Look over objectives and try those types of

Look over objectives and try those types of problems on past worksheets and quizzes, especially if you know you are weak on it • Come in for help before the test for extra help or to retake quizzes! • These are the “clicker” questions from this unit – Ans: (2) B (3) B (4) A (5) C (6) A (7) D (8) D (9) A (10) B (11) B (12) D (13) C (14) B (15) B (16) D (17) D (18) D (19) D (20) B (21) D (22) C (23) C (24) D – Notably missing from these are questions about identifying types of reactions and why we balance equations and how you know an equation is balanced

What are the products in the following chemical equation: Zn + Cu. SO 4 Zn. SO 4 + Cu A. ) zinc and copper B. ) zinc sulfate and copper C. ) zinc and copper (II) sulfate D. ) only zinc

Given the incomplete equation: 4 Fe + 3 O 2 2 X Which compound is represented by X? A. ) Fe. O B. ) Fe 2 O 3 C. ) Fe 3 O 2 D. ) Fe 3 O 4

Ca. SO 4 + Al. Cl 3 Al 2(SO 4)3 + Ca. Cl 2 What is the coefficient of Al 2(SO 4)3 when the equation is completely balanced using the smallest whole-number coefficients? A. ) 1 B. ) 2 C. ) 3 D. ) 4

Given the incomplete equation: 2 N 2 O 5(g) Which set of products completes and balances the incomplete equation? A. ) 2 N 2(g) + 3 H 2(g) B. ) 2 N 2(g) + 2 O 2(g) C. ) 4 NO 2(g) + O 2(g) D. ) 4 NO(g) + 5 O 2(g)

Given the unbalanced equation: Al + O 2 Al 2 O 3 When this equation is completely balanced using smallest whole numbers, what is the sum of the coefficients? A. ) 9 B. ) 7 C. ) 5 D. ) 4

If you rewrite the following word equation as a balanced chemical equation, what will the coefficient and symbol for fluorine be? nitrogen trifluoride nitrogen + fluorine A. ) 6 F 2 B. ) F 3 C. ) 6 F D. ) 3 F 2
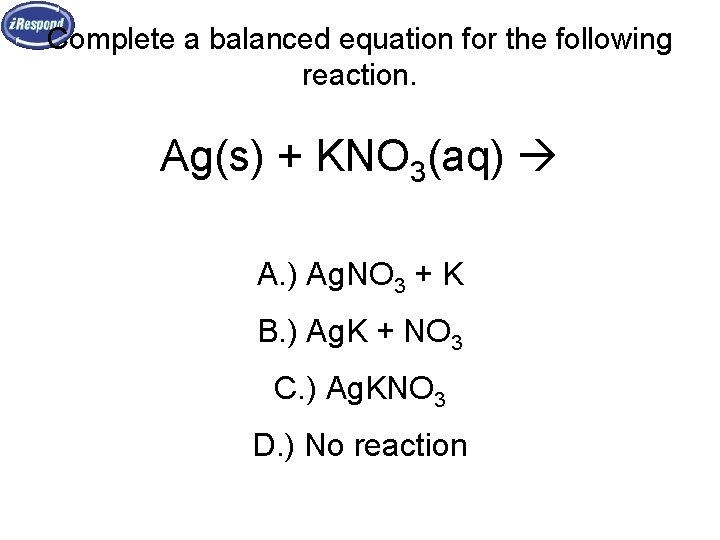
Complete a balanced equation for the following reaction. Ag(s) + KNO 3(aq) A. ) Ag. NO 3 + K B. ) Ag. K + NO 3 C. ) Ag. KNO 3 D. ) No reaction
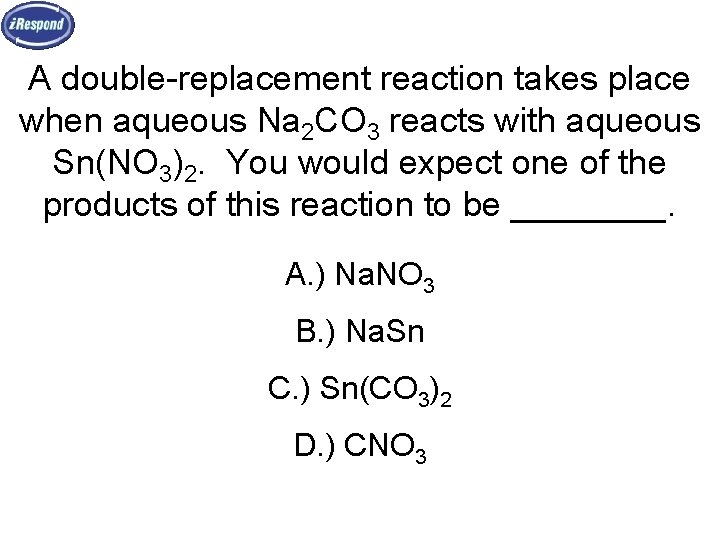
A double-replacement reaction takes place when aqueous Na 2 CO 3 reacts with aqueous Sn(NO 3)2. You would expect one of the products of this reaction to be ____. A. ) Na. NO 3 B. ) Na. Sn C. ) Sn(CO 3)2 D. ) CNO 3
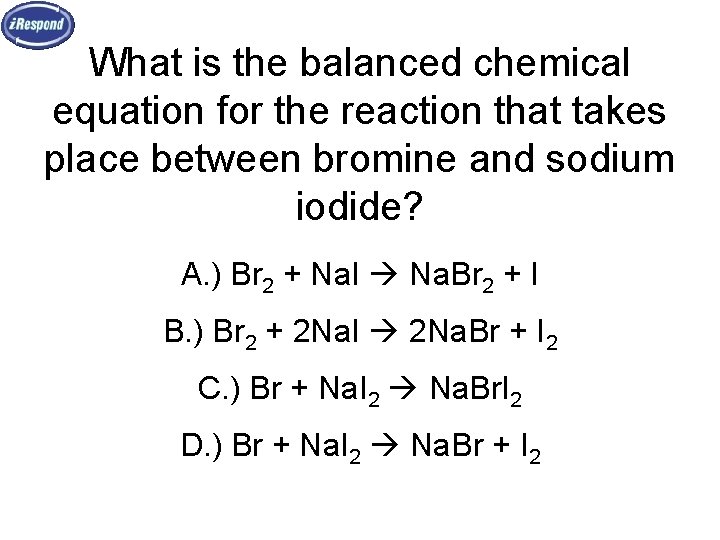
What is the balanced chemical equation for the reaction that takes place between bromine and sodium iodide? A. ) Br 2 + Na. I Na. Br 2 + I B. ) Br 2 + 2 Na. I 2 Na. Br + I 2 C. ) Br + Na. I 2 Na. Br. I 2 D. ) Br + Na. I 2 Na. Br + I 2
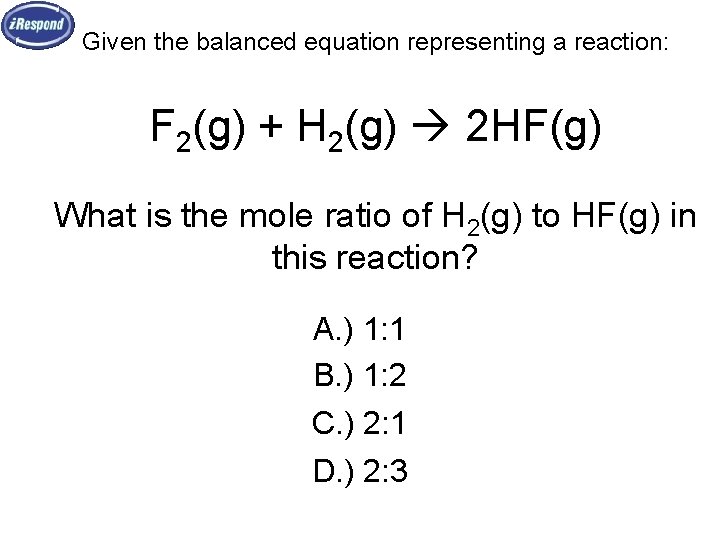
Given the balanced equation representing a reaction: F 2(g) + H 2(g) 2 HF(g) What is the mole ratio of H 2(g) to HF(g) in this reaction? A. ) 1: 1 B. ) 1: 2 C. ) 2: 1 D. ) 2: 3
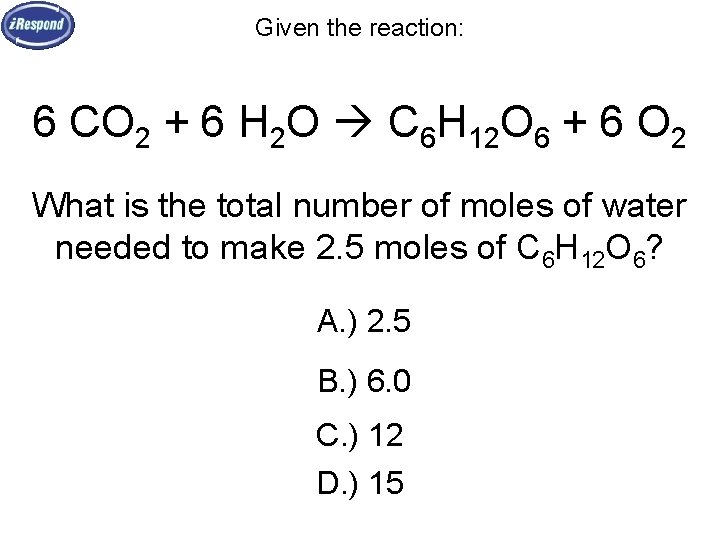
Given the reaction: 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 What is the total number of moles of water needed to make 2. 5 moles of C 6 H 12 O 6? A. ) 2. 5 B. ) 6. 0 C. ) 12 D. ) 15
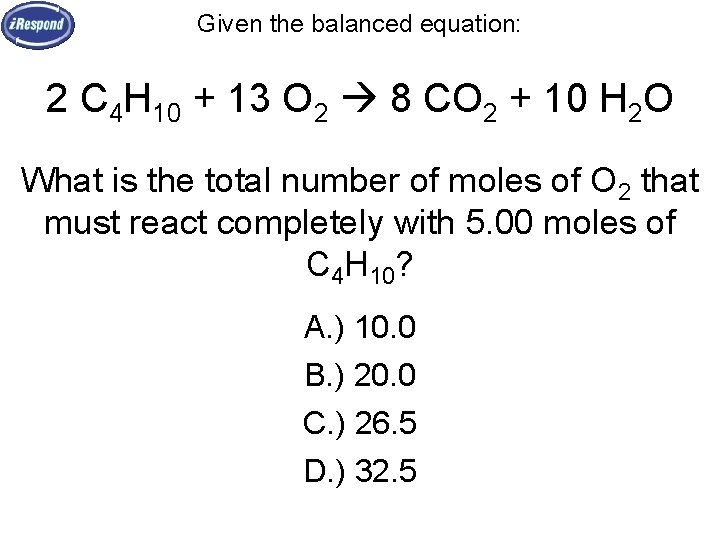
Given the balanced equation: 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O What is the total number of moles of O 2 that must react completely with 5. 00 moles of C 4 H 10? A. ) 10. 0 B. ) 20. 0 C. ) 26. 5 D. ) 32. 5

2 Pb(NO 3)2(s) 2 Pb. O(s) + 4 NO 2(g) + O 2(g) How many grams of oxygen are produced when 11. 5 grams of NO 2 is formed? A. ) 1. 00 g B. ) 2. 00 g C. ) 2. 88 g D. ) 32. 0 g
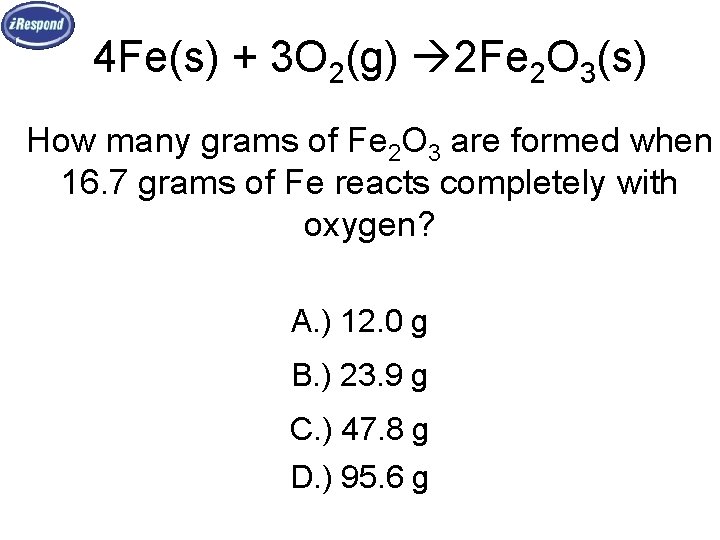
4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) How many grams of Fe 2 O 3 are formed when 16. 7 grams of Fe reacts completely with oxygen? A. ) 12. 0 g B. ) 23. 9 g C. ) 47. 8 g D. ) 95. 6 g
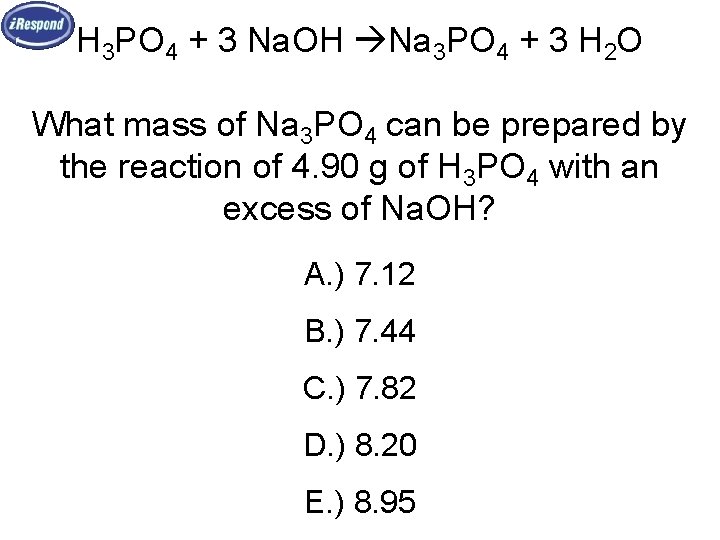
H 3 PO 4 + 3 Na. OH Na 3 PO 4 + 3 H 2 O What mass of Na 3 PO 4 can be prepared by the reaction of 4. 90 g of H 3 PO 4 with an excess of Na. OH? A. ) 7. 12 B. ) 7. 44 C. ) 7. 82 D. ) 8. 20 E. ) 8. 95
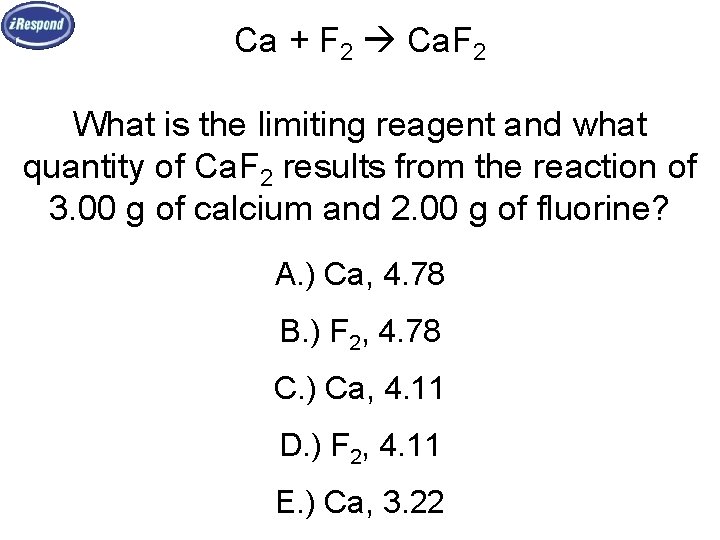
Ca + F 2 Ca. F 2 What is the limiting reagent and what quantity of Ca. F 2 results from the reaction of 3. 00 g of calcium and 2. 00 g of fluorine? A. ) Ca, 4. 78 B. ) F 2, 4. 78 C. ) Ca, 4. 11 D. ) F 2, 4. 11 E. ) Ca, 3. 22

Once the limiting reactant is all used up in a reaction, the left-over reactant is known as A. ) the product B. ) the limited reactant C. ) the limiting reactant D. ) the excess reactant

The reactant you run out of first is known as A. ) the running reactant B. ) the excess reactant C. ) the absent reactant D. ) the limiting reactant

When we use a balanced equation to predict the number of grams of a product in a reaction, we get the A. ) product yield B. ) theoretical yield C. ) percent yield D. ) actual yield

In an experiment, students determined theoretical yield to be 20 grams. Their actual yield was 15 grams. What was their percent yield? A. ) 133% B. ) 1. 33% C. ) 0. 75% D. ) 75%

In an experiment, students determined theoretical yield to be 12 grams. Their percent yield was 90 percent. What was their actual yield? A. ) 13. 3 g B. ) 5. 3 g C. ) 10. 8 g D. ) 7. 5 g
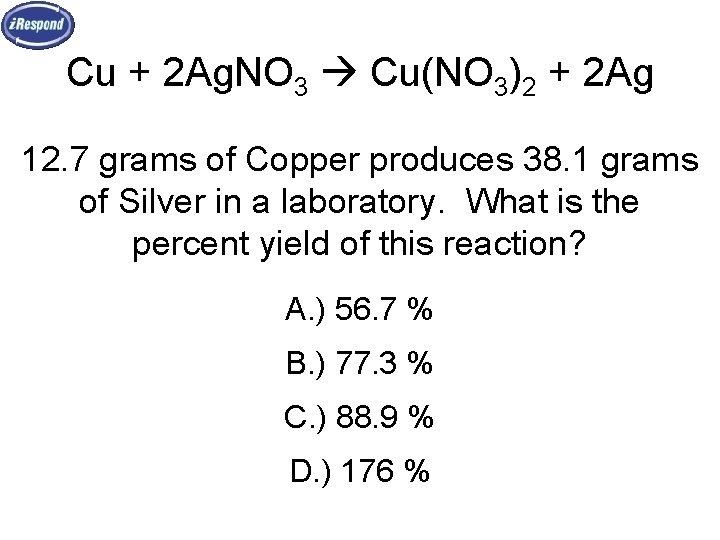
Cu + 2 Ag. NO 3 Cu(NO 3)2 + 2 Ag 12. 7 grams of Copper produces 38. 1 grams of Silver in a laboratory. What is the percent yield of this reaction? A. ) 56. 7 % B. ) 77. 3 % C. ) 88. 9 % D. ) 176 %

4 NH 3 + 5 O 2 4 NO + 6 H 2 O The reaction of 0. 68 g of NH 3 with excess O 2 according to the following reaction yields 0. 98 g of NO. What is the percent yield? A. ) 72% B. ) 74% C. ) 78% D. ) 82% E. ) 86%
- Slides: 24