Logarithms Logarithms Logarithms to various bases red is

Logarithms
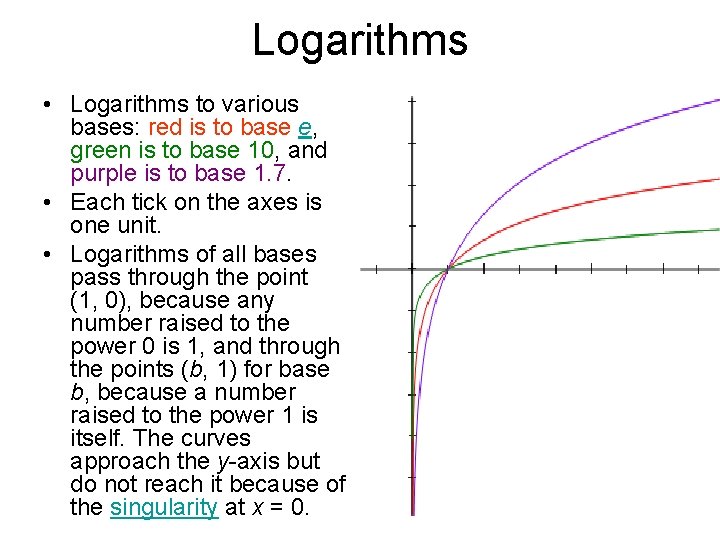
Logarithms • Logarithms to various bases: red is to base e, green is to base 10, and purple is to base 1. 7. • Each tick on the axes is one unit. • Logarithms of all bases pass through the point (1, 0), because any number raised to the power 0 is 1, and through the points (b, 1) for base b, because a number raised to the power 1 is itself. The curves approach the y-axis but do not reach it because of the singularity at x = 0.

Definition • The log of any number is the power to which the base must be raised to give that number. • log(10) is 1 and log(100) is 2 (because 102 = 100). • Example log 2 X = 8 28 = X X = 256

Example 1 • • “ 10 to the” is also the anti-log (opposite) log x 10 = X
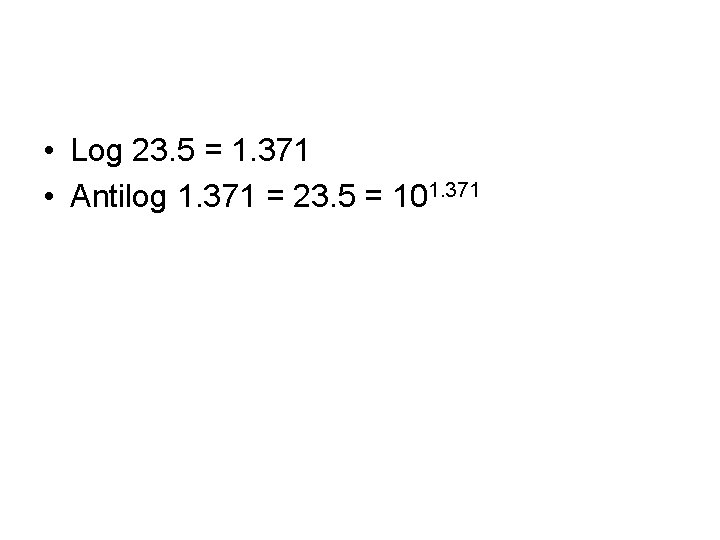
• Log 23. 5 = 1. 371 • Antilog 1. 371 = 23. 5 = 101. 371

Logs used in Chem • The most prominent example is the p. H scale, but many formulas that we use require to work with log and ln. • The p. H of a solution is the -log([H+]), where square brackets mean concentration.

Example 2 Review Log rules • log X = 0. 25 • Raise both side to the power of 10 (or calculating the antilog) 10 log x = 100. 25 X = 1. 78

Example 3 Review Log Rules • Logc (am) = m logc(a) • Solve for x 3 x = 1000 • Log both sides to get rid of the exponent log 3 x = log 1000 x log 3 = log 1000 x = log 1000 / log 3 x = 6. 29

Multiplying and Dividing logs • log a x log b = log (a+b) • log a/b = log (a-b) • This holds true as long as the logs have the same base.

Problem 1 • 2 log (x) – log 10 - 3 = 0

Solution Try It Out Problem 1 Solution

Problem 2 • 3. 5 = ln 5 x
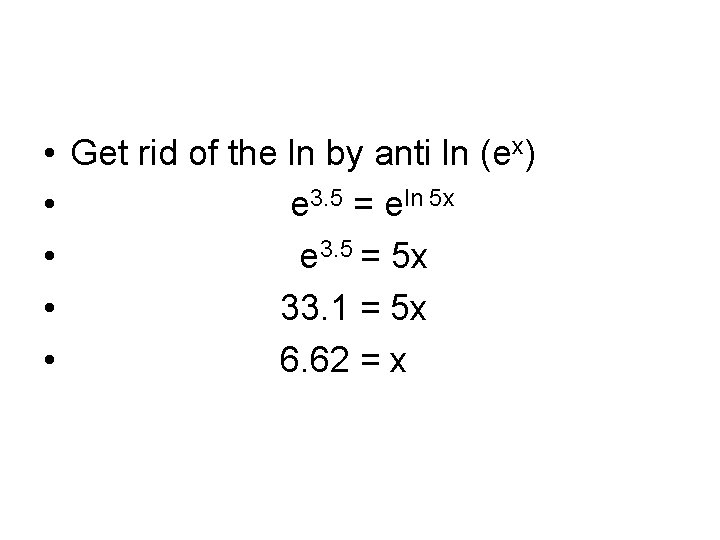
• • • Get rid of the ln by anti ln (ex) e 3. 5 = eln 5 x e 3. 5 = 5 x 33. 1 = 5 x 6. 62 = x

Negative Logarithms • We recall that 10 -1 means 1/10, or the decimal fraction, 0. 1. • What is the logarithm of 0. 1? • SOLUTION: 10 -1 = 0. 1; log 0. 1 = -1 • Likewise 10 -2 = 0. 01; log 0. 01 = -2

Natural Logarithms • The natural log of a number is the power to which e must be raised to equal the number. e =2. 71828 • natural log of 10 = 2. 303 • e 2. 303= 10 ln 10 = 2. 303 • e ln x = x

SUMMARY Common Logarithm Natural Logarithm log xy = log x ln xy = ln x + + log y ln y log x/y = log ln x/y = ln x - log y ln y log xy = y log ln xy = y ln x x log x 1/y = (1/y )log x ln x 1/y =(1/y)ln x

In summary Number Exponential Expression Logarithm 1000 103 3 100 102 2 10 101 1 1 100 0 1/10 = 0. 1 10 -1 -1 1/100 = 0. 01 10 -2 -2 1/1000 = 0. 001 10 -3 -3

Simplify the following expression log 59 + log 23 + log 26 • We need to convert to “Like bases” (just like fraction) so we can add • Convert to base 10 using the “Change of base formula” • (log 9 / log 5) + (log 3 / log 2) + (log 6 / log 2) • Calculates out to be 5. 535
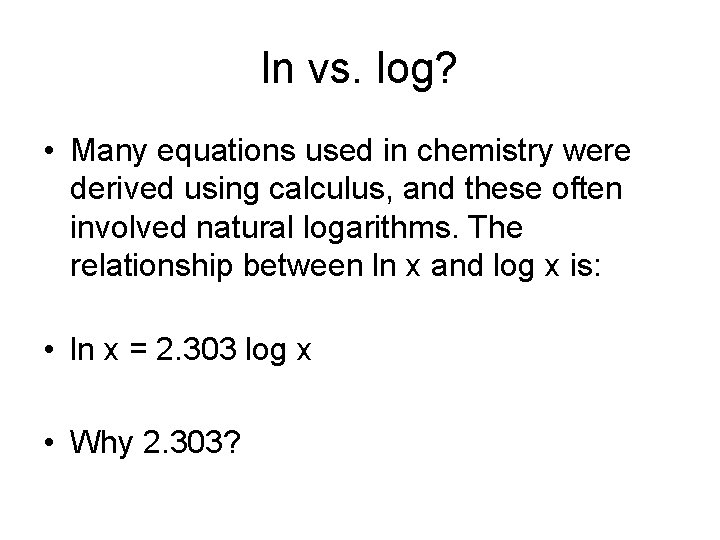
ln vs. log? • Many equations used in chemistry were derived using calculus, and these often involved natural logarithms. The relationship between ln x and log x is: • ln x = 2. 303 log x • Why 2. 303?

What’s with the 2. 303; • Let's use x = 10 and find out for ourselves. • Rearranging, we have (ln 10)/(log 10) = number. • We can easily calculate that ln 10 = 2. 302585093. . . or 2. 303 and log 10 = 1. So, substituting in we get 2. 303 / 1 = 2. 303. Voila!

Sig Figs and logs • For a measured quantity, the number of digits after the decimal point equals the number of sig fig in the original number • 23. 5 measured quantity 3 sig fig • Log 23. 5 = 1. 371 3 sig fig after the decimal point

More log sig fig examples • log 2. 7 x 10 -8 = -7. 57 The number has 2 significant figures, but its log ends up with 3 significant figures. • ln 3. 95 x 106 = 15. 189 the number has 5 3

OK – now how about the Chem. • LOGS and Application to p. H problems: • p. H = -log [H+] • What is the p. H of an aqueous solution when the concentration of hydrogen ion is 5. 0 x 10 -4 M? • p. H = -log [H+] = -log (5. 0 x 10 -4) = - (-3. 30) • p. H = 3. 30
![Inverse logs and p. H • p. H = -log [H+] • What is Inverse logs and p. H • p. H = -log [H+] • What is](http://slidetodoc.com/presentation_image_h/a8b7c894af52d65e38d52c67f3e3e10b/image-24.jpg)
Inverse logs and p. H • p. H = -log [H+] • What is the concentration of the hydrogen ion concentration in an aqueous solution with p. H = 13. 22? • p. H = -log [H+] = 13. 22 log [H+] = -13. 22 [H+] = inv log (-13. 22) [H+] = 6. 0 x 10 -14 M (2 sig. fig. )
- Slides: 24