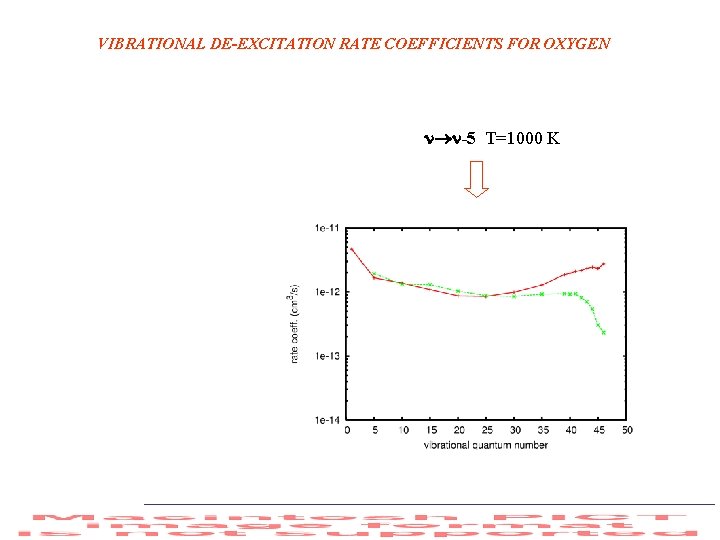
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN -5 T=1000 K
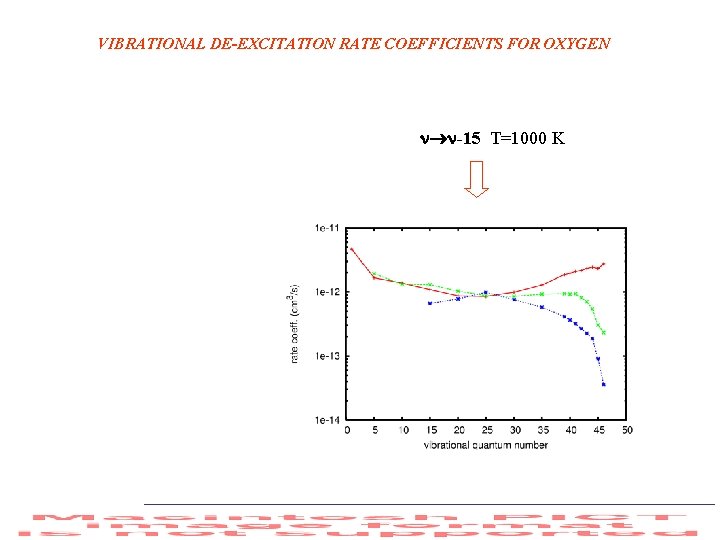
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN -15 T=1000 K
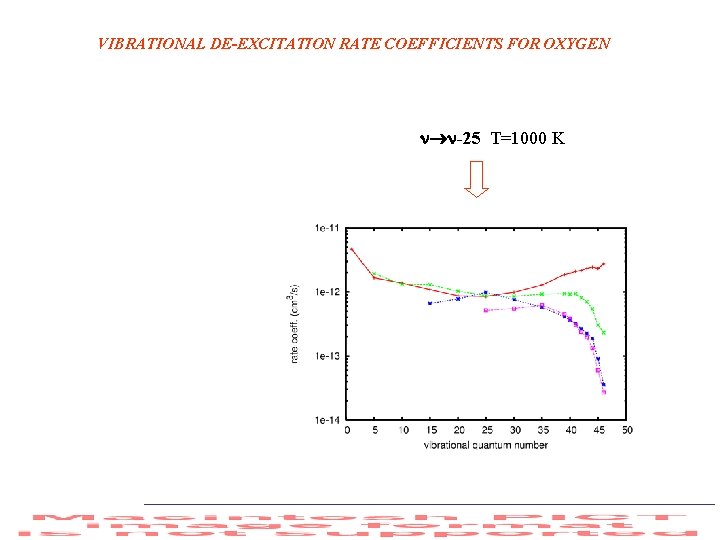
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN -25 T=1000 K
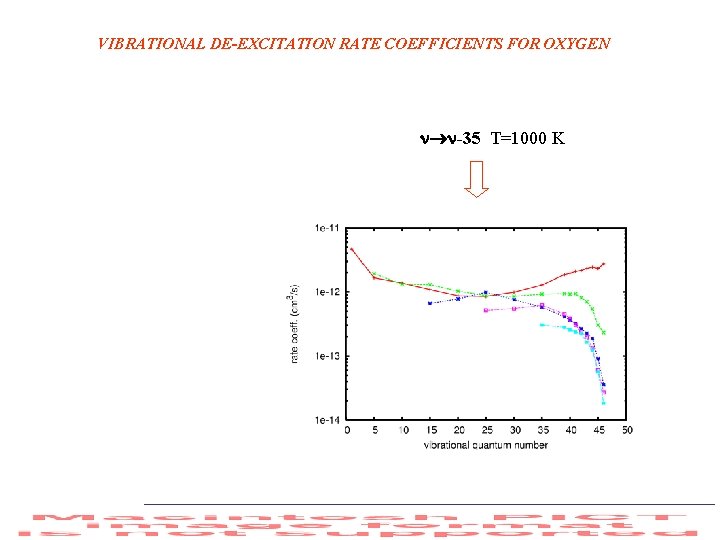
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN -35 T=1000 K
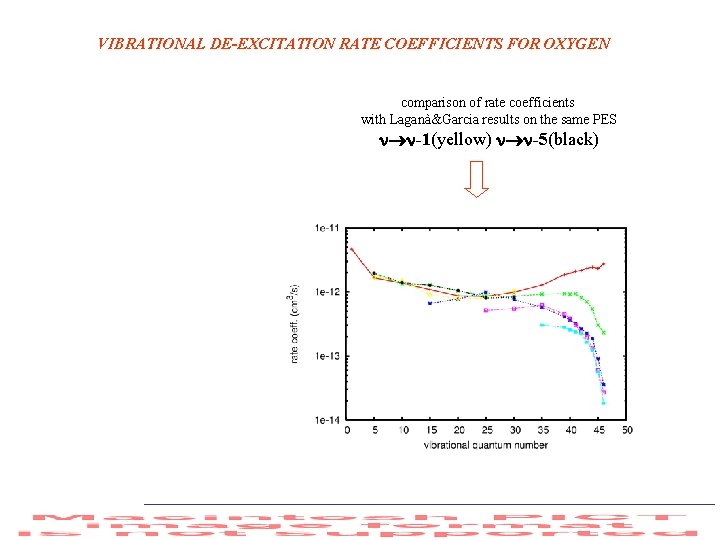
VIBRATIONAL DE-EXCITATION RATE COEFFICIENTS FOR OXYGEN comparison of rate coefficients with Laganà&Garcia results on the same PES -1(yellow) -5(black)
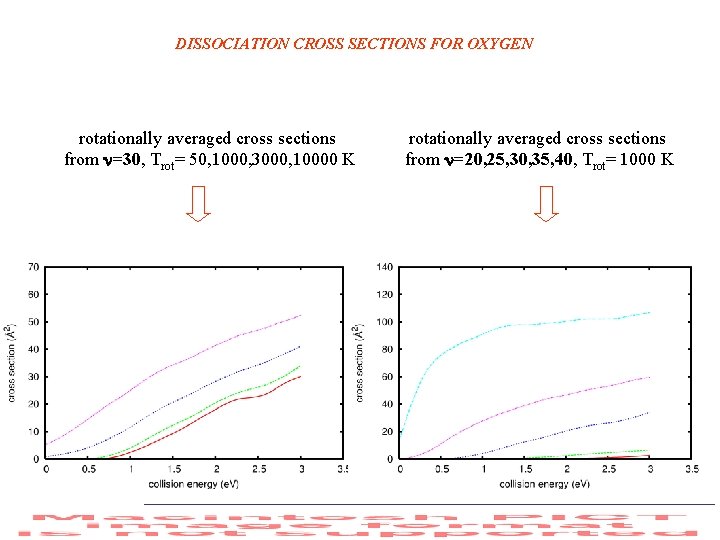
DISSOCIATION CROSS SECTIONS FOR OXYGEN rotationally averaged cross sections from =30, Trot= 50, 1000, 3000, 10000 K rotationally averaged cross sections from =20, 25, 30, 35, 40, Trot= 1000 K

TOTAL DISSOCIATION CROSS SECTIONS FOR OXYGEN !! COMPARISON with some EXPERIMENTAL FITS!! • Our rate is similar to that of Shatalov within ± 13% over the whole interval 1000 -10000 K • NF: no correction factor • VF: variable factor
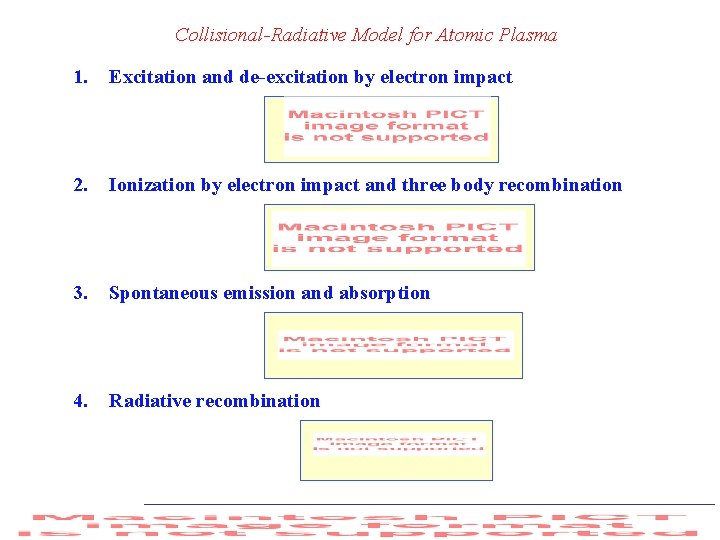
Collisional-Radiative Model for Atomic Plasma 1. Excitation and de-excitation by electron impact 2. Ionization by electron impact and three body recombination 3. Spontaneous emission and absorption 4. Radiative recombination
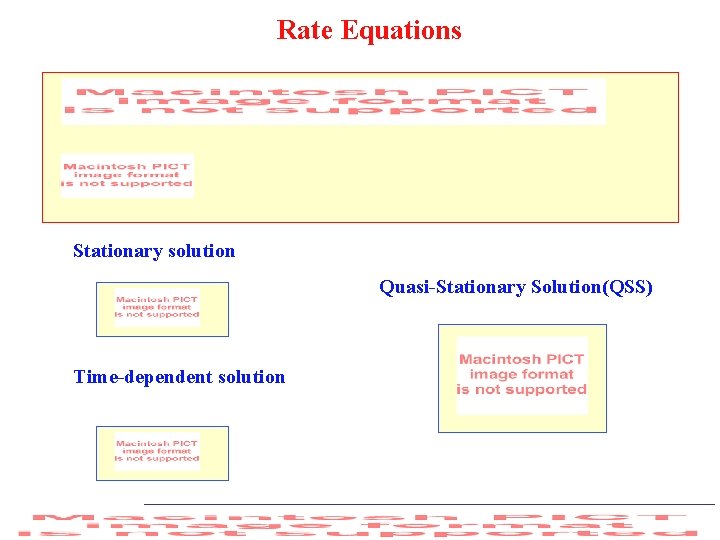
Rate Equations Stationary solution Quasi-Stationary Solution(QSS) Time-dependent solution
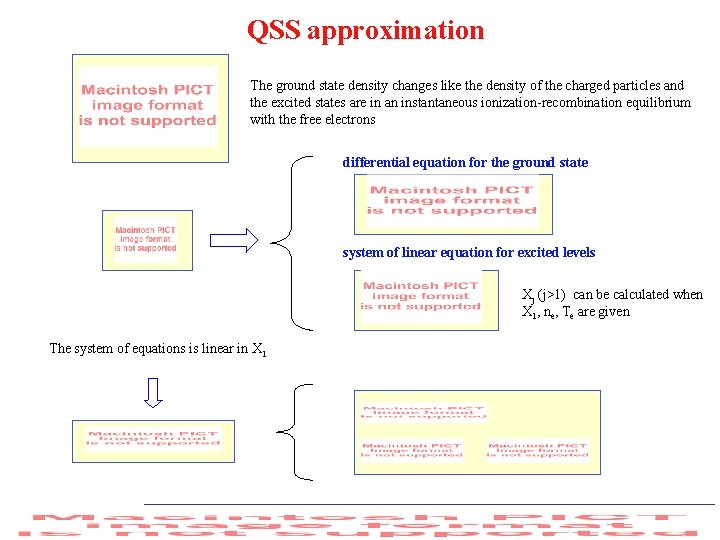
QSS approximation The ground state density changes like the density of the charged particles and the excited states are in an instantaneous ionization-recombination equilibrium with the free electrons differential equation for the ground state system of linear equation for excited levels Xj (j>1) can be calculated when X 1, ne, Te are given The system of equations is linear in X 1

CR for Atomic Nitrogen Plasma: Energy-level Model

CR for Atomic Nitrogen Plasma with QSS Xi vs level energy Te=5800 K Te=11600 K Te=17400 K
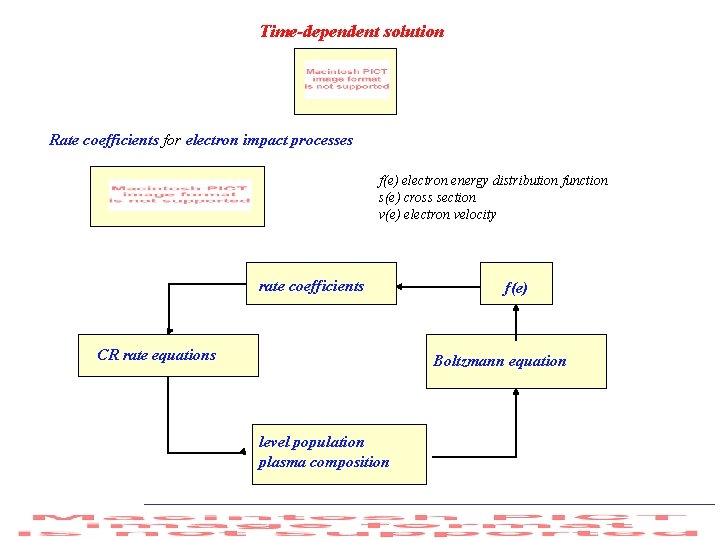
Time-dependent solution Rate coefficients for electron impact processes f(e) electron energy distribution function s(e) cross section v(e) electron velocity rate coefficients CR rate equations f(e) Boltzmann equation level population plasma composition

ATOMIC HYDROGEN PLASMA P=100 Torr, Tg=30000 K, Te(t=0)=1000 K density (cm-3) vs time(s) c H+ =c e- =10 -8 , c. H=1, c. H(1)=1, c. H(i)=0 Xi = ni/ni. SB vs time(s) i>1 Te vs time(s)

c. H(i)/g(i) vs Ei eedf(e. V-3/2) vs E
- Slides: 79