Low energy photocatalytic glycerol conversion to valuable products

Low energy photocatalytic glycerol conversion to valuable products via Bi 2 O 3 polymorphs in the presence of H 2 O 2 Paphada Limpachanangkula, Licheng Liub, Mali Hunsomc, Benjapon Chalermsinsuwana, d a Fuels b Research Center, Department of Chemical Technology, Faculty of Science, Chulalongkorn University, Bangkok 10330, Thailand CAS Key Laboratory of Bio-based Materials, Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Qingdao 266101, Shandong, China c Associate d Center Fellow of Royal Society of Thailand (AFRST), Bangkok 10300, Thailand of Excellence on Petrochemical and Materials Technology, Chulalongkorn University, Bangkok 10330, Thailand MHMK 1808 October 24, 2018
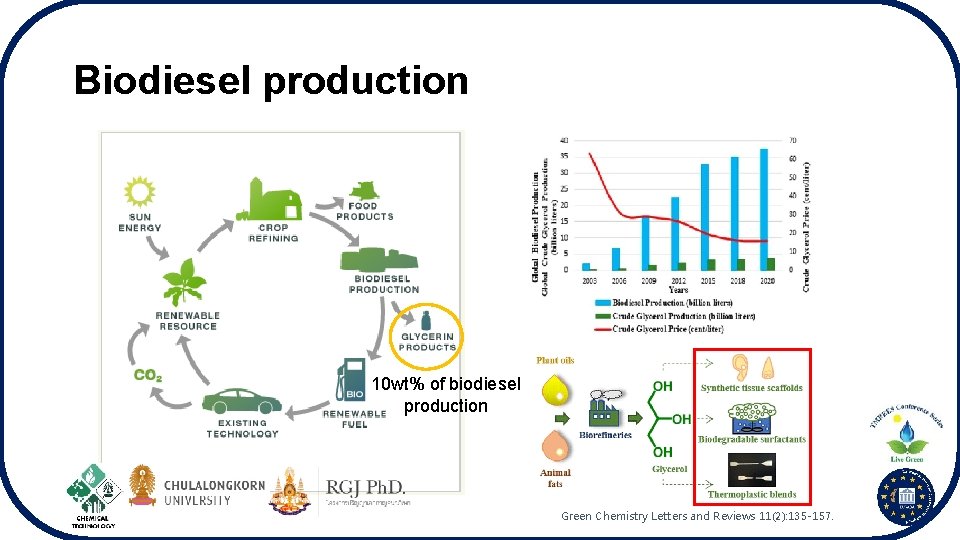
Biodiesel production 10 wt% of biodiesel production Green Chemistry Letters and Reviews 11(2): 135 -157.
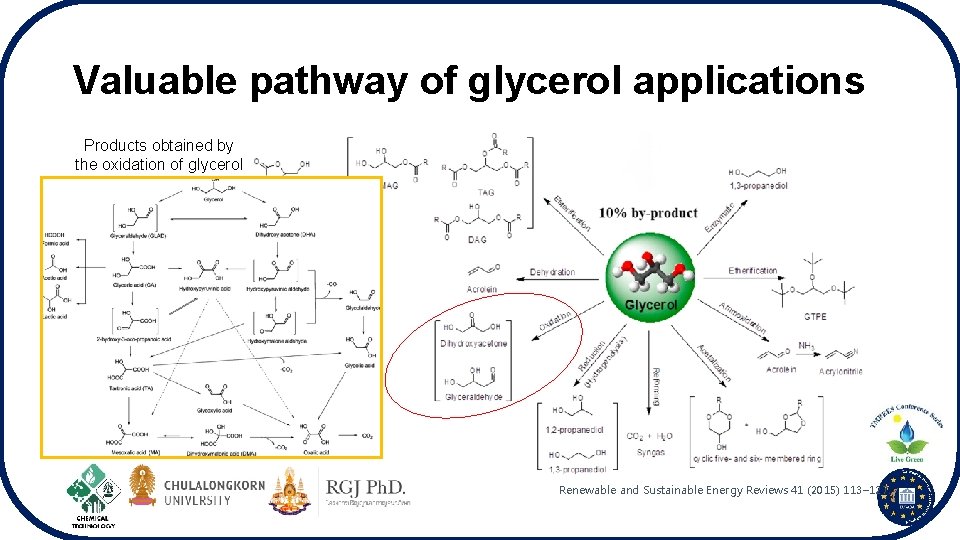
Valuable pathway of glycerol applications Products obtained by the oxidation of glycerol Renewable and Sustainable Energy Reviews 41 (2015) 113– 127.

Band edge position of photocatalysts Conduction band (CB): more negative potential than the reduction potential of the chemical species that will be reduced Nanoscale, 2013, 5, 8326 -8339. Valence band (VB): more positively than the oxidation potential of the chemical species that will be oxidized
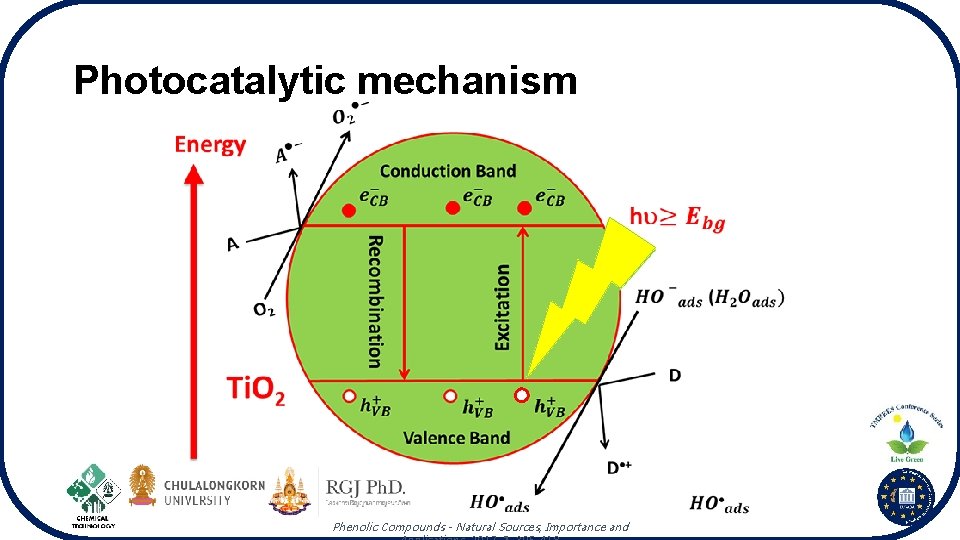
Photocatalytic mechanism Phenolic Compounds - Natural Sources, Importance and
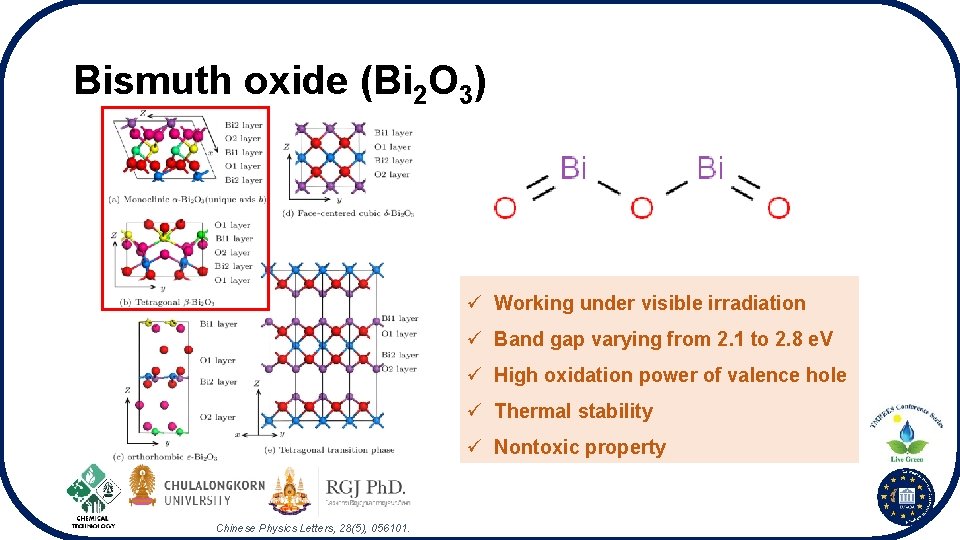
Bismuth oxide (Bi 2 O 3) ü Working under visible irradiation ü Band gap varying from 2. 1 to 2. 8 e. V ü High oxidation power of valence hole ü Thermal stability ü Nontoxic property Chinese Physics Letters, 28(5), 056101.
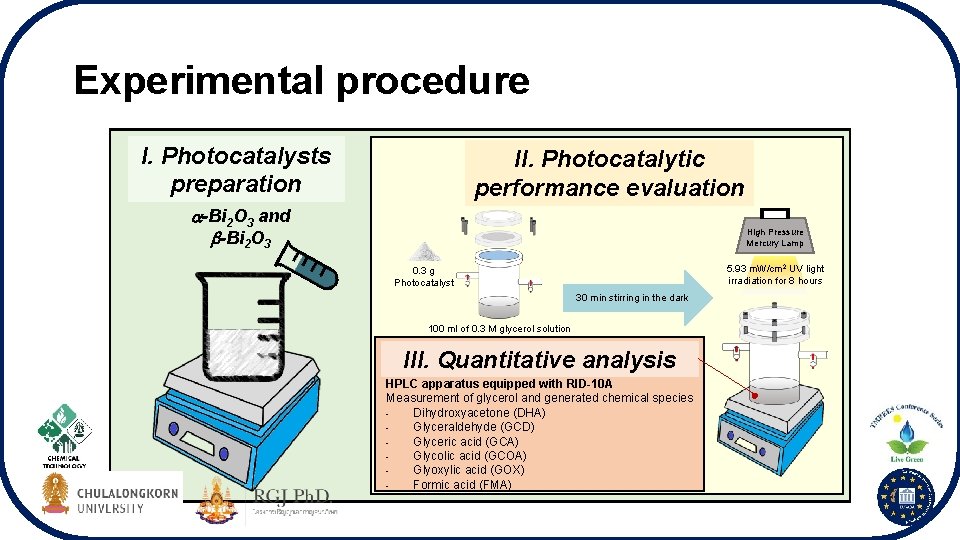
Experimental procedure I. Photocatalysts preparation II. Photocatalytic performance evaluation -Bi 2 O 3 and -Bi 2 O 3 High Pressure Mercury Lamp 5. 93 m. W/cm 2 UV light irradiation for 8 hours 0. 3 g Photocatalyst 30 min stirring in the dark 100 ml of 0. 3 M glycerol solution III. Quantitative analysis HPLC apparatus equipped with RID-10 A Measurement of glycerol and generated chemical species Dihydroxyacetone (DHA) Glyceraldehyde (GCD) Glyceric acid (GCA) Glycolic acid (GCOA) Glyoxylic acid (GOX) Formic acid (FMA)
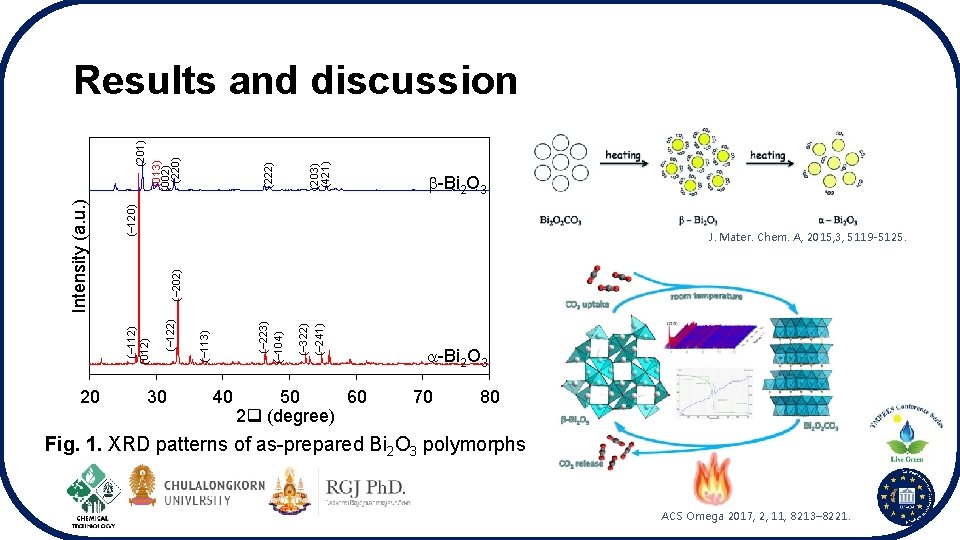
20 (203) (421) (222) ( 120) -Bi 2 O 3 30 40 ( 322) ( 241) ( 223) ( 104) ( 113) ( 122) ( 202) J. Mater. Chem. A, 2015, 3, 5119 -5125. ( 112) (012) Intensity (a. u. ) (201) (013) (002) (220) Results and discussion 50 60 2 (degree) -Bi 2 O 3 70 80 Fig. 1. XRD patterns of as-prepared Bi 2 O 3 polymorphs ACS Omega 2017, 2, 11, 8213– 8221.
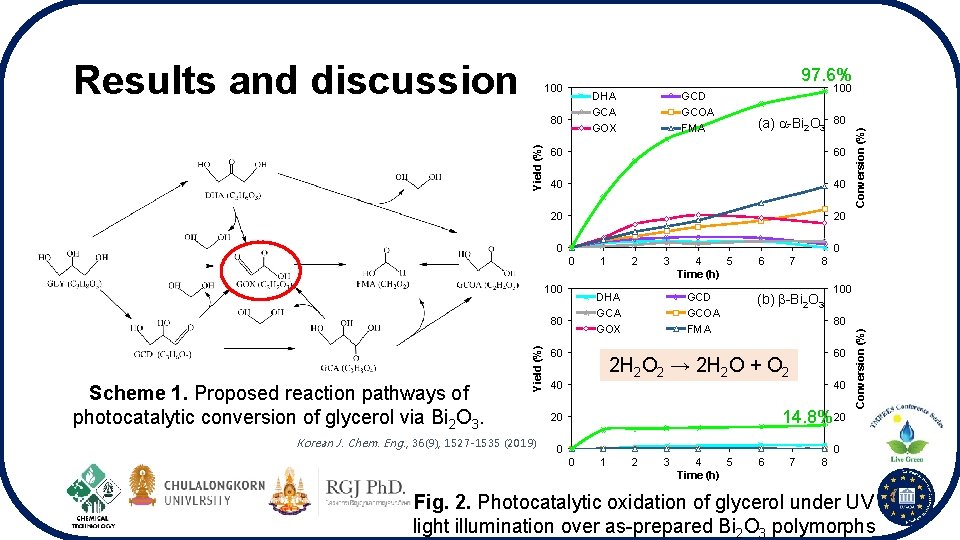
97. 6% DHA GCA GOX Yield (%) 80 80 40 40 20 20 0 0 1 2 3 DHA GCA GOX 80 Yield (%) (a) -Bi 2 O 3 60 100 Korean J. Chem. Eng. , 36(9), 1527 -1535 (2019) 100 60 0 Scheme 1. Proposed reaction pathways of photocatalytic conversion of glycerol via Bi 2 O 3. GCD GCOA FMA 60 4 5 Time (h) GCD GCOA FMA 6 7 8 (b) -Bi 2 O 3 100 80 60 2 H 2 O 2 → 2 H 2 O + O 2 40 Conversion (%) 100 40 14. 8% 20 20 0 Conversion (%) Results and discussion 0 0 1 2 3 4 5 Time (h) 6 7 8 Fig. 2. Photocatalytic oxidation of glycerol under UV light illumination over as-prepared Bi 2 O 3 polymorphs

Conclusions • The low energy photocatalytic performance of the different Bi 2 O 3 polymorphs was investigated via glycerol oxidation under irradiation of UV light in the presence of H 2 O 2 • Different phase of Bi 2 O 3 photocatalysts showed different glycerol transformation performance • -Bi 2 O 3 displayed greater photocatalytic activity for glycerol conversion almost 6. 6 times higher than that of -Bi 2 O 3 because H 2 O 2 can be cleaved to form OH radicals for glycerol oxidation • -Bi 2 O 3 system produced more valuable products especially GOX which is a significant building block for many industrial organic molecules • -Bi 2 O 3 favored H 2 O 2 decomposition that inhibited OH radicals production to oxidize glycerol
- Slides: 10