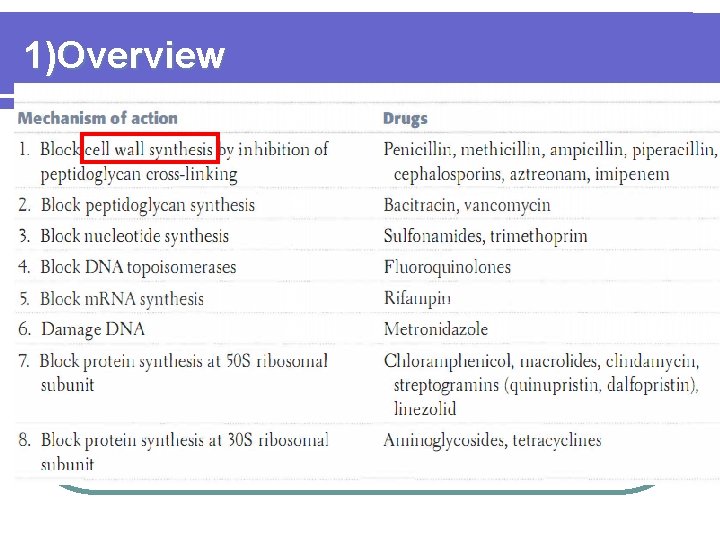
Logical Line 1Overview 2Resistance of Lactam Antibiotics 3Classification


Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective

Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective
1)Overview

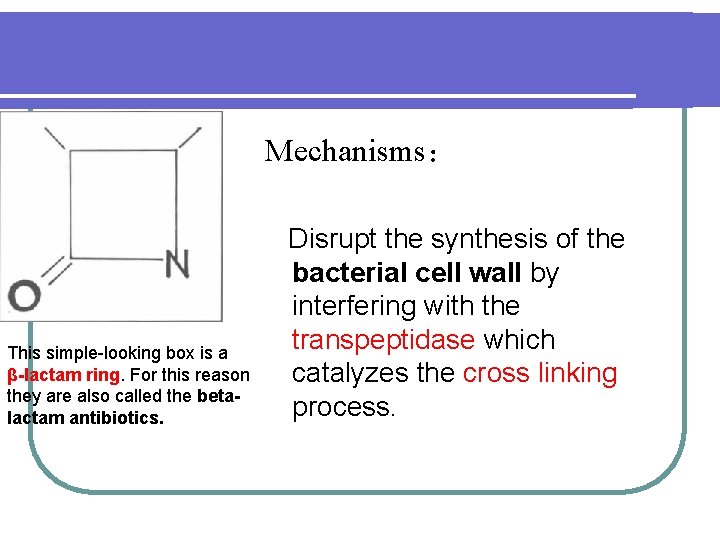
Mechanisms: This simple-looking box is a β-lactam ring. For this reason they are also called the betalactam antibiotics. Disrupt the synthesis of the bacterial cell wall by interfering with the transpeptidase which catalyzes the cross linking process.
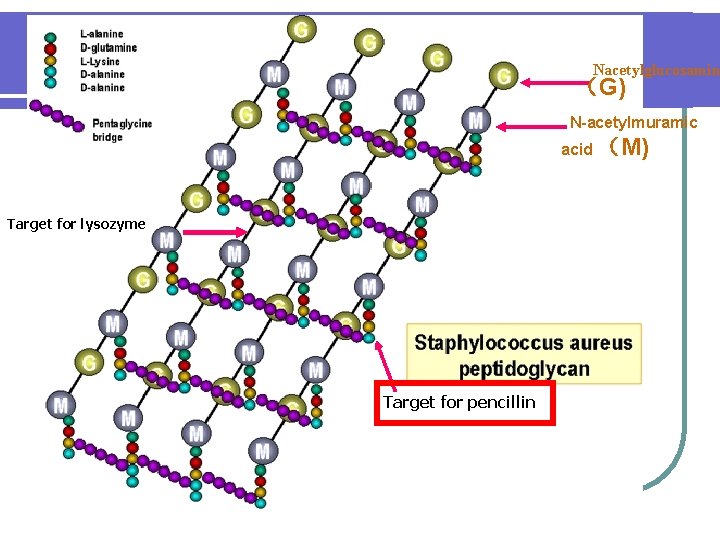
Nacetylglucosamin (G) N-acetylmuramic acid (M) Target for lysozyme Target for pencillin


Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective

2)Resistance of β- Lactam Antibiotics Four primary mechanisms by which bacteria can overcome β-lactam antibiotics: Production of β-lactamase enzymes (ii) Changes in the active site of PBPs (iii) Decreased expression of outer membrane proteins(OMPs) (iv) Efflux Pumps (i)

β-lactamase ! l Nearly 70 years ago, the first β-lactamase enzyme was identified in Bacillus (Escherichia) coli before the clinical use of penicillin. l E. P. Abraham and E. Chain described the phenomenon, but they are unable to isolate it. l 4 years later, Kirby successfully extracted these cellfree “penicillin inactivators” from Staphylococcus aureus. The emergence of a significant clinical problem

β-lactamase ! l The growing number of β-lactam antibiotics has since increased the selective pressure on bacteria, promoting the survival of organisms with multiple β-lactamases. l Currently, more than 850β-lactamases are identified.

Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3) Mechanism & Classification of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective
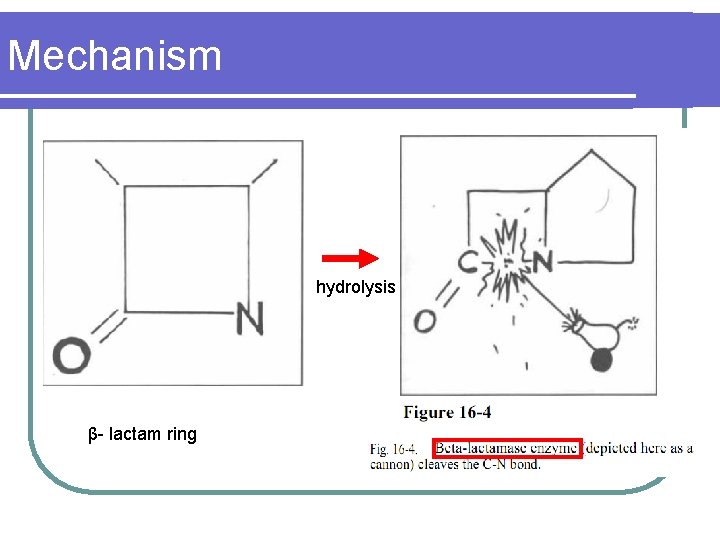
Mechanism hydrolysis β- lactam ring
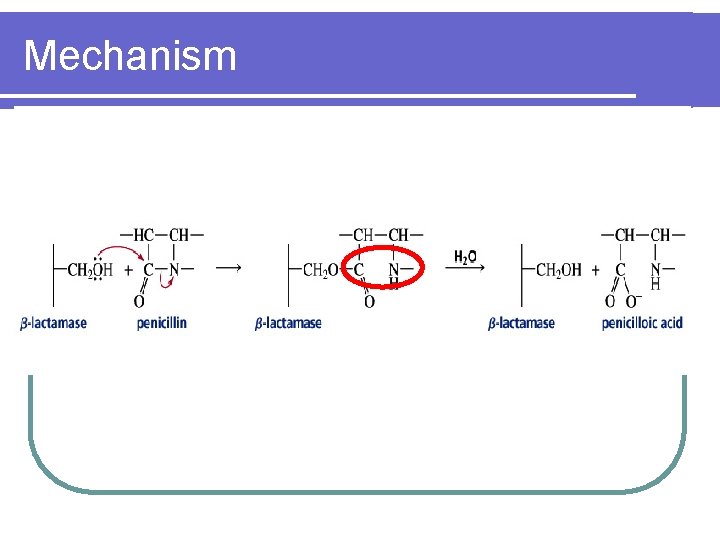
Mechanism
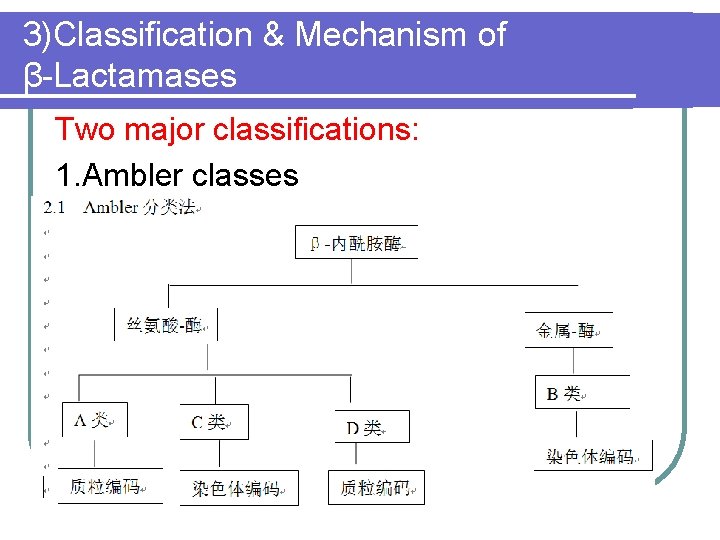
3)Classification & Mechanism of β-Lactamases Two major classifications: 1. Ambler classes

2. Bush-Jacoby-Medeiros groups

eg. l Class A serine β-lactamases l Class A ESBLs l Class A serine carbapenemases l Class B metallo-β-lactamases l Class C serine cephalosporinases l Class D serine oxacillinases

Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective

4)β- Lactamase Inhibitor Circumventing β- Lactamases-mediated resistent in 2 ways: (i) Create substrates that reversibly and/or irreversibly bind the enzyme with high affinity but form unfavorable steric interactions as the acyl-enzyme. (ii) Develop mechanism-based or irreversible “suicide inhibitors”.

β- Lactamase Inhibitors Clavulanic acid ---the first β-lactamase inhibitor ---isolated from Streptomyces clavuligerus in the 1970 s ---showed little antimicrobial activity alone, but when combined with amoxicillin, amoxicillin clavulanate significantly lowered the amoxicillin MICs (against S. aureus, K. pneumoniae, Proteus mirabilis, and E. coli) l Sulbactam l Tazobactam Sulbactam and tazobactam are penicillinate sulfones that were later developed by the pharmaceutical industry as synthetic compounds in 1978 and 1980, respectively l

l All three β-lactamase inhibitor compounds share structural similarity with penicillin; l are effective against many susceptible organisms expressing class A ; l are generally less effective against class B, C, and D β-lactamases

Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor( combination) 6)Lessons Learned & A Prespective

5)β- Lactam- β-Lactamase Inhibitor( combination) Amoxicillin-clavulanate (ii) Ticarcillin-clavulanate (iii) Ampicillin-sulbactam (iv) Piperacillin-tazobactam (i)

Logical Line 1)Overview 2)Resistance of β- Lactam Antibiotics 3)Classification & Mechanism of β-Lactamases 4)β- Lactamase Inhibitor 5)β- Lactam- β-Lactamase Inhibitor(combination) 6)Lessons Learned & A Prespective

6)Lessons Learned & A Prespective “Ideal inhibitor” High affininity for the active site Mimicking the “natural substrate” Stabilizing interactions in the active site Reaction chemistry that slows deacylation and favors activation over inhibitor hydrolysis (v) Rapid cell penetration (vi) Low propensity to induce β-lactamase production (vii) Identification of measureable biological correlates of β-lactamase inhibition in the cell. (viii) In the meantime, use what we have. (i) (iii) (iv)

A Perspective The main challenge in β-lactamase inhibitor development is : Discovering novel inhibitors with activity against a broad spectrum of inhibitorsusceptible and-resistant enzymes from multiple classes.

A Perspective New inhibitors must also target carbapenemases (serine and metallo-), as carbapenems are still the most potent β-lactams available for clinical use. Development of “second-generation” inhibitors that target resistant β-lactamases

Fighting against…… β- lactamase inhibitor Resistance to β- lactamase inhibitor ……………… A long way to go…………. .

“Is the perfect β-lactamase inhibitor an unattainable goal? ” “Perhaps. Yet, we anticipate with excitement the achievements in the next 3 decades and suspect that better chemistries and combinations will ultimately be found. ”
- Slides: 30