LPHAGLUCOSIDASE LPHA AMYLASE INHIBITORY ACTIVITY SAURABH MARU ASSISTANT

ΑLPHA-GLUCOSIDASE & ΑLPHA –AMYLASE INHIBITORY ACTIVITY SAURABH MARU ASSISTANT PROFESSOR (PHARMACOLOGY) SCHOOL OF PHARMACY & TECHNOLOGY MANAGEMENT, SVKM’S NMIMS, SHIRPUR

INTRODUCTION • Mainly two carbohydrate hydrolyzing enzymes (α-amylase and αglucosidase) are responsible for postprandial hyperglycemia. • α-amylase begins the process of carbohydrate digestion by hydrolysis of 1, 4 -glycosidic linkages of polysaccharides (starch, glycogen) to disaccharides • α-glucosidase catalyzes the disaccharides to monosaccharides, which leads to postprandial hyperglycemia. • Hence, inhibitors of α-amylase and α-glucosidase – Control of hyperglycemia by delaying carbohydrate digestion – Consequently reduce the postprandial plasma glucose level.
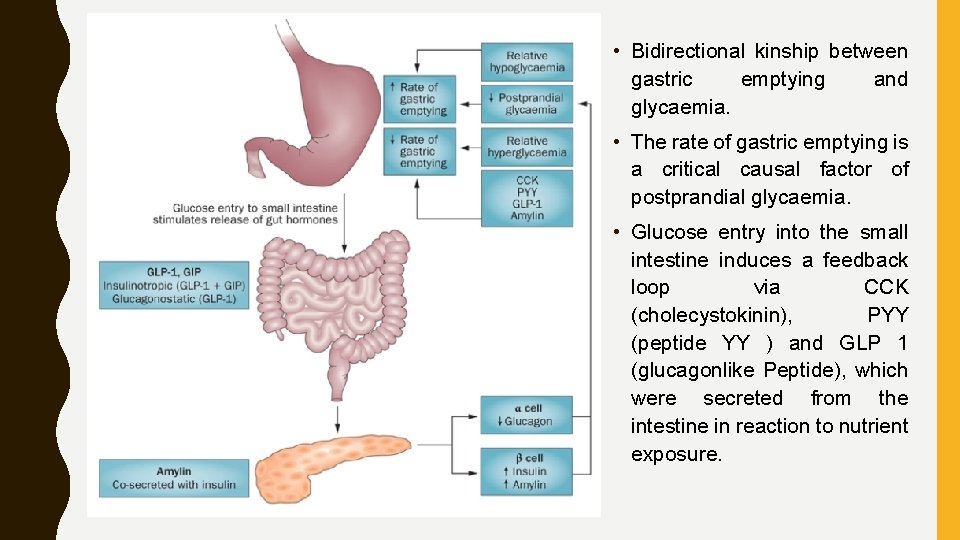
• Bidirectional kinship between gastric emptying and glycaemia. • The rate of gastric emptying is a critical causal factor of postprandial glycaemia. • Glucose entry into the small intestine induces a feedback loop via CCK (cholecystokinin), PYY (peptide YY ) and GLP 1 (glucagonlike Peptide), which were secreted from the intestine in reaction to nutrient exposure.
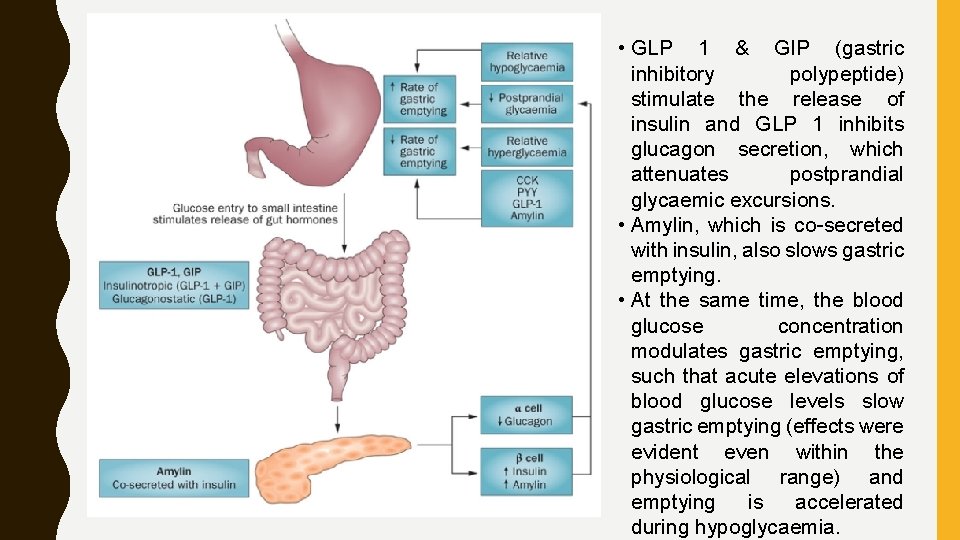
• GLP 1 & GIP (gastric inhibitory polypeptide) stimulate the release of insulin and GLP 1 inhibits glucagon secretion, which attenuates postprandial glycaemic excursions. • Amylin, which is co-secreted with insulin, also slows gastric emptying. • At the same time, the blood glucose concentration modulates gastric emptying, such that acute elevations of blood glucose levels slow gastric emptying (effects were evident even within the physiological range) and emptying is accelerated during hypoglycaemia.

CONTROL OF POSTPRANDIAL HYPERGLYCEMIA • The pancreas secretes a measured quantity of insulin at dissimilar intervals in body. • Increase in insulin release from the pancreas as blood glucose levels were elevated follow Masshe meals; however, in type 2 diabetics this functioning was deficient. • This dysfunction leads to cause hyperglycemia, which was an important and early defect in type 2 diabetics and was due to poor suppression of endogenous glucose formation due to no early-phase insulin response • The pathophysiology of postprandial hyperglycemia is represented in figure. • The determinants of postprandial blood glucose levels were the presence of insulin and the entry of glucose from the gut. • However, disposing glucose in peripheral tissues as well as decreased down regulation of hepatic glucose output were the significant contributors

• Importantly, the mealtime hyperglycemia might be a more accurate predictor for glycated haemoglobin A 1 c (Hb. A 1 c) level analysis and of cardiovascular mortality than fasting hyperglycemia. • Postprandial hyperglycemia has been connected to cardiovascular complications, even though Hb. A 1 c values were within non-diabetic range, whereas increased fasting plasma glucose levels were not independently linked to increased cardiovascular disease risk • To overcome microvascular and macrovascular complications good glycemic control was one of the cornerstones • This finding concentrated for more attention on control of postprandial hyperglycemia and should think of having an oral antidiabetic drug to control postprandial hyperglycemia so as to achieve an early phase insulin release without causing late hyperinsulinemia or an increased risk of hypoglycaemia

• However, available oral antidiabetic drugs have limitations of either being contraindicated or with dose reduction in patients with compromised renal function especially diabetes associated with nephropathy. • αG inhibitors were reported to contribute in reducing Hb. A 1 c in individuals by targeting postprandial hyperglycemia and without dose alteration for those with renal impairment • To overcome it, in type 2 diabetics, αG inhibitor drugs like acarbose & miglitol could block action of enzyme αG in small intestine that broke down carbohydrate into glucose. • Therefore, glucose entered bloodstream more slowly, giving the pancreas additional time to secrete enough insulin to handle it. • Moreover, αG inhibitors were reported to be useful in elderly patients and in those with mild to moderate renal function impairments when other anti-diabetic agents were contraindicated.

• It potentially provided anotherapeutic option for patients with T 2 DM in which glycemic control is inadequate, despite diet alone or with pharmacological therapy with sulfonylurea and biguanide. • αG inhibitors reported to improve postprandial hyperglycemia and fasting blood sugar in type 2 diabetics who were uncontrolled, despite on diet control and taking other oral antidiabetic agents
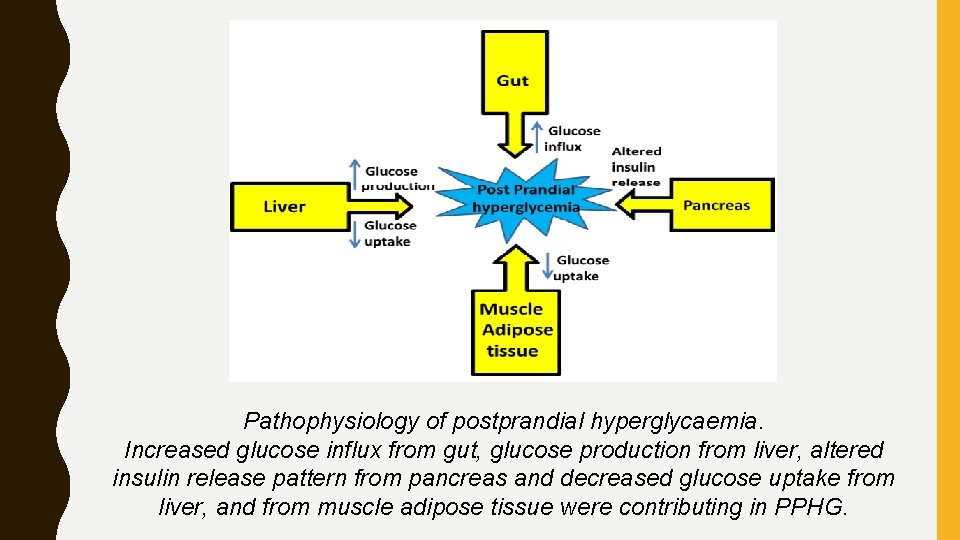
Pathophysiology of postprandial hyperglycaemia. Increased glucose influx from gut, glucose production from liver, altered insulin release pattern from pancreas and decreased glucose uptake from liver, and from muscle adipose tissue were contributing in PPHG.

INTESTINAL ABSORPTION AND DIGESTION OF CARBOHYDRATES • Carbohydrates were the most abundant biological molecules in nature. • They delivered a broad range of uses, including supplying a substantial fraction of the dietary calories for most beings, representing as a storage form of energy in the liver, muscle and dishing out as cell membrane components that mediated some forms of intercellular communication. • Carbohydrates could be classified as monosaccharides, disaccharides, oligosaccharides and polysaccharides based on the number of carbon atoms they contain or monosaccharide units attached to their structure

DIGESTION OF DIETARY CARBOHYDRATES • The main sites of dietary carbohydrate digestion were the mouth and intestinal lumen. • This phenomenon of digestion was catalysed by enzymes known as glycoside hydrolases (glycosidases) that hydrolyze glycosidic bonds. • Glycosidases enzyme that catalyzes the hydrolysis of a bond joining a sugar of a glycoside to an alcohol or another sugar unit. • The terminal products of carbohydrate digestion – Monosaccharides, glucose, galactose and fructose • Primarily absorbed by segment of small intestine • The major dietary polysaccharides were of – Plant (starch, composed of amylose and amylopectin) and – Animal (glycogen) origin.

• During chewing, ptyalin or salivary αA (SAA) acts briefly on dietary starch and glycogen, hydrolyzing random α (1 -4) bonds. • Carbohydrate digestion resulting from enzymatic action comprised a mixture of short, and unbranched oligosaccharides known as dextrins, • Since split amylopectin and glycogen also contain α-(1 -6) bonds, which αA cannot hydrolyze, Carbohydrate digestion standstills provisionally in stomach, because strong acidity inactivates SAA enzyme activity. • Acidic stomach chime reaches small intestine and get neutralized by bicarbonate secreted by the pancreas, and pancreatic αA continues the process of starch digestion (digestion initiated in saliva). • Ultimate digestive processes occurred mainly in the mucosal lining of proximal jejunum, where sucrase and isomaltase were brush border enzyme and are activities of a single protein which was cleaved into binary functional subunits, and forming (called as) sucrase-isomaltase complex.

• Maltase formed a comparable complex with an exoglucosidase (glucoamylase) that cleaves α (1 -4) glycosidic bonds in dextrins. • These enzymes were secreted through, and remain associated with, the luminal side of the brush border membranes of the intestinal mucosal cells. • For instance, isomaltase cut α (1 -6) bond in isomaltose and maltase cleaves maltose and maltotriose, each producing glucose, • Sucrase cleaves sucrose constructing glucose and fructose, and • Lactase (β-galactosidase) cleaves lactose producing galactose and glucose
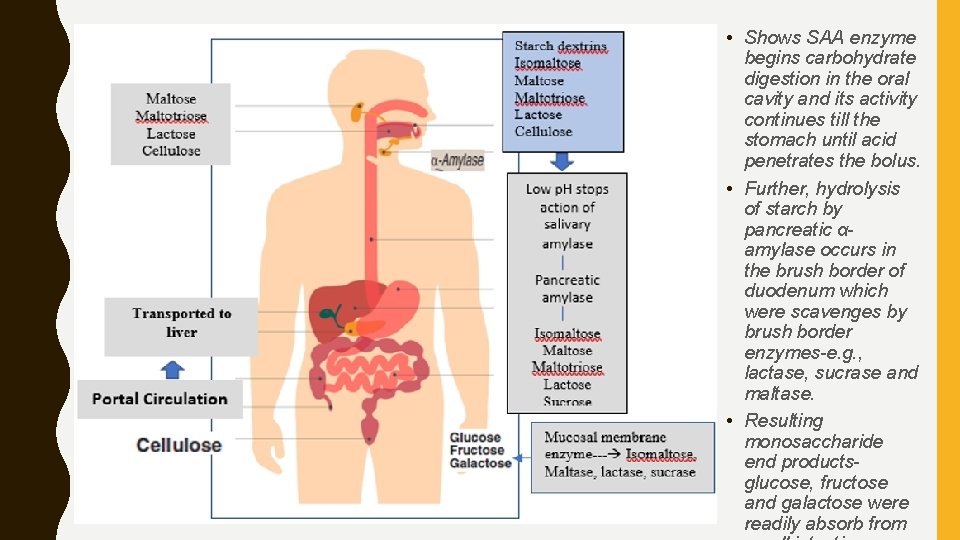
• Shows SAA enzyme begins carbohydrate digestion in the oral cavity and its activity continues till the stomach until acid penetrates the bolus. • Further, hydrolysis of starch by pancreatic αamylase occurs in the brush border of duodenum which were scavenges by brush border enzymes-e. g. , lactase, sucrase and maltase. • Resulting monosaccharide end productsglucose, fructose and galactose were readily absorb from

IN-VITRO ASSAY α-amylase inhibitory activity • In a 96 -well plate, reaction mixture containing 50 μl phosphate buffer (100 m. M, p. H = 6. 8), 10 μl α–amylase (2 U/ml), and 20 μl of varying concentrations of extract and fractions (0. 1, 0. 2, 0. 3, 0. 4, and 0. 5 mg/ml) was preincubated at 37°C for 20 min. • Then, the 20 μl of 1% soluble starch (100 m. M phosphate buffer p. H 6. 8) was added as a substrate and incubated further at 37°C for 30 min; 100 μl of the DNS color reagent was then added and boiled for 10 min. • The absorbance of the resulting mixture was measured at 540 nm using Multiplate Reader.

• Acarbose at various concentrations (0. 1– 0. 5 mg/ml) was used as a standard. • Without test (extract and fractions) substance was set up in parallel as control and each experiment was performed in triplicates. • The results were expressed as percentage inhibition, which was calculated using the formula, • Inhibitory activity (%) = (1 − As/Ac) × 100 • Where, – As is the absorbance in the presence of test substance and – Ac is the absorbance of control.

α-glucosidase inhibitory activity • In a 96 -well plate, reaction mixture containing 50 μl phosphate buffer (100 m. M, p. H = 6. 8), 10 μl alpha-glucosidase (1 U/ml), and 20 μl of varying concentrations of extract and fractions (0. 1, 0. 2, 0. 3, 0. 4, and 0. 5 mg/ml) was preincubated at 37°C for 15 min. • Then, 20 μl P-NPG (5 m. M, p-nitrophenyl-alpha-Dglucopyranoside) was added as a substrate and incubated further at 37°C for 20 min. • The reaction was stopped by adding 50 μl Na 2 CO 3 (0. 1 M). • The absorbance of the released p-nitrophenol was measured at 405 nm using Multiplate Reader.

• Acarbose at various concentrations (0. 1– 0. 5 mg/ml) was included as a standard. • Without test substance was set up in parallel as a control and each experiment was performed in triplicates. • The results were expressed as percentage inhibition, which was calculated using the formula, • Inhibitory activity (%) = (1 − As/Ac) × 100 • Where, – As is the absorbance in the presence of test substance and – Ac is the absorbance of control.

Extraction and Fractionation • Dried powdered (500 g) material was first subjected to cold maceration to extract thermolabile constituents if any with 70% v/v ethanol for 24 h. • Extract was filtered, and the marc was further subjected for soxhlation (95% v/v ethanol). Filtrates of both maceration and soxhlation were combined and concentrated using a rotary evaporator (IKA RV 10) at 40°C under reduced pressure, which yields total extract of 40 g and 46 g. • Fractionation of A. caudatum extract was carried out as per Cos et al. , with minor modifications [Figure 1]. • Alcoholic extract was dispersed in 5% w/v citric acid and washed with dichloromethane. Dichloromethane layer was separated and it was concentrated to 1/3 rd volume using rotary evaporator at 40°C under reduced pressure.

• Concentrated dichloromethane layer was partitioned with 90% v/v methanol and petroleum ether (1: 1) to get fraction 1, (F 1, 9. 65 g) and fraction 2, (F 2, 7. 15 g). • Aqueous layer was concentrated to half and p. H adjusted to 9. 0 with 10% ammonium hydroxide. Aqueous layer washed with dichloromethane, which gives fraction 3 and fraction 4 (F 3, 0. 584, and F 4, 16. 45 g). • Same procedure was used for fractionation of C. argentea extract and percentage yield of the fractions were F 1 10. 15 g, F 2 9. 76 g, F 3 0. 593 g, and F 4 18. 65 g, respectively.


RESULTS • In the study, hydro-alcoholic extract and four fractions of A. caudatum and C. argentea were evaluated for their inhibitory effect on αamylase and α-glucosidase enzymes by in-vitro method. • The hydro-alcoholic extract and its fractions 1, 2, 3, and 4 of A. caudatum (at a concentration of 0. 5 mg/ml) exhibited 32. 42, 46. 25, 61. 45, 20. 12, and 26. 04 α-amylase inhibitory activity [Figure 2] and 36. 42, 47. 25, 63. 45, 29. 22, and 32. 34 α-glucosidase inhibitory activity [Figure 3], respectively. • Whereas, C. argentea hydro-alcoholic extract and Fractions 1, 2, 3, and 4 exhibited 22. 42, 21. 04, 29. 04, 30. 12, and 59. 45 α-amylase inhibitory activity [Figure 4], 37. 62, 31. 04, 37. 24, 29. 34, and 61. 45 α-glucosidase inhibitory activity, respectively [Figure 5].

• Acarbose was used as a standard reference drug, which showed αamylase inhibitory activity with an IC 50 value of 0. 108 mg/ml and αglucosidase inhibitory activity with an IC 50 value of 0. 083 mg/ml. • Among all, fraction 2 of A. caudatum and fraction 4 of C. argentea has shown best enzyme inhibitory activity with an IC 50 value 0. 241 and 0. 211 (α-amylase and α-glucosidase) [Table 1] and 0. 294 and 0. 249 mg/ml (αamylase and α-glucosidase) [Table 2] which were comparable with that of acarbose.


DISCUSSION • Previously, the antidiabetic activity of A. caudatum and C. argentea has been reported in the literature. • The ethanolic extract of A. caudatum whole plant shown antidiabetic activity with 200 mg/kg b. w in streptozotocin-induced diabetic rats. • Ethanolic extract of C. argentea roots and seeds with 250 and 500 mg/kg b. w reduced hyperglycemia in streptozotocin and alloxan-induced diabetic rats. • There was no information available in the literature about the in-vitro (αamylase and α-glucosidase inhibitory activity) antidiabetic studies of these two plants. • Hence, the present study aimed to evaluate α-amylase and α-glucosidase inhibitory activity of hydro-alcoholic extract and its fractions of A. caudatum and C. argentea.

• After fractionation of the hydro-alcoholic extract, steroids and lipids were found in fraction 1, phenolics and terpenoids were found in fraction 2. Fraction 3 was shown positive results for alkaloids and fraction 4 shown positive results for flavonoids. • Based on the results obtained, fraction 2 was showed highest inhibitory potential than extract and other fractions. • Whereas, phytochemical investigation of C. argentea showed positive results for alkaloids, phenolics, triterpenoids, flavonoids, steroids, saponins, and tannins. • After fractionation of the hydro-alcoholic extract, steroids, and lipids were found in fraction 1, phenolics, and terpenoids were found in fraction 2. • Fraction 3 was shown positive results for alkaloids and fraction 4 shown appreciable levels of flavonoids.

• Many bioactive compounds from different plants have been reported to have hypoglycemic effect, in that mostly phenolics and triterpenoids such as oleanane, ursane, lupane, and flavonoids have a positive correlation as antidiabetic agents. • The presence of triterpenoids and phenolics in fraction 2 might have attributed to the highest enzyme inhibition activity compared to other fractions in A. caudatum. • Hence, the triterpenoids of this plant may be responsible for enzyme inhibitory activity.

• Apart from that polyphenolic compounds were found in fraction 2, may interact or inhibit specific positions in enzymes thereby reducing the potency of α-amylase and α-glucosidase. • The presence of flavonoid compounds in fraction 4 of C. argentea may act against diabetes mellitus either through their capacity to avoid glucose absorption or to improve glucose tolerance by competitive inhibition of sodium-dependent glucose transporter-1. • Another possible mechanism followed by flavonoid compounds (luteolin, kaempferol, chrysin, and galangin) to control blood glucose levels is the inhibition of α-amylase and α-glucosidase activity in the intestine. • Due to above reasons, fraction 2 of A. caudatum and fraction 4 of C. argentea showed comparable results with that of acarbose.

• With the help of results in correlation with previous reports it can be hypothesized that the significant enzyme inhibitory activity of fraction 2 and fraction 4 may interfere or delay the absorption of dietary carbohydrates as well as disaccharides in the small intestine, leading to the suppression of meal-induced increase of plasma glucose. • Hence, it may useful in the management of T 2 D. Based on the lead fractions obtained from in-vitro studies, we are going to plan an invivo study for further confirmation of the obtained results.

Limitations of the Study • Active compounds isolation from the fractions and its structural elucidation by nuclear magnetic resonance spectroscopy may helpful to develop newer antidiabetic agents. • Here our target was an in-vitro evaluation of α-amylase and αglucosidase inhibitory activity of the hydro-alcoholic extract, and fractions of A. caudatum and C. argentea.

Conclusion • The results of the present study prove that the fraction 2 of the A. caudatum and fraction 4 of the C. argentea are effective α-amylase and α-glucosidase inhibitors, which may helpful to reduce the postprandial glucose levels. • However, the principle compounds responsible for the inhibitory action of α-amylase and α-glucosidase need to be further identified and characterized. • This may be useful for the development of new antidiabetic agents from native plant resources.

THANK YOU
- Slides: 32