Lorcaserin Improves Renal Outcomes in Obese and Overweight

Lorcaserin Improves Renal Outcomes in Obese and Overweight Patients in the CAMELLIA-TIMI 61 Trial E. A. Bohula, S. D. Wiviott, J. P. Dwyer, S. E. Inzucchi, D. K. Mc. Guire, K. Im, S. A. Murphy, W. Miao, C. Perdomo, T. Patel, S. R. Smith, A. C. Keech, M. S. Sabatine, & B. M. Scirica on behalf of the CAMELLIA-TIMI 61 Investigators An Academic Research Organization of Brigham and Women’s Hospital and Harvard Medical School

Background • Obesity is associated with an increased risk of developing kidney disease • Unclear if mechanism is causal or via associated comorbidities • Lifestyle modification & bariatric surgery improves albuminuria, but has mixed effects on e. GFR • Renal effects of pharmacologically-mediated weight loss not well described An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School
Lorcaserin • Selective agonist of serotonin (5 HT)-2 C receptor • Hypothalamic activation of the POMC (pro-opiomelanocortin) pathway → appetite suppression • Based on phase 3 studies testing weight loss efficacy, approved for use in the US for chronic weight management An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School Lorcaserin + 5 HT 2 CR Image modified from Marx J. Science. 2003; 299: 846 -849.

Trial Schema Median Follow up: 3. 3 yrs Obese or Overweight (BMI≥ 27 kg/m 2) Established CV disease* or T 2 DM & other CV risk factors† N = 12, 000 Exercise & Reduced-Calorie Diet Lorcaserin 10 mg BID RANDOMIZE 1: 1 DOUBLE BLIND Stratified by CV disease or CV RF PLACEBO Follow up visits Q 3 mo x 2 yr then Q 4 mo Primary CV Safety EP: MACE (CV death, MI, CVA) Primary Metabolic Efficacy EP: Incident T 2 DM in pts with pre-DM *CAD, PAD or cerebrovascular disease; †T 2 DM with ≥ 1 of following: HTN, HL, hs. CRP>3, e. GFR 30 -60, albuminuria An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School Bohula EA et al. Am Heart J 2018; 202: 39 -48
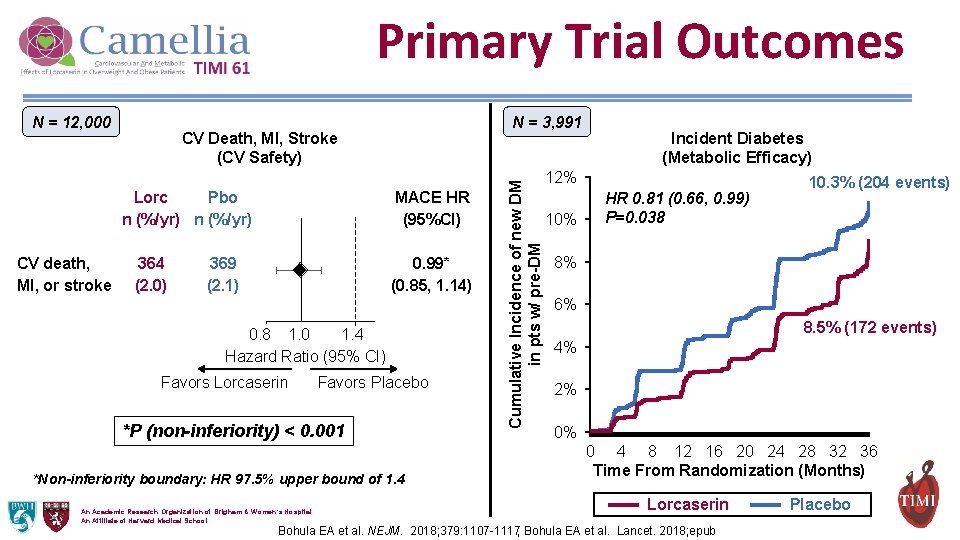
Primary Trial Outcomes CV Death, MI, Stroke (CV Safety) Lorc Pbo n (%/yr) CV death, MI, or stroke N = 3, 991 364 (2. 0) MACE HR (95%CI) 369 (2. 1) 0. 99* (0. 85, 1. 14) 0. 8 1. 0 1. 4 Hazard Ratio (95% CI) Favors Lorcaserin Favors Placebo *P (non-inferiority) < 0. 001 *Non-inferiority boundary: HR 97. 5% upper bound of 1. 4 An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School Cumulative Incidence of new DM in pts w/ pre-DM N = 12, 000 Incident Diabetes (Metabolic Efficacy) 12% 10% HR 0. 81 (0. 66, 0. 99) P=0. 038 10. 3% (204 events) 8% 6% 8. 5% (172 events) 4% 2% 0% 0 4 8 12 16 20 24 28 32 36 Time From Randomization (Months) Lorcaserin Bohula EA et al. NEJM. 2018; 379: 1107 -1117; Bohula EA et al. Lancet. 2018; epub Placebo

Renal Outcomes • Pre-specified Primary Renal Composite Outcome – Persistent new or worsening micro- or macro-albuminuria* • Micro- (UACR ≥ 30 mg/g) or macro-albuminuria (≥ 300 mg/g) – Persistent new or worsening CKD* • Increase in CKD stage by KDIGO staging – – Persistent doubling of serum creatinine* End-stage renal disease (ESRD) = dialysis or e. GFR<15 Renal transplant Renal death • Other Pre-specified Renal Endpoints – Persistent e. GFR decline by ≥ 30% or ≥ 40%* An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School *On ≥ 2 consecutive measurements ≥ 30 days apart KDIGO CKD Stage e. GFR UACR 0 ≥ 60 None 1 ≥ 90 ≥ 30 2 60 -89 ≥ 30 3 30 -59 ≥ 30 4 15 -29 ≥ 30 5 <15 or dialysis ≥ 30 e. GFR by CKD-EPI (ml/min/1. 73 m 2); UACR by spot urinary albumin to creatinine ratio(mg/g)
![Baseline Characteristics Characteristic (N=12, 000) Age (median, IQR) Male Value 64 [58, 69] 64 Baseline Characteristics Characteristic (N=12, 000) Age (median, IQR) Male Value 64 [58, 69] 64](http://slidetodoc.com/presentation_image_h2/733b88d32e091f22ad0b759c0a0f983f/image-7.jpg)
Baseline Characteristics Characteristic (N=12, 000) Age (median, IQR) Male Value 64 [58, 69] 64 Weight in kg (median, IQR) 102 [90, 116] BMI in kg/m 2 (median, IQR) 35 [32, 39] Established CV Disease / Multiple CV RF Hypertension DM / Pre-DM / No DM e. GFR in ml/min/1. 73 m 2 (median, IQR) >=90 60 -<90 <60 UACR in mg/g (median, IQR) <30 30 -<300 (microalbuminuria) >=300 (macroalbuminuria) An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School 75 / 25 90 57 / 33 / 10 76 [63 -89] 24 57 19 7. 0 [3. 9 -19. 0] 82 15 3. 2 % unless otherwise specified. Pooled data; no differences between treatment arms (p≥ 0. 05)
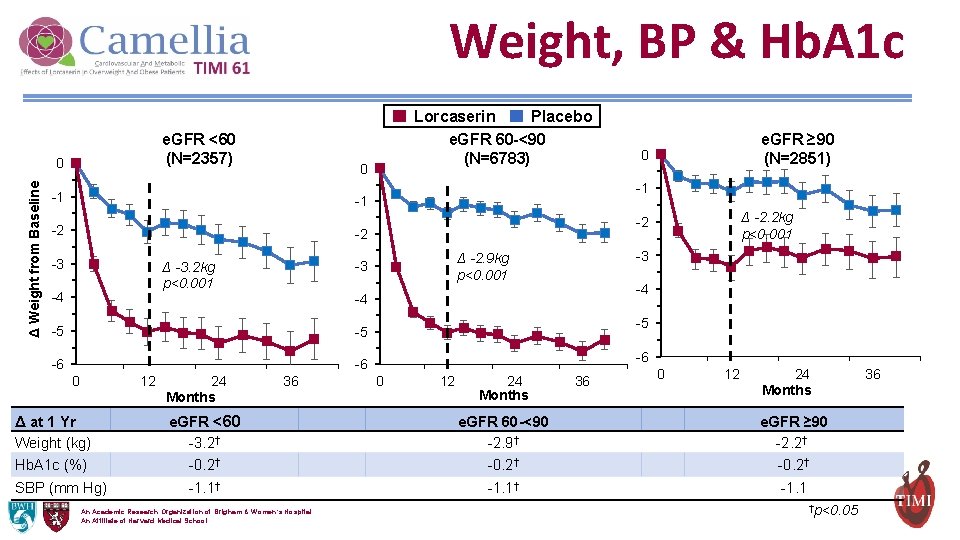
Weight, BP & Hb. A 1 c e. GFR <60 (N=2357) Δ Weight from Baseline 0 Lorcaserin Placebo e. GFR 60 -<90 (N=6783) 0 -1 -1 -1 -2 -4 -3 Δ -2. 9 kg p<0. 001 -3 Δ -3. 2 kg p<0. 001 -4 -4 -5 -5 -6 -6 0 12 Δ at 1 Yr Weight (kg) Hb. A 1 c (%) SBP (mm Hg) 24 Months 36 Δ -2. 2 kg p<0. 001 -2 -2 -3 e. GFR ≥ 90 (N=2851) 0 -5 -6 0 12 24 Months 36 0 12 24 Months e. GFR <60 -3. 2† -0. 2† e. GFR 60 -<90 -2. 9† -0. 2† e. GFR ≥ 90 -2. 2† -0. 2† -1. 1† -1. 1 An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School †p<0. 05 36
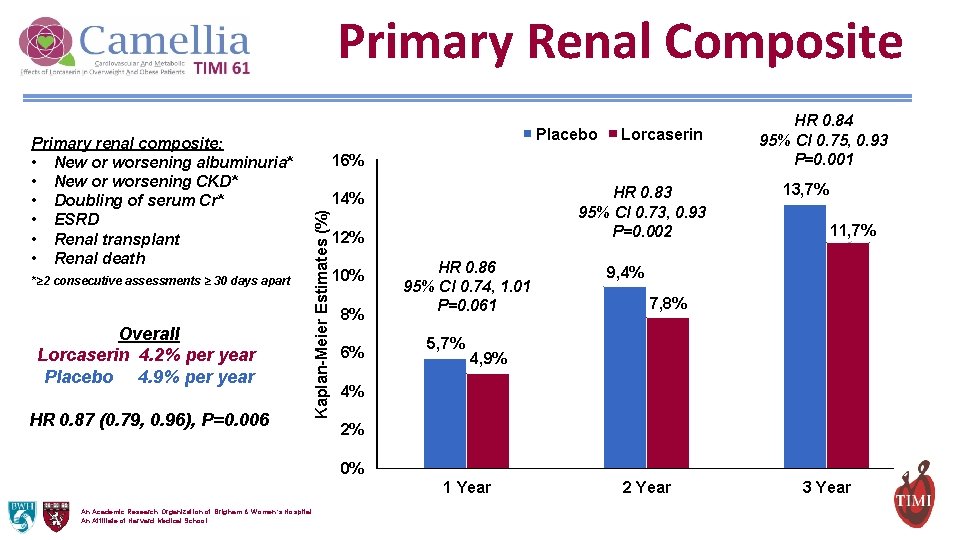
Primary Renal Composite *≥ 2 consecutive assessments ≥ 30 days apart Overall Lorcaserin 4. 2% per year Placebo 4. 9% per year HR 0. 87 (0. 79, 0. 96), P=0. 006 Lorcaserin 16% HR 0. 83 95% CI 0. 73, 0. 93 P=0. 002 14% Kaplan-Meier Estimates (%) Primary renal composite: • New or worsening albuminuria* • New or worsening CKD* • Doubling of serum Cr* • ESRD • Renal transplant • Renal death Placebo 12% 10% 8% 6% HR 0. 86 95% CI 0. 74, 1. 01 P=0. 061 5, 7% HR 0. 84 95% CI 0. 75, 0. 93 P=0. 001 13, 7% 11, 7% 9, 4% 7, 8% 4, 9% 4% 2% 0% 1 Year An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School 2 Year 3 Year
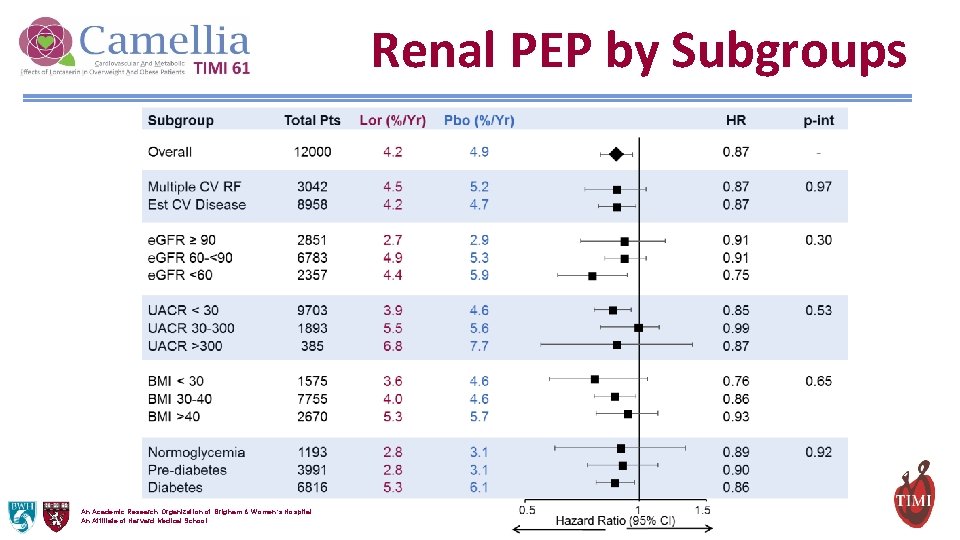
Renal PEP by Subgroups An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School
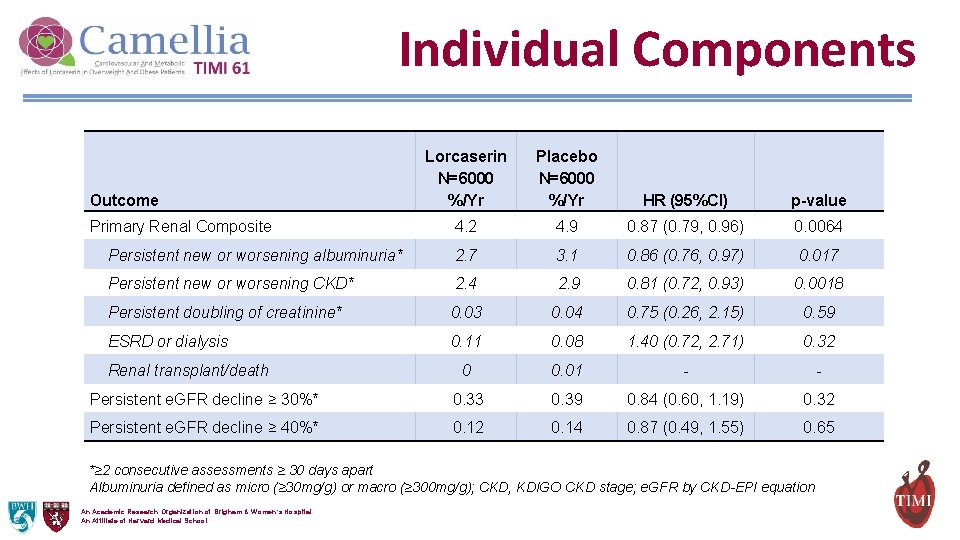
Individual Components Lorcaserin N=6000 %/Yr Placebo N=6000 %/Yr HR (95%CI) p-value 4. 2 4. 9 0. 87 (0. 79, 0. 96) 0. 0064 Persistent new or worsening albuminuria* 2. 7 3. 1 0. 86 (0. 76, 0. 97) 0. 017 Persistent new or worsening CKD* 2. 4 2. 9 0. 81 (0. 72, 0. 93) 0. 0018 Persistent doubling of creatinine* 0. 03 0. 04 0. 75 (0. 26, 2. 15) 0. 59 ESRD or dialysis 0. 11 0. 08 1. 40 (0. 72, 2. 71) 0. 32 0 0. 01 - - Persistent e. GFR decline ≥ 30%* 0. 33 0. 39 0. 84 (0. 60, 1. 19) 0. 32 Persistent e. GFR decline ≥ 40%* 0. 12 0. 14 0. 87 (0. 49, 1. 55) 0. 65 Outcome Primary Renal Composite Renal transplant/death *≥ 2 consecutive assessments ≥ 30 days apart Albuminuria defined as micro (≥ 30 mg/g) or macro (≥ 300 mg/g); CKD, KDIGO CKD stage; e. GFR by CKD-EPI equation An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School
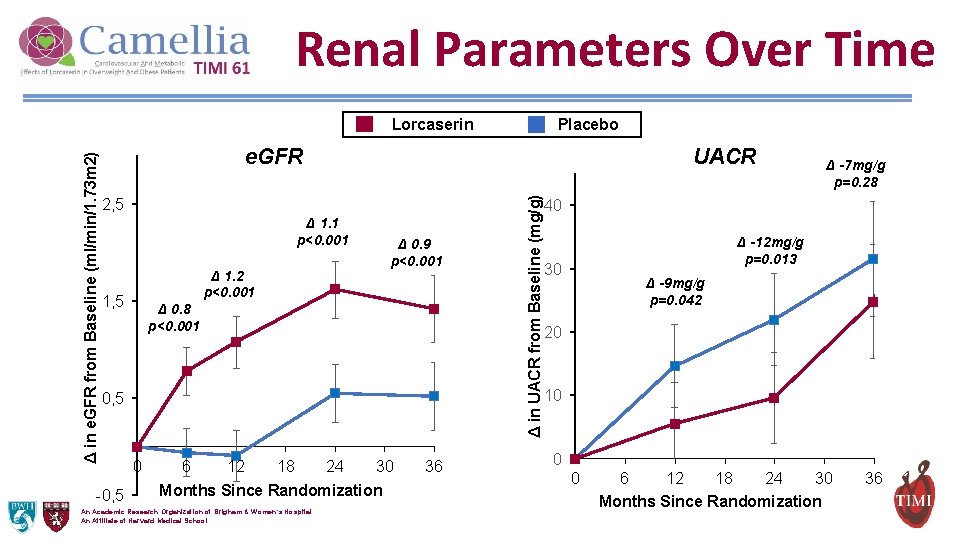
Renal Parameters Over Time Placebo e. GFR UACR 2, 5 Δ 1. 1 p<0. 001 Δ 0. 9 p<0. 001 Δ 1. 2 p<0. 001 1, 5 Δ 0. 8 p<0. 001 0, 5 -0, 5 0 6 12 18 24 30 Months Since Randomization An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School 36 Δ in UACR from Baseline (mg/g) Δ in e. GFR from Baseline (ml/min/1. 73 m 2) Lorcaserin Δ -7 mg/g p=0. 28 40 Δ -12 mg/g p=0. 013 30 Δ -9 mg/g p=0. 042 20 10 0 0 6 12 18 24 30 Months Since Randomization 36

Summary On a background of lifestyle interventions in overweight or obese patients at high CV risk, lorcaserin: • Resulted in modest, durable improvement in risk factors, including weight, Hb. A 1 c and blood pressure • ↓ incident & progressive of renal disease by e. GFR and albuminuria • Findings consistent over time and across subgroups An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School

Conclusion Taken together, the findings of CV safety, glycemic benefit and now improved renal status support the role of lorcaserin as an adjunct to lifestyle modification for chronic management of weight and metabolic health. An Academic Research Organization of Brigham & Women’s Hospital An Affiliate of Harvard Medical School

Lorcaserin and Renal Outcomes in Obese and Overweight Patients in the CAMELLIA-TIMI 61 Trial Benjamin M. Scirica, Erin A. Bohula, Jamie P. Dwyer, Arman Qamar, Silvio E. Inzucchi, Darren K. Mc. Guire, Anthony C. Keech MD, Steven R. Smith, Sabina A. Murphy, Kyungah Im, Lawrence A. Leiter, Milan Gupta, Tushar Patel, Wenfeng Miao, Carlos Perdomo, Marc P. Bonaca, Christian T. Ruff, Marc S. Sabatine, and Stephen D. Wiviott for the CAMELLIA-TIMI 61 Steering Committee and Investigators An Academic Research Organization of Brigham and Women’s Hospital and Harvard Medical School
- Slides: 15