Low Temperature Sterilization Technology Overview q Brief History

Low Temperature Sterilization Technology

Overview q Brief History of Sterilization q Why Low Temperature Sterilization? q Low Temperature Technology Evolution q Vaporized Hydrogen Peroxide Sterilizers 2 TSO 3 Inc. 2016. All Rights Reserved.

Brief History of Sterilization
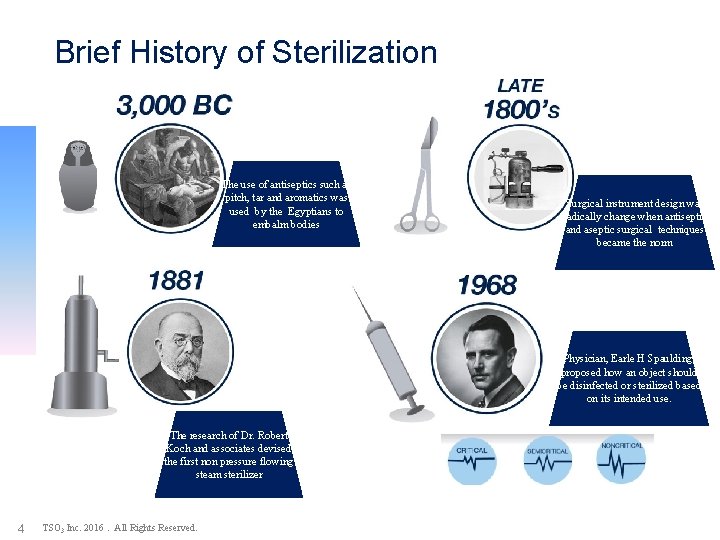
Brief History of Sterilization The use of antiseptics such as pitch, tar and aromatics was used by the Egyptians to embalm bodies Surgical instrument design was radically change when antiseptic and aseptic surgical techniques became the norm Physician, Earle H Spaulding, proposed how an object should be disinfected or sterilized based on its intended use. The research of Dr. Robert Koch and associates devised the first non pressure flowing steam sterilizer 4 TSO 3 Inc. 2016. All Rights Reserved.

Brief History of Sterilization 1994 Dr. William Rutala worked with the CDC to define the Characteristics of an Ideal Sterilization Method 5 TSO 3 Inc. 2016. All Rights Reserved.

Characteristics of an Ideal Sterilization Method 6 • High efficacy - the agent should be virucidal, bactericidal tuberculocidal, fungicidal. and sporicidal. • Rapid activity - ability to achieve sterilization quickly. • Strong penetrability - ability to penetrate common medical device packaging materials and penetrate into the interior of device lumens. • Material compatibility - produce negligible changes in either the appearance or function of processed items and packaging materials, even after repeated cycling. • Nontoxic - present no health risk to the operator or to the patient and pose no hazard to the environment. • Organic material resistance - withstand reasonable organic material challenge without loss of efficacy. • Adaptability - suitable for large or small (point of use) installations. • Monitoring capability - monitored easily and accurately with physical. chemical. and biological process monitors. • Cost-effectiveness - reasonable cost for installation and for routine operation. TSO 3 Inc. 2016. All Rights Reserved.

Sterilization Methods • Steam • Ethylene Oxide "Gas" Sterilization • Hydrogen Peroxide Gas Plasma • Peracetic Acid Sterilization • Vaporized Hydrogen Peroxide (VHP) • Ozone • VHP/Ozone • Flash Sterilization • Ionizing Radiation • Dry-Heat Sterilizers • Formaldehyde Steam • Infrared Radiation • Vaporized Peracetic Acid 7 TSO 3 Inc. 2016. All Rights Reserved.

Why Low Temperature Sterilization?

Why low temperature sterilization? “Many components of today’s advanced surgical tools cannot tolerate the high heat of steam sterilizers. Demand for lowtemperature alternatives has driven manufacturers to create safer, faster low-temperature sterilizers. Markets and Markets predicts that the technology will become an essential element of ORs and central sterile processing departments in the next few years. ” OR Today 2012 9 TSO 3 Inc. 2016. All Rights Reserved.

Why Low Temperature Sterilization? Allows creativity and innovation in device design which allow surgeons to reach places, and create opportunities for less invasive surgical techniques. . . By the use of plastics, adhesives, electronic technology and other materials/technologies that are either destroyed or suffer reduced life in steam sterilization technologies. 10 TSO 3 Inc. 2016. All Rights Reserved.
Market Growth Drivers on e i i t r a e g ul r p n u s io po t l e a l v a i u b s p a lo o v g p n i g g ” n y n s i l g a u ow g ag b r m i r G n e n i i p m u ow s n r “ i f G h o t ce ow r n G le a v e r P S) I M s( 11 TSO 3 Inc. 2016. All Rights Reserved.
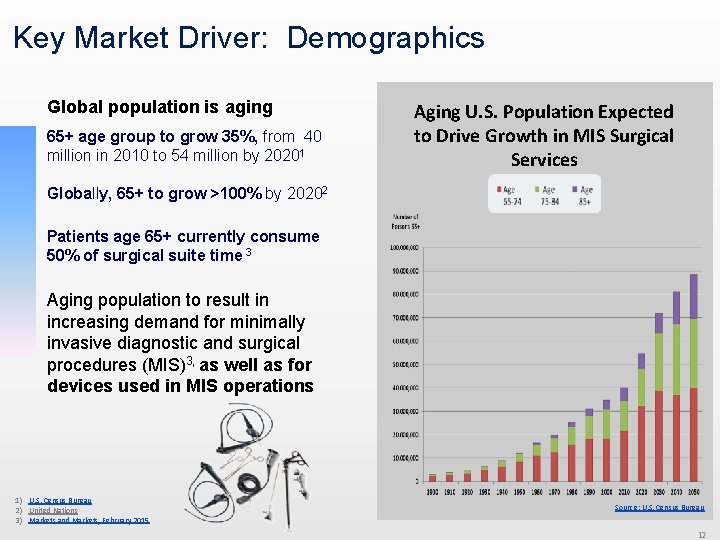
Key Market Driver: Demographics Global population is aging 65+ age group to grow 35%, from 40 million in 2010 to 54 million by 20201 Aging U. S. Population Expected to Drive Growth in MIS Surgical Services Globally, 65+ to grow >100% by 20202 Patients age 65+ currently consume 50% of surgical suite time 3 Aging population to result in increasing demand for minimally invasive diagnostic and surgical procedures (MIS)3, as well as for devices used in MIS operations 1) U. S. Census Bureau 2) United Nations 3) Markets and Markets, February 2015 Source: U. S. Census Bureau 12
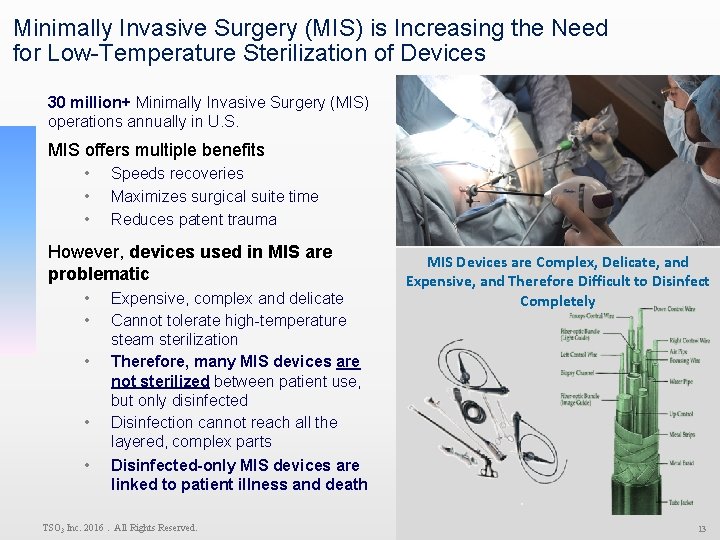
Minimally Invasive Surgery (MIS) is Increasing the Need for Low-Temperature Sterilization of Devices 30 million+ Minimally Invasive Surgery (MIS) operations annually in U. S. MIS offers multiple benefits • • • Speeds recoveries Maximizes surgical suite time Reduces patent trauma However, devices used in MIS are problematic • • • Expensive, complex and delicate Cannot tolerate high-temperature steam sterilization Therefore, many MIS devices are not sterilized between patient use, but only disinfected Disinfection cannot reach all the layered, complex parts MIS Devices are Complex, Delicate, and Expensive, and Therefore Difficult to Disinfect Completely Disinfected-only MIS devices are linked to patient illness and death TSO 3 Inc. 2016. All Rights Reserved. 13

The Problem with Disinfection-only: Superbug & Endoscope Connection SUPERBUG = Antimicrobial Resistant Bacteria • At least 2 million Americans suffer from antibiotic-resistant bacteria annually – 23, 000 die 1 • Half of all bugs that cause infections after surgery are antibiotic resistant 2 Superbug, CRE, has become resistant to most available antibiotics, resulting in death in up to 50% of patients who become infected 1 CRE infections increasingly prevalent and linked to use of endoscopes, including seven incidents and two deaths at UCLA Medical Center 3 1) Source: Centers for Disease Control (CD) 2) The Lancet Infectious Diseases journal, as reported by Time, October 2015 3) Associated Press, February 20, 2015 14

Low Temperature Technology Evolution
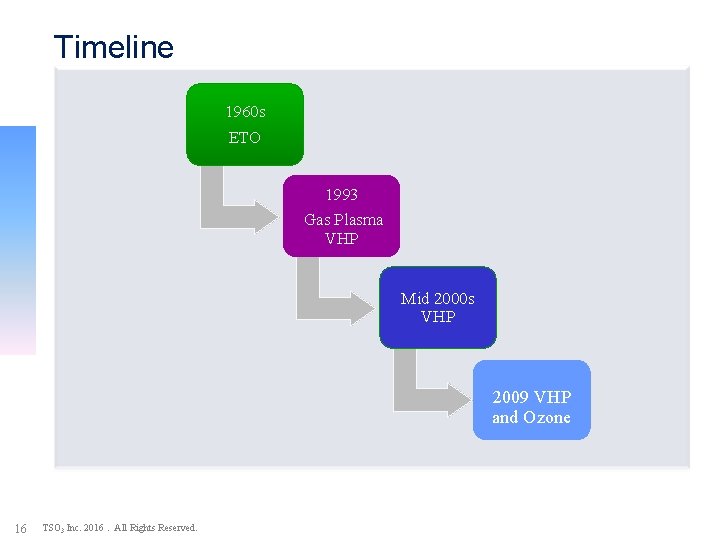
Timeline 1960 s ETO 1993 Gas Plasma VHP Mid 2000 s VHP 2009 VHP and Ozone 16 TSO 3 Inc. 2016. All Rights Reserved.

Approved Low Temperature Sterilization Processes (Canada and US) 17 Low Temperature Process Major Supplier Brand ETO 3 M Steri-Vac™ Gas Plasma VHP Advanced Sterilization Products STERRAD® VHP Steris V-PRO® ma. X VHP and Ozone Getinge Infection Control STERIZONE® TSO 3 Inc. 2016. All Rights Reserved.
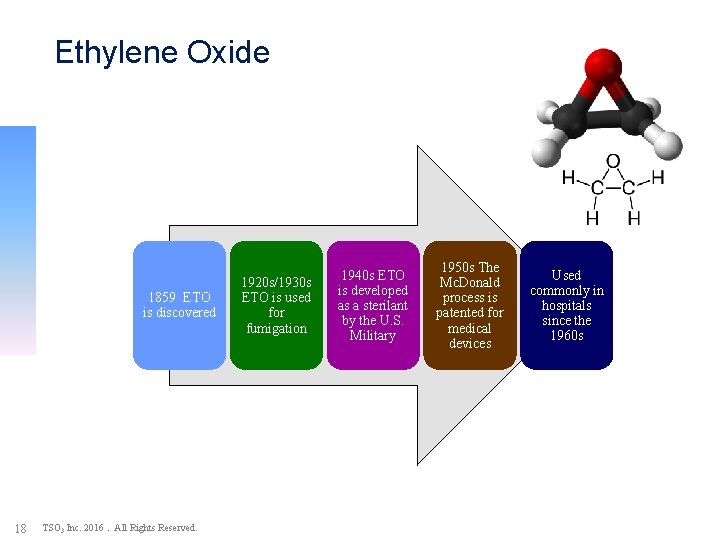
Ethylene Oxide (EO) Sterilization: Timeline of Events 1859 ETO is discovered 18 TSO 3 Inc. 2016. All Rights Reserved. 1920 s/1930 s ETO is used for fumigation 1940 s ETO is developed as a sterilant by the U. S. Military 1950 s The Mc. Donald process is patented for medical devices Used commonly in hospitals since the 1960 s

Ethylene Oxide Pros q Kills just about everything q Excellent Penetration q Works well for lumened devices q Good compatibility with most materials and packaging Cons q Kills just about everything q Toxic, hazardous carcinogenic linked to cancer q Flammable at room temperature q Requires personnel and room monitoring q Takes a really long time due to aeration requirements (12 – 18 hrs) 19 TSO 3 Inc. 2016. All Rights Reserved.

Gas Plasma (Vaporized Hydrogen Peroxide) q Gas plasma sterilization was patented in 1987 and was cleared for market under the STERRAD brand (ASP) in 1993 and is now in more than 60 countries. q Often referred to as gas plasma sterilization but the kill is accomplished by vaporized is H 2 O 2. q An electrical charge is applied to the vapor field to generate low temperature plasma. q Plasma participates in breaking down of the residual hydrogen peroxide. 20 TSO 3 Inc. 2016. All Rights Reserved.

Gas Plasma Sterilization 1. Vacuum 2. Injection 3. Diffusion 4. Plasma 5. Vent 21 TSO 3 Inc. 2016. All Rights Reserved.

Hydrogen Peroxide (plasma, VHP) Pros q Broad material and device compatibility q Fast cycle times q Better inventory management (compared to ETO) q Non-toxic by products q Low temperature, low moisture environment 22 TSO 3 Inc. 2016. All Rights Reserved. Cons q Struggles with long lumens q Inability to process liquids, powders, or strong absorbers (e. g. , cellulose) q Devices/material not compatible with deep vacuum q Limited capacity. Multiple cycles necessary to cover full range of devices

VAPORIZED H 202 Sterilizers

Why is Hydrogen Peroxide Important? Hydrogen Peroxide Addressed Need for Safer, More Cost Effective Sterilization Chemical Used in Medical Device Reprocessing ETO Ethylene Oxide (100%, 90/10, etc. – all forms) • Toxic • Carcinogenic • Slow • Environmental pollutant H 2 O 2 Hydrogen Peroxide (VHP, Plasma, etc. ) • Quick • Environmentally safe • However, typically expensive, with strict loading limitations and monitoring TSO 3 Inc. 2016. All Rights Reserved. Relative Market Share Over Time 24

VHP Sterilizers 1 st Generation STERRAD® 100 S STERRAD ® 100 NX • 2 cycles at 90% (original) & 2 optional cycles at 59% 2 nd Generation Steris (AMSCO) V-PRO ® ma. X • 3 Cycles Latest Technology TSO 3 Inc. 2016. All Rights Reserved. STERIZONE ® VP 4 Sterilizer • 1 Cycle for all devices • 75 lb load capacity 25

STERRAD® 100 NX® • Low temperature sterilization process at 50°C (122° F) • Uses vaporized hydrogen peroxide for the terminal sterilization of packaged, resuseable medical devices • Followed by creation of a low energy plasma field to break apart the peroxide vapor. • Returns to atmospheric pressure by use of HEPA filitered room air. • Releases only water vapor and oxygen (no drain required). No toxic residues. 26

STERIS V-PRO®max • Low temperature sterilization process at < 55°C (131° F) • Uses vaporized hydrogen peroxide for the terminal sterilization of packaged, resuseable medical devices • No secondary means of reducing hydrogen peroxide residual. • Returns to atmospheric pressure by use of HEPA filtered room air. • Releases only water vapor and oxygen (no drain required). No toxic residues. 27

STERIZONE® VP 4 Sterilizer • Low temperature sterilization process at 41ºC ± 3°C • Uses vaporized hydrogen peroxide for the terminal sterilization of packaged, resuseable medical devices • Followed by injection of ozone and dwell, to aid in the breakdown of residual H 2 O 2 on device surfaces. • Ozone contributes additional lethality to the process. • Releases only water vapor and oxygen (no drain required). No toxic residues. Note: Only sterilizer approved to sterilizer multichannel endoscopes up to 4 channels. 28 TSO 3 Inc. 2016. All Rights Reserved.
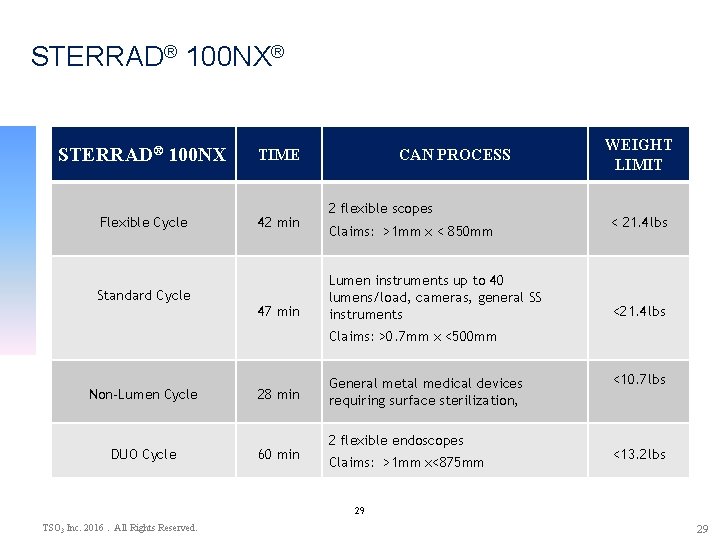
STERRAD® 100 NX® STERRAD® 100 NX TIME Flexible Cycle 42 min Standard Cycle 47 min CAN PROCESS 2 flexible scopes Claims: >1 mm x < 850 mm Lumen instruments up to 40 lumens/load, cameras, general SS instruments WEIGHT LIMIT < 21. 4 lbs <21. 4 lbs Claims: >0. 7 mm x <500 mm Non-Lumen Cycle 28 min DUO Cycle 60 min General metal medical devices requiring surface sterilization, 2 flexible endoscopes Claims: >1 mm x<875 mm <10. 7 lbs <13. 2 lbs 29 TSO 3 Inc. 2016. All Rights Reserved. 29
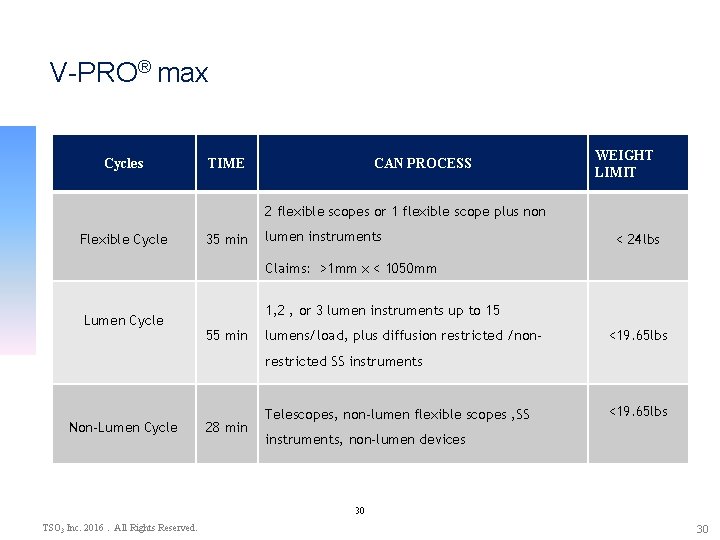
V-PRO® max Cycles TIME CAN PROCESS WEIGHT LIMIT 2 flexible scopes or 1 flexible scope plus non Flexible Cycle 35 min lumen instruments < 24 lbs Claims: >1 mm x < 1050 mm Lumen Cycle 1, 2 , or 3 lumen instruments up to 15 55 min lumens/load, plus diffusion restricted /non- <19. 65 lbs restricted SS instruments Non-Lumen Cycle 28 min Telescopes, non-lumen flexible scopes , SS <19. 65 lbs instruments, non-lumen devices 30 TSO 3 Inc. 2016. All Rights Reserved. 30

STERIZONE® VP 4 Sterilizer Cycles Time Can Process ID x Dia. Up to 3 single channel flexible endoscopes in the presence of other packaged medical devices or ≥ 1. 0 mm id x ≤ 989 mm up to 2 double channel and one single channel for a maximum of 5 total channels Single Cycle 46 minutes* Rigid channel devices including single channel and double rigid channel endoscopes Multi-Channel flexible endoscopes up to 4 channels such as air, water, working, etc. (e. g. colonoscope, gastroscopes) ≥ 0. 7 mm x ≤ 500 mm ≥ 2. 0 mm x ≤ 575 mm ≥ 1. 2 mm x ≤ 1955 m ≥ 1. 45 mm x ≤ 3500 mm Mixed loads of general instrumentation up to 75 lb. consisting of nonlumened endoscopes and instruments. • Cycle time varies based on composition of the load and load conditions. **Only the Olympus® EVIS EXERA II Duodenovideoscope TJF-Q 180 V in Canada and EU. The US FDA 510 k does not include this scope. 31 TSO 3 Inc. 2016. All Rights Reserved.
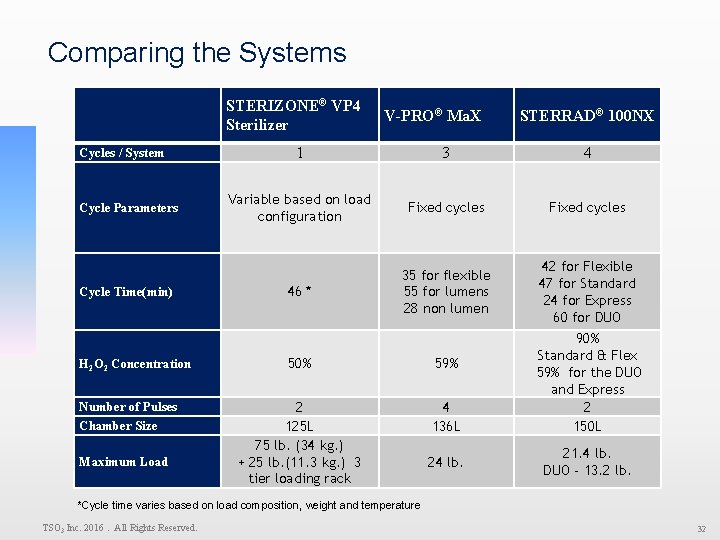
Comparing the Systems Cycles / System Cycle Parameters Cycle Time(min) H 2 O 2 Concentration Number of Pulses Chamber Size Maximum Load STERIZONE® VP 4 Sterilizer V-PRO® Ma. X STERRAD® 100 NX 1 3 4 Variable based on load configuration Fixed cycles 46 * 35 for flexible 55 for lumens 28 non lumen 42 for Flexible 47 for Standard 24 for Express 60 for DUO 50% 59% 2 125 L 75 lb. (34 kg. ) + 25 lb. (11. 3 kg. ) 3 tier loading rack 4 136 L 24 lb. 90% Standard & Flex 59% for the DUO and Express 2 150 L 21. 4 lb. DUO - 13. 2 lb. *Cycle time varies based on load composition, weight and temperature TSO 3 Inc. 2016. All Rights Reserved. 32
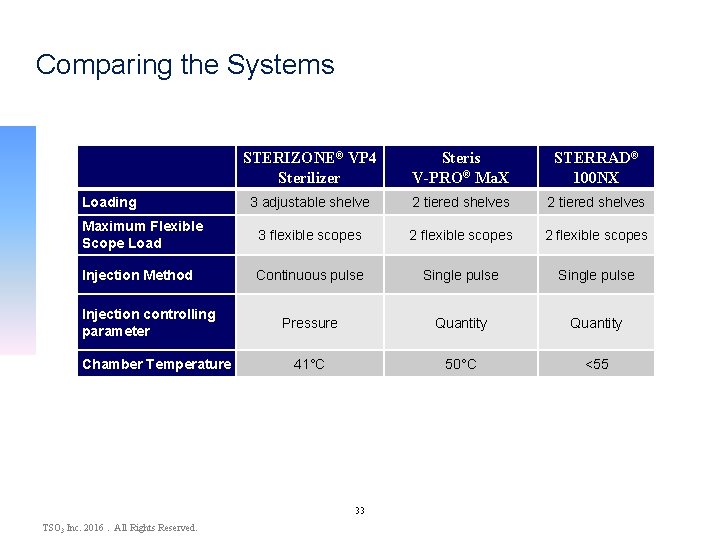
Comparing the Systems STERIZONE® VP 4 Sterilizer Steris V-PRO® Ma. X STERRAD® 100 NX 3 adjustable shelve 2 tiered shelves Maximum Flexible Scope Load 3 flexible scopes 2 flexible scopes Injection Method Continuous pulse Single pulse Pressure Quantity 41°C 50°C <55 Loading Injection controlling parameter Chamber Temperature 33 TSO 3 Inc. 2016. All Rights Reserved.
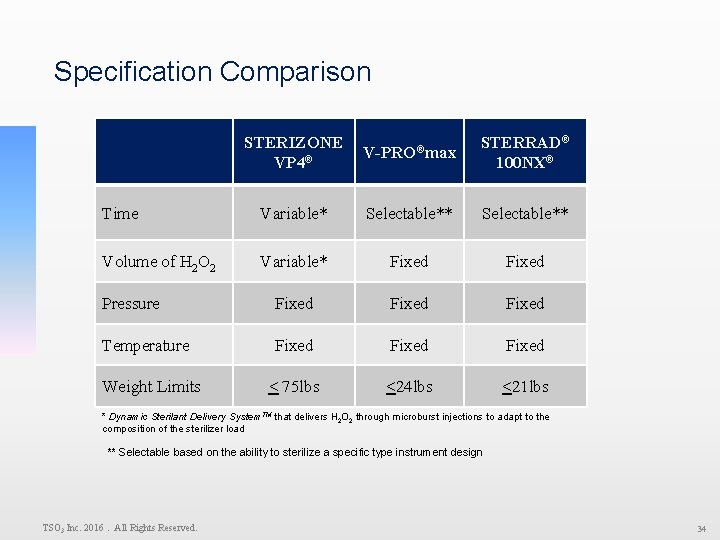
Specification Comparison STERIZONE V-PRO®max ® VP 4 STERRAD® 100 NX® Time Variable* Selectable** Volume of H 2 O 2 Variable* Fixed Pressure Fixed Temperature Fixed < 75 lbs <24 lbs <21 lbs Weight Limits * Dynamic Sterilant Delivery System. TM that delivers H 2 O 2 through microburst injections to adapt to the composition of the sterilizer load ** Selectable based on the ability to sterilize a specific type instrument design TSO 3 Inc. 2016. All Rights Reserved. 34

Creating the Improved Standard in Healthcare Sterile Reprocessing™ Thank you! Questions?

Copyright @ 2016 TSO 3 Inc. All rights reserved for all countries. No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form, be it electronically, mechanically, or by any other means such as photocopying, recording or otherwise, without the prior written permission of TSO 3 Inc. The TSO 3 logo and STERIZONE® are trademarks registered with the US Patent and Trademark Office. The STERIZONE® VP 4 Sterilizer is patented in the US under US Pat. 9, 101, 679; in Canada under Pat. 2, 767, 726; and in Europe under Pat. EP 2, 601, 976. Other patents applications pending. Corresponding foreign patents and patent applications pending. The information contained in this presentation is subject to change without notice. 36
- Slides: 36