LUNG CANCER SCREENING Albert A Rizzo MD FACP

LUNG CANCER SCREENING Albert A. Rizzo, MD FACP FCCP Section Chief Pulmonary/ Critical Care Medicine Christiana Care Health System, Newark De & Immediate Past-Chair National Board of Directors American Lung Association September 2012

Disclosures • • • Pfizer – Speakers’ Bureau and Research Novartis – Speakers’ Bureau and Research Boehringer Ingelheim – Researach CSL Behring – Consulting Intermune - Research

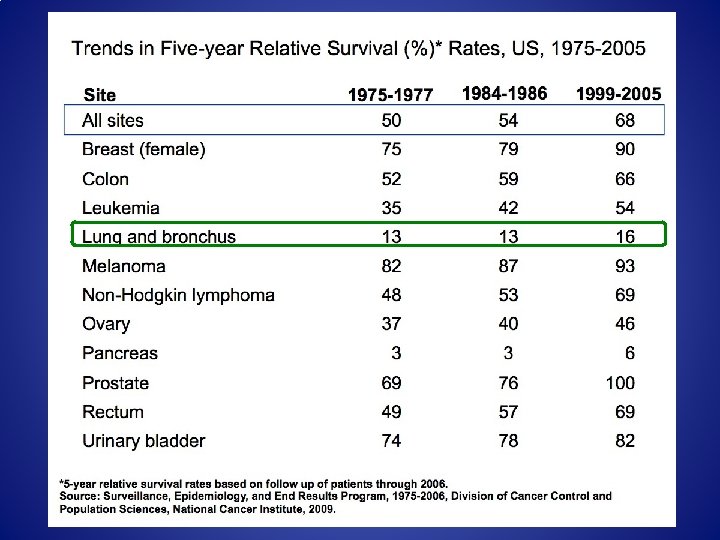
Some initial facts • In the year 2012, ACS estimates 260, 000 new cases of lung cancer and 160, 000 lung cancer related deaths • Worldwide in 2008, 1. 4 million lung cancer deaths • 75% of patients with lung cancer present with symptoms due to advanced local or metastatic disease not amenable to cure • 5 yr survival of 16%

What is Screening? • Screening for cancer means testing for cancer before there any symptoms. • Screening for some types of cancer has reduced deaths by early detection and treatment. Now there is a test that can reduce death from lung cancer through early detection. • The best way to prevent lung cancer is to never smoke or stop smoking now. If your patients are still smoking, talk to them about ways you can help them quit smoking.

Reasons That Screening in Lung Cancer would be effective High morbidity and mortality Significant prevalence (0. 5 -2. 2%) Identifiable risk factors (allow targeting) Lengthy pre-clinical phase Therapy is more effective in early stage disease

A Guiding Principle of Lung Cancer Screening • Make sure that the benefits of treating the smaller number of patients who will receive a lung cancer diagnosis through screening outweigh the harms that could occur from screening a large population of healthy individuals.

Ideal Screening Test High Sensitivity High Specificity Safe and Acceptable Low Cost Decrease mortality and/or improve quality of life

Outcomes to be assessed Cancer detection rates Stage at detection Survival Disease-specific mortality Overall mortality

Potential Harms of Screening Detection of benign abnormalities Radiation risks Prolonged follow-up Overdiagnosis

Lung Cancer Screening Trials

Understanding terms… Survival = # alive with lung cancer # with lung cancer Mortality = # lung cancer deaths # screened
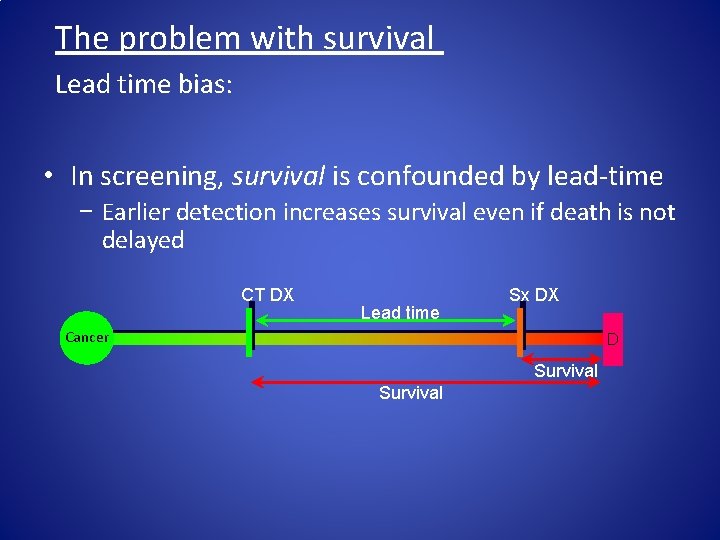
The problem with survival Lead time bias: • In screening, survival is confounded by lead-time − Earlier detection increases survival even if death is not delayed CT DX Lead time Sx DX Cancer D Survival
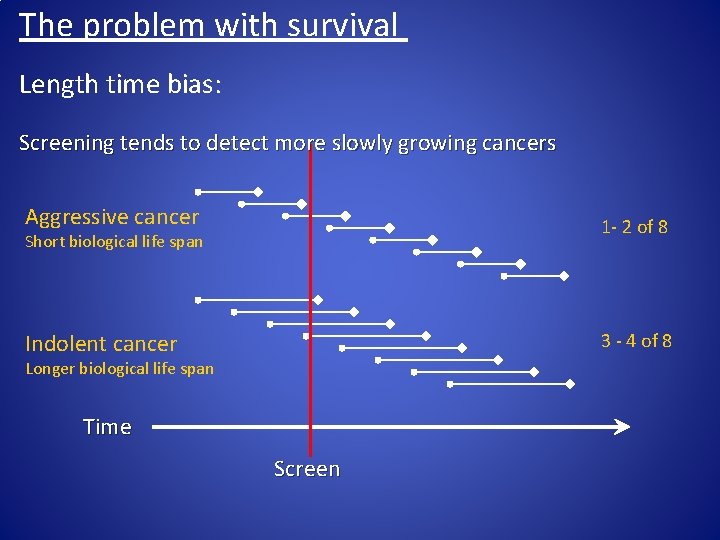
The problem with survival Length time bias: Screening tends to detect more slowly growing cancers Aggressive cancer 1 - 2 of 8 Indolent cancer 3 - 4 of 8 Short biological life span Longer biological life span Time Screen
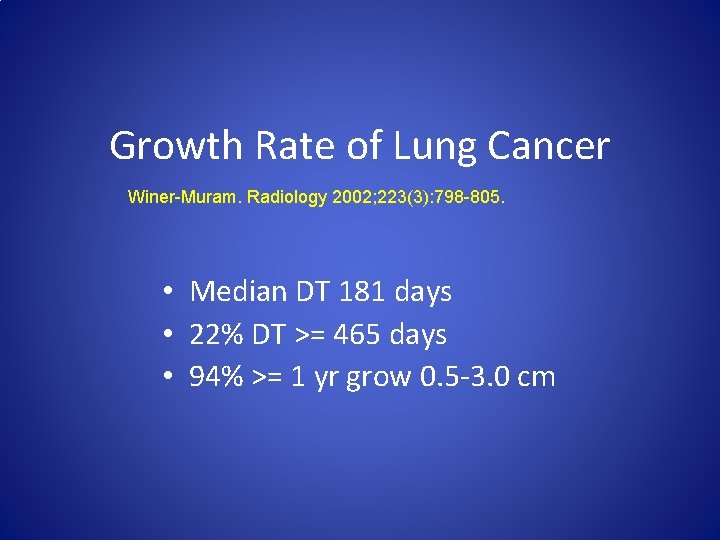
Growth Rate of Lung Cancer Winer-Muram. Radiology 2002; 223(3): 798 -805. • Median DT 181 days • 22% DT >= 465 days • 94% >= 1 yr grow 0. 5 -3. 0 cm
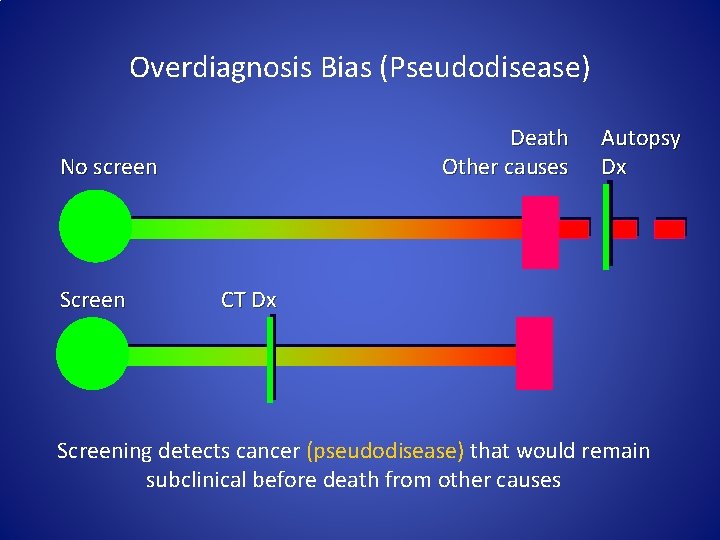
Overdiagnosis Bias (Pseudodisease) Death Other causes No screen Screen Autopsy Dx CT Dx Screening detects cancer (pseudodisease) that would remain subclinical before death from other causes

Effects of Overdiagnosis • Falsely increases sensitivity of test • Falsely increases PPV of test • Falsely increases incidence • Falsely improves stage distribution • Falsely improves case survival • Does not decrease pop mortality

Volunteer Bias May not represent general population More concerned they have increased risk More than usually health conscious (lower risk)
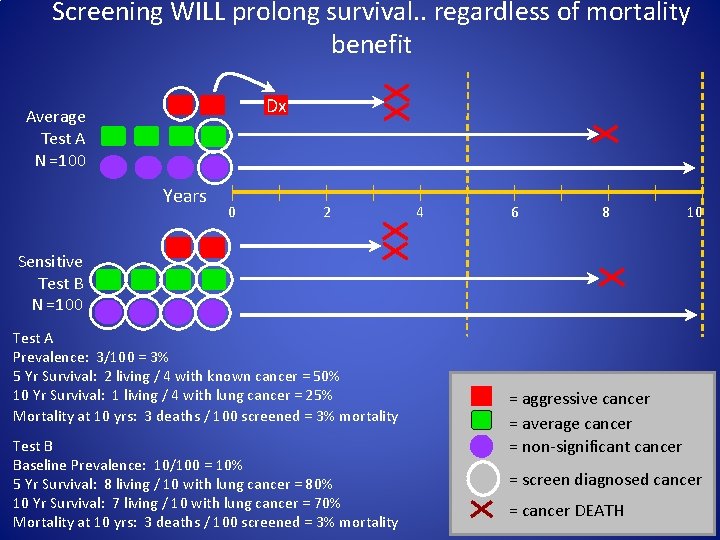
Screening WILL prolong survival. . regardless of mortality benefit Dx Average Test A N =100 Years 0 2 4 6 8 10 Sensitive Test B N =100 Test A Prevalence: 3/100 = 3% 5 Yr Survival: 2 living / 4 with known cancer = 50% 10 Yr Survival: 1 living / 4 with lung cancer = 25% Mortality at 10 yrs: 3 deaths / 100 screened = 3% mortality Test B Baseline Prevalence: 10/100 = 10% 5 Yr Survival: 8 living / 10 with lung cancer = 80% 10 Yr Survival: 7 living / 10 with lung cancer = 70% Mortality at 10 yrs: 3 deaths / 100 screened = 3% mortality = aggressive cancer = average cancer = non-significant cancer = screen diagnosed cancer = cancer DEATH
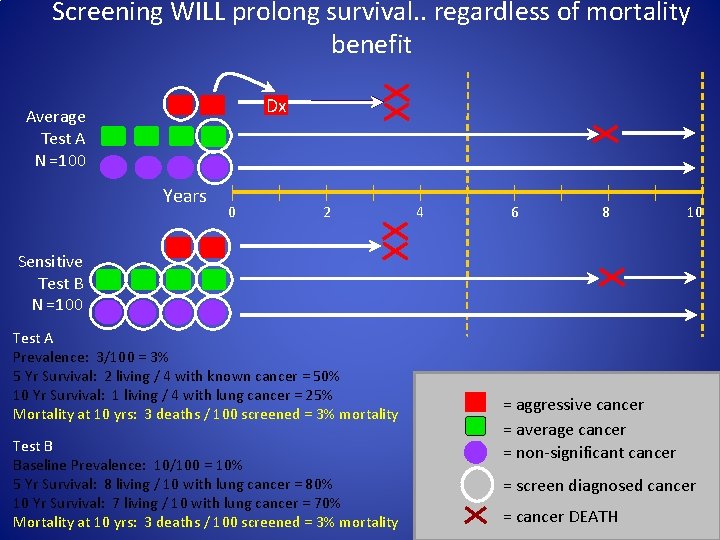
Screening WILL prolong survival. . regardless of mortality benefit Dx Average Test A N =100 Years 0 2 4 6 8 10 Sensitive Test B N =100 Test A Prevalence: 3/100 = 3% 5 Yr Survival: 2 living / 4 with known cancer = 50% 10 Yr Survival: 1 living / 4 with lung cancer = 25% Mortality at 10 yrs: 3 deaths / 100 screened = 3% mortality Test B Baseline Prevalence: 10/100 = 10% 5 Yr Survival: 8 living / 10 with lung cancer = 80% 10 Yr Survival: 7 living / 10 with lung cancer = 70% Mortality at 10 yrs: 3 deaths / 100 screened = 3% mortality = aggressive cancer = average cancer = non-significant cancer = screen diagnosed cancer = cancer DEATH

CT-detected Lung Cancers will Have ↑ survival • Lead-time: advance time of Dx even absent a delay in death • Screening selects for biologically favorable cancers (length bias) • Some proportion = overdiagnosis (overtreatment) − Biologically benign behavior − Death from competing cause • True ↑ in longevity indeterminate

History of lung cancer screening Early lung cancer screening with sputum and CXRs in 1970’s Radiology 2011
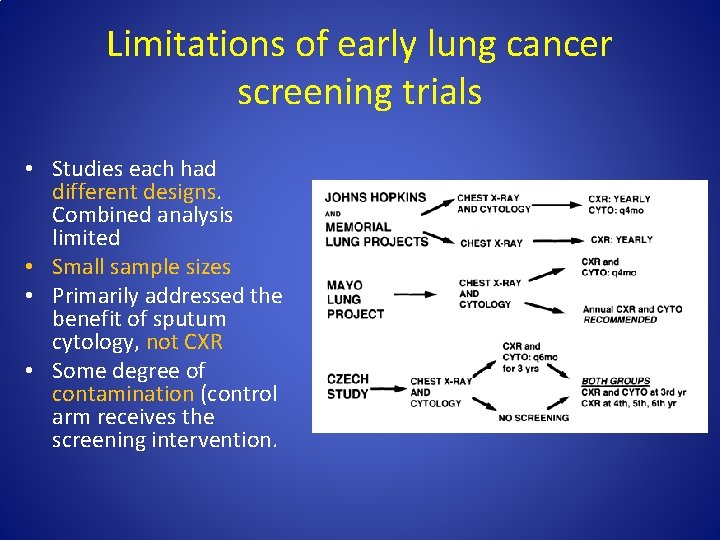
Limitations of early lung cancer screening trials • Studies each had different designs. Combined analysis limited • Small sample sizes • Primarily addressed the benefit of sputum cytology, not CXR • Some degree of contamination (control arm receives the screening intervention.
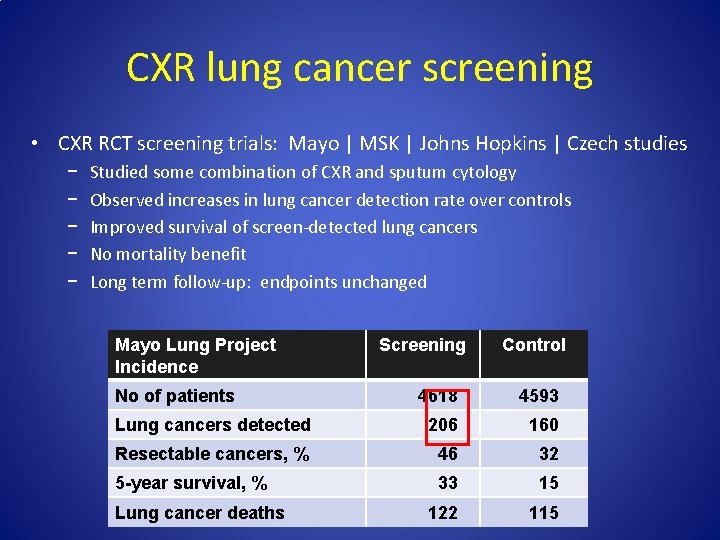
CXR lung cancer screening • CXR RCT screening trials: Mayo | MSK | Johns Hopkins | Czech studies − − − Studied some combination of CXR and sputum cytology Observed increases in lung cancer detection rate over controls Improved survival of screen-detected lung cancers No mortality benefit Long term follow-up: endpoints unchanged Mayo Lung Project Incidence No of patients Screening Control 4618 4593 Lung cancers detected 206 160 Resectable cancers, % 46 32 5 -year survival, % 33 15 122 115 Lung cancer deaths

Early Low Dose CT lung cancer screening Trials • Early low dose CT screening: Single arm studies − ELCAP: Henschke CI et al. Lancet 1999; 354: 99 -105. − Japanese Studies • Sobue et al. J Clin Oncol. 2002; 20: 911 -920 • Sone et al. British J Cancer 2001; 84: 25 -32 • Nawa et al. Chest. 2002; 122: 15 -20 − Mayo Study. Swensen SJ et al. Radiology 2005; 235: 259265
![Early low dose CT screening: Single arm studies US Trial N [+] Screens Total Early low dose CT screening: Single arm studies US Trial N [+] Screens Total](http://slidetodoc.com/presentation_image_h/cc9d997c0823cf4789ec16fd4dfa3a92/image-25.jpg)
Early low dose CT screening: Single arm studies US Trial N [+] Screens Total Lung Ca Stage I NSCLC ELCAP CT Prevalence 1000 233 (23%) 27 (2. 7%) 23 (85%) ELCAP CXR Prevalence 1000 68 (7%) * 7(0. 3%) 4 (57%) 7 (2 interval) * 50% were false shadows ; positive screens are actually 3. 5% Incidence Year 1 -2 • • • 2. 5% 5 Trial design: Single arm—subjects received both annual CXR and CT screening Eligibility: Asymptomatic | > 60 yrs | > 10 pack yrs Trial endpoints: The frequencies of • • 1184 Nodule detection Nodules representing malignancy Malignant nodules that are curable ELCAP DID NOT ADDRESS MORTALITY, BUT DEMOSTRATED THE IMPORTANCE OF CT OVER CXR IN LUNG CANCER DETECTION Henschke CI et al. Lancet 1999; 354: 99 -105 Henschke CI et al. Cancer 2001; 92: 153 -159

Early low dose CT screening: CONCLUSIONS • CT detection rates − CT has ~6 -fold higher nodule detection rate than CXR − CT has ~3 -fold higher cancer detection rate than CXR − Benign nodules = majority of detected nodules (> 90%) • CT has 5 -fold increase in resectable lung cancers • No proven benefit in lung cancer mortality True stage shift which requires not only an increase in earlystage disease but a concomitant decrease in late-stage disease when compared with a control population.

N LS T National Lung Screening Trial National Cancer Institute
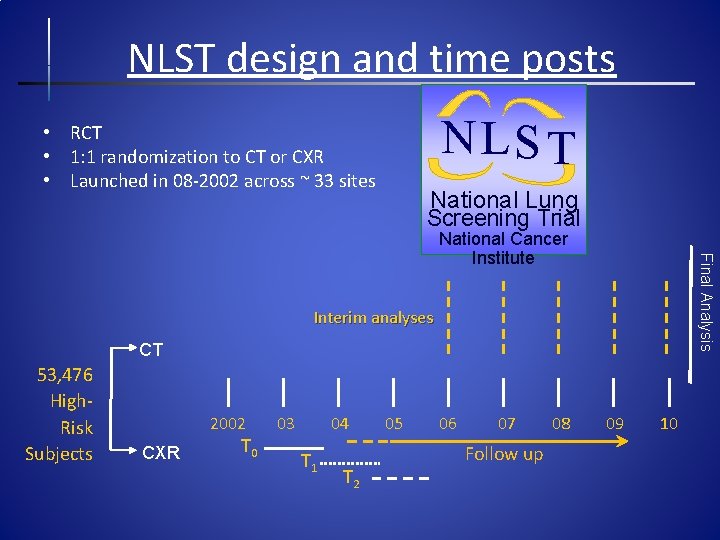
NLST design and time posts NLS T • RCT • 1: 1 randomization to CT or CXR • Launched in 08 -2002 across ~ 33 sites National Lung Screening Trial Final Analysis National Cancer Institute Interim analyses CT 53, 476 High. Risk Subjects 2002 CXR T 0 03 04 T 1 T 2 05 06 07 Follow up 08 09 10

Standardized Eligibility § Males | Females N LS T § 55 -74 Yrs § Asymptomatic § Current or former smokers ≥ 30 pack yrs National Lung § Former smokers have quit within ≤ 15 yrs National Cancer Institute § No prior lung cancer Screening Trial § No cancer within past 5 yrs § No chest CT w/in prior 18 months

Endpoints • Lung cancer specific mortality − 90% power to detect a 20% mortality reduction with LDCT − Compliance: 85% CT | 80% CXR − Contamination: 5% CT | 10% CXR • All cause mortality • Medical resource utilization • ACRIN secondary aims: − Short | long term effects on smoking habits and beliefs − Cost-effectiveness − Specimen biorepository for molecular biomarkers of risk | early Dz NLST Research Team. Radiology 2011; 258(1): 243 -253.
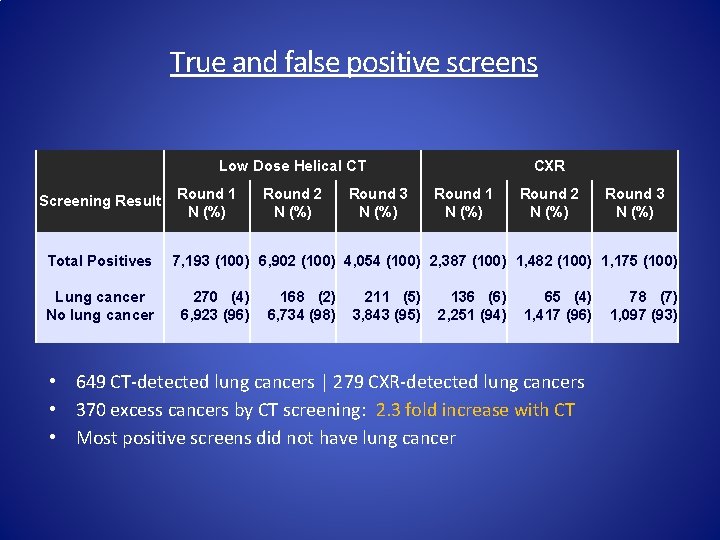
True and false positive screens Low Dose Helical CT Screening Result Total Positives Lung cancer No lung cancer Round 1 N (%) Round 2 N (%) Round 3 N (%) CXR Round 1 N (%) Round 2 N (%) Round 3 N (%) 7, 193 (100) 6, 902 (100) 4, 054 (100) 2, 387 (100) 1, 482 (100) 1, 175 (100) 270 (4) 6, 923 (96) 168 (2) 6, 734 (98) 211 (5) 3, 843 (95) 136 (6) 2, 251 (94) 65 (4) 1, 417 (96) • 649 CT-detected lung cancers | 279 CXR-detected lung cancers • 370 excess cancers by CT screening: 2. 3 fold increase with CT • Most positive screens did not have lung cancer 78 (7) 1, 097 (93)
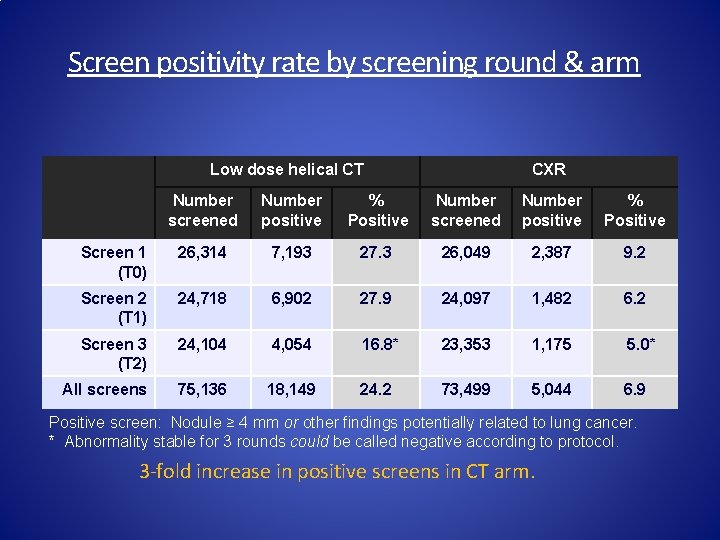
Screen positivity rate by screening round & arm Low dose helical CT CXR Number screened Number positive % Positive Screen 1 (T 0) 26, 314 7, 193 27. 3 26, 049 2, 387 9. 2 Screen 2 (T 1) 24, 718 6, 902 27. 9 24, 097 1, 482 6. 2 Screen 3 (T 2) 24, 104 4, 054 16. 8* 23, 353 1, 175 5. 0* All screens 75, 136 18, 149 24. 2 73, 499 5, 044 6. 9 Positive screen: Nodule ≥ 4 mm or other findings potentially related to lung cancer. * Abnormality stable for 3 rounds could be called negative according to protocol. 3 -fold increase in positive screens in CT arm.
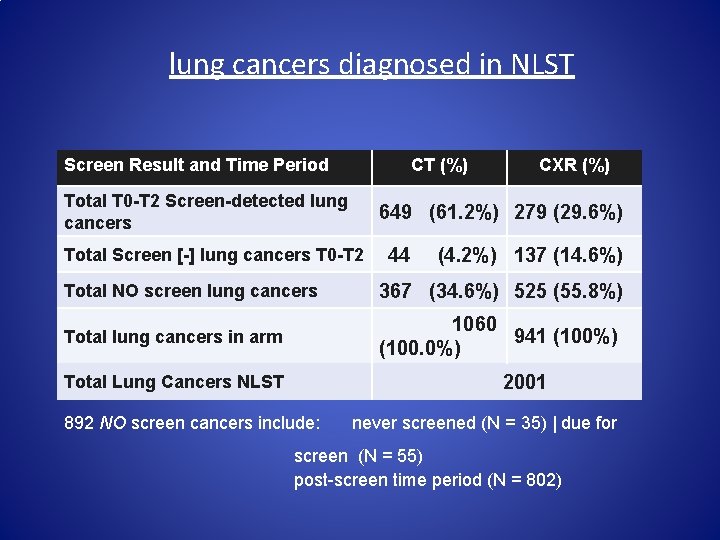
lung cancers diagnosed in NLST Screen Result and Time Period CT (%) Total T 0 -T 2 Screen-detected lung cancers CXR (%) 649 (61. 2%) 279 (29. 6%) Total Screen [-] lung cancers T 0 -T 2 44 (4. 2%) 137 (14. 6%) Total NO screen lung cancers 367 (34. 6%) 525 (55. 8%) Total lung cancers in arm 1060 941 (100%) (100. 0%) 2001 Total Lung Cancers NLST 892 NO screen cancers include: never screened (N = 35) | due for screen (N = 55) post-screen time period (N = 802)
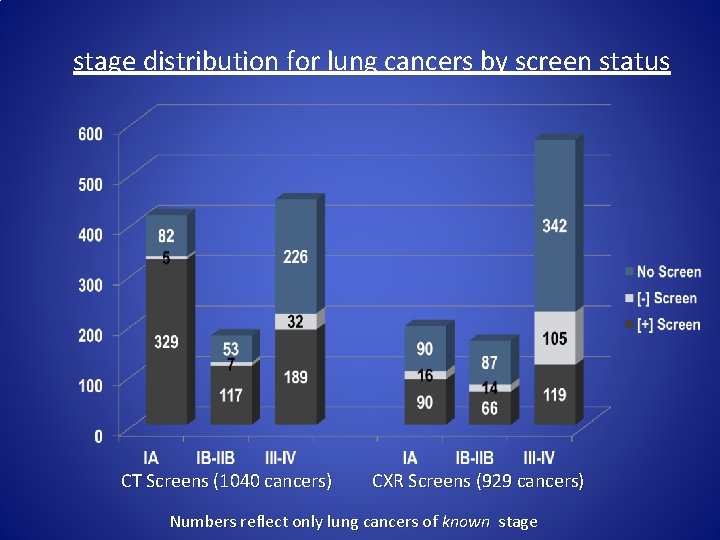
stage distribution for lung cancers by screen status CT Screens (1040 cancers) CXR Screens (929 cancers) Numbers reflect only lung cancers of known stage
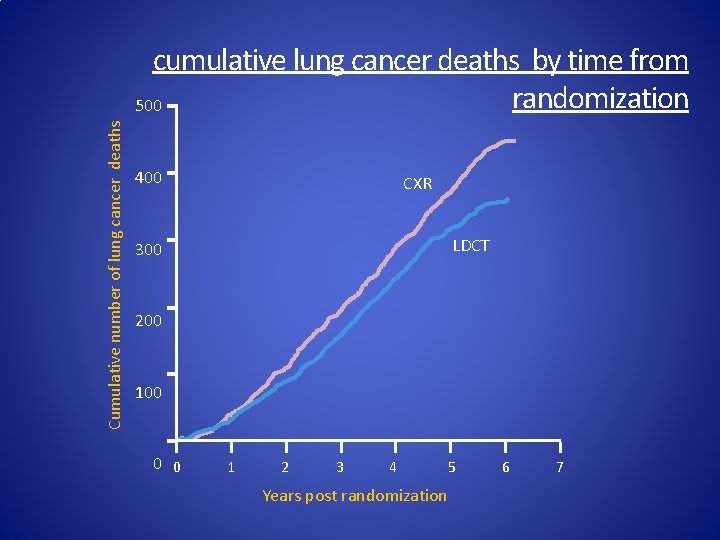
Cumulative number of lung cancer deaths cumulative lung cancer deaths by time from randomization 500 400 CXR LDCT 300 200 100 0 0 1 2 3 4 Years post randomization 5 6 7
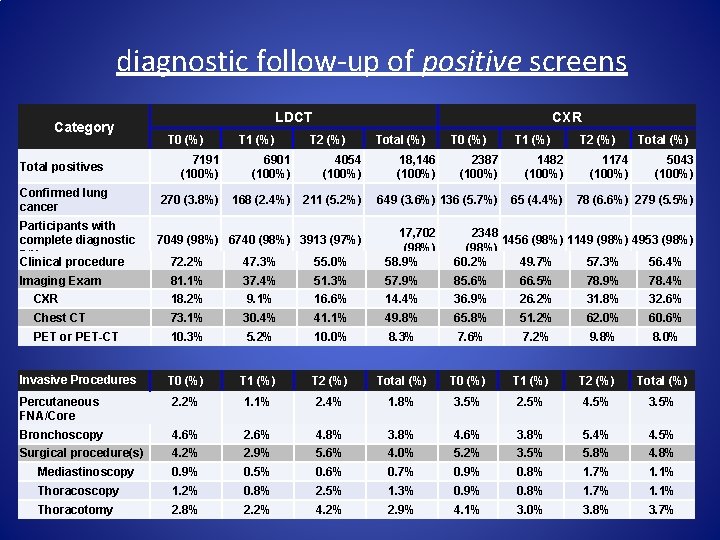
diagnostic follow-up of positive screens Category LDCT T 0 (%) T 1 (%) CXR T 2 (%) 7191 (100%) 6901 (100%) 4054 (100%) Confirmed lung cancer 270 (3. 8%) 168 (2. 4%) 211 (5. 2%) Participants with complete diagnostic F/U Clinical procedure 7049 (98%) 6740 (98%) 3913 (97%) Total positives Total (%) 18, 146 (100%) T 0 (%) T 1 (%) 2387 (100%) 1482 (100%) 649 (3. 6%) 136 (5. 7%) 65 (4. 4%) T 2 (%) 1174 (100%) Total (%) 5043 (100%) 78 (6. 6%) 279 (5. 5%) 72. 2% 47. 3% 55. 0% 17, 702 (98%) 58. 9% 81. 1% 37. 4% 51. 3% 57. 9% 85. 6% 66. 5% 78. 9% 78. 4% CXR 18. 2% 9. 1% 16. 6% 14. 4% 36. 9% 26. 2% 31. 8% 32. 6% Chest CT 73. 1% 30. 4% 41. 1% 49. 8% 65. 8% 51. 2% 62. 0% 60. 6% PET or PET-CT 10. 3% 5. 2% 10. 0% 8. 3% 7. 6% 7. 2% 9. 8% 8. 0% Invasive Procedures T 0 (%) T 1 (%) T 2 (%) Total (%) Percutaneous FNA/Core 2. 2% 1. 1% 2. 4% 1. 8% 3. 5% 2. 5% 4. 5% 3. 5% Bronchoscopy 4. 6% 2. 6% 4. 8% 3. 8% 4. 6% 3. 8% 5. 4% 4. 5% Surgical procedure(s) 4. 2% 2. 9% 5. 6% 4. 0% 5. 2% 3. 5% 5. 8% 4. 8% Mediastinoscopy 0. 9% 0. 5% 0. 6% 0. 7% 0. 9% 0. 8% 1. 7% 1. 1% Thoracoscopy 1. 2% 0. 8% 2. 5% 1. 3% 0. 9% 0. 8% 1. 7% 1. 1% Thoracotomy 2. 8% 2. 2% 4. 2% 2. 9% 4. 1% 3. 0% 3. 8% 3. 7% Imaging Exam 2348 1456 (98%) 1149 (98%) 4953 (98%) 60. 2% 49. 7% 57. 3% 56. 4%
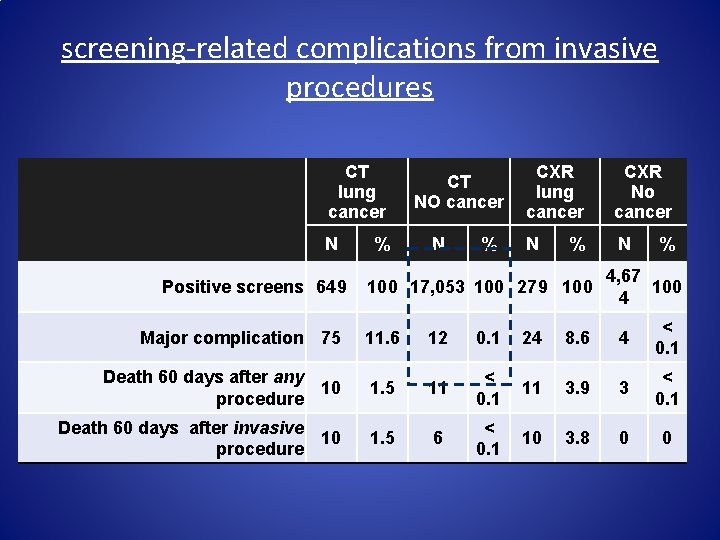
screening-related complications from invasive procedures CT lung cancer N Positive screens 649 % CT NO cancer N % CXR lung cancer CXR No cancer N N % 100 17, 053 100 279 100 % 4, 67 100 4 Major complication 75 11. 6 12 0. 1 24 8. 6 4 < 0. 1 Death 60 days after any 10 procedure 1. 5 11 < 0. 1 11 3. 9 3 < 0. 1 Death 60 days after invasive 10 procedure 1. 5 6 < 0. 1 10 3. 8 0 0

NLST Summary • CT-detects more lung cancers than CXR x 2. 3 folds • True stage shift observed in CT arm • 20% lung cancer mortality reduction CT vs. CXR − Absolute risk reduction = 0. 4% (AR CT= 1. 3% | CXR = 1. 7%) • Few major complications • NNS (Number needed to screen) : 320 − NNS (Breast Cancer): US: 238, NZ: 781 • NCI_2012 and J med Screen, 2001; 8(3): 114 -5 • Need for diagnostic algorithm to decrease false positives

Take Away Points from NSLT • Population was younger than the general population • Population was fairly well educated • Population included more former smokers • Operative mortality was low (1%)
Rational for Lung Cancer Screening

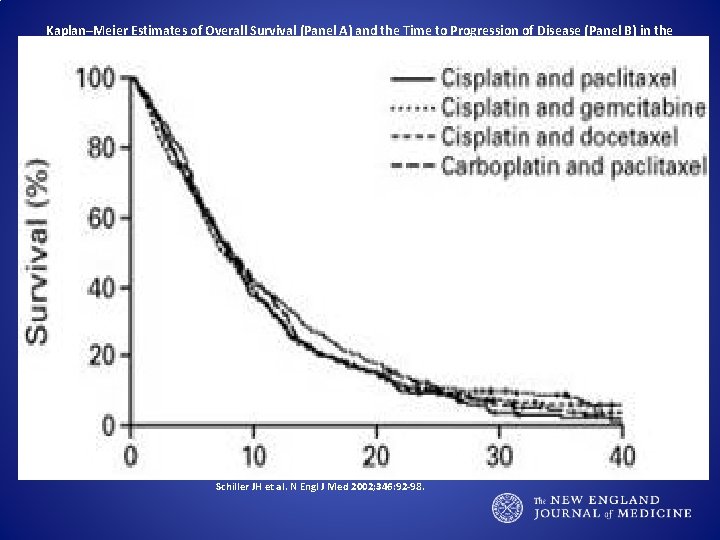
Kaplan–Meier Estimates of Overall Survival (Panel A) and the Time to Progression of Disease (Panel B) in the Study Patients, According to the Assigned Treatment. Schiller JH et al. N Engl J Med 2002; 346: 92 -98.

FY 2010 Federal Research Dollars Per Death Federal spending from the combined FY 2010 research dollars of the National Cancer Institute, Department of Defense and Centers for Disease Control and Prevention.

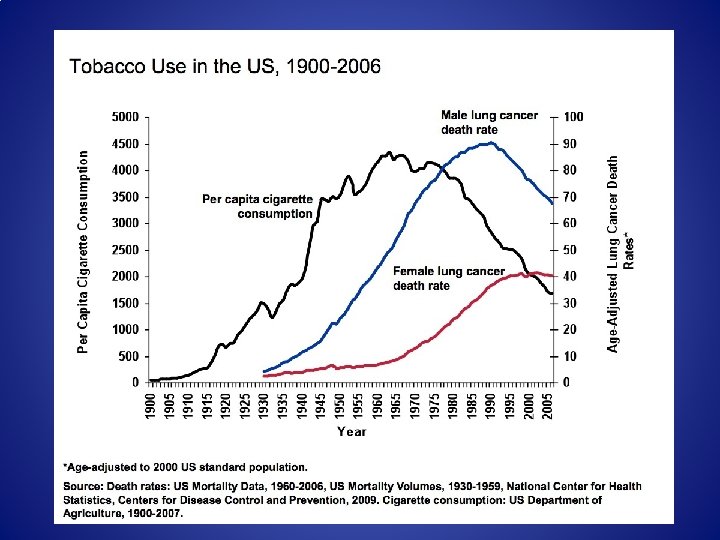
85% of all lung cancers are linked to cigarette use.
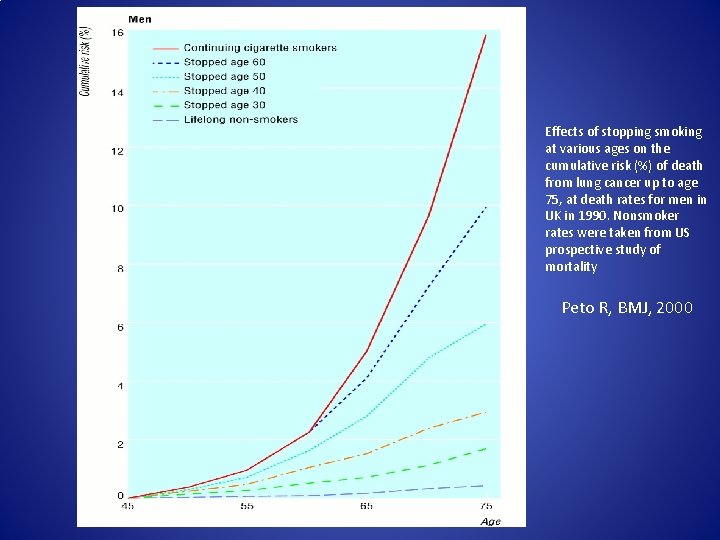
Effects of stopping smoking at various ages on the cumulative risk (%) of death from lung cancer up to age 75, at death rates for men in UK in 1990. Nonsmoker rates were taken from US prospective study of mortality Peto R, BMJ, 2000

Cost of Lung Cancer Care • Annual Cost of Lung Cancer treatment in US – $10 billion • Estimated Annual treatment cost – $21, 000 per patient – $47, 000 per patient for those who live more than one year Heathcare Mang Sci 1999, J Clin Oncol 1997
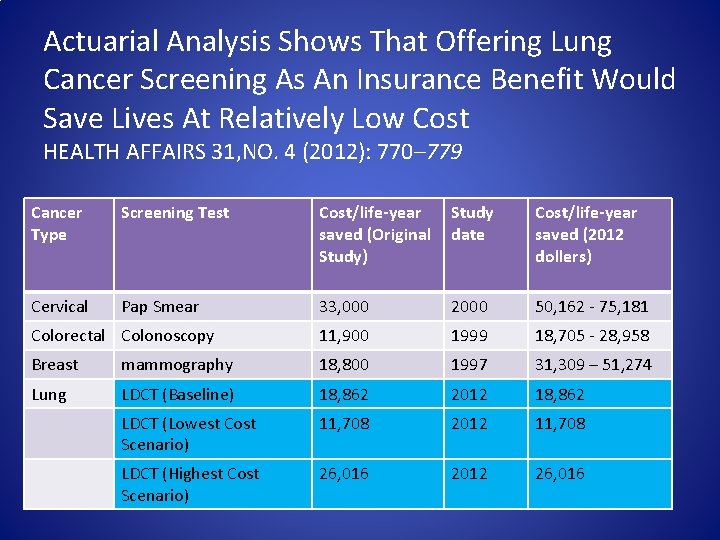
Actuarial Analysis Shows That Offering Lung Cancer Screening As An Insurance Benefit Would Save Lives At Relatively Low Cost HEALTH AFFAIRS 31, NO. 4 (2012): 770– 779 Cancer Type Screening Test Cost/life-year saved (Original Study) Study date Cost/life-year saved (2012 dollers) Cervical Pap Smear 33, 000 2000 50, 162 - 75, 181 Colorectal Colonoscopy 11, 900 1999 18, 705 - 28, 958 Breast mammography 18, 800 1997 31, 309 – 51, 274 Lung LDCT (Baseline) 18, 862 2012 18, 862 LDCT (Lowest Cost Scenario) 11, 708 2012 11, 708 LDCT (Highest Cost Scenario) 26, 016 2012 26, 016

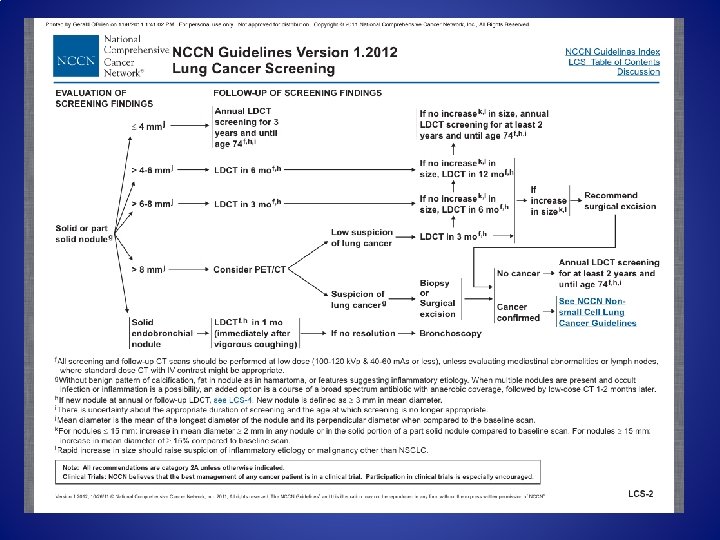
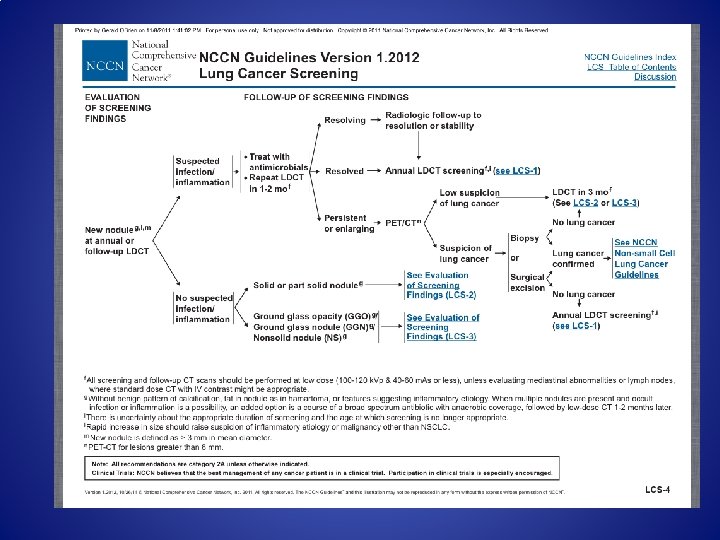
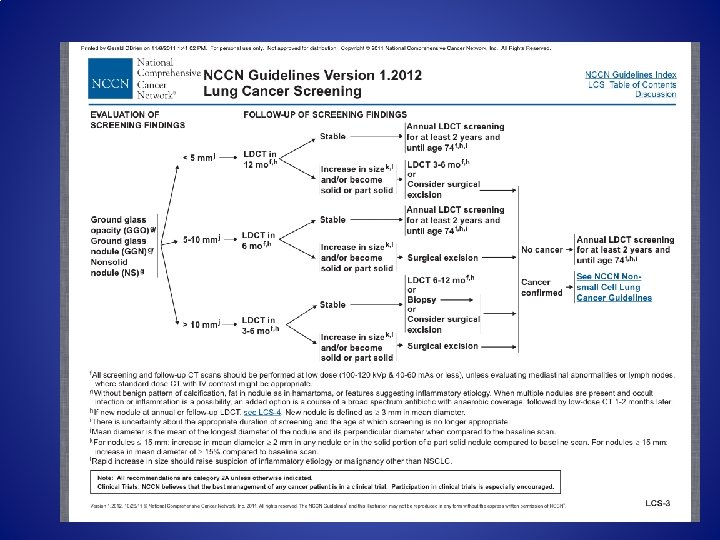
NCCN Guidelines: Lung Cancer Screening October 26, 2011 • • • NCCN. Org • Lung cancer screening with CT should be part of a program of care and should not be performed in isolation as a free standing test. The risks and benefits of lung cancer screening should be discussed with the individual before doing a screening LDCT scan. It is recommended that institutions performing lung cancer screening use a multidisciplinary approach that may include specialties such as radiology, pulmonary medicine, thoracic oncology, and thoracic surgery. Management of downstream testing and follow-up of small nodules are imperative and may require establishment of administrative processes to ensure the adequacy of follow-up.
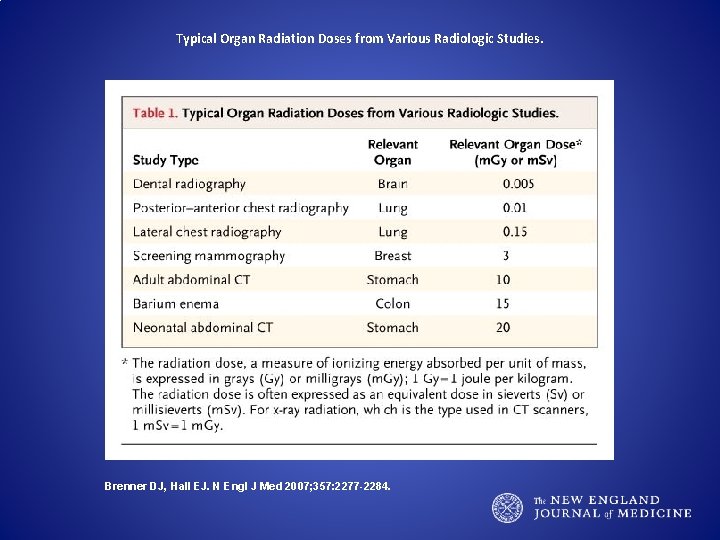
Typical Organ Radiation Doses from Various Radiologic Studies. Brenner DJ, Hall EJ. N Engl J Med 2007; 357: 2277 -2284.

Ionizing Radiation Linear No Threshold Model Roentgens (R) vs Grays (Gy) vs Sieverts (Sv) 3 m. Sv – annual background dose 1000 m. Sv – 4 -5% risk of fatal cancer – Myeloma, leukemia, lung, thyroid, breast • CXR 0. 1 m. Sv, CT chest 8 m. Sv, LDCT chest 1. 5 m. Sv • •

Institute of Medicine • Use of CT scanning increased three fold since 1993 • Top environmental causes of breast cancer – CT scanning – Postmenopausal Hormone Therapy • 30% of all CT scans “medically unnecessary” • Justification, Optimization, Limitation
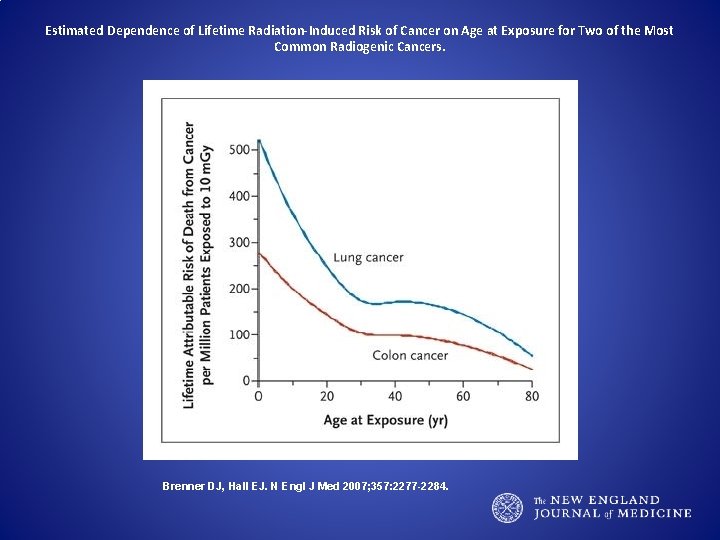
Estimated Dependence of Lifetime Radiation-Induced Risk of Cancer on Age at Exposure for Two of the Most Common Radiogenic Cancers. Brenner DJ, Hall EJ. N Engl J Med 2007; 357: 2277 -2284.

NLST complications (partial list) § Major complications – – – Acute respiratory failure Respiratory arrest Prolonged mechanical ventilation Cardiac arrest | MI | CHF Hemothorax requiring tube Empyema Bronchopulmonary fistula Bronchial stump leak Injury to vital organ | vessel Thromboembolic complications Death • Intermediate complications − Respiratory distress − Mucostasis bronchoscopy − Cardiac arrhythmia needing attention − Cardiac ischemia | ST elevation − Pleural effusion − Pneumothorax needing chest tube − Fever | infection | sepsis | pneumonia − Transfusion for blood loss − Steroid-induced diabetes − Hospitalization post procedure − Vocal cord paralysis − Pain requiring referral to specialist

Lung Cancer Screening: American Lung Association Recommendations Low-dose CT screening should be recommended for those people who meet National Lung Cancer Screening Trail criteria: • current or former smokers, aged 55 to 74 years a smoking history of at least one pack a day for 30 years (30 pack-years ) • no history of lung cancer • Individuals should not receive a chest X-ray for lung cancer screening • Low-dose CT screening should NOT be recommended for everyone

Should my patient be screened? v Prior to appointment: Questions to think about – Does my patient meet the NLST criteria or another high-risk profile that makes them good candidates for screening? – What are the benefits/risks for my patient if screened? – What are my referral options? Where do I refer? v During the average medical visit: ~12 minutes – – – Take a complete health history and determine possible co-morbidities (conduct spirometry? ) Advocate smoking cessation Educate about symptoms of underlying lung disease Discuss the benefits /risks and possible procedures associated with the screening process Discuss costs of screening – health insurance reimbursement, time and personal costs If recommending screening, discuss • Availability – low dose CT is available and will be done, high quality machine and staff • Low Dose CT /qualified center with expertise to follow up after test • Follow up care through multidisciplinary approach. v Points to Remember: – – – Do not offer a Chest X-ray as an option for screening Refer patients only to qualified centers, those providing low-dose CT scans and a multi-disciplinary team for follow-up Ensure that patients know the difference between a screening test and screening process.

Screening Referrals Refer your patient to institutions that have experience in conducting Low Dose CT scans, as well as, using the latest CT technology. • Make sure that the facility uses “best practices” for lung cancer screening • There should be a link to an expert multidisciplinary team that can provide followup for evaluation of nodules. • Discussion of results and follow-up is key.

Making an Individual Decision to Get Screened: Questions to Ask Your Doctor • Understanding – Are you considered high risk for lung cancer? • NLST high risk group • Genetics • Smoking History – pack years • Other exposures – occupational, etc. – What is your current health status? Do you have co-morbidities like COPD? – What are my screening options? – What does the screening process entail? • Risks – What are some of the complications of the diagnosis procedure? • False Positives • Negative results do not absolutely rule out the chance for cancer incidence – What is the cumulative exposure to radiation? – What are the unknowns?

Making an Individual Decision to Get Screened: Questions to Ask Your Doctor, con’t v Costs – Does my health insurance company reimburse for CT scans? – How much does a CT scan cost? What about the cost of follow up procedures? – How will screening affect my quality of life? (living with indeterminate diagnosis) – How much time and personal costs will I spend throughout the screening
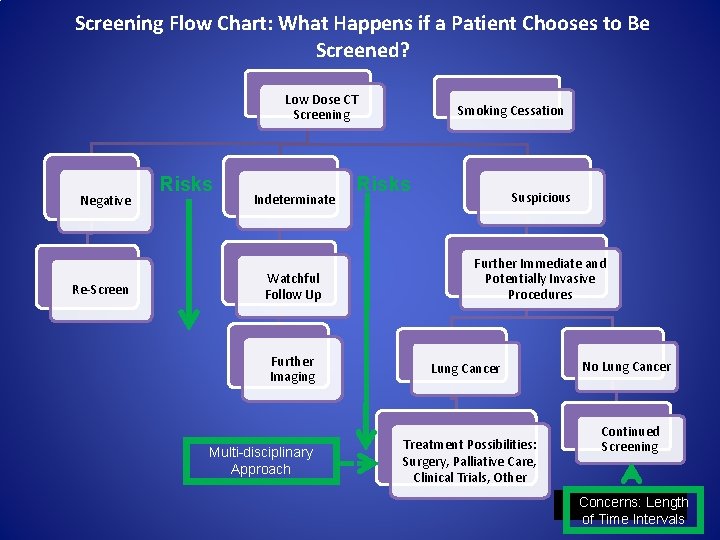
Screening Flow Chart: What Happens if a Patient Chooses to Be Screened? Low Dose CT Screening Negative Re-Screen Risks Indeterminate Watchful Follow Up Further Imaging Multi-disciplinary Approach Smoking Cessation Risks Suspicious Further Immediate and Potentially Invasive Procedures Lung Cancer Treatment Possibilities: Surgery, Palliative Care, Clinical Trials, Other No Lung Cancer Continued Screening Concerns: Length of Time Intervals

Some Remaining Issues • • • Cost effectiveness Screening frequency Population targets Criteria for “positive” findings Follow up protocols to decrease false-positive evaluations

THANK YOU QUESTIONS?
- Slides: 66